Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.117464
Revised: January 11, 2026
Accepted: February 5, 2026
Published online: March 20, 2026
Processing time: 97 Days and 16 Hours
Diabetic ketoacidosis (DKA) resulting from type 2 diabetes mellitus (T2DM) is less common, and the factors associated with adverse outcomes and mortality are not well established based on large-scale studies.
To identify the risk factors for adverse outcomes and mortality in treated patients with DKA in T2DM.
Retrospective analysis of patients admitted to a tertiary-care hospital in the United Kingdom with DKA and T2DM for inpatient management between January 2010 to September 2024 to identify the clinical profile, demographic features, and laboratory parameters impacting treatment outcomes and survival.
Four hundred and sixty-four patients were included. Of these 395 (85.13%) were White, 266 (57.3%) were males with a mean age at presentation of 61.3 (17.6) years, median inpatient hospital stay of 5 (interquartile range: 3-10.3) days, and a mean glycated HbA1c of 89.3 (30) mmol/mol. The 30-day and 90-day mortality were 13.4% and 11% respectively after the index DKA event. The long-term survival after the DKA event was only 58.6%. Presence of cerebrovascular disease [odds ratio (OR): 6.75; 95%CI: 0.76-12.74], use of sodium glucose cotransporter 2 inhibitors (OR: 5.8; 95%CI: 1.32-9.62), chronic obstructive pulmonary disease (OR: 3.6; 95%CI: 2.14-6.44), higher national early warning score 2 score (OR: 1.14; 95%CI: 0.10-2.18) and low systolic blood pressure (OR: -0.18; 95%CI: -0.32 to -0.04) at admission were the significant predictors of longer inpatient stay. Coexistent peripheral vascular disease (PVD; OR: 46.43) and congestive heart failure (CHF; OR: 30.83), and lower estimated glomerular filtration rate (eGFR; OR: 0.98) were the important predictors of mortality during the hospital treatment. The significant predictors on 30-day mortality were: Age (OR: 1.06), eGFR (0.97) and index of multiple deprivation (IMD) decile (0.74). The factors associated with long-term mortality risk were dementia (20.54-fold higher), continued use of sulfonylurea/met
DKA carries a serious risk of mortality in both the short and long term in T2DM patients. Factors such as older age, dementia, PVD, CHF, low eGFR define the riskiest groups. These groups of patients may benefit from closer follow-up and more aggressive metabolic and comorbidity management after discharge.
Core Tip: Diabetic ketoacidosis (DKA) resulting from type 2 diabetes mellitus (T2DM) is less common. A retrospective analysis of 464 patients from a tertiary-care United Kingdom hospital with DKA and T2DM identified that mortality rates at 30 and 90 days were 13.4% and 11% respectively after the index DKA event. The survival after the DKA event was only 58.6% on long-term follow-up. Coexistent peripheral vascular disease, congestive heart failure, and lower estimated glo
- Citation: Chandrabalan V, Howcroft T, Alkhalifah MK, Younis N, Aldhafiri E, Pappachan JM. Diabetic ketoacidosis in patients with type 2 diabetes: Risk factors for mortality and adverse outcomes. World J Exp Med 2026; 16(1): 117464
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/117464.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.117464
Diabetic ketoacidosis (DKA) occurs as an acute, serious metabolic decompensation in patients with diabetes mellitus requiring emergency hospitalisation for treatment and associated with a high risk of multiorgan complications and mortality. DKA occurs as a consequence of absolute or relative insulin deficiency and therefore is a usual metabolic complication in patients with type 1 diabetes mellitus (T1DM)[1]. Insulin deficiency results in excess hepatic glucose output (from increased gluconeogenesis and glycogenolysis) along with impaired glucose uptake by skeletal muscle and adipose tissue associated with symptomatic hyperglycemia (polydipsia and polyurea) seen in DKA[2]. Insulin deficiency along with activation of other counterregulatory hormones (especially glucagon) result in adipolysis with the release of free fatty acids, the metabolism of which causes production of ketoacids and metabolic acidosis of DKA.
Type 2 diabetes mellitus (T2DM) usually results from insulin resistance (IR) and patients are often hyperinsulinemic because of IR. Although hyperinsulinemia often protects patients with T2DM from DKA, there can be situations where the IR and relative insulin deficiency can occasionally worsen as in severe infections, inflammatory states, and in conditions with acute metabolic stress (with higher insulin requirement due to severe IR) when these patients also develop DKA[2-4]. Marked insulin deficiency occurs in some patients with advanced stages of T2DM due to β-cell failure and with pathobiological similarity to T1DM making them prone to develop DKA[5]. In the recent years, euglycemic ketoacidosis resulting from the use of sodium glucose cotransporter 2 inhibitors (SGLT-2i) has been an important reason for development of DKA in patients with T2DM[6-8].
As DKA is metabolic emergency associated with several complications like acute kidney injury (AKI)[9], electrolyte imbalance[10], respiratory failure requiring intubation[11], cerebral edema[12], and even death[13,14], it is crucial to identify the risk factors for DKA in every patient with diabetes. Because of the lower risk of DKA, patients with T2DM are not often assessed for this potentially lethal complication in usual clinical practice. As the incidence and prevalence of T2DM is increasing exponentially in recent years due to the obesity pandemic globally, it is very important to identify patients who are at risk of DKA to optimise management strategies. This study is such an attempt to identify the risk factors and complications associated with DKA in a large teaching hospital in the United Kingdom.
This is a retrospective cohort study conducted at the Lancashire Teaching Hospitals NHS Trust (LTHTR) to investigate the clinical and laboratory profile, and the treatment outcomes of all patients admitted with DKA and T2DM for inpatient management between January 2010 to September 2024. The diabetes service of LTHTR provides appropriate diabetic care for patients residing in the Central Lancashire and South Cumbria regions of the United Kingdom, with a population of about 0.4 million people. The study had the appropriate institutional review board clearance (No. DIAB/CA/2021-22/03).
All adult patients (age ≥ 18 years) with T2DM admitted with DKA during the above period were screened for inclusion. The Joint British Diabetes Society criteria for DKA were used to identify patients (capillary glucose > 11 mmol/L, capillary ketone > 3 mmol/L or urine ketone ≥ ++ and venous pH < 7.35 and/or bicarbonate < 15 mmol/L). When patients had been on SGLT-2i class of medications, even those with plasma glucose level of < 11 mmol/L also was considered as having DKA when other criteria were met in presence of an anion gap > 10.
LTHTR have transformed routinely collected clinical data from multiple electronic health record (EHR) systems into the Observational Medical Outcomes Partnership Common Data Model (OMOP CDM) for nearly 2 million patients. This database has been accredited to standards specified by the European Health Data Evidence Network (EHDEN) and metadata is available on the EHDEN Portal as Incrementing Database for Research in Lancashire. Data from January 2010 was used for this study reflecting the improving data capture, quality and density in our EHR over time.
Patients with a diagnosis of T2DM who developed DKA during the study period. Those who had treatment for DKA with data available on the clinical records for assessing the prespecified primary outcome measures.
DKA patients with other types of diabetes (T1DM, pancreatitis-related diabetes, diagnosis of T2DM uncertain), and incomplete data in the clinical records to assess primary outcomes.
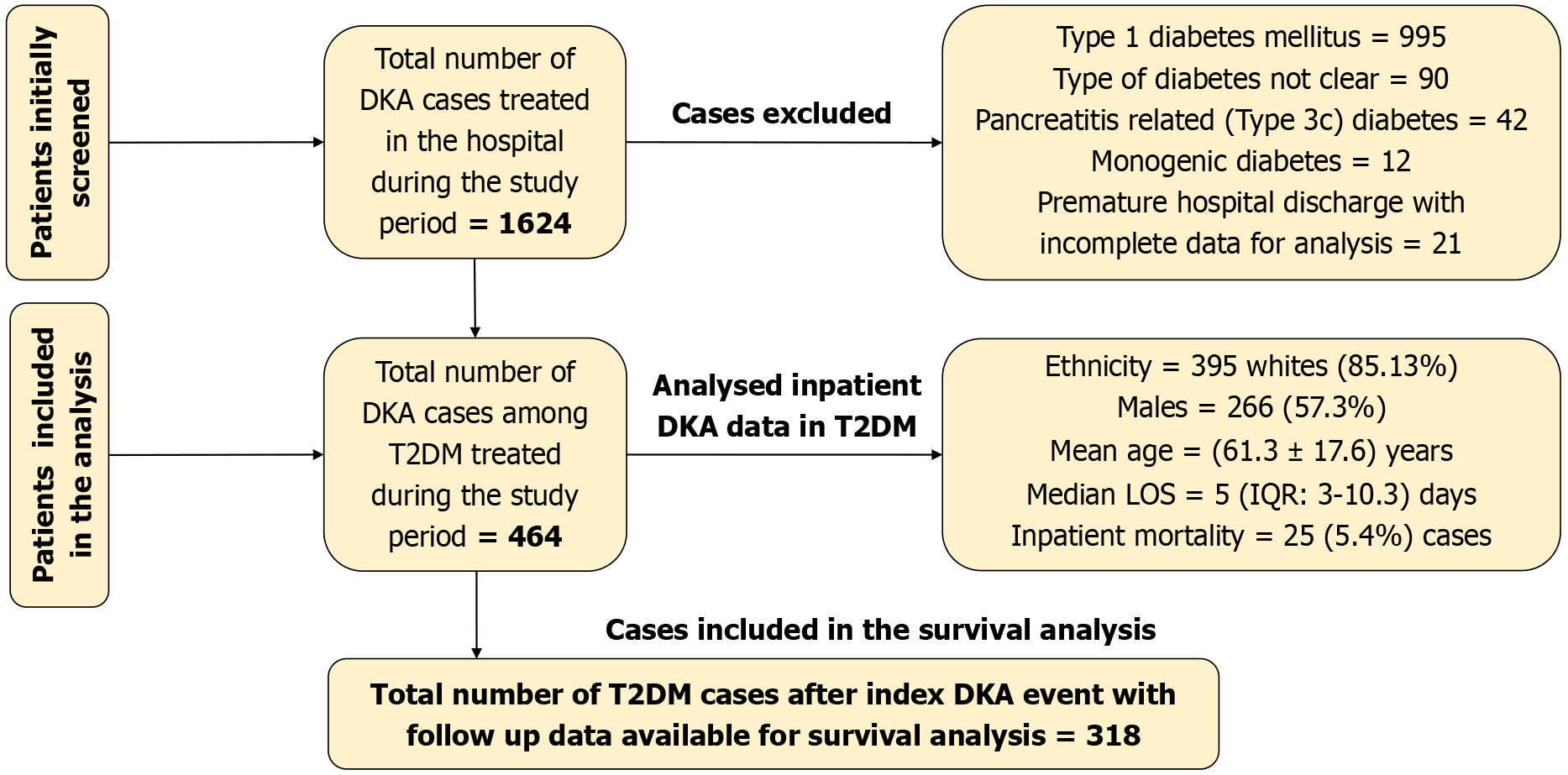
All patients with a diagnosis of T2DM who were admitted to hospital with DKA to during the study period were identified using diagnostic codes. Demographic information (age, sex, and ethnic background), associated comorbidities, clinical observations and laboratory information at the time of admission with DKA, the duration of inpatient hospital stays, and death during, within 30 days and longer term during follow up were also recorded. For various analyses, we only included cases with meaningful data during follow-up to obtain statistical validity. A STROBE diagram of study selection process is shown in the Figure 1.
Primary outcome measures: The primary outcomes assessed were the completion of DKA treatment with resolution of acidosis and ketonemia and/or death following DKA.
Secondary outcome measures: Length of hospital stay (LOS) and the contribution of various patient characteristics including the clinical/Laboratory parameters during admission, demographic variables, presence of comorbidities, and treatment modalities on death or duration of hospital stay.
All analysis was conducted with LTHTR’s Trusted Research Environment using Python programming language (https://www.python.org/) and the statsmodels statistical analysis library (https://www.statsmodels.org/). Charlson comor
A total of 464 patients with T2DM were admitted to hospital with DKA during the study period. 266 (57.3%) were males and 395 (85.13%) were White with the remainder other ethnic groups [Asians 41 (8.84%), Afro-Caribbean 10 (2.1%) and ethnicity not recorded 18 (3.88%)]. Mean age at presentation was 61.3 ± 17.6 years and median age was 60 (51-76) years. Table 1 shows the comorbidities recorded and Table 2 shows the baseline clinical and lab features. The Figure 1 below shows the study flow diagram.
| Comorbidity | Number of cases | Prevalence (%) |
| COPD | 96 | 20.69 |
| AKI | 80 | 17.24 |
| Cerebrovascular disease | 69 | 14.87 |
| Congestive heart failure | 39 | 8.41 |
| Cancer | 35 | 7.54 |
| Acute MI | 31 | 6.68 |
| Peptic ulcer | 29 | 6.25 |
| Dementia | 28 | 6.03 |
| Hypertension | 21 | 4.53 |
| Peripheral vascular disease | 18 | 3.88 |
| Metastatic cancer | 11 | 2.37 |
| Rheumatoid arthritis | 11 | 2.37 |
| Mild liver disease | 7 | 1.51 |
| Lab parameter | Mean (SD) | Median (25th-75th percentiles) |
| HbA1c (mmol/mol) | 89.5 (30) | 88.5 (51-130) |
| NEWS 2 score | 3.6 (3) | 5 (3-15) |
| Potassium | 3.6 (0.7) | 3.6 (3.2-4.0) |
| eGFR | 56.4 (26.5) | 56.5 (34-85.3) |
| Systolic BP | 106 (21) | 103 (95-114) |
| pH | 7.2 (0.2) | 7.3 (7.1-7.3) |
The median LOS was 5 (3-10.3) days and the mean was 8.7 ± 10.3 days. Cerebrovascular disease, use of SGLT-2i, chronic obstructive pulmonary disease (COPD), and higher national early warning score (NEWS) 2 score and low systolic blood pressure (BP) at admission were the significant predictors of longer inpatient stay, while sex, and medical conditions like AKI, hypertension, rheumatoid disease and cancer did not affect the length of stay. Table 3 shows the factors influencing LOS. This linear regression model was aimed to identify factors associated with LOS in a hospital setting, using a variety of clinical, demographic, and biochemical variables.
| Predictor | Coefficient | 95%CI | P value | Interpretation |
| Intercept | -19.14 | (-110.98 to 72.70) | 0.679 | Baseline length of stay (reference group) |
| Cerebrovascular disease | 6.75 | (0.76 to 12.74) | 0.028 | Adds about 6.7 days to LOS |
| Those who were on SGLT-2 inhibitors | 5.80 | (1.32 to 9.62) | 0.035 | Adds about 5.8 days to LOS |
| COPD | 3.60 | (2.14 to 6.44) | 0.04 | Adds about 3.6 days to LOS |
| Higher NEWS 2 score | 1.14 | (0.10 to 2.18) | 0.033 | Each NEWS 2 point adds about 1.14 days to LOS |
| Systolic BP | -0.18 | (-0.32 to -0.04) | 0.014 | Lower systolic BP predicts longer LOS |
| Acute myocardial infarction | -6.06 | (-12.15 to 0.03) | 0.051 | Shortens LOS by about 6 days (only borderline significance as P > 0.05 and confidence interval crosses 0) |
Interestingly, lower systolic BP at admission (below 120/80 mm Hg) was also significantly associated with longer hospitalisation, with each mmHg reduction predicting an additional 0.18 days of stay. Lower systolic BP usually implies circulatory volume depletion in patients with DKA indicating more severe disease. This also likely reflects the increased complexity and monitoring required in hypotensive patients. Conversely, a recent myocardial infarction was associated with a shorter LOS (about 6 days), though this finding was borderline significant and may be influenced by early discharge pathways (for treatment in other hospitals for coronary interventions), or mortality bias.
The model also found that individuals whose race was not recorded had longer stays (about 10 days), suggesting that missing or poorly classified demographic data may correlate with unmeasured clinical or social complexity. However, most other demographic variables (such as sex, age, and other race groups) and diabetes medications (except SGLT-2i) did not demonstrate significant associations with LOS.
Twenty-five (5.4%) patients died during hospital admission while 37 (8%) died within 30 days and 51 (11%) died within 90 days of the index event. At last follow-up, 272 patients (58.6%) were alive, corresponding to 192 deaths.
This logistic regression model identified several variables significantly associated with in-hospital mortality. The overall model fit was strong, with a pseudo-R2 of 0.438 and a highly significant likelihood ratio test (P < 0.000001), indicating that the model explains a meaningful portion of the variability in patient outcomes (Table 4).
| Predictor | Coefficient | Std. error | P value | 95%CI | Odds ratio |
| Congestive heart failure | 3.43 | 0.989 | 0.001 | 1.49 to 5.37 | 30.83 |
| Peripheral vascular disease | 3.84 | 1.288 | 0.003 | 1.31 to 6.36 | 46.43 |
| Use of metformin pre-admission | -2.39 | 0.71 | 0.001 | -3.78 to -0.99 | 0.09 |
| Minimum eGFR value (indicating baseline kidney function) | -0.023 | 0.011 | 0.038 | -0.044 to -0.001 | 0.98 |
Congestive heart failure (CHF) was the strongest positive predictor: Patients with CHF had over 30 times higher odds of death in hospital compared to those without, even when adjusting for age, comorbidities, medications, and other laboratory values. Similarly, peripheral vascular disease (PVD) increased the odds of death by over 46 times, highlighting the substantial risk these vascular conditions confer.
In contrast, the use of metformin prior to admission was associated with a 91% reduction in the odds of death. This strong protective effect remained significant after adjusting for diabetes complications and other treatments, suggesting either a therapeutic benefit or that metformin use may serve as a proxy for better baseline health. Finally, lower estimated glomerular filtration rate (eGFR) values (worse kidney function) were also associated with increased mortality risk, with each unit increase in eGFR decreasing the odds of death by approximately 2%.
Other predictors, such as age, dementia, and NEWS 2 scores, showed suggestive trends but did not reach statistical significance in this model.
This logistic regression model examined the predictors of 30-day mortality (Table 5) after index hospitalisation among 318 patients (with available data). The model demonstrated statistically significant overall performance (log-likelihood ratio P < 0.000001) and identified four key predictors.
| Predictor | Coefficient | Std. error | P value | 95%CI | Odds ratio |
| Intercept | -3.92 | 1.452 | 0.007 | -6.77 to -1.08 | 0.02 |
| IMD decile (deprivation index) | -0.31 | 0.104 | 0.003 | -0.51 to -0.10 | 0.74 |
| Age | 0.058 | 0.02 | 0.003 | 0.020 to 0.097 | 1.06 |
| Minimum eGFR value | -0.033 | 0.012 | 0.005 | -0.057 to -0.010 | 0.97 |
The age of the patient was a significant positive predictor: For each additional year of age, the odds of dying within 30 days increased by approximately 6%. Similarly, lower values of eGFR, a measure of kidney function, were associated with a higher mortality risk. Each unit increase in eGFR reduced the odds of 30-day mortality by about 3%, underscoring the importance of renal function as a mortality determinant.
Importantly, patients from more deprived areas (lower IMD decile) also had higher odds of dying, with each unit increase in the IMD decile (indicating less deprivation) associated with a 26% reduction in 30-day mortality risk. This finding highlights the continuing influence of socioeconomic factors on clinical outcomes.
The intercept was also statistically significant, representing the log-odds of death for a reference patient (male, average covariate values), but is not directly interpretable in isolation.
Other variables such as sex, heart failure, vascular disease, and length of stay were not significantly associated with 30-day mortality in this sample, although their inclusion improved the overall model fit.
We performed an analysis of 318 patients, with follow up data (Table 6) available in our records, to check the longer-term survival after index hospitalisation with DKA. This Cox regression model (with data available in our database) revealed several significant predictors of mortality risk over time among 105 patients, all of whom experienced the event of interest (death). The concordance index of 0.73 suggests the model had strong discriminative ability for survival outcomes.
| Predictor | Coefficient | Exp (coefficient) | Std. error | 95%CI | P value |
| Age | 0.04 | 1.04 | 0.01 | 0.02 to 0.06 | < 0.005 |
| Use of metformin | 1.89 | 6.62 | 0.53 | 0.85 to 2.93 | < 0.005 |
| Use of sulfonylureas | 2.13 | 8.39 | 0.66 | 0.83 to 3.42 | < 0.005 |
| Cerebrovascular disease | -1.08 | 0.34 | 0.44 | -1.94 to -0.22 | 0.01 |
| Dementia | 3.02 | 20.54 | 0.67 | 1.72 to 4.33 | < 0.005 |
Age was a consistent and significant predictor: Each additional year of age was associated with a 4% increase in hazard of death. Two antidiabetic therapies (prior to index hospitalisation) showed opposing effects: Use of sulfonylureas was associated with an 8.4-fold increase in hazard, and metformin use with a 6.6-fold increase, although the latter might reflect confounding due to treatment assignment or underlying severity. Though it appears paradoxical at first glance that metformin use appears to be strongly protective for in-hospital mortality (OR = 0.09) and increases the HR in the long-term mortality model, these findings are probably not implying a cause-effect relationship but may be related to patient selection and treatment periods.
Dementia was associated with a striking 20.5-fold increase in hazard, indicating a major survival disadvantage in this population. In contrast, patients with cerebrovascular disease (CEVD) demonstrated a 66% lower hazard, which may reflect early discharge, end-of-life care outside hospital, or misclassification of cause of death. These findings underscore the need for tailored clinical monitoring and support for older adults, especially those with cognitive impairment or complex diabetes regimens.
The study identified that the inpatient mortality due to DKA in patients with T2DM was 5.4% with another 8% of cases dying within 30 days (overall 30-day mortality 13.4%) and 11% within 90 days of the index DKA event. We also found that the long-term survival after the DKA event is only 58.6%. Presence of cerebrovascular disease (OR: 6.75), use of SGLT-2 inhibitors (OR: 5.8), COPD (OR: 3.6), higher NEWS 2 score at admission (OR: 1.14) and systolic BP (OR: -0.18) were the significant predictors of longer inpatient stay. Coexistent PVD (OR: 46.43) and CHF (OR: 30.83), eGFR (OR: 0.98) and metformin use (OR: 0.09) were the important predictors of mortality during the hospital treatment. The significant predictors on 30-day mortality were: Higher age (OR: 1.06), eGFR (0.97) and IMD decile (0.74). The factors associated with long-term mortality risk were dementia (20.54 higher), use of older antidiabetic agents like sulphonylurea and metformin, and age (4% higher with each additional year).
Historically, DKA has been identified as a significant risk factor for immediate and long-term complications including excess mortality. A recent study by Song and Frier[15] suggested that the mortality risk is higher among patients with T2DM compared to those with T1DM who develop DKA. They found a mortality rate of 30.8% with median time of death 43 days. As we were unable to identify the time of death among those who died after 90 days of hospital discharge, we are unable to assess the median time of death in all cases. However, we observed a lower overall mortality rate of only 24.4% at 90 days of the index DKA event, while the mortality rate reported by Song and Frier[15] was apparently higher. Though we observed a lower in hospital mortality rate of 5.4% compared to 6.4% observed from a recent study reported from Israel[16], we observed a much higher mortality rate with 19% of cases dying within 90 days of the index DKA event, while only 7.7% of deaths were observed in the Israeli cohort even with a comparable mean age of 63 (13 years) of patients in both cohorts. Varying demographic and patient related factors such as different comorbidity burden specific populations could have influenced this wide discrepancy in the mortality rates observed in different studies. Moreover, wide use of disease modifying agents like SGLT-2i with mortality benefits in managing T2DM in the later part of the study period would have impacted the adverse survival in our cohort. The wide variation in the mortality rates was also observed in a very recent systematic literature review examining the burden of DKA among T2DM patients across the world[17].
We found a shorter LOS (mean 8.7 days and median 5 days) compared to a much longer period of stay (13.4 ± 17.6) days in a large study data on DKA among T2DM patients reported from Australia between 2002 and 2016[17,18]. Better treatment facilities in recent years for patients with DKA and lower mean age (61.3 ± 17.6) years at presentation in our cohort would probably explain this discrepancy. Several factors associated with prolongation of hospital stay in DKA were identified in different studies[19-21]. Again there have been wide discrepancies in the reasons for prolonged hospitalization such as low pH at admission, higher HbA1c, electrolyte disturbances, higher Charlson comorbidity index, need for intensive care unit admission, older age, male sex, and obesity were the predictors of longer hospital stays in these studies. However, we observed that cerebrovascular disease, use of SGLT-2 inhibitors, COPD, and higher NEWS 2 score and lower systolic BP at admission were the predictors of longer hospital stay, while race, sex, and medical comorbidities such as MI, AKI, rheumatoid disease and cancer had no effects.
Our observation of a 4% excess risk of mortality with each year of increasing age after the index DKA event is especially important as potential preventive strategies can be sought in such older individuals. As we observed poor metabolic control with a high mean HbA1c (89.5 ± 30 mmol/mol) in our cohort, prompt glycemic control could have potentially improved the long-term survival or at least prevented several complications associated with diabetes. Long-term follow-up (34 years) data from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study indicated that DKA was associated with an excess risk of all-cause mortality (HR: 9.13), recurrent DKA (HR: 3.24), advanced kidney disease (HR: 2.10), major adverse cardiac events (HR: 1.66), severe hypoglycemia (HR: 1.53) and advanced neuropathy (HR: 1.49) compared to those without an incident DKA[22]. Though this study is on patients with T1DM, we have to bear in mind that poorly controlled T2DM clinically behaves similar to T1DM with acute metabolic complications including DKA. Therefore, our cohort with very high HbA1c would have been insulinopenic with higher immediate and long-term morbidity and mortality risks.
Poorer survival among those with dementia, heart failure, and PVD among our DKA cohort imply that these subgroups should be managed more rigorously to reduce the mortality risk. Liu et al[23] showed that dementia is associated with a 30% excess long-term mortality risk among patients admitted with a hyperglycemic complication such as DKA in T2DM patients. Poorer survival in those with PVD observed in our cohort could be related to adverse cardiovascular outcomes in patients with diabetes as evidenced by previous studies[24,25]. The apparent excess mortality observed among those treated with sulphonylureas and biguanides could be explained by the fact that patients receiving newer antidiabetic medications such as glucagon-like peptide-1 receptor agonists and SGLT-2i in our cohort would have had survival advantage over the older agents as these drugs reduce overall and cardiovascular mortality as shown in multiple studies[26-28].
We acknowledge the following limitations to our study. While the use of the OMOP CDM accelerated evidence generation, retrospective analysis of routinely collected data from EHRs is a limitation with incomplete data capture for some of the clinicopathological measures of interest. Moreover, all diagnostic coding data in the database were derived from data entered by the hospital’s coding department, there could have been occasional errors in relation to faulty or missed codes. We were also unable to get data on the causes of death after 90 days of discharge. Moreover, the apparently protective effects on mortality conferred by metformin use (inpatient mortality) and presence CEVD (long term mortality) could be artefactual due to the peculiarity of our data and should be interpreted with caution in making clinical practice decisions.
However, the study provides results from a large data with a very long follow-up period of over 15 years which make our observations unique. The analyzed clinical and laboratory parameters provide a reasonably robust evidence-base for the immediate and longer-term survival, the risk factors for acute complications, and the LOS in this study cohort to strengthen our current understanding on the management of T2DM and DKA.
The inpatient mortality due to DKA in patients with T2DM was comparatively low at 5.4% compared with previously reported rates with an overall 90-day death risk of 24.4% after the index DKA event. The long-term survival following a DKA event is only 58.6% at 15 years. DKA carries a serious risk of mortality in both the short and long term in T2DM patients. Factors such as higher age, presence of dementia, PVD, CHF, and low eGFR define the highest-risk group. These patients may benefit from closer follow-up and more aggressive metabolic/comorbidity management after discharge.
| 1. | Veauthier B, Levy-Grau B. Diabetic Ketoacidosis: Evaluation and Treatment. Am Fam Physician. 2024;110:476-486. [PubMed] |
| 2. | Lizzo JM, Goyal A, Kaur J. Adult Diabetic Ketoacidosis. 2025 Nov 30. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 3. | Roberts CGP, Athinarayanan SJ, Ratner RE, Umpierrez GE. Illnesses associated with ketosis including diabetic ketoacidosis during very low carbohydrate and ketogenic diets. Diabetes Obes Metab. 2025;27:2531-2539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 4. | Wang L, Meng X, Tang Y, Hao Y. Altered association between cortisol and adrenocorticotropic hormone levels in the early stage of type 2 diabetic ketoacidosis. Front Endocrinol (Lausanne). 2025;16:1418357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 5. | Donath MY, Halban PA. Decreased beta-cell mass in diabetes: significance, mechanisms and therapeutic implications. Diabetologia. 2004;47:581-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 284] [Article Influence: 12.9] [Reference Citation Analysis (1)] |
| 6. | Baek HS, Jeong C, Yang Y, Lee J, Lee J, Lee SH, Cho JH, Sohn TS, Son HS, Yoon KH, Lee EY. Diabetic Ketoacidosis as an Effect of Sodium-Glucose Cotransporter 2 Inhibitor: Real World Insights. Diabetes Metab J. 2024;48:1169-1175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 7. | Zhao Z, Zhao F, Zhang Y, Hu X, Li J, Tian C, Jin P, Liu D. Risk factors of dapagliflozin-associated diabetic ketosis/ketoacidosis in patients with type 2 diabetes mellitus: A matched case-control study. Diabetes Res Clin Pract. 2023;196:110236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 8. | Cohen B, Harris YT, Schulman-Rosenbaum R. Sodium-Glucose Cotransporter 2 Inhibitors Should Be Avoided for the Inpatient Management of Hyperglycemia. Endocr Pract. 2024;30:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 9. | Meena J, Yadav J, Kumar J, Dawman L, Tiewosh K, Mittal A, Kumar R, Dayal D. Incidence, predictors, and short-term outcomes of acute kidney injury in children with diabetic ketoacidosis: a systematic review. Pediatr Nephrol. 2023;38:2023-2031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 10. | Szabó GV, Szigetváry C, Turan C, Engh MA, Terebessy T, Fazekas A, Farkas N, Hegyi P, Molnár Z. Fluid resuscitation with balanced electrolyte solutions results in faster resolution of diabetic ketoacidosis than with 0.9% saline in adults - A systematic review and meta-analysis. Diabetes Metab Res Rev. 2024;40:e3831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 11. | Dunn BK, Coore H, Bongu N, Brewer KL, Kumar D, Malur A, Alkhalisy H. Treatment Challenges and Controversies in the Management of Critically Ill Diabetic Ketoacidosis (DKA) Patients in Intensive Care Units. Cureus. 2024;16:e68785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (3)] |
| 12. | Hamud AA, Mudawi K, Shamekh A, Kadri A, Powell C, Abdelgadir I. Diabetic ketoacidosis fluid management in children: systematic review and meta-analyses. Arch Dis Child. 2022;107:1023-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 13. | Maharjan J, Pandit S, Arne Johansson K, Khanal P, Karmacharya B, Kaur G, Kumar Aryal K. Effectiveness of interventions for emergency care of hypoglycaemia and diabetic ketoacidosis: A systematic review. Diabetes Res Clin Pract. 2024;207:111078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 14. | Pannu AK, Saroch A, Singla V, Sharma N, Dutta P, Jain A, Angrup A. Clinical spectrum, etiology and outcome of infectious disease emergencies in adult diabetic patients in northern India. Diabetes Metab Syndr. 2020;14:921-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 15. | Song SH, Frier BM. Severe hypoglycaemia and diabetic ketoacidosis in adults presenting to a hospital emergency department: Adverse prognostic markers for survival in type 2 diabetes and the role of SGLT2 inhibitors. Diabet Med. 2025;42:e15466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 16. | Keler M, Vlasov P, Elkan M, Koren S, Koren R. Hospitalization Outcomes of Patients with Type 2 Diabetes Mellitus Complicated with Diabetic Ketoacidosis. Isr Med Assoc J. 2024;26:566-571. [PubMed] |
| 17. | Wysham C, Bindal A, Levrat-Guillen F, Kostadinova D, Poon Y. A systematic literature review on the burden of diabetic ketoacidosis in type 2 diabetes mellitus. Diabetes Obes Metab. 2025;27:2750-2767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 18. | Jones HC, Kiburg KV, Lee MH, O'Neal DN, MacIsaac RJ. Trends in diabetic ketoacidosis in Victoria, Australia 2002-2016. BMC Endocr Disord. 2024;24:128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 19. | Ata F, Khan AA, Khamees I, Iqbal P, Yousaf Z, Mohammed BZM, Aboshdid R, Marzouk SKK, Barjas H, Khalid M, El Madhoun I, Bashir M, Kartha A. Clinical and biochemical determinants of length of stay, readmission and recurrence in patients admitted with diabetic ketoacidosis. Ann Med. 2023;55:533-542. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 20. | Saadeh NA, Hammouri HM, Zahran DJ. Diabetic Ketoacidosis in Northern Jordan: Seasonal Morbidity and Characteristics of Patients. Diabetes Metab Syndr Obes. 2023;16:3057-3064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 21. | Khan AA, Ata F, Iqbal P, Bashir M, Kartha A. Clinical and biochemical predictors of intensive care unit admission among patients with diabetic ketoacidosis. World J Diabetes. 2023;14:271-278. [PubMed] [DOI] [Full Text] |
| 22. | Budhram DR, Bapat P, Bakhsh A, Abuabat MI, Verhoeff NJ, Mumford D, Orszag A, Jain A, Cherney DZI, Fralick M, Weisman A, Lovblom LE, Perkins BA. Prognostic Implications of Diabetic Ketoacidosis in Adults on Long-term Mortality and Diabetes-Related Complications. Can J Diabetes. 2024;48:462-470.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 23. | Liu CL, Lin MY, Hwang SJ, Liu CK, Lee HL, Wu MT. Association of hyperglycemia episodes on long-term mortality in type 2 diabetes mellitus with vascular dementia: A population-based cohort study. J Diabetes Complications. 2019;33:123-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 24. | Kim C, Yang YS, Ryu GW, Choi M. Risk factors associated with amputation-free survival for patients with peripheral arterial disease: a systematic review. Eur J Cardiovasc Nurs. 2021;20:295-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 25. | Agnelli G, Belch JJF, Baumgartner I, Giovas P, Hoffmann U. Morbidity and mortality associated with atherosclerotic peripheral artery disease: A systematic review. Atherosclerosis. 2020;293:94-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 163] [Article Influence: 27.2] [Reference Citation Analysis (1)] |
| 26. | Lee MMY, Sattar N, Pop-Busui R, Deanfield J, Emerson SS, Inzucchi SE, Mann JFE, Marx N, Mulvagh SL, Poulter NR, Badve SV, Pratley RE, Perkovic V, Buse JB, McGuire DK; SOUL Trial Investigators. Cardiovascular and Kidney Outcomes and Mortality With Long-Acting Injectable and Oral Glucagon-Like Peptide 1 Receptor Agonists in Individuals With Type 2 Diabetes: A Systematic Review and Meta-analysis of Randomized Trials. Diabetes Care. 2025;48:846-859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 25.0] [Reference Citation Analysis (2)] |
| 27. | Kunutsor SK, Seidu BS, Seidu S. Cardiovascular effectiveness of newer glucose-lowering agents, with and without baseline lipid-lowering therapy in type 2 diabetes: A systematic meta-analysis of cardiovascular outcome trials and real-world evidence. Prim Care Diabetes. 2024;18:589-598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 28. | Zhang M, Lin C, Cai X, Jiao R, Bai S, Li Z, Lv F, Yang W, Liu G, Yang X, Ji L. One or two? Comparison of the cardiorenal effects between combination therapy and monotherapy with SGLT2i or GLP1RA. Diabetes Obes Metab. 2025;27:806-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |