Published online Sep 9, 2021. doi: 10.5492/wjccm.v10.i5.301
Peer-review started: April 6, 2021
First decision: June 5, 2021
Revised: June 17, 2021
Accepted: August 24, 2021
Article in press: August 24, 2021
Published online: September 9, 2021
Processing time: 154 Days and 20.8 Hours
Panton-Valentine leukocidin (PVL) is an exotoxin secreted by Staphylococcus aureus (S. aureus), responsible for skin and soft tissue infections. As a cause of severe necrotising pneumonia, it is associated with a high mortality rate. A rare entity, the epidemiology of PVL S. aureus (PVL-SA) pneumonia as a complication of influenza coinfection, particularly in young adults, is incompletely understood.
An adolescent girl presented with haemoptysis and respiratory distress, deteriorated rapidly, with acute respiratory distress syndrome (ARDS) and profound shock requiring extensive, prolonged resuscitation, emergency critical care and venovenous extracorporeal membrane oxygenation (ECMO). Cardiac arrest and a rare complication of ECMO cannulation necessitated intra-procedure extracorporeal cardiopulmonary resuscitation, i.e., venoarterial ECMO. Coordinated infectious disease, microbiology and Public Health England engagement identified causative agents as PVL-SA and influenza A/H3N2 from bronchial aspirates within hours. Despite further complications of critical illness, the patient made an excellent recovery with normal cognitive function. The coordinated approach of numerous multidisciplinary specialists, nursing staff, infection control, specialist cardiorespiratory support, hospital services, both adult and paediatric and Public Health are testimony to what can be achieved to save life against expectation, against the odds. The case serves as a reminder of the deadly nature of PVL-SA when associated with influenza and describes a rare complication of ECMO cannulation.
PVL-SA can cause severe ARDS and profound shock, with influenza infection. A timely coordinated multispecialty approach can be lifesaving.
Core Tip: We present a case of profound vasoplegic shock and acute respiratory distress syndrome in a healthy adolescent after a mild prodromal illness. Acute cardiorespiratory failure persisted despite aggressive resuscitation and vasoactive support. Cardiac arrest and complicated extracorporeal membrane oxygenation (ECMO) cannulation necessitated emergency venoarterial-ECMO during cardiopulmonary resuscitation. Early respiratory samples confirmed H3N2 influenza and Panton-Valentine leukocidin-Staphylococcus aureus (PVL-SA) pneumonia: A rare and serious manifestation of the PVL-SA, usually associated with less severe skin and soft-tissue infections. The patient’s ultimate survival and recovery depended on the extraordinary interplay and rapid utilisation of multidisciplinary teams which we highlight for the benefit of other services to ensure optimal outcomes, even against the odds.
- Citation: Cuddihy J, Patel S, Mughal N, Lockie C, Trimlett R, Ledot S, Cheshire N, Desai A, Singh S. Near-fatal Panton-Valentine leukocidin-positive Staphylococcus aureus pneumonia, shock and complicated extracorporeal membrane oxygenation cannulation: A case report. World J Crit Care Med 2021; 10(5): 301-309
- URL: https://www.wjgnet.com/2220-3141/full/v10/i5/301.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v10.i5.301
Panton-Valentine leukocidin (PVL), an exotoxin produced by specific strains of Staphylococcus aureus (S. aureus), is primarily responsible for skin and soft tissue infections[1,2]. Complications can include necrotising pneumonia and associated acute respiratory distress syndrome (ARDS). PVL-S. aureus (PVL-SA) pneumonia is characterised by pyrexia and haemoptysis and is often preceded by an influenza-like illness[3]. It has a poor prognosis and high mortality, even in young patients.
United Kingdom guidance states that individuals under the age of 16 years should be admitted to Paediatric Intensive Care Units (PICU); however, a proportion are admitted to Adult Intensive Care Units (AICU) instead[4]. We report a case of near-fatal necrotising PVL-SA pneumonia in an adolescent patient co-infected with influenza treated by adult emergency services requiring the utilisation of the multidisciplinary team. The extremely rapid deterioration in this patient highlights the urgency of diagnosis, treatment and utilisation of specialist services, including extracorporeal membrane oxygenation (ECMO) service to optimise patient outcomes.
A 15-year-old girl, on a school trip to London from the Middle East, presented to the emergency room (ER) with 3 h of progressive shortness of breath and chest pain, after a one-day history of cough, difficulty breathing with new sudden haemoptysis.
One week before admission, some of her school friends had developed Influenza-like symptoms. The patient reported coryzal symptoms for one week before her trip but still felt well enough to travel. There was no history of cutaneous injury or pathology.
The patient was previously healthy with no past medical history. There was no history of immunodeficiency in her or her family.
There was no history of immunodeficiency in her or her family. The patient had no known drug or food allergies.
On arrival in the ER, auscultation revealed crackles and reduced air entry in the lower right hemithorax. She coughed dark blood and became suddenly more distressed. She was tachypnoeic at 50 breaths/min and saturating at 92% on room air. Her heart rate was 120 beats/min, blood pressure 105/74 mmHg, and she was afebrile with cold extremities.
Initial arterial blood gas analysis showed a pH of 7.42 (normal range: 7.35-7.45), PaCO2 of 4.0 kPa (normal range: 4.7-6.0 kPa) and PaO2 of 7.2 kPa (normal range: 10.5-13.5 kPa) despite supplementary oxygen. Blood lactate was 1.7 mmol/L (normal range: < 1.0 mmol/L), sodium bicarbonate level 19.5 mmol/L (normal range: 22-29 mmol/L) and base excess -5 mmol/L (normal range: -2-2 mmol/L). Of note, the patients’ blood work up demonstrated a leukopaenia with neutrophil count 0.8 × 109/L (reference range 2.0-7.1 × 109/L). and c-reactive protein was 3.7 mg/L (reference range: < 10 mg/L). Blood film analysis was not performed. Full blood count and c-reactive protein results from initial laboratory investigation are presented in Table 1.
| Test | Unit | Value |
| White blood cell count | × 109/L | 1.5 |
| Haemoglobin | g/L | 146 |
| Haematocrit | L/L | 0.45 |
| Platelet count | × 109/L | 129 |
| Neutrophil count | × 109/L | 0.8 |
| Lymphocyte count | × 109/L | 0.6 |
| Monocyte count | × 109/L | 0 |
| Eosinophil count | × 109/L | 0 |
| Basophil count | × 109/L | 0 |
| C-reactive protein | mg/L | 0.6 |
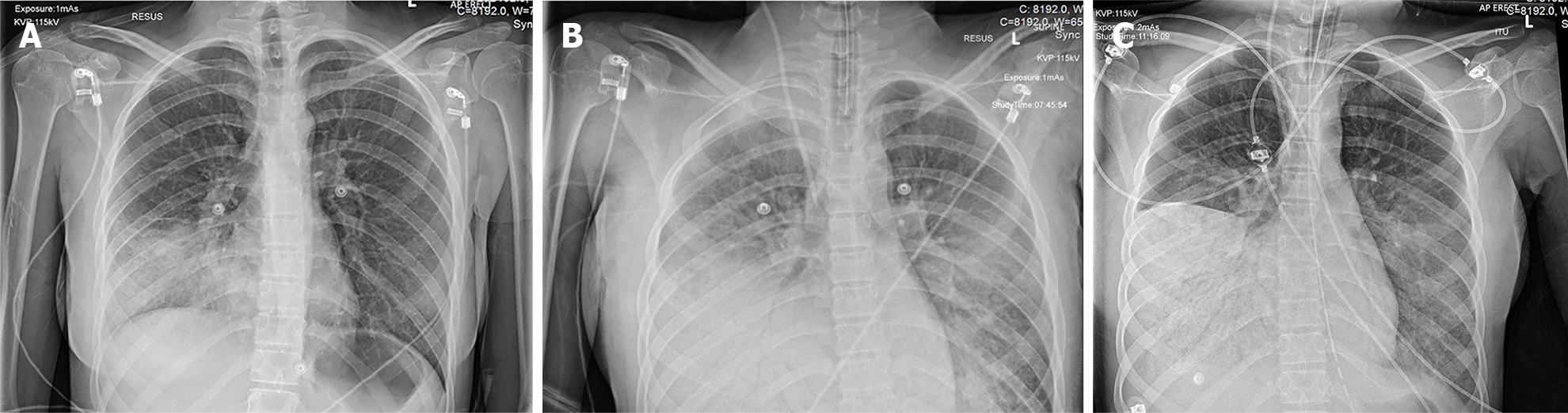
Plain erect radiographs of the chest taken during the initial period of the patient’s admission to hospital before transfer to the severe acute respiratory failure (SARF) centre are shown in Figure 1. These radiographs, and subsequent cross-section computed tomography of the chest performed at the SARF, showed rapidly progressive consolidation and four-quadrant opacification consistent with the other features of ARDS[5].
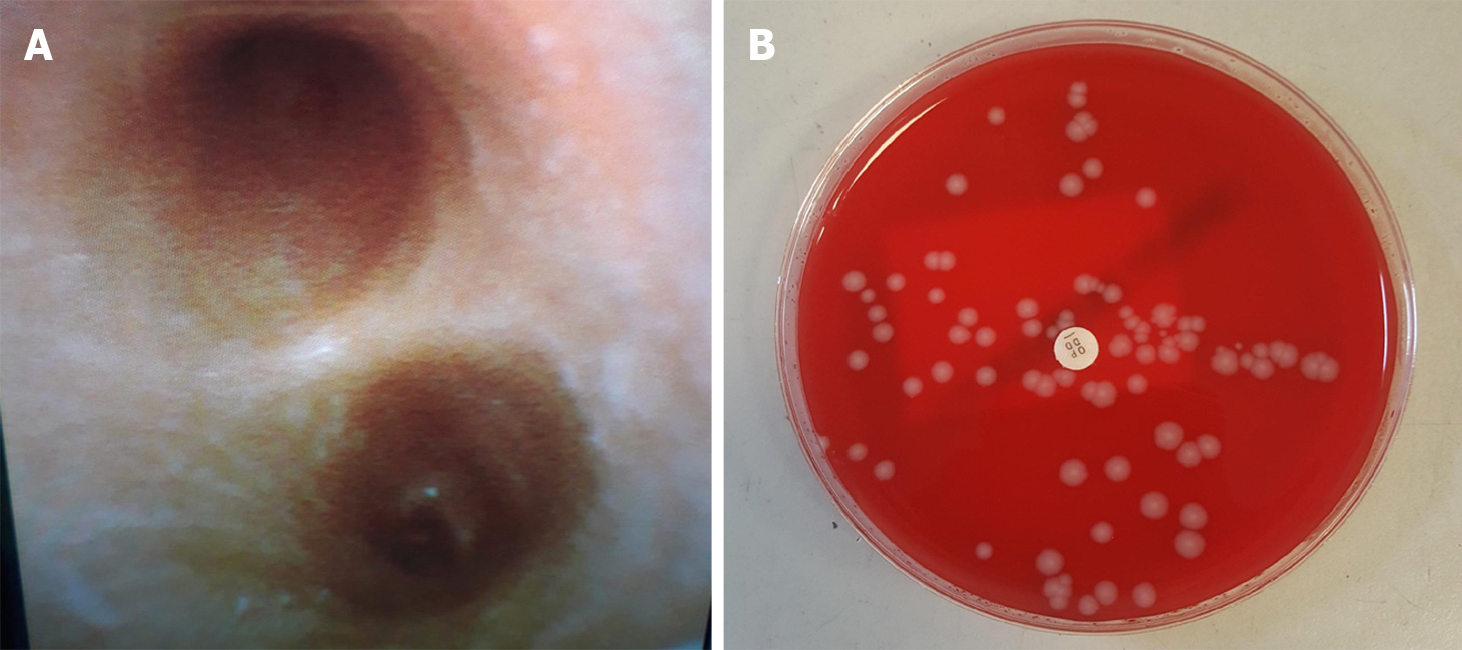
Diagnostic and therapeutic bronchoscopy was performed, demonstrating copious yellow, protein-rich plasma-like secretions with over 300mL fluid suctioned from her bronchial tree. A white speckled appearance of the bronchial mucosa was apparent (Figure 2A). Bronchial washings were taken.
The patient was otherwise healthy with no previous surgery or trauma; therefore, an infective respiratory pathogen was suspected early in the patient’s attendance. The presenting prodromal symptoms lead to consideration of a viral source, whilst the rapid deterioration with refractory septic shock raised the clinical suspicion of a coexistent bacterial pathogen. Other causes of vasoplegic shock were considered, including toxic shock syndrome but excluded based on history and clinical examination.
Given the travel history, suspicion of Middle East respiratory syndrome coronavirus (MERS-CoV) was high; therefore, investigations and therapy targeted this possibility, with personal protective equipment provided for staff. The concern regarding a potential emerging pathogen instigated quick microbiology/infectious disease (ID) advice and involvement of the Public Health England (PHE) laboratories.
Other commonly occurring community pathogens were considered, and the clinical narrative of contacts with family and fellow students with influenza-like symptoms placed this high on the differential list. The patient underwent rapid respiratory viral testing, which identified influenza A/H3N2. The same cultures grew S. aureus sensitive to methicillin (Figure 2B). The strain was confirmed as PVL-SA from PHE reference laboratory using whole-genome sequencing. All samples were negative for MERS-CoV.
Surviving sepsis protocols were implemented within 1 h following arrival to ER, and therapy escalated following deterioration[6]. This was initially intravenous (IV) ceftriaxone 2 g once daily, enteral azithromycin 500 mg once daily, and enteral oseltamivir 75 mg once daily. Antimicrobials were escalated to increase the bioavailability of gram-positive cover. IV linezolid 600 mg twice daily, IV clindamycin 900 mg four times a day, IV ceftriaxone 2 g once daily and IV clarithromycin 500 mg twice daily were all initiated approximately 6 h after presentation. This change followed advice from ID and microbiology teams to include toxin inhibition and improve lung penetration as per national guidelines. Emergency lifesaving therapy was required and initiated before any diagnostic test results were available, with escalation directed by the ID and microbiology team.
After a short period of non-invasive ventilation, rapid deterioration necessitated emergency intubation and mechanical ventilation, and the patient developed refractory shock whilst in the ER. Management of refractory shock required significant fluid resuscitation with high dose adrenaline (0.2 μg/kg/min) and noradrenaline (1.2 μg/kg/min) to maintain minimum survivable blood pressure. Bedside transthoracic echocardiography guided fluid resuscitation and demonstrated very hyperdynamic but notably underfilled ventricles. The patient remained cold peripherally with a peripheral capillary refill time of more than 5 s. Fluid resuscitation included serial 20% human albumin solution and crystalloids. Referral to the regional paediatric intensive care team was made, but retrieval was not feasible for several hours due to other clinical emergencies. The regional adult SARF referral centre was contacted for respiratory venovenous (VV)-ECMO support and retrieval.
After 6 h of resuscitation in ER, the patient was transferred to the AICU. The decision to admit was made based on the lack of PICU availability and the ongoing profound hypoxaemia, mixed metabolic and respiratory acidosis and noradrenaline/ adrenaline dependent shock. Her clinical condition deteriorated with progressively worsening haemodynamic instability and hypoxaemia. She was persistently desaturating to 60%-75% with maximal volume-controlled ventilation on 100% oxygen, with an increased heart rate to 160 beats/min. She received approximately 6000 mL of crystalloid and albumin fluid resuscitation on admission to AICU. Shock dose IV corticosteroid (100 mg hydrocortisone) was administered, and she received 2 g/kg IV immunoglobulin (IVIg)[6]. The patient remained profoundly hypoxaemic with ongoing poor air entry to the right lung. Bronchoalveolar fluid aspiration was performed, with transient improvements in oxygen saturations, before the recurrence of alveolar flooding and samples sent for rapid laboratory analysis.
Despite these measures, the patient continued to deteriorate with worsening haemodynamic instability and hypoxaemia. She was taken to theatre to instigate VV-ECMO. Full ECMO cannulation protocols were followed, and vascular pre-assessment was performed. Simultaneous cannulation of the right jugular and left and right femoral veins was complicated when the left femoral cannula became stuck, unable to be fully advanced or removed. Following vascular cut down by an assisting cardiothoracic surgeon to remove the cannula, the distal part of the cannula snapped, with proximal 10 cm remaining in the vessel as a foreign body. Haemorrhage was controlled manually then haemostasis was achieved by suture. The patient lost cardiac output. Cardiopulmonary resuscitation (CPR) was initiated, and immediate cannulation of the right femoral artery during extracorporeal CPR allowed venoarterial (VA)-ECMO to be established, resulting in the return of systemic output. During this time in theatre, aggressive fluid resuscitation continued, totalling 2000 mL crystalloid, six units of packed red cells, two pools cryoprecipitate, one bag of fresh frozen plasma and two pools of platelets, as guided by ultrasound assessment of central venous filling. From arrival in ER to her transfer to the ECMO centre, over approximately 16 h, the patient received an estimated 12.2 L of resuscitation fluids (230 mL/kg). The resuscitation process was adjusted from ER to ICU to maintain a life-sustaining blood pressure whilst reducing the very high levels of vasopressors and inotropes to minimise the risk of iatrogenic arrhythmias whilst maintaining acceptable cardiac output and oxygen delivery. Despite the measures described, adequate life-sustaining blood pressures were not adequately maintained, and high fluid volumes were required. At all times dynamic assessments of fluid responsiveness were used to guide resuscitation.
During her acute admission, there was involvement from the paediatric, anaesthetic, adult critical care, ECMO retrieval, cardiothoracic surgical, general surgical, ID, haematology and theatre teams. This, alongside the implementation of specialist shock pathways in a timely manner, was crucial to a subsequently positive outcome in a near-fatal case.
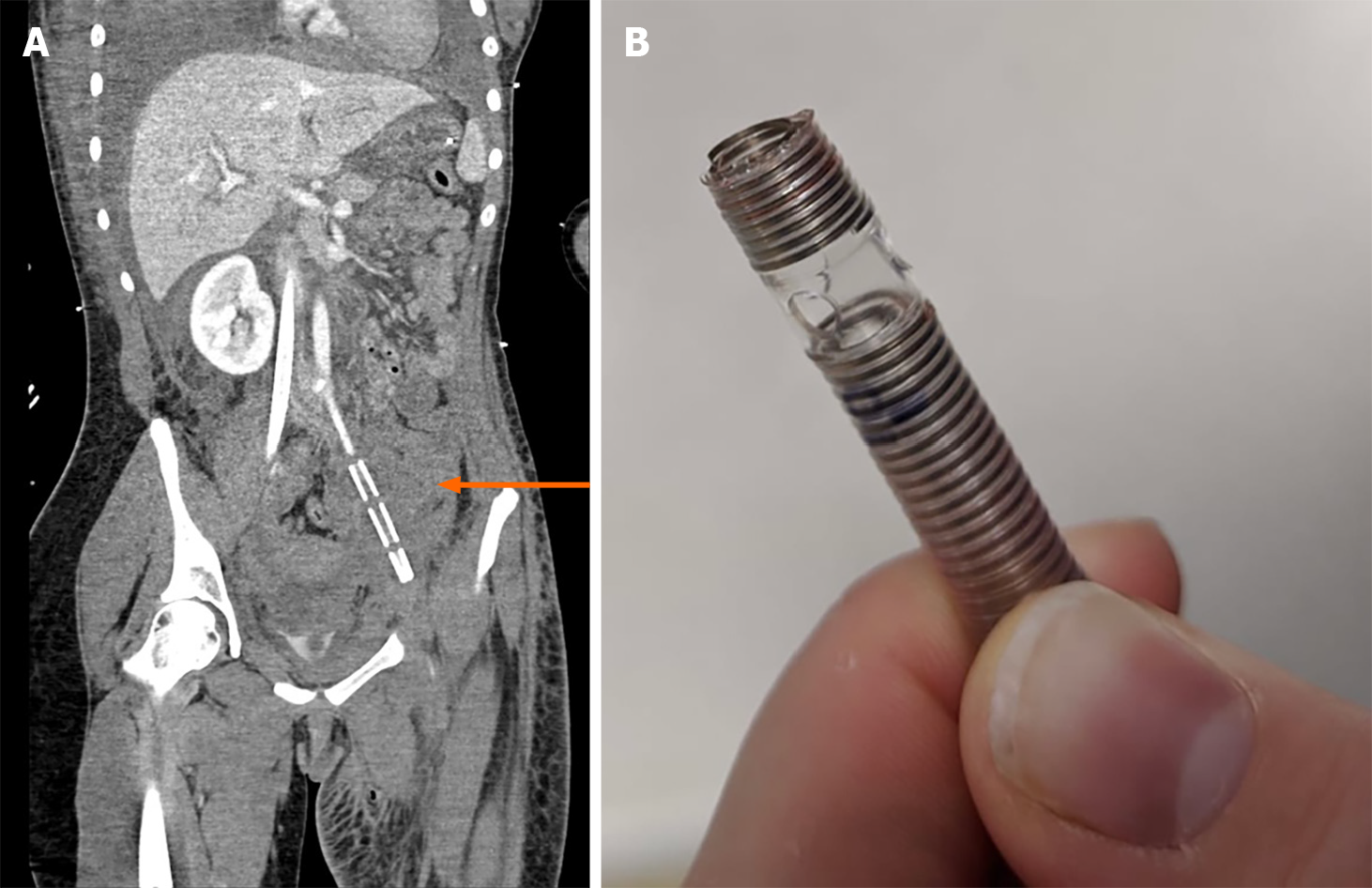
Following the establishment of VA-ECMO and transfer to the regional SARF centre, further contrast imaging demonstrated the retained cannula in the left common iliac artery (Figure 3). There was thread-like blood flow to the left leg. She developed ischaemia of the distal limbs worse on the left, with disseminated intravascular coagulation and multiple organ failure. Consequently, VA-ECMO was converted to VV-ECMO, removing the arterial cannula to improve the chances of perfusion and leg preservation. Collateral circulation developed with conservative management, and adequate lower limb perfusion occurred. After one month on VV-ECMO, a further three weeks on AICU and six weeks on PICU, the patient was successfully extubated with good neurological function. She made steady progress with respiratory and functional rehabilitation, although requiring a prolonged period of renal replacement therapy. Three months after her initial presentation, she was transferred to a non-tertiary United Kingdom hospital where she was recovering well with intensive physiotherapy assisted mobilisation. Mobility was improving, and a persistent left anterior lower leg skin wound eventually healed. The retained section of ECMO cannula remained in situ, causing no further clinical issue and is being followed up. At the time of this report, 18 months on, she is at home making good progress with ongoing community rehabilitation services. She is independently mobility following successful skin grafting to the left lower leg for non-healing wounds (Figure 4).
S. aureus secretes six cytolytic toxins, of which the best known is PVL. It is composed of two proteins and encoded by two genes lukS-PV and lukF-PV[7,8]. S. aureus produces pore-forming cytotoxins allowing bacteria to replicate inside host cells, preventing activation of the innate immune response. This can lead to cell lysis due to its ability to form pores in the cytoplasmic membrane. PVL is a strain of S. aureus that can cause severe skin and soft-tissues infections. Invasive infections can result in rare but rapidly fatal pneumonia in young and healthy individuals[9,10]. Initially presenting with fever, haemoptysis, and leukopaenia, PVL-SA pneumonia can quickly progress to ARDS with a high mortality rate[1,3].
Adolescents aged 12-19 years often have complex physical and psychological changes which require special consideration. Wood et al[11] reported that AICU staff suggest adolescents are to be transferred to PICU when they have more than one system failure. Our patient weighed 53 kg and was considered physiologically adult. There was a clear discussion and establishment of collaborative care throughout her stay on AICU. This extended to her and her family with ongoing psychological support and a family liaison team throughout.
Vasoplegic shock (distributive) can be encountered in different clinical scenarios, including septic shock, toxic shock syndrome, post-cardiac bypass surgery, burns and trauma[12]. Fluid bolus therapy (FBT) is the mainstay of treatment in paediatric sepsis; however, clinical research in this area is challenging[13]. In 2018, Gelbart[14] reported that FBT had been used in paediatric sepsis management for several years without overarching evidence to support its appropriate use. Increasing attention is now turning to evidence that suggests harm can come from excessive fluid therapy. Under-resuscitation can lead to multiple organ dysfunction. However, over-resuscitation may result in pulmonary or peripheral oedema[15]. The United Kingdom resuscitation council guidelines for paediatric advanced life support[16] states that restricted fluid therapy with isotonic crystalloid may be more favourable than the more profuse use of fluids in some forms of septic shock. Vigorous fluids and albumin were administered to our patient at the known cost of potential pulmonary oedema to replace lost circulating volume and stabilise life-threatening cardiovascular collapse.
Increasingly, doctors are open to early discussions of extracorporeal life support (ECLS)[17]. During the H1N1 influenza A pandemic in 2009, due to the acute effects of severe respiratory failure, a third of patients admitted to AICU required ECLS and led to the creation of the United Kingdom national SARF ECMO service[17]. Noah et al[18] summarised four case reports with patients on ECMO for respiratory failure secondary to PVL-SA. Of these cases, two of the patients were 15 and 17-years-old, and both were successfully discharged to their local hospitals following eight and nineteen days on ECMO, respectively. Haider et al[19] reported a case of a 12-year-old boy who developed PVL-SA pneumonia. The patient rapidly developed respiratory failure and died due to secondary cardiac arrest. The initiation of ECMO, despite complications, allowed time for other interventions to benefit our patient, particularly antimicrobial therapy. Without ECMO, judicious resuscitation and tertiary critical care, she would almost certainly have died. Further, the rapid intervention of an experienced cardiothoracic surgeon to prevent potential massive blood loss following ECMO complications highlights the importance of having the right people in the right place, at the right time.
Establishing the correct antimicrobial regimen is essential. The United Kingdom Health Protection Agency guideline[20] recommends using empirical broad-spectrum antimicrobials that suppress toxin production, such as clindamycin, linezolid and rifampicin. IV flucloxacillin is not recommended due to low necrotic tissue penetration, particularly as it may increase the PVL toxin production above the minimal inhibitory concentration. IVIg is recommended at a dose of 2 g/kg.
In summary, a healthy adolescent presented with worsening respiratory symptoms, rapidly progressing to profound fulminant shock and severe ARDS. This was secondary to Influenza A/H3N2 and PVL-SA pneumonia. In profound septic shock, consideration of both viral and bacterial infections should initiate rapid diagnostic tests where possible, in combination with appropriate and early anti-infective cover. Prompt intubation and ventilation, management of septic shock and ARDS with early fluid and vasopressor/inotropic resuscitation, IV antimicrobials, and IVIg plus extracorporeal support leading to high level tertiary critical care provided the framework for her remarkable survival, despite complicated ECMO cannulation and cardiac arrest. Early involvement of ID and microbiology specialists, along with PHE support, is required for optimal patient outcomes and staff protection when dealing with suspected high-risk infective pathogens. This case demonstrates the importance of collaboration between multidisciplinary teams and specialist centres with prompt ECMO referral in similar patients. It further highlights the importance of early ECMO initiation for suspected PVL-SA. It reveals the challenges of emergency management of critically unwell adolescents in centres without PICU availability. Patients like this require a multidisciplinary approach and the utilisation of various specialities to ensure positive outcomes. Finally, it is an example of what is achievable through coordinated and timely cooperation between acute care specialities, disciplines, medical services and agencies in emergency lifesaving situations.
We wish to acknowledge and thank the clinical and nursing staff for their exceptional teamwork in managing an extremely unwell patient under extraordinary circumstances. We acknowledge and thank the national severe acute respiratory failure extracorporeal membrane oxygenation service, adult and paediatric critical care, surgical, perfusion (Ms Rosie Smith), nursing, allied health professional and psychology teams at the Royal Brompton Hospital for their expertise and care. Special thanks to our patient and her parents for their permission to use her case to advance understanding of this condition for the benefit of others.
| 1. | Gillet Y, Issartel B, Vanhems P, Fournet JC, Lina G, Bes M, Vandenesch F, Piémont Y, Brousse N, Floret D, Etienne J. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet. 2002;359:753-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1100] [Cited by in RCA: 1031] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 2. | British Association of Dermatologists. Panton Valentine Leukocidin Staphylococcus Aureus (PVL-SA) Skin Infection. 2019. [cited 18 February 2021]. Available from: https://www.bad.org.uk/shared/get-file.ashx?id=179&itemtype=document. |
| 3. | Lavoue S, Le Gac G, Gacouin A, Revest M, Sohier L, Mouline J, Jouneau S, Flecher E, Tattevin P, Tadié JM. Extracorporeal circuit for Panton-Valentine leukocidin-producing Staphylococcus aureus necrotizing pneumonia. Med Mal Infect. 2016;46:314-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Wood D, Goodwin S, Pappachan J, Davis P, Parslow R, Harrison D, Ramnarayan P. Characteristics of adolescents requiring intensive care in the United Kingdom: A retrospective cohort study. J Intensive Care Soc. 2018;19:209-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526-2533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2524] [Cited by in RCA: 4399] [Article Influence: 314.2] [Reference Citation Analysis (0)] |
| 6. | Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, Kumar A, Sevransky JE, Sprung CL, Nunnally ME, Rochwerg B, Rubenfeld GD, Angus DC, Annane D, Beale RJ, Bellinghan GJ, Bernard GR, Chiche JD, Coopersmith C, De Backer DP, French CJ, Fujishima S, Gerlach H, Hidalgo JL, Hollenberg SM, Jones AE, Karnad DR, Kleinpell RM, Koh Y, Lisboa TC, Machado FR, Marini JJ, Marshall JC, Mazuski JE, McIntyre LA, McLean AS, Mehta S, Moreno RP, Myburgh J, Navalesi P, Nishida O, Osborn TM, Perner A, Plunkett CM, Ranieri M, Schorr CA, Seckel MA, Seymour CW, Shieh L, Shukri KA, Simpson SQ, Singer M, Thompson BT, Townsend SR, Van der Poll T, Vincent JL, Wiersinga WJ, Zimmerman JL, Dellinger RP. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017;43:304-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4894] [Cited by in RCA: 4169] [Article Influence: 463.2] [Reference Citation Analysis (6)] |
| 7. | Kaneko J, Kamio Y. Bacterial two-component and hetero-heptameric pore-forming cytolytic toxins: structures, pore-forming mechanism, and organization of the genes. Biosci Biotechnol Biochem. 2004;68:981-1003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 231] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 8. | Cunnington A, Brick T, Cooper M, Danin J, Hunt D, Jeanes A, Kearns AM, Nolan M, Lyall H. Severe invasive Panton-Valentine Leucocidin positive Staphylococcus aureus infections in children in London, UK. J Infect. 2009;59:28-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Shallcross LJ, Fragaszy E, Johnson AM, Hayward AC. The role of the Panton-Valentine leucocidin toxin in staphylococcal disease: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13:43-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 378] [Cited by in RCA: 329] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 10. | Monecke S, Slickers P, Ellington MJ, Kearns AM, Ehricht R. High diversity of Panton-Valentine leukocidin-positive, methicillin-susceptible isolates of Staphylococcus aureus and implications for the evolution of community-associated methicillin-resistant S. aureus. Clin Microbiol Infect. 2007;13:1157-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 88] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 11. | Wood D, Geoghegan S, Ramnarayan P, Davis PJ, Tume L, Pappachan JV, Goodwin S, Wray J. Where Should Critically Ill Adolescents Receive Care? J Intensive Care Med. 2020;35:1271-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 12. | Lambden S, Creagh-Brown BC, Hunt J, Summers C, Forni LG. Definitions and pathophysiology of vasoplegic shock. Crit Care. 2018;22:174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 202] [Cited by in RCA: 166] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 13. | Glassford NJ, Gelbart B, Bellomo R. Coming full circle: thirty years of paediatric fluid resuscitation. Anaesth Intensive Care. 2017;45:308-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 14. | Gelbart B. Fluid Bolus Therapy in Pediatric Sepsis: Current Knowledge and Future Direction. Front Pediatr. 2018;6:308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | Greene N, Bhananker S, Ramaiah R. Vascular access, fluid resuscitation, and blood transfusion in pediatric trauma. Int J Crit Illn Inj Sci. 2012;2:135-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 16. | Maconochie I, Bingham B, Skellett S. Guidelines: paediatric advanced life support. 2015. [cited 18 February 2021]. Available from: https://www.resus.org.uk/resuscitation-guidelines/paediatric-advanced-life-support/. |
| 17. | Gaffney AM, Wildhirt SM, Griffin MJ, Annich GM, Radomski MW. Extracorporeal life support. BMJ. 2010;341:c5317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Noah MA, Dawrant M, Faulkner GM, Hill AM, Harvey C, Hussain A, Jenkins DR, Nichani S, Peek GJ, Sosnowski AW, Firmin RK. Panton-Valentine leukocidin expressing Staphylococcus aureus pneumonia managed with extracorporeal membrane oxygenation: experience and outcome. Crit Care Med. 2010;38:2250-2253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Haider S, Wright D. Panton-Valentine leukocidin Staphylococcus causing fatal necrotising pneumonia in a young boy. BMJ Case Rep. 2013;2013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Health Protection Agency. Guidance on the diagnosis and management of PVL-associated Staphylococcus aureus infections (PVL-SA) in England. 2008. [cited 18 February 2021]. Available from: http://www.hpa.org.uk/webc/HPAwebFile/HPAweb_C/1218699411960. |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Unsolicited manuscript
Specialty type: Critical care medicine
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bizzoca D, Wan Y S-Editor: Wang LL L-Editor: A P-Editor: Li X