Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.115246
Revised: October 28, 2025
Accepted: January 6, 2026
Published online: March 9, 2026
Processing time: 145 Days and 12.3 Hours
Generalized anxiety disorder (GAD) is a prevalent psychiatric condition in children and adolescents, frequently linked to difficulties in daily functioning and academic performance. Increasing evidence suggests that immune dysregulation and neuroinflammatory pathways contribute to its pathophysiology, yet studies examining circulating cytokines and related biomarkers in pediatric populations are still scarce.
To investigate serum levels of C-reactive protein (CRP), tumor necrosis factor-like weak inducer of apoptosis, neopterin, interleukin-6, tumor necrosis factor-alpha, interferon-gamma, and zinc in drug-naïve children and adolescents with GAD, to gain insight into potential immune-related alterations.
The observational case-control design was conducted, including 25 drug-naïve patients with ‘pure’ GAD and 30 healthy controls aged 8-18 years. Psychiatric diagnoses were confirmed by the Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version, Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition and the Revised Child Anxiety and Depression Scale. Venous blood samples were analyzed using enzyme-linked immunosorbent assay, nephelometry, and electro-chemiluminescence assays. Group comparisons were performed using the Mann-Whitney U test and the χ2 test, and correlations were assessed using Spearman's correlation.
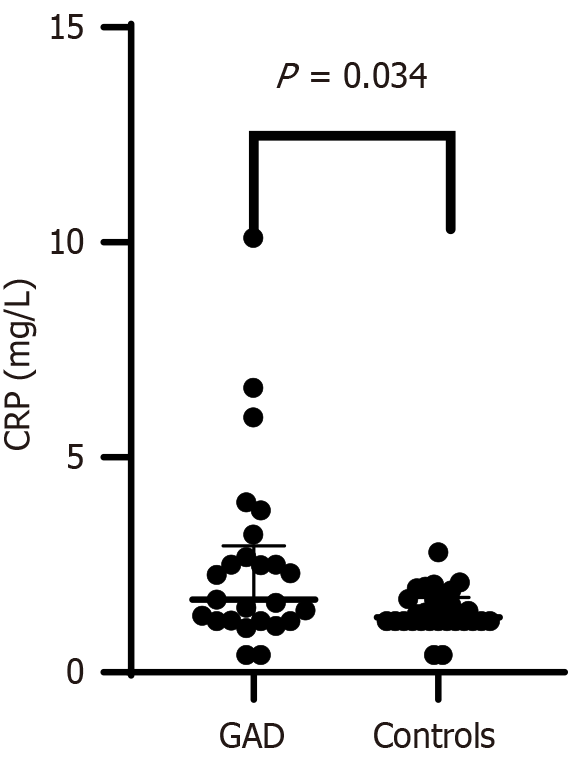
CRP levels were significantly higher in the GAD group than controls (median 1.7 mg/L vs 1.3 mg/L, P = 0.034, r = 0.32). No significant group differences were observed for tumor necrosis factor-alpha tumor necrosis factor-like weak inducer of apoptosis, interleukin-6, interferon-gamma, zinc, or neopterin. Time since symptom onset was positively correlated with total anxiety severity (r = 0.729, P = 0.001). Receiver operating characteristic analysis showed modest diagnostic value for CRP (area under the curve = 0.665, 95% confidence interval: 0.511-0.820).
This study demonstrates a moderate elevation in CRP levels in drug-naïve patients with GAD. Although no change in other inflammatory markers was observed in our study, this may suggest a low-grade inflammatory response. The early detection of symptoms is still critical, and more longitudinal studies with larger samples are necessary further to elucidate the role of inflammation in childhood anxiety pathophysiology.
Core Tip: This study investigated the potential relationship between generalized anxiety disorder (GAD) and systemic inflammation in drug-naïve children and adolescents. Significantly higher C-reactive protein levels compared to healthy controls suggest a low-grade inflammatory process may be present in pediatric GAD. In contrast, no significant group differences were found in other circulatory immune markers. This selective pattern of elevation suggests that specific components of the inflammatory response may be activated in a limited manner in GAD. Furthermore, the positive correlation between clinical symptom severity and illness duration supports the potential impact of early diagnosis and intervention on symptom progression. However, larger sample sizes and longitudinal studies are needed to define causal relationships and clinical outcomes more robustly.
- Citation: Ünal K, Taş Torun Y, Erol ME, Kurt ZK, Özbaş C. Could high serum C-reactive protein levels serve as a biomarker for children and adolescents diagnosed with generalized anxiety disorder? World J Clin Pediatr 2026; 15(1): 115246
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/115246.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.115246
Generalized anxiety disorder (GAD) is an anxiety disorder characterized by persistent and uncontrollable worry lasting at least six months. GAD is among the most common anxiety disorders in children and adolescents. The reported prevalence of GAD ranges from 2.2% to 3.6%[1,2]. GAD symptoms manifest in both psychological and somatic ways. While GAD patients frequently report psychological symptoms such as restlessness, easy fatigability, difficulty concentrating, irritability, and sleep disturbances, patients also frequently report somatic symptoms such as palpitations, sweating, and tremors[3]. In the current literature, GAD is found to coexist with other anxiety disorders, depression, phobias, attention deficit hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), substance abuse, oppositional defiant disorder, learning disabilities, and language disorders[4].
The etiology of GAD is not fully understood, but it is believed to result from a complex interplay between genetic and environmental factors[5]. Current evidences suggest that neuroinflammation contributes to the pathogenesis of many psychiatric disorders, and neuroimmune dysfunction in the central nervous system may underlie these conditions. In this context, pro-inflammatory cytokines such as interleukin (IL)-1β, IL-2, IL-6, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ have been implicated in psychiatric disorders[6-8].
Moreover, members of the TNF superfamily have emerged as central modulators of central nervous system pathology[9]. Tumor necrosis factor-like weak inducer of apoptosis (TWEAK) is a cytokine belonging to the TNF superfamily and initiates signal transduction by binding to the fibroblast growth factor-inducible 14 receptors on the cell surface. It activates multiple signaling cascades, leading to the induction of pro-inflammatory cytokines and amplification of neuroinflammatory responses[10,11]. Dysregulation of TWEAK signaling has been observed in psychiatric disorders such as bipolar disorder, schizophrenia, and attention-deficit/hyperactivity disorder, suggesting a potential role in neuropsychiatric pathophysiology[10-13].
Closely related to this immune activation, a macrophage-derived metabolite, neopterin, is a marker of cellular immune response. Elevated neopterin levels have been reported in psychiatric disorders, underscoring the relevance of immune-mediated mechanisms in psychiatric conditions[14,15].
Immune dysregulation involving TWEAK signaling and neopterin release may contribute to the pathogenesis of GAD. To our knowledge, no prior study has simultaneously evaluated C-reactive protein (CRP), TWEAK, and neopterin along with canonical cytokines in a drug-naïve pediatric GAD sample; this study addresses that gap. This study aims to address this gap by evaluating the serum levels of CRP, TWEAK, neopterin, IL-6, TNF-α, IFN-γ, and zinc in drug-naïve youth with “pure” GAD. We hypothesize that alterations in these markers may provide novel insights into immune mechanisms underlying GAD and inform its clinical course.
The current study was conducted in accordance with the ethical principles of the Declaration of Helsinki. This re
This study was conducted between January 1, 2023 and January 1, 2025, at the Gazi University (Faculty of Medicine). Participants were selected from the Gazi University (Faculty of Medicine) hospital’s child and adolescent psychiatry outpatient clinic, and samples were analyzed at the Department of Medical Biochemistry of the Gazi University Hospital. The researcher conducted a semi-structured psychiatric interview for the patient group to ensure compliance with the inclusion and exclusion criteria.
Inclusion criteria for the patient group were as follows: Children and adolescents aged between 8 and 18 who were diagnosed with GAD based on the Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version, Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) and Revised Child Anxiety and Depression Scales (RCADS).
Exclusion criteria for all participants were: (1) A diagnosis of neurological disorders (e.g., cerebral palsy, epilepsy), metabolic disorders (e.g., phenylketonuria), diabetes mellitus, cancer, allergic, rheumatologic, inflammatory, or autoimmune diseases; (2) History of an infectious disease within the last 4 weeks; (3) Use of anti-inflammatory medications within the previous 4 weeks; (4) Immunosuppressive treatment in the past 6 months; (5) History of substance use in the last 3 months; (6) A clinical suspicion of pediatric autoimmune neuropsychiatric disorders associated with streptococci; (7) Children meeting obesity criteria [body mass index (BMI) ≥ 95th percentile]; and (8) Any primary psychiatric diagnosis other than GAD. Only ‘pure’ GAD patients meeting DSM-5 criteria without comorbid psychiatric disorders were included. These exclusions were prospectively applied to reduce confounding by metabolic or medication effects.
In this study, a child and adolescent psychiatrist performed all clinical assessments and scale scoring. A single researcher conducted a semi-structured psychiatric interview with the patient group to assess eligibility according to the inclusion and exclusion criteria. The researchers constructed a socio-demographic questionnaire to gather data on the socio-demographic status of the participants, such as gender, education, number of brothers and sisters, family structure, age, parents’ education, and economic condition.
Clinical assessment was carried out with standardized diagnostic tools. The Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version, DSM-5, is a semi-structured tool designed to assess current and lifetime mental illnesses in children and adolescents aged 6-18 years and was updated in 2016 to align with the DSM-5 diagnostic criteria. Validity and reliability of the Turkish version were established in 2019 by Ünal et al[16], and the same was used in the current study to diagnose GAD.
In addition, the RCADS is a 47-item scale that can be utilized for screening and assessment of anxiety and depressive disorders in children and adolescents. It has parent and self-report forms. The RCADS provides a total anxiety score, as well as subscale scores for specific subscales, including separation anxiety, generalized anxiety, panic disorder, social phobia, and major depressive disorder. Validity and reliability the Turkish versions were established by Gormez et al[17] in 2017. RCADS was used in the present study to assess symptom severity and quantify participants’ clinical profiles.
After an overnight fast, venous blood samples were collected from untreated GAD patients and controls, and the samples were centrifuged at 4000 rpm for 15 minutes to separate the serum for biochemical analysis. Samples showing signs of hemolysis or icterus were carefully excluded to maintain data integrity. Serum samples were stored at -80 °C until the day of analysis.
Serum TWEAK (Catalog No. E1820Hu), neopterin (Catalog No. E3155Hu), and IFN-γ (Catalog No. E0105Hu) levels were measured using commercial enzyme-linked immunosorbent assay (ELISA) kits (Bioassay Technology Laboratory, Zhejiang, China) according to the manufacturer’s instructions. TNF-α concentrations were determined using diagnostic-grade ELISA kits (DIAsource ImmunoAssays S.A., Louvain-la-Neuve, Belgium). Serum IL-6 levels were measured by electro-chemiluminescence immunoassay using an Elecsys analyzer (Roche Diagnostics, Rotkreuz, Switzerland). CRP concentrations were determined by nephelometry using an IMAGE 800 analyzer (Beckman Coulter Inc., IN, United States).
The lower limits of quantification for serum neopterin, TWEAK, IFN-γ, TNF-α, IL-6, and CRP were 0.061 nmol/L, 5.51 mg/L, 0.49 ng/mL, 0.7 pg/mL, 1.5 pg/mL, and 0.8 mg/L, respectively. The corresponding measurement ranges were 0.1-38 nmol/L for neopterin, 10-4000 mg/L for TWEAK, 1-400 ng/mL for IFN-γ, and 7-518 pg/mL for TNF-α. The intra- and inter-assay coefficients of variation for all assays were < 8% and < 10%, respectively, except for the TNF-α assay, which demonstrated intra-assay coefficients of variation < 7% and inter-assay coefficients of variation < 5%.
Diagnostic reference ranges were provided by the assay manufacturers as follows: TNF-α, 4.6-12.4 pg/mL; IL-6, < 7 pg/mL; and CRP, 0-5 mg/L. CRP and zinc were analyzed using clinically validated diagnostic platforms, whereas TNF-α was measured with a research-grade but analytically validated ELISA (DIAsource ImmunoAssays S.A., Louvain-la-Neuve, Belgium). These methods ensured high analytical precision (coefficients of variation < 5% for diagnostic assays and < 7% intra-assay, < 5% inter-assay for the TNF-α ELISA), supporting the overall reliability of the biochemical measurements.
In this study, statistical analyses were performed with the use of IBM SPSS Statistics version 23.0 (IBM Corp., Chicago, IL, United States) to ensure methodological rigor and accuracy. Descriptive statistics were initially used to summarize characteristics of the study sample. Categorical variables were presented as n (%). For numerical data, central tendency and dispersion were reported as a mean ± SD in cases of normally distributed variables, or as median with interquartile range (IQR) for skewed distributions. The Kolmogorov-Smirnov test was used to formally assess the normality of numerical variables, guiding the selection of subsequent statistical tests. Associations between categorical variables were analyzed using the χ2 test. Correlation analyses were conducted to investigate the relationships between clinical scores. The clinical instruments used included the RCADS Clinical Interview Version (RCADS-CV), specifically the GAD subscale and the Total Anxiety score, along with the RCADS Parent Version counterparts. Pearson’s or Spearman’s correlation coefficients were calculated depending on the distribution of the data. The strength of the correlation, represented by ‘r’, was interpreted based on the following standardized scale: 0.0-0.39 for a weak or no relationship, 0.40-0.69 for a moderate relationship, 0.70-0.89 for a strong relationship, and 0.90-0.99 for a powerful relationship.
Further, the diagnostic value of CRP for the identification of GAD was assessed using Receiver operating characteristic (ROC) curve analysis. The area under the ROC curve was calculated to reflect the overall discriminatory power of CRP, and optimal threshold values were determined for clinical use. For all statistical tests, the level of statistical significance was set to less than 0.05 (P < 0.05), and all estimates reported were at a 95% confidence interval (CI).
For sample size, which examined serum CRP levels in individuals with GAD, was considered[18]. Based on the effect size value suggested by Kohen (effect size f = 0.73), an alpha error (P-value) of 0.05, and a 1-beta error (power) of 0.80, a total of 50 participants were determined to be sufficient to test the null hypothesis. Considering non-response and missing data, an additional 10% was added, resulting in a total of 55 participants. G*Power version 3.1.9.7 (Universität Düsseldorf, Germany) was used for analyses[19].
The socio-demographic and clinical characteristics of the 25 drug-naïve GAD patients and 30 healthy controls are detailed in Table 1. The two groups were comparable across several variables, including age [median (IQR) GAD: 160 (135-185) months; controls: 154 (128-177) months], gender, BMI percentile, smoking status, coronavirus disease 2019 history, and family structure (P > 0.05). A notable difference was observed in family psychiatric history. The GAD group had a significantly higher prevalence of psychiatric illness within the family compared to the control group (P = 0.019). The median time since symptom onset (i.e., duration of anxiety symptoms as reported by the child or caregiver prior to diagnosis) for GAD patients was 11 (6-25) months. As expected, RCADS-CV scores for all subscales were significantly higher in the GAD group compared to the control group. The comprehensive socio-demographic, clinical, and RCADS scores for all participants are presented in Table 1.
| Parameters | GAD group (n = 25) | Control group (n = 30) | P value |
| Gender | 0.128 | ||
| Female | 18 (72) | 15 (50) | |
| Male | 7 (28) | 15 (50) | |
| Family history of chronic illnesses | 13 (52.0) | 21 (65.6) | 0.298 |
| Family history of psychiatric disorders | 14 (56) | 8 (26.7) | 0.019 |
| Smoking (yes) | 1 (3.8) | 0 (0) | 0.464 |
| COVID-19 history (yes) | 12 (46.2) | 12 (40.0) | 0.64 |
| Parents together (yes) | 23 (88.5) | 26 (86.7) | 0.839 |
| Age (years) | 160 (135-185) | 154 (128-177) | 0.271 |
| BMI percentile | 45 (22-80) | 61 (19.1-82) | 0.622 |
| Time since symptom onset (months) | 11 (6-25) | ||
| RCADS-CV scores | |||
| SAD | 60 (53-70) | 44.5 (41-51) | < 0.001 |
| GAD | 59 (54-68) | 42 (39-47) | < 0.001 |
| PD | 68 (52-80) | 43.5 (39-52) | < 0.001 |
| SP | 54 (42-72) | 39 (33-44) | < 0.001 |
| OCD | 60 (52-69) | 46.5 (38-51.3) | < 0.001 |
| MDD | 61 (44-75) | 42.5 (38-52) | < 0.001 |
| Total | 65 (51-75) | 41 (37-47) | < 0.001 |
Biochemical analyses were conducted on 25 patients with GAD and 30 healthy controls. The study investigated serum levels of several biomarkers, including CRP, TNF-α, TWEAK, IL-6, IFN-γ, zinc, and neopterin. All biochemical measures were reported as median (IQR).
A significant finding emerged in the analysis of serum CRP levels. The median CRP level in the GAD group was 1.7 (1.2-2.94) mg/L, which was significantly higher than the median level of 1.3 (1.2-1.7) mg/L observed in the control group (P = 0.034). This result is visually represented in Figure 1.
In contrast to CRP, no statistically significant differences were found between the GAD and control groups for the other measured biomarkers. The median values for TNF-α, TWEAK, IL-6, IFN-γ, zinc, and neopterin were comparable between the two groups (P > 0.05; Table 2).
| GAD group (n = 25) | Control group (n = 30) | P value1 | |
| CRP (mg/L) | 1.7 (1.2-2.94) | 1.3 (1.2-1.7) | 0.034 |
| TNF-α (pg/mL) | 7.9 (6.4-11.0) | 9.3 (7.7-11.0) | 0.466 |
| TWEAK (mg/L) | 1111 (893-1889) | 1236 (1009-1380) | 0.980 |
| IL-6 (pg/mL) | 2.8 (0.75-3.99) | 2.8 (0.8-4.0) | 0.615 |
| IFN-γ (ng/mL) | 59.4 (38.4-95.4) | 61.6 (54-84) | 0.628 |
| Zinc (μg/dL) | 94 (82-104) | 100 (93-112) | 0.073 |
| Neopterin (nmol/L) | 3.66 (1.84-4.74) | 2.9 (2.2-3.9) | 0.674 |
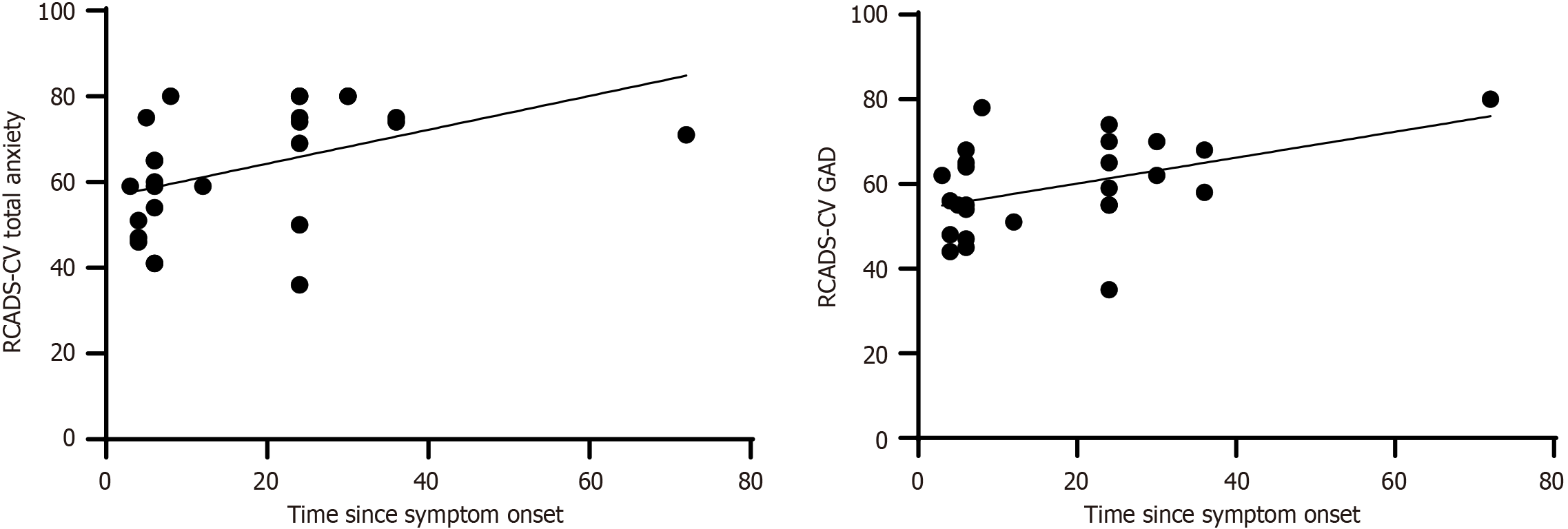
Correlations between CRP, TNF-α, TWEAK, IL-6, IFN-γ, zinc, and neopterin levels and time since symptom onset, RCADS-CV GAD, and RCADS-CV total anxiety scores were examined in the patient group. A positive correlation was found between time since symptom onset and the RCADS-CV total anxiety score (r = 0.729, P = 0.001), and a positive correlation was also found between the RCADS-CV GAD score (r = 0.642, P < 0.001).
The correlations between RCADS-CV GAD, RCADS-CV total anxiety, and time since symptom onset in the GAD group are presented in Figure 2. No significant correlations were found between other biochemical measurements and clinical parameters.
ROC curve analysis was done to assess the ability of serum CRP levels to distinguish between the GAD and control groups. The area under the curve (AUC) was 0.665 (95%CI: 0.511-0.820, P = 0.036), showing a statistically significant but modest difference. At the best threshold value found using the Youden index, CRP had a sensitivity of 64% and a specificity of 66% for telling GAD patients apart from healthy controls.
In this study, the socio-demographic, clinical, and biochemical characterization of drug-naïve GAD children and adolescents was researched. Consistent with previous evidence suggesting a link between anxiety disorders and immune dysregulation, we observed elevated serum CRP levels among the patient group. In contrast, other inflammatory and metabolic measures did not significantly differ from controls.
Our finding of elevated CRP in child GAD is consistent with prior work, but narrows the susceptibility to confounding factors. Copeland et al[20] found that, while the association between GAD and CRP remained significant in unadjusted models, it was partially mediated following adjustment for covariates such as BMI and medication use. By prospectively excluding individuals with obesity (BMI ≥ 95th percentile) and patients on anti-inflammatory or psychotropic medications, our study reduces the influence of these confounders. These methodologic niceties strengthen the conclusion that the CRP elevation observed here is intrinsically linked to GAD rather than a secondary metabolic or pharmacologic effect.
This result aligns with an emerging body of research implicating low-grade systemic inflammation in anxiety disorders. A recent meta-analysis confirmed that CRP concentrations are modestly but significantly elevated in GAD compared to controls, consistent with findings across related psychiatric conditions such as major depressive disorder, where the inflammatory signal is even more robust[21]. Significantly, the median CRP concentration in our GAD cohort (1.7 mg/L) falls within the range considered indicative of moderate cardiovascular risk and low-grade systemic inflammation (1-3 mg/L)[22]. These findings collectively support the hypothesis that altered immune function may contribute to the pathophysiology of GAD in youth.
Another important finding of this study is the absence of significant group differences in other inflammatory and biochemical markers, including TNF-α, TWEAK, IL-6, IFN-γ, zinc, and neopterin. Rather than representing a trivial null result, this pattern may provide insight into the specific biological mechanisms of GAD.
The broader psychiatric literature highlights striking heterogeneity in these markers: For example, serum TWEAK levels have been reported as significantly reduced in ADHD and in subsets of bipolar disorder during both manic episodes and remission[10,23]. At the same time, other studies demonstrated elevated TWEAK and soluble tumor necrosis factor receptor-1 levels in bipolar patients[12]. In schizophrenia, no significant difference in overall TWEAK levels was observed, but male patients had significantly lower plasma TWEAK compared to controls[9]. These divergent findings underscore that TWEAK does not behave uniformly across psychiatric conditions, but may instead reflect disorder- and context-specific immune signaling.
A similar pattern of heterogeneity can also be observed for neopterin. In our study, there was no difference in the neopterin levels between GAD patients and controls. However, this biomarker has been shown to be increased in various other psychiatric disorders, such as autism spectrum disorder[24-26], ADHD[27], and major depressive disorder[28], as well as in pre-treatment schizophrenia patients[29]. By contrast, in post-traumatic stress disorder, both pre-dexa
Cytokine findings also provide additional information. Although IL-6 and TNF-α were not significantly elevated in our GAD group in our study, other studies have observed proinflammatory cytokine activation in psychiatric populations. For example, IL-6 has been reported to be significantly higher in ADHD[32], and schizophrenia[33]. On the other hand, in euthymic bipolar disorder patients, no significant differences in IL-6 levels were observed highlighting the situation-dependent nature of cytokine changes. Additionally, bipolar manic patients showed increased TNF-α and altered pro/anti-inflammatory cytokine ratios (IL-6/IL-4, TNF-α/IL-4, IL-2/IL-4, IFN-γ/IL-4) compared to controls[34], which may support the concept of immune imbalance rather than uniform cytokine elevation. Therefore, our null findings for IL-6 and TNF-α in GAD may indicate subtler changes in anxiety compared to mood and psychotic disorders, suggesting that immune dysregulation in GAD may involve a less robust or non-uniform inflammatory response compared to other major psychiatric conditions.
In addition to these basic pro-inflammatory cytokines, there is increasing evidence that the failure to find significant group differences in markers such as TNF-α, IL-6, and IFN-γ does not rule out immune pathology in psychiatric disease. Instead, it could be a reflection of the heterogeneity in immune activation in anxiety and mood spectrum disorders. Experimental studies have established that cytokine responses to stimulation, particularly IL-6 can be associated with psychological and somatic symptoms, indicating that immune dysregulation may manifest differently across subgroups[35]. Moreover, research on metabolic-immune interactions has shown that cytokines such as IL-6 act as bidirectional mediators linking inflammation and metabolism. At the same time, TNF-α and IL-6 play central roles in systemic inflammatory processes, including cachexia[36]. Collectively, these findings imply that the lack of statistically significant group differences in TNF-α, IL-6, TWEAK, IFN-γ, or neopterin should not be interpreted as an absence of immune involvement but rather as a reflection of disorder-specific immune regulation or limited statistical power. Supporting evidence from other pediatric psychiatric studies also points to diagnosis-specific immune patterns. Other pediatric psychiatric studies also validate diagnosis-specific immune patterns. For example, current investigations among drug-naïve children and adolescents with ADHD revealed that TWEAK and CRP levels were dissimilar from controls. However, cytokines such as TNF-α, IL-6, IFN-γ, zinc, and neopterin were not significantly different[37]. These findings, in combination with the present results, suggest immune dysregulation in pediatric psychiatric disorders may vary by disorder and symptom dimension rather than following a uniform inflammatory process.
The positive correlation between time since symptom onset and symptom severity (RCADS-CV total anxiety: r = 0.729, P = 0.001; RCADS-CV GAD: r = 0.642, P < 0.001) is consistent with the chronic and insidious nature of GAD in children and adolescents. Symptoms typically emerge over time and, if not treated, may persist into adulthood and form the basis for additional psychiatric comorbidities. Such a correlation would suggest that chronic psychological stress exposure could have a role in symptom chronicity and possibly even in mild systemic inflammatory activation. But as the study was cross-sectional, causality is not inferred, and unmeasured factors such as chronic exposure to stress, subclinical comorbid illness, or environmental exposures may have been behind this association. These findings cumulatively underscore the importance of early recognition and prompt treatment to minimize symptom burden and improve long-term functioning.
In the ROC analysis, serum CRP had an AUC of 0.665 (95%CI: 0.511-0.820), indicating poor discriminative capability according to published interpretive criteria[38,39]. Statistically significant, but indicative of low diagnostic accuracy, the value is consistent with the modest sensitivity (64%) and specificity (66%) identified at the best cut-off point. In
Despite these strengths, there are some limitations to note. In ROC analysis, serum CRP was 0.665 (95%CI: 0.511-0.820), indicating poor to low-moderate discriminative power and therefore should be considered to confer only modest diagnostic utility. However, CRP was measured using a clinically standardized nephelometric assay which is a diagnostic test. Another notable limitation is the absence of clinically established reference ranges for certain markers such as TWEAK and neopterin, which complicates the clinical interpretation of the results. Therefore, our findings highlight that these biomarkers should currently be considered research tools, and their potential roles as biological indicators in GAD require confirmation in larger, independent cohorts. Although lifestyle-related factors such as diet, sleep quality, and physical activity were not formally quantified, all participants were recruited under standardized morning fasting conditions, and individuals with obesity or recent infections were excluded, likely reducing major metabolic variability. Furthermore, while the GAD group had a higher rate of family psychiatric history, this variable did not show any significant association with biomarker levels within the group. Nevertheless, future studies should incorporate structured assessments of lifestyle factors and familial psychiatric burden to elucidate their influence on inflammatory status better.
Future research should therefore prioritize larger longitudinal designs to clarify the temporal relationship between inflammation and GAD. While the present study observed elevated CRP without neopterin elevation, this profile could reflect disorder-specific heterogeneity in immune signaling. Expansion of the biomarker panel through high-throughput metabolomics and proteomics, and integration of such findings with neuroimaging data from circuits implicated in GAD (e.g., amygdala-prefrontal networks), would yield a more integrated view of the disorder’s neuroimmune signature.
This study provides preliminary evidence linking elevated CRP, a marker of low-grade systemic inflammation, to GAD in children and adolescents. While this finding supports a growing body of literature, the limited diagnostic performance of CRP highlights the critical distinction between its biological relevance and clinical utility as a standalone biomarker.
We sincerely thank all children and parents for their participation in this study. The authors gratefully acknowledge the Academic Writing Practice and Research Center of Gazi University for providing professional academic language editing services.
| 1. | Mohammadi MR, Pourdehghan P, Mostafavi SA, Hooshyari Z, Ahmadi N, Khaleghi A. Generalized anxiety disorder: Prevalence, predictors, and comorbidity in children and adolescents. J Anxiety Disord. 2020;73:102234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 2. | Stein MB, Sareen J. Clinical practice. Generalized Anxiety Disorder. N Engl J Med. 2015;373:2059-2068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 187] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 3. | Terlizzi EP, Villarroel MA. Symptoms of Generalized Anxiety Disorder Among Adults: United States, 2019. NCHS Data Brief. 2020;1-8. [PubMed] |
| 4. | Mutluer T, Gorker I, Akdemir D, Ozdemir DF, Ozel OO, Vural P, Tufan AE, Karacetin G, Arman AR, Fis NP, Demirci E, Ozmen S, Hesapcioglu ST, Oztop D, Tural U, Aktepe E, Aksu H, Ardic UA, Basgul S, Bilac O, Coskun M, Celik GG, Demirkaya SK, Dursun OB, Durukan I, Fidan T, Gokcen C, Gormez V, Gundogdu OY, Herguner S, Kandemir H, Kilic BG, Kilincaslan A, Nasiroglu S, Sapmaz SY, Sahin N, Tahiroglu AY, Toros F, Unal F, Yazici IP, Yazici KU, Isik U, Ercan ES. Prevalence, comorbidities and mediators of childhood anxiety disorders in urban Turkey: a national representative epidemiological study. Soc Psychiatry Psychiatr Epidemiol. 2023;58:919-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 5. | Keeton CP, Kolos AC, Walkup JT. Pediatric generalized anxiety disorder: epidemiology, diagnosis, and management. Paediatr Drugs. 2009;11:171-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Gibney SM, Drexhage HA. Evidence for a dysregulated immune system in the etiology of psychiatric disorders. J Neuroimmune Pharmacol. 2013;8:900-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 176] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 7. | Najjar S, Pearlman DM, Alper K, Najjar A, Devinsky O. Neuroinflammation and psychiatric illness. J Neuroinflammation. 2013;10:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 357] [Cited by in RCA: 534] [Article Influence: 41.1] [Reference Citation Analysis (0)] |
| 8. | Kim YK, Na KS, Myint AM, Leonard BE. The role of pro-inflammatory cytokines in neuroinflammation, neurogenesis and the neuroendocrine system in major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2016;64:277-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 349] [Cited by in RCA: 474] [Article Influence: 47.4] [Reference Citation Analysis (0)] |
| 9. | Tatlıdil Yaylacı E, Yüksel RN, Ünal K, Altunsoy N, Cingi M, Yalçın Şahiner Ş, Aydemir MÇ, Göka E. TNF-related weak inducer of apoptosis (TWEAK) levels in schizophrenia. Psychiatry Res. 2015;229:755-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Işık Ü, Kılıç F, Demirdaş A, Aktepe E, Aydoğan Avşar P. Serum Tumor Necrosis Factor-Like Weak Inducer of Apoptosis (TWEAK) Levels Are Decreased in Children with Attention-Deficit/Hyperactivity Disorder. Psychiatr Danub. 2022;34:51-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 11. | Winkles JA. The TWEAK-Fn14 cytokine-receptor axis: discovery, biology and therapeutic targeting. Nat Rev Drug Discov. 2008;7:411-425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 478] [Cited by in RCA: 475] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 12. | Barbosa IG, Vaz GN, Rocha NP, Machado-Vieira R, Ventura MRD, Huguet RB, Bauer ME, Berk M, Teixeira AL. Plasma Levels of Tumor Necrosis Factor Superfamily Molecules Are Increased in Bipolar Disorder. Clin Psychopharmacol Neurosci. 2017;15:269-275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Cingi Yirün M, Yirün O, Ünal K, Yüksel RN, Altunsoy N, Tatlidil Yaylaci E, Aydemir MÇ, Göka E. Serum TNF-related weak inducer of apoptosis (TWEAK) and TNF-related apoptosis-inducing ligand (TRAIL) levels of patients with bipolar disorder in manic episode, in remission and healthy controls. Psychiatry Res. 2017;257:338-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Atmaca M, Kuloglu M, Tezcan E, Onal S, Ustundag B. Neopterin levels and dexamethasone suppression test in posttraumatic stress disorder. Eur Arch Psychiatry Clin Neurosci. 2002;252:161-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Klaus F, Guetter K, Schlegel R, Seifritz E, Rassi A, Thöny B, Cathomas F, Kaiser S. Peripheral biopterin and neopterin in schizophrenia and depression. Psychiatry Res. 2021;297:113745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Ünal F, Öktem F, Çetin Çuhadaroğlu F, Çengel Kültür SE, Akdemir D, Foto Özdemir D, Çak HT, Ünal D, Tıraş K, Aslan C, Kalaycı BM, Aydos BS, Kütük F, Taşyürek E, Karaokur R, Karabucak B, Karakök B, Karaer Y, Artık A. [Reliability and Validity of the Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version, DSM-5 November 2016-Turkish Adaptation (K-SADS-PL-DSM-5-T)]. Turk Psikiyatri Derg. 2019;30:42-50. [PubMed] |
| 17. | Gormez V, Kılınçaslan A, Orengul AC, Ebesutani C, Kaya I, Ceri V, Nasıroglu S, Filiz M, Chorpita B. Psychometric properties of the Turkish version of the Revised Child Anxiety and Depression Scale - Child Version in a clinical sample. Psychiatry Clin Psychopharmacol. 2017;27:84-92. [RCA] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 101] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 18. | Nayek S, Ghosh S. A comparative study of serum C-reactive protein in patients with Generalised Anxiety Disorder and Depression. J Med Res. 2018;4:123-131. [DOI] [Full Text] |
| 19. | Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39:175-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51559] [Cited by in RCA: 37807] [Article Influence: 1989.8] [Reference Citation Analysis (0)] |
| 20. | Copeland WE, Shanahan L, Worthman C, Angold A, Costello EJ. Generalized anxiety and C-reactive protein levels: a prospective, longitudinal analysis. Psychol Med. 2012;42:2641-2650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 101] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 21. | Costello H, Gould RL, Abrol E, Howard R. Systematic review and meta-analysis of the association between peripheral inflammatory cytokines and generalised anxiety disorder. BMJ Open. 2019;9:e027925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 177] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 22. | Singh B, Goyal A, Patel BC. C-Reactive Protein: Clinical Relevance and Interpretation. 2025 May 3. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 23. | Karadağ H, Saygılı G, Yüksel R, Usta MB, Topçuoğlu C, Erzin G. Serum TNF- Related Weak Inducer of Apoptosis (TWEAK), TNF- Related Apoptosis-Inducing Ligand (TRAIL) Levels in Patients with Bipolar Depression, Major Depression and a Healthy Control Group. Psychiatr Danub. 2021;33:314-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 24. | Zhao HX, Yin SS, Fan JG. High plasma neopterin levels in Chinese children with autism spectrum disorders. Int J Dev Neurosci. 2015;41:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 25. | Bodur S, Ceylan MF, Iseri E, Sener S, Yucel AA. Serum neopterin levels in patients with autism. Int J Dev Disabil. 2014;60:109-115. [DOI] [Full Text] |
| 26. | Sweeten TL, Posey DJ, McDougle CJ. High blood monocyte counts and neopterin levels in children with autistic disorder. Am J Psychiatry. 2003;160:1691-1693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 103] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 27. | Ceylan MF, Uneri OS, Guney E, Ergin M, Alisik M, Goker Z, Senses Dinc G, Karaca Kara F, Erel O. Increased levels of serum neopterin in attention deficit/hyperactivity disorder (ADHD). J Neuroimmunol. 2014;273:111-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 28. | Celik C, Erdem M, Cayci T, Ozdemir B, Ozgur Akgul E, Kurt YG, Yaman H, Isintas M, Ozgen F, Ozsahin A. The association between serum levels of neopterin and number of depressive episodes of major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:372-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 29. | Chittiprol S, Venkatasubramanian G, Neelakantachar N, Babu SV, Reddy NA, Shetty KT, Gangadhar BN. Oxidative stress and neopterin abnormalities in schizophrenia: a longitudinal study. J Psychiatr Res. 2010;44:310-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 30. | Kuloğlu M, Atmaca M, Onal S, Geçici O, Bulut V, Tezcan E. Neopterin levels and dexamethasone suppression test in obsessive-compulsive disorder. Psychiatry Res. 2007;151:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 31. | Özkan Y, Kandemir H, Sapmaz ŞY, Taneli F. TGF-β1, neopterin, tetrahydrobiopterin, and nitric oxide levels in pediatric obsessive-compulsive disorder. J Obsessive Compuls Relat Disord. 2021;29:100649. [DOI] [Full Text] |
| 32. | Tezcan ME, Balcı EK. The relationship between inflammatory blood cells and executive functions in medication-free children with ADHD. Genel Tıp Dergisi. 2025;35:232-241. [DOI] [Full Text] |
| 33. | Kunz M, Ceresér KM, Goi PD, Fries GR, Teixeira AL, Fernandes BS, Belmonte-de-Abreu PS, Kauer-Sant'Anna M, Kapczinski F, Gama CS. Serum levels of IL-6, IL-10 and TNF-α in patients with bipolar disorder and schizophrenia: differences in pro- and anti-inflammatory balance. Braz J Psychiatry. 2011;33:268-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 34. | Kim YK, Jung HG, Myint AM, Kim H, Park SH. Imbalance between pro-inflammatory and anti-inflammatory cytokines in bipolar disorder. J Affect Disord. 2007;104:91-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 283] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 35. | Wang WY, Liu N, Qi XX, Han B, Sun JN, Chen ZL, Wang MW, Wang YY. Predictive effect of lipopolysaccharide-stimulated inflammatory cytokines on symptoms of generalized anxiety disorder. World J Psychiatry. 2024;14:1308-1318. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 36. | Agudelo LZ, Femenía T, Orhan F, Porsmyr-Palmertz M, Goiny M, Martinez-Redondo V, Correia JC, Izadi M, Bhat M, Schuppe-Koistinen I, Pettersson AT, Ferreira DMS, Krook A, Barres R, Zierath JR, Erhardt S, Lindskog M, Ruas JL. Skeletal muscle PGC-1α1 modulates kynurenine metabolism and mediates resilience to stress-induced depression. Cell. 2014;159:33-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 422] [Cited by in RCA: 596] [Article Influence: 49.7] [Reference Citation Analysis (3)] |
| 37. | Torun YT, Kurt ZK, Ünal K, Ibrahimkhanlı L, Özbaş C. Could Low Serum TWEAK Levels Serve as a Biomarker for Children and Adolescents Diagnosed with ADHD, Specifically the Predominantly Inattentive Subtype? Clin Psychopharmacol Neurosci. 2025;23:648-657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 38. | Hanley JA, McNeil BJ. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology. 1982;143:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16948] [Cited by in RCA: 12340] [Article Influence: 280.5] [Reference Citation Analysis (5)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/