Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.114953
Revised: November 13, 2025
Accepted: January 5, 2026
Published online: March 9, 2026
Processing time: 155 Days and 4.2 Hours
Acute diarrhea is the most common gastrointestinal disease in children under five years of age and is associated with significant morbidity and mortality risks. Lactose-free formulas may be beneficial for hospitalized patients.
To compare the efficacy, safety, and cost-effectiveness of a medium-chain triglyceride-containing lactose-free formula (MLF) with a standard lactose-free formula (SLF) in children hospitalized with acute diarrhea.
In this randomized, double-blind, controlled trial, children aged 6-24 months were randomized to receive MLF or SLF. The primary outcomes were the duration until diarrhea resolution and the frequency of stools after enrollment. Secondary outcomes included length of hospital stay, treatment costs, and caregiver satis
Forty-four children completed the study (MLF = 22, SLF = 22). The median duration of diarrhea after enrollment did not differ significantly between the groups, 24.0 hours [interquartile range (IQR): 21.5] vs 45.0 hours (IQR: 49.0); P = 0.293. The reduction in stool frequency was also comparable [7.5 (IQR: 5.8) vs 4.0 (IQR: 3.8) times/day; P = 0.083]. The Hodges-Lehmann estimated median difference in diarrhea duration was -4 hours (95% confidence interval: -26.0 to 2.0), supporting the noninferiority of MLF. The median length of hospital stay was 2 days in both groups. Formula costs were significantly lower in the MLF group (227.9 Thai Baht vs 373.6 Thai Baht; P < 0.001). Caregiver satisfaction, product acceptance, and safety profiles were comparable between the groups.
MLF demonstrated noninferior clinical outcomes and safety compared to SLF, with a significant reduction in formula-related costs. MLF could be an appropriate alternative for the management of acute diarrhea in young children, especially in resource-constrained settings.
Core Tip: This randomized controlled trial compared a medium-chain triglyceride-containing lactose-free formula (MLF) with a standard lactose-free formula in children hospitalized with acute diarrhea. MLF showed noninferior efficacy and safety compared to standard lactose-free formula, with similar clinical outcomes and caregiver satisfaction, but at significantly lower cost. MLF may therefore serve as a cost-effective alternative for managing acute diarrhea in young children, particularly in resource-limited settings.
- Citation: Phavichitr N, Tantibhaedhyangkul R, Dumrisilp T, Prachuapthunyachart S, Chanpreecha W, Ratanamongkol P, Chongsrisawat V. Clinical outcomes of two lactose-free formulas in the management of acute childhood diarrhea. World J Clin Pediatr 2026; 15(1): 114953
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/114953.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.114953
Diarrheal diseases are major causes of morbidity and mortality among children under five years of age worldwide and can lead to severe dehydration, malnutrition, and impaired development. Annually, an estimated 1.7 billion cases occur, with the highest burden in low- and middle-income countries[1]. In Thailand, the incidence of acute diarrhea was highest among children aged 0-4 years, reaching 3929.48 per 100000 people in 2022[2]. Management of acute diarrhea in children prioritizes rehydration and nutritional support[3]. Acute infectious diarrhea may damage enterocytes at the tips of intestinal villi, which can result in secondary lactase deficiency and symptoms such as osmotic diarrhea, bloating, and more frequent bowel movements[4]. The European Society of Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) suggests dietary modifications for infants and young children with severe watery diarrhea who are not breastfed, are suspected of having lactose intolerance, or are hospitalized. Switching to lactose-free formulas may be beneficial[5]. Systematic reviews support the use of lactose-free diets to manage acute diarrhea in children, and such diets may decrease both diarrhea duration and treatment failure rates, especially in hospitalized patients[6,7].
There is considerable variability in the composition, cost, and accessibility of lactose-free formulas across healthcare settings. In resource-limited settings, the availability, affordability, and cultural acceptability of specific products are important considerations. A medium-chain triglyceride (MCT)-containing, lactose-free formula produced locally is widely accessible at low cost. This formula has been used for three decades in children with digestive and absorptive disorders, including those requiring enteral nutrition. Despite its long-term use, published research evaluating its efficacy specifically during episodes of acute diarrhea is lacking. This study primarily aimed to compare the duration and severity of diarrhea between two lactose-free formulas in hospitalized children aged 6 months to 24 months. Secondary objectives included assessing formula acceptability, caregiver satisfaction, and associated formula costs until recovery from diarrhea.
This study was a double-blind, randomized, controlled, multicenter trial conducted in the pediatric departments of Phramongkutklao Hospital, King Chulalongkorn Memorial Hospital, and Bhumibol Adulyadej Hospital in Bangkok, Thailand. We enrolled hospitalized patients aged 6 months to 24 months with acute diarrhea admitted between September 14, 2023, and May 30, 2025. The exclusion criteria included severe dehydration; bloody mucous stools; the use of antibiotics, probiotics, or antidiarrheal agents within seven days before enrollment; cow’s milk protein allergy; exclusive breastfeeding; severe malnutrition; chronic gastrointestinal disorders such as inflammatory bowel disease; and other chronic conditions. The study was reviewed and approved by relevant Institutional Review Boards and conducted in accordance with Good Clinical Practices, the Declaration of Helsinki and its subsequent amendments, and national regulations. Informed consent was obtained from all participants’ caregivers prior to enrollment. The study was registered with the Thai Clinical Trials Registry, No. TCTR20240706013.
In this study, a noninferiority design was used, and the formula for two independent means was used to compare the mean diarrhea duration between groups. Sample size determination was based on the formula for comparing two independent means, with calculations performed using the sample size calculator[8]. The power analysis assumed a two-sided significance level of α = 0.05, 80% power, a standard deviation of 28.8 hours, and a margin of 12 hours, as derived from a previous study of lactose-free formula[9]. The margin reflects clinically acceptable differences in lactose-free formula efficacy. Allowing for a 15% dropout rate, a total of 42 participants (21 per group) were targeted to ensure adequate power and statistical reliability.
Eligible participants were randomized using a computerized block of four randomization methods, stratified by study site location. Both groups received lactose-free formula at a standard concentration of 20 kilocalories per fluid ounce. The intervention group received an MCT-containing, lactose-free formula (MLF) (PAN-ENTERAL®, Thai Otsuka Pharmaceutical Co., Ltd., Bangkok, Thailand), and the control group received a standard lactose-free formula (SLF) (ENFALAC A+ LactoFree Care 360° DHA plusTM, Mead Johnson Nutrition, Chonburi, Thailand). The composition of macronutrients in the research formulas is shown in Table 1. The volume of formula intake was appropriate for each child’s age, supplemented with breast milk or age-appropriate food. All participants received standard medical care for acute childhood diarrhea.
| Nutrients (g/100 kcal) | MCT-containing, lactose-free formula (MLF) | Standard lactose-free formula (SLF) |
| Protein | 3.0 | 2.1 |
| Source | Casein | Milk protein |
| Fat | 5.0 | 5.4 |
| Source | MCT oil, soy oil | Palm olein, soy oil, coconut oil, high oleic safflower oil, DHA, ARA |
| Carbohydrate | 10.7 | 10.6 |
| Source | Dextrin, sucrose | Solid corn syrup |
| Caloric density (calories per ounce) | 20 | 20 |
Upon enrollment, participants underwent a physical examination and anthropometric measurements (weight, length, and head circumference), and nutritional status was assessed using World Health Organization Anthro software version 3.2, a tool developed by the World Health Organization[10]. Stool consistency was scored using the modified Bristol Stool Form Scale for Children[11]. Additionally, the frequency of vomiting and dietary intake over the preceding 24 hours was recorded. Stool samples were collected for analysis of viral pathogens (Simple Rota-Adeno-Noro, OPERON, Zaragoza, Spain) and bacterial cultures. Bowel movements were monitored every two hours during hospitalization, and the frequency and consistency of the stools were recorded. Episodes of vomiting and other gastrointestinal symptoms were recorded. Intake data, including the volume of formula, breast milk, and complementary foods, were also collected. At least one of the following criteria was required for diarrhea recovery: Fewer than 3 bowel movements per day, normal stool consistency (modified Bristol Stool Form Scale for Children score: 3-4), and no watery stool for at least 12 hours. Discharge from the hospital was at the discretion of the attending physician. Prior to discharge, anthropometric parameters and body temperature were reassessed. All children were provided with the assigned formula until the follow-up visit, which was approximately 7 days after discharge, with a permissible deviation of up to ± 3 days. During this visit, weight and gastrointestinal symptoms were reassessed. The caregivers completed the Perceived Maternal Parenting Self-Efficacy Questionnaire and product acceptance questionnaire using the Visual Analogue Scale.
All statistical analyses were performed using an intention-to-treat approach. The distribution of continuous variables was assessed using the Shapiro-Wilk test. Variables with a normal distribution are reported as the means with standard deviations, while nonnormally distributed variables are expressed as medians with interquartile ranges (IQR). Between-group comparisons of continuous variables were conducted using Student’s t test or the Mann-Whitney U test, depending on the distribution of the data. For variables analyzed with the Mann-Whitney U test, the Hodges-Lehmann median difference and its corresponding 95% confidence interval (CI) were calculated to estimate the magnitude and precision of the between-group difference. Categorical variables were summarized as frequencies and percentages and were compared using the χ2 test. For continuous variables measured across multiple time points, between-group comparisons at each time point were performed using the Mann-Whitney U test. A two-sided P value < 0.05 was considered to indicate statistical significance. All the statistical analyses were conducted using IBM SPSS Statistics, version 30.0 (IBM Corp., Armonk, NY, United States).
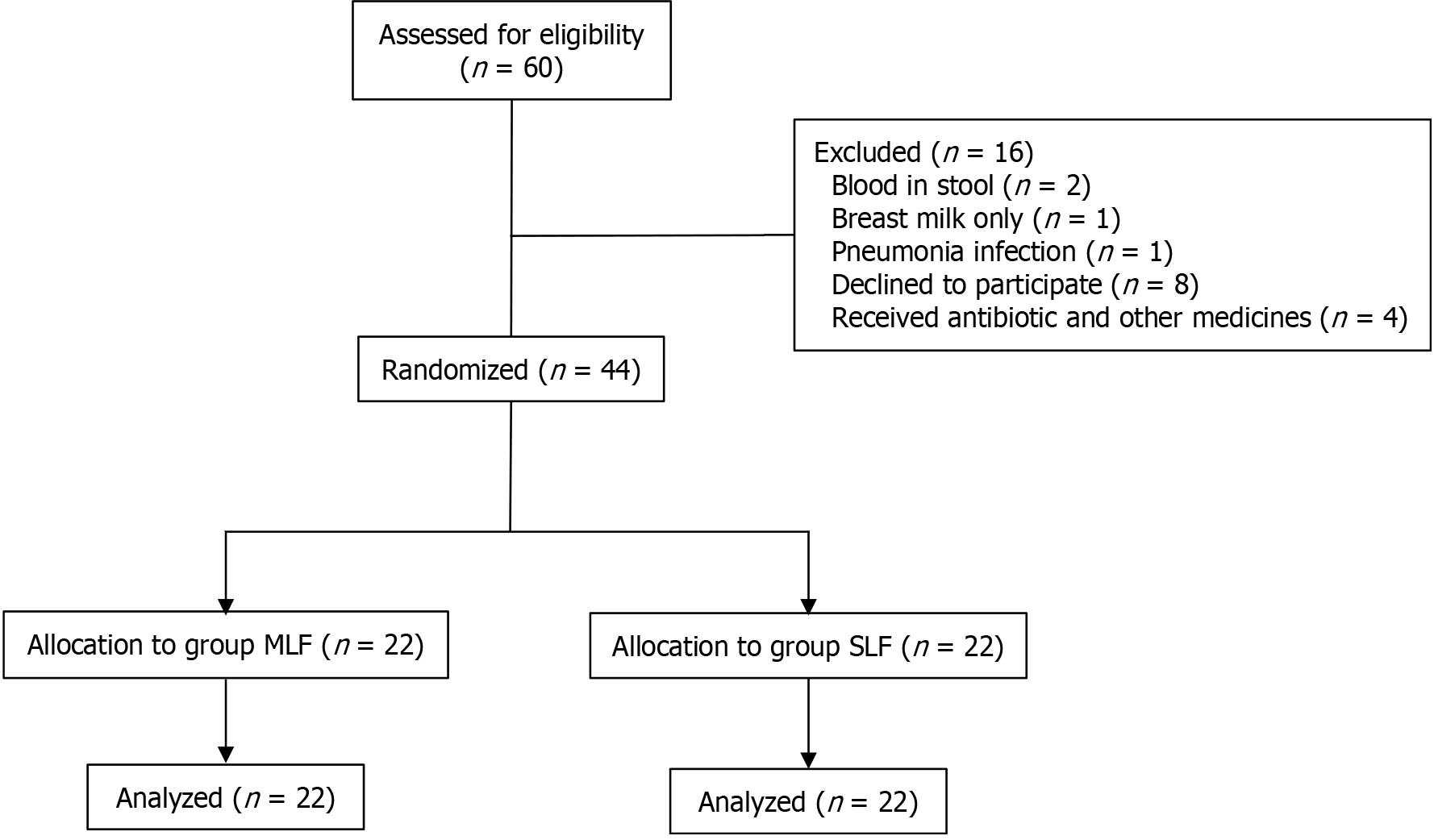
A total of 44 children aged 6-24 months with acute diarrhea were randomized, with 22 allocated to group MLF and 22 to group SLF. The Consolidated Standards of Reporting Trials flow diagram of the study is shown in Figure 1. Comparisons of demographic data (Table 2) revealed no significant intergroup differences in median age, sex distribution, body weight, length, or head circumference. The diarrhea parameters, including stool frequency, stool consistency, and median duration, were identical during the 24 hours before enrollment. The categories of infectious etiologies were classified as viral, bacterial, or unidentified infections without significant disparity in distribution across groups. The utilization of medications was comparable between the two study groups. No statistically significant differences were observed in the administration of antiemetic medications between the two groups (P > 0.05).
| Variable | Group MLF (n = 22) | Group SLF (n = 22) | P value |
| 1Gender female | 10.0 (45.5) | 10.0 (45.5) | 1.000 |
| Age (months) | 14.5 (10.5) | 14.5 (9.8) | 0.605 |
| 16-12 months | 10.0 (45.5) | 9.0 (40.9) | 0.761 |
| 1≥ 13 months | 12.0 (54.5) | 13.0 (59.1) | |
| Body weight on admission (g) | 9950.0 (2991.3) | 9900.0 (2340.0) | 0.366 |
| 2Body length on admission (cm) | 76.8 ± 7.7 | 78.3 ± 6.3 | 0.208 |
| 2Head circumference (cm) | 45.0 ± 3.3 | 45.0 ± 2.5 | 0.328 |
| Diarrhea parameters before enrollment | |||
| Duration of diarrhea (days) | 1.0 (1.0) | 2.0 (1.0) | 0.139 |
| Stool frequency in 24 hours (times) | 9.0 (5.5) | 7.5 (4.8) | 0.493 |
| Stool consistency in 24 hours (mBSFS-C score) | 5.0 (0.0) | 5.0 (0.0) | 0.639 |
| 3Infection by stool assessment | |||
| 1Viral infection (Rotavirus, adenovirus, norovirus) | 8.0 (38.0) | 7.0 (33.3) | 0.506 |
| 1Bacteria (Salmonella spp., Aeromonas spp.) | 4.0 (19.1) | 3.0 (14.3) | |
| 1Unidentified infection | 9.0 (42.9) | 11.0 (52.4) | |
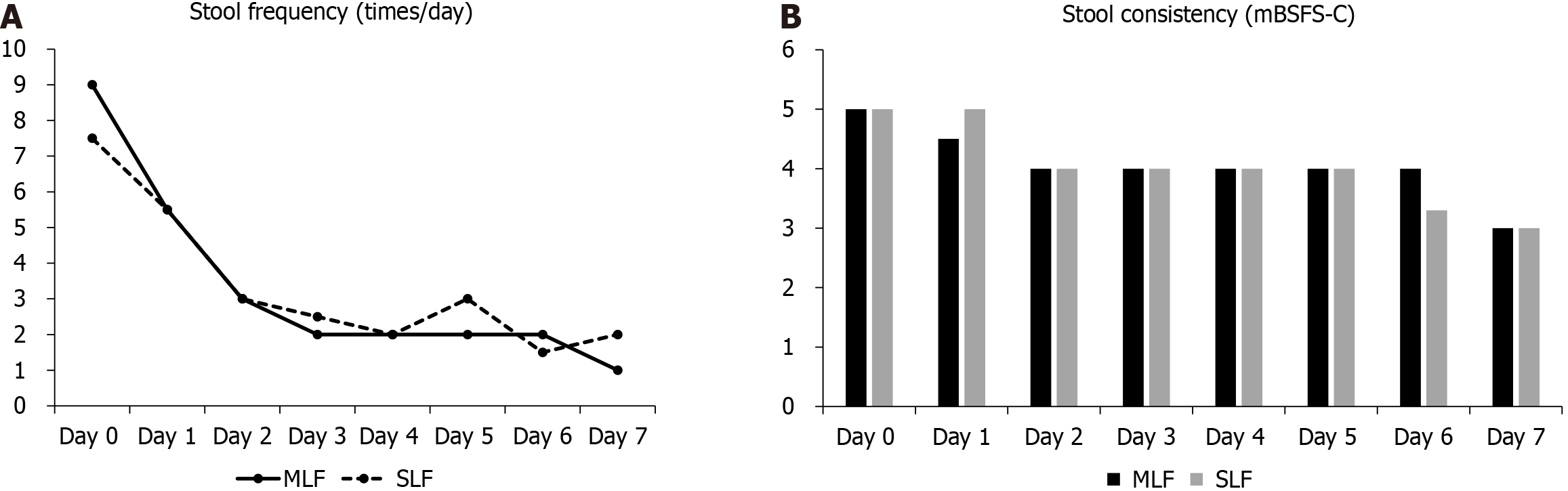
There was no statistically significant difference in the reduction in stool frequency from baseline to discharge between the two groups (P = 0.083). Group MLF experienced a median change of -7.5 times per day (IQR: 5.8), whereas group SLF experienced a median change of -4.0 times per day (IQR: 3.8), as shown in Table 3. No statistically significant difference in diarrhea duration was observed between the two groups (P = 0.293). The median duration of diarrhea in group MLF was 24.0 hours (IQR: 21.5), whereas that in group SLF was 45.0 hours (IQR: 49.0). The Hodges-Lehmann estimate of the median difference in diarrhea duration between groups was -4 hours (95%CI: -26.0 to 2.0). The median length of hospitalization was 2 days (IQR: 1.0) in both groups. The daily stool frequency and stool consistency from Day 0 to Day 7 were not significantly different between the groups at any time point, as shown in Figure 2. Both groups demonstrated a progressive reduction in stool frequency and improvement in stool consistency over the course of treatment.
| Group MLF (n = 22) | Group SLF (n = 22) | P value | |
| Diarrhea improvement outcomes | |||
| Change of stool frequency on discharge day compared to baseline (times/day) | -7.5 (5.8) | -4.0 (3.8) | 0.083 |
| Duration of diarrhea (hours) | 24.0 (21.5) | 45.0. (49.0) | 0.293 |
| Length of hospitalization (days) | 2.0 (1.0) | 2.0 (1.0) | 0.825 |
| Other outcomes | |||
| Amount of consumed formula until recovery (mL) | 900.0 (1291.6) | 1102.5 (696.3) | 0.391 |
| Cost of consumed formula until recovery (THB) | 227.9 (0.0) | 373.6 (0.0) | < 0.001 |
No significant changes in body weight from baseline were observed during hospitalization [group MLF: 0.0 g (IQR: 470.0) vs group SLF: 39.0 g (IQR 687.5), P = 0.684] or at the follow-up visit [group MLF: -60.0 g (IQR 410.0) vs group SLF: 100.0 g (IQR 852.5), P = 0.573]. The incidence of vomiting decreased over time in both groups, with no significant differences at any time point. No other gastrointestinal adverse events, including bloating or constipation, were reported in either group throughout the study. The median product acceptance score was comparable at 4.0 (IQR: 1.8) for group MLF and 4.0 (IQR: 1.0) for group SLF, P = 0.350 (with the highest satisfaction score being 5 points). Perceived Maternal Parenting Self-Efficacy Questionnaire scores also did not significantly differ between groups [79.0 (IQR: 13.0) vs 74.0 (IQR: 8.00), P = 0.144]. There was no significant difference between the groups in terms of the median volume of products consumed until recovery. The median formula intake was 900 mL (IQR: 1291.6) in group MLF and 1102.5 mL (IQR: 696.3) in group SLF (P = 0.391). The cost of the product used was significantly lower in group MLF (227.9 Thai Baht vs 373.6 Thai Baht, P < 0.001), as calculated in accordance with the product price list from IMS® Thailand healthcare index 2024.
Subgroup analysis comparing the viral and nonviral infection groups revealed no statistically significant differences in diarrhea outcomes between the MLF and SLF groups. In the viral infection subgroup (n = 15), in group MLF, the median change in stool frequency was -8.0 (IQR: 7.8) times/day vs -3.0 (IQR: 0.5) in group SLF (P = 0.287), with diarrhea duration trends favoring group MLF (22 hours vs 68 hours, P = 0.162). Stool consistency scores showed comparable improvement trajectories between groups across all time points. For nonviral infections (n = 27), there was no significant difference (P = 0.156) between the two groups, but compared with group SLF (-4.0, IQR: 3.5 times/day), group MLF showed a trend toward a higher change in stool frequency (-8.0, IQR: 5.0 times/day). The duration of diarrhea was similar between the groups (44 hours vs 45 hours, P = 0.903).
Acute diarrhea remains a significant health problem for children under five years of age. Hospitalization may place financial burdens on families, particularly in resource-limited settings. Compared with standard treatments such as oral rehydration alone, dietary modifications, including the use of lactose-free formulas, have been suggested to reduce stool output, lower the risk of persistent diarrhea, and accelerate recovery[12]. Expert guidelines recommend early feeding with lactose-free or low-lactose diets in non-breastfed children to support gut recovery, digestive enzyme function, and weight gain[13].
In this randomized controlled trial, no statistically significant differences in clinical outcomes were observed between the two lactose-free formulas studied. The median difference in diarrhea duration was -4.0 hours (95%CI: -26.0 to 2.0), with the upper bound below the prespecified noninferiority margin of 12 hours. This confirms the noninferiority of MLF compared with SLF. Although not statistically significant, the group MLF tended to have a shorter diarrhea duration (P = 0.293) and a greater reduction in stool frequency (7.5 times/day vs 4.0 times/day, P = 0.083). Intake volume and caregiver-reported satisfaction were comparable between the groups, but due to its significantly lower cost, MLF might be considered a clinically viable and economical option. The 39% lower cost of MLF suggests that it could help reduce the cost of care for hospitalized children with acute diarrhea in settings where treatment cost and affordability are major considerations.
Many studies compare lactose-free formulas with lactose-containing formulas; however, our study is among the first to focus on two lactose-free formulas. The diarrhea durations observed in both groups in this study aligned with previous research in similar settings[14-16]. The lower proportion of rotavirus infections compared with that reported in the data from Thailand in 2004 (12% vs 50%)[17] may partially explain the relatively mild and brief diarrheal episodes in our cohort. Increased rotavirus vaccination over the past two decades and changes in viral epidemiology have likely contributed to this shift[18].
The composition of MLF differs from that of most SLFs, namely, in its fat component, which contains 50% of the component as MCT. This MCT composition was originally designed for children with digestive or absorptive disorders because of its easy absorption and fast metabolism[19]. Excessive MCT can cause gastrointestinal symptoms and osmotic diarrhea, but we detected no adverse gastrointestinal events in this study. This finding is consistent with previous studies reporting that moderate doses of MCT are generally well tolerated[20]. Additionally, MLF has casein as its sole protein component, which may raise concerns about curd formation and its appropriateness of use in infants. Recent data, however, suggest that casein-based formulas are well tolerated and nutritionally adequate in infants[21,22]. No differences in weight gain were observed between the groups, which is consistent with previous studies on acute diarrhea management[14,15]. Nonetheless, these findings may be due to the short intervention and follow-up periods of the study.
The strengths of this study are that it emphasized the comparison of the clinical effectiveness of two lactose-free formulas differing in fat and protein compositions (among other less remarkable compositions), as well as its integration of cost analysis. Our findings revealed that MLF was clinically effective and safe for treating childhood acute diarrhea, cost-saving, and comparably acceptable to patients and caregivers. These factors suggest the potential use of MLF in hospitalized children with acute diarrhea. Importantly, all participants tolerated the assigned formula equally, and no dropouts due to feeding intolerance occurred.
Limitations include the limited generalizability of the results to more severe patients due to the specific inclusion/exclusion criteria (for example, severely dehydrated or chronically ill patients were not enrolled). Furthermore, mal
This randomized controlled trial demonstrated that MLF was noninferior to SLF in managing acute diarrhea among hospitalized children and offered significant cost advantages. These results support its consideration as a viable dietary management option in resource-limited settings.
The authors gratefully acknowledge the invaluable assistance of the laboratory staff, nurses, and research assistants at Phramongkutklao Hospital, King Chulalongkorn Memorial Hospital, and Bhumibol Adulyadej Hospital for data collection. We also extend our sincere thanks to Thai Otsuka Pharmaceutical Co., Ltd., for providing the study formulas for this research.
| 1. | World Health Organization. Diarrhoeal disease. 2024. [cited 3 August 2025]. Available from: https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease. |
| 2. | Department of Disease Control. Annual Epidemiological Surveillance Report 2022. [cited 3 August 2025]. Available from: https://apps-doe.moph.go.th/boeeng/annual/Annual/Annual_Report_2565.pdf. |
| 3. | World Health Organization. Guideline on management of pneumonia and diarrhoea in children up to 10 years of age. Geneva: World Health Organization; 2024. [cited 3 August 2025]. Available from: https://iris.who.int/handle/10665/380059. |
| 4. | Usai-Satta P, Scarpa M, Oppia F, Cabras F. Lactose malabsorption and intolerance: What should be the best clinical management? World J Gastrointest Pharmacol Ther. 2012;3:29-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 55] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (5)] |
| 5. | Guarino A, Ashkenazi S, Gendrel D, Lo Vecchio A, Shamir R, Szajewska H; European Society for Pediatric Gastroenterology, Hepatology, and Nutrition; European Society for Pediatric Infectious Diseases. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/European Society for Pediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe: update 2014. J Pediatr Gastroenterol Nutr. 2014;59:132-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 382] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 6. | MacGillivray S, Fahey T, McGuire W. Lactose avoidance for young children with acute diarrhoea. Cochrane Database Syst Rev. 2013;2013:CD005433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Gaffey MF, Wazny K, Bassani DG, Bhutta ZA. Dietary management of childhood diarrhea in low- and middle-income countries: a systematic review. BMC Public Health. 2013;13 Suppl 3:S17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Sample Size Calculator. Sample Size Estimation in Clinical Research: from Randomized Controlled Trials to Observational Studies. [cited 14 June 2025]. Available from: https://riskcalc.org/samplesize/. |
| 9. | Abdulrazzaq S, Jaafar FA, Mohammed ZA. Lactose versus Lactose Free Regimen in Children with Acute Diarrhea. Open Access Maced J Med Sci. 2021;9:1279-1282. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | World Health Organization. Child growth standards. 2011. [cited 14 June 2025]. Available from: https://www.who.int/tools/child-growth-standards/software. |
| 11. | Lane MM, Czyzewski DI, Chumpitazi BP, Shulman RJ. Reliability and validity of a modified Bristol Stool Form Scale for children. J Pediatr. 2011;159:437-441.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 113] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 12. | Putri AMN, Witarto AP, Yofi FA, Suasti NMA, Alawiyah K. Efficacy of lactose-free formula in acute diarrheal management for children under 5 years: a systematic review and meta-analysis. Paediatr Indones. 2024;64:233-43. [DOI] [Full Text] |
| 13. | Consortium G; Dutt S, Bhattacharya A, Fotedar A, Ray C, Dhekne J, Srinivas S. Expert recommendations on feeding practices of milk diets in acute infant diarrhea in India: a collaborative position statement. Med Res Arch. 2024;12. [DOI] [Full Text] |
| 14. | Saneian H, Yaghini O, Modaresi M, Razmkhah N. Lactose-free compared with lactose-containing formula in dietary management of acute childhood diarrhea. Iran J Pediatr. 2012;22:82-86. [PubMed] |
| 15. | Nguyen RN, Bui NQ, Thai DN. Efficacy of Lactose-Free Milk in Treating Acute Gastroenteritis in Vietnamese Children: A Randomized Controlled Trial. Cureus. 2024;16:e61178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Nitayakul P, Jeephet K, Ngeonmak T. Efficacy of lactose-free formula as a 24-hour management approach for acute diarrhea among children. J Southeast Asian Med Res. 2023;7:e0175. [DOI] [Full Text] |
| 17. | Simakachorn N, Tongpenyai Y, Tongtan O, Varavithya W. Randomized, double-blind clinical trial of a lactose-free and a lactose-containing formula in dietary management of acute childhood diarrhea. J Med Assoc Thai. 2004;87:641-649. [PubMed] |
| 18. | Charoenwat B, Suwannaying K, Paibool W, Laoaroon N, Sutra S, Thepsuthammarat K, Sirirattanakul S. The impact of rotavirus vaccination on acute diarrhea in Thai children under 5 years of age in the first year of universal implementation of rotavirus vaccines in the National Immunization Program (NIP) in Thailand: a 6-year analysis. BMC Public Health. 2023;23:2109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 19. | Hong SJ. Types of Special Infant Formulas Marketed in Korea and Their Indications. Pediatr Gastroenterol Hepatol Nutr. 2018;21:155-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 20. | Tanchoco CC, Cruz AJ, Rogaccion JM, Casem RS, Rodriguez MP, Orense CL, Hermosura LC. Diet supplemented with MCT oil in the management of childhood diarrhea. Asia Pac J Clin Nutr. 2007;16:286-292. [PubMed] |
| 21. | Descallar FB, Roy D, Wang X, Zhu P, Ye A, Liang Y, Pundir S, Singh H, Acevedo-Fani A. Investigation of the gastric digestion behavior of commercial infant formulae using an in vitro dynamic infant digestion model. Front Nutr. 2024;11:1507093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 22. | Huppertz T, Chia LW. Milk protein coagulation under gastric conditions: A review. Int Dairy J. 2021;113:104882. [RCA] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 23. | Nyeko R, Kalyesubula I, Mworozi E, Bachou H. Lactose intolerance among severely malnourished children with diarrhoea admitted to the nutrition unit, Mulago hospital, Uganda. BMC Pediatr. 2010;10:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/