Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.112088
Revised: August 9, 2025
Accepted: October 28, 2025
Published online: March 9, 2026
Processing time: 232 Days and 18.6 Hours
Bilirubin-induced neurologic dysfunction (BIND) remains a serious complication of severe neonatal hyperbilirubinemia, especially in resource-limited settings. While total serum bilirubin (TSB) is widely used for risk stratification, the bili
To compare the diagnostic accuracy of the B/A ratio vs TSB for predicting acute BIND in neonates.
We conducted a systematic review and meta-analysis of observational studies evaluating the B/A ratio and TSB for predicting BIND in neonates (≥ 35 weeks' gestational age). Data sources included PubMed, EMBASE, Cochrane Central, and Google Scholar. Pooled standardized mean difference (SMD), sensitivity, spe
Five studies involving a total of 1022 neonates were included, with a male-to-female ratio of approximately 57:40. The SMD for B/A ratio was 1.71 (95%CI: 1.00–2.41, P < 0.0001), and for TSB it was 1.68, both showing strong associations with BIND. Meta-regression revealed no significant difference in predictive value between the two biomarkers (P = 0.96).
Both TSB and the B/A ratio are comparably effective in predicting BIND. While the B/A ratio may provide in
Core Tip: Bilirubin-induced neurologic dysfunction (BIND) remains a preventable cause of neonatal brain injury, yet early risk stratification is challenging. We conducted a meta-analysis of observational studies using clinical criteria to diagnose BIND and found that elevated serum bilirubin/albumin (B/A) ratio is significantly associated with increased risk of BIND. This association remained robust across subgroup and meta-regression analyses. Our findings support the B/A ratio as a simple, accessible biomarker that may improve clinical prediction of BIND and guide early intervention in jaundiced neonates.
- Citation: Ahmad N, Ahmed U, Mohy Ud Din G, Habib M, Luqman MS, Talha M, Saifullah M, Rana I. Comparison of bilirubin albumin ratio and total serum bilirubin for predicting neurological dysfunction in newborns: A meta-analysis. World J Clin Pediatr 2026; 15(1): 112088
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/112088.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.112088
Acute bilirubin-induced neurologic dysfunction (BIND) encompassing acute bilirubin encephalopathy as well as chronic kernicterus remains a crucial and critical concern in term and late preterm infants. Although neonatal jaundice is common; affecting almost 60%-80% of newborns worldwide[1], its progression to severe hyperbilirubinemia and eventually neurotoxicity carries substantial risk of morbidity and mortality and thus eventually long-term neurodevelopmental impairments[2]. Despite recent advances in field of neonatology such cases continue to occur at a steadfast rate even in high income countries demonstrated by the incidence of peak total bilirubin > 25 mg/dL being 1 in 2480 live births in Canada and relatively are markedly much more prevalent in low-resource settings[3]. Studies have shown kernicterus affecting up to 0.7 per 1000 live births in parts of Africa, Asia and Latin America, vs 0.1/1000 in high-income regions, leading to approximately 1.2 deaths per 1000 births in low-income countries in comparison to around 0.01 per 1000 in high-income[4]. These epidemiologic data underscore the global burden of severe neonatal jaundice and the need for reliable predictors for the early detection and management of those infants which are at risk to develop acute BIND.
The pathophysiology of bilirubin neurotoxicity is clustered around unbound bilirubin. Bilirubin is produced from heme catabolism and circulates mostly bound to albumin; only the small unbound (free) fraction (Bf) can cross the blood-brain barrier, this concentration is believed to dictate the degree of neurotoxicity associated with hyperbilirubinemia[5]. When bilirubin production exceeds albumin-binding capacity, free bilirubin rises and penetrates the central nervous system, where it interacts with neurons and glial cells to cause neuronal cell apoptosis[6]. In resource limited settings one should take in account the co-morbid factors like prematurity, sepsis and acidosis that reduce albumin binding thus further elevating the free bilirubin and exacerbate the risk of developing BIND. Thus, the neurotoxic potential of hyperbilirubinemia depends not only on total bilirubin levels but critically on the balance between bilirubin and its carrier protein. Because free bilirubin measurement is not routinely available at the bedside, the bilirubin/albumin (B/A) ratio has been proposed as an indirect biomarker of neurotoxicity risk[7]. The B/A ratio effectively normalizes bilirubin concentration to the infant’s binding capacity. Early studies have tested this idea; Ardakani et al[8] found that a B/A cutoff of 8 mg/dL per g predicted acute BIND with 100% sensitivity and specificity of 94%, vs a total bilirubin cutoff of 25 mg/dL having 100% sensitivity, but only 85% specificity. They suggested that using B/A alongside total serum bilirubin (TSB) can improve specificity and prevent unnecessary invasive procedures including exchange transfusions. Similarly, Iskander et al[7] observed that both TSB and B/A were strong predictors of bilirubin encephalopathy, but B/A does not necessarily improve prediction over TSB alone. These and other data provide the rationale for formally comparing B/A to TSB in predicting acute BIND.
Given the global impact of severe neonatal hyperbilirubinemia, particularly in resource-poor settings where delayed screening and therapy persist, kernicterus remains a leading cause of preventable neonatal death and disability[1]. A recent review highlighted ongoing high rates of avoidable exchange transfusions and related complications in low-income and middle-income countries, along with persistent cases of extreme hyperbilirubinemia even in developed settings, leading to considerable healthcare burdens. These observations underscore the critical need for enhanced risk stratification strategies to more effectively direct interventions and mitigate adverse outcomes on a global scale. American Academy of Pediatrics (AAP) guidelines provide hour-specific TSB thresholds (the Bhutani nomogram zones) with built-in adjustments for risk factors such as isoimmune hemolysis, G6PD deficiency, and gestational age, but rely solely on TSB without incorporating albumin or binding capacity. In fact, the latest AAP 2022 revision raised phototherapy thresholds to reduce apparent overtreatment[9], but still does not address low albumin scenarios where free bilirubin could be dangerously elevated at moderate TSB levels. This gap justifies exploring whether the B/A ratio might improve risk assessment beyond existing protocols. Therefore, this study aimed to compare the predictive accuracy of the B/A ratio vs TSB in identifying neonates at risk of developing acute BIND. The primary objective was to determine whether the B/A ratio offers improved sensitivity and specificity over TSB alone in predicting acute BIND in a neonatal population.
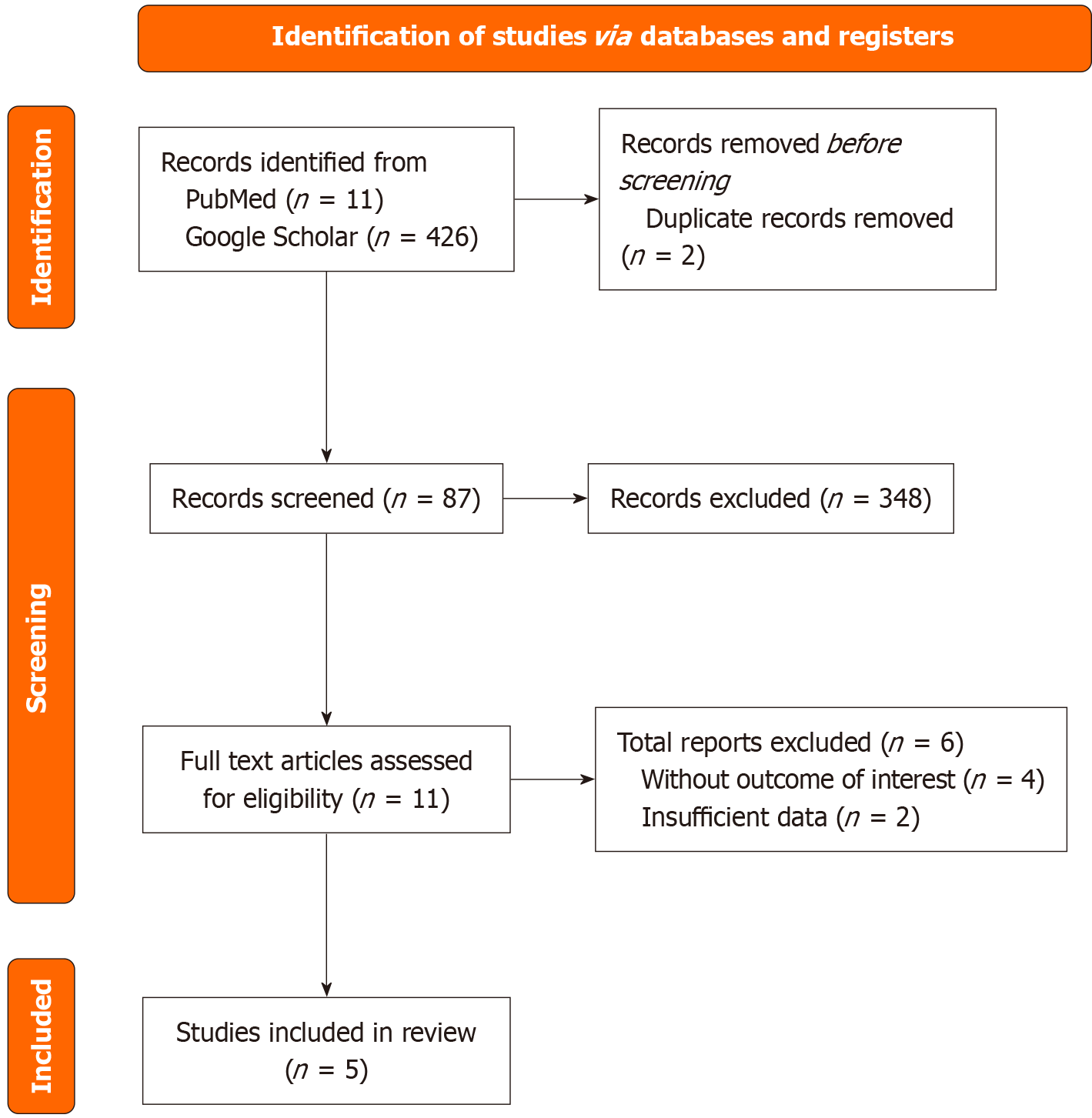
This systematic review and meta-analysis were undertaken in alignment with the methodological standards set forth in the Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy and are reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses of Diagnostic Test Accuracy (PRISMA-DTA) stu
We included all observational studies (cross-sectional or prospective cohorts) that assessed the diagnostic accuracy of the bilirubin-albumin (B/A) ratio in predicting BIND among neonates. Eligible studies were required to provide sufficient data to construct a 2 × 2 contingency table comparing B/A ratio thresholds with the occurrence of BIND as defined by clinical or neuroimaging criteria. We included studies irrespective of publication status, language, or geographic location. Exclusion criteria included case reports, reviews, editorials, conference abstracts without primary data, and studies using a reference standard not based on clinical diagnosis of BIND.
A comprehensive literature search was conducted in PubMed, EMBASE, Cochrane Central, and Google Scholar from inception to July 2020, with an updated search in August 2022. Reference lists of all eligible full-text articles and previous systematic reviews were manually screened for additional relevant studies. Grey literature and unpublished data were searched using Google and the Index Medicus for the South-East Asia Region.
The following search strategy was adapted for each database: (1) PubMed: (“bilirubin-albumin ratio” OR “bilirubin albumin binding” OR “bilirubin binding affinity”) AND (“neonate” OR “newborn” OR “infant”) AND (“BIND” OR “kernicterus” OR “bilirubin encephalopathy”); (2) Cochrane Central: “bilirubin-albumin ratio” AND “neonate” AND “neurologic dysfunction”; and (3) Google Scholar: “bilirubin albumin ratio” AND “BIND”. The complete search strategy is available in the Supplementary material[7,8,10-13].
Two reviewers independently screened titles and abstracts utilizing Rayyan software. Full-text publications meeting initial criteria were subsequently evaluated for eligibility, with any discrepancies resolved through consensus.
A standardized, pilot-tested form was used for data extraction by one reviewer and independently verified by a second reviewer. Extracted data included: (1) Study characteristics (author, year, country, design); (2) Population characteristics (gestational age, birth weight, inclusion criteria); (3) Index test details (B/A ratio threshold, measurement method); (4) Reference standard (clinical diagnosis of BIND, neuroimaging); and (5) Diagnostic accuracy data (true positives, false positives, true negatives, false negatives).
Where multiple B/A thresholds were reported, each was recorded separately. Discrepancies were resolved through discussion or consultation with a third reviewer.
The methodological quality and risk of bias of included studies were assessed independently by two reviewers using the Quality Assessment of Diagnostic Accuracy Studies-2 tool, customized to the review question. The four domains evaluated were: (1) Patient selection; (2) Index test; (3) Reference standard; and (4) Flow and timing. Predefined signaling questions were used to judge the risk of bias and applicability concerns, and any disagreements were resolved through consensus or arbitration by a third reviewer. The detailed criteria and customized tool are available in Supplementary material[7,8,10-13].
All analyses were conducted using R (version 4.2.2; R Foundation for Statistical Computing, Vienna, Austria). Pooled sensitivity and specificity (with 95%CI) were obtained by fitting a bivariate random-effects meta-analysis model[14]. From this model we derived the summary receiver operating characteristic curve and its area under the curve, and we calculated the pooled diagnostic odds ratio with 95%CI. To compare the two index tests, mixed-effects bivariate meta-regression was performed by including test type (TSB vs B/A) as a covariate in the model. Heterogeneity of the pooled estimates was assessed using the I2 statistic (to quantify between-study inconsistency) and by visual inspection of forest plots[14]; no additional subgroup or meta-regression analyses were performed to explore heterogeneity due to the small number of studies and consistency of test thresholds. Likewise, no formal assessment of publication bias was conducted (e.g. Deeks’ funnel plot asymmetry test was not applied). The review was conducted and reported in accordance with the PRISMA-DTA guidelines[15].
A total of five studies with 1022 neonates were included in this meta-analysis, showing a male-to-female ratio of roughly 57:40[8,10-13]. Each study provided diagnostic accuracy information for both TSB and the B/A ratio, creating 10 comparisons in total.
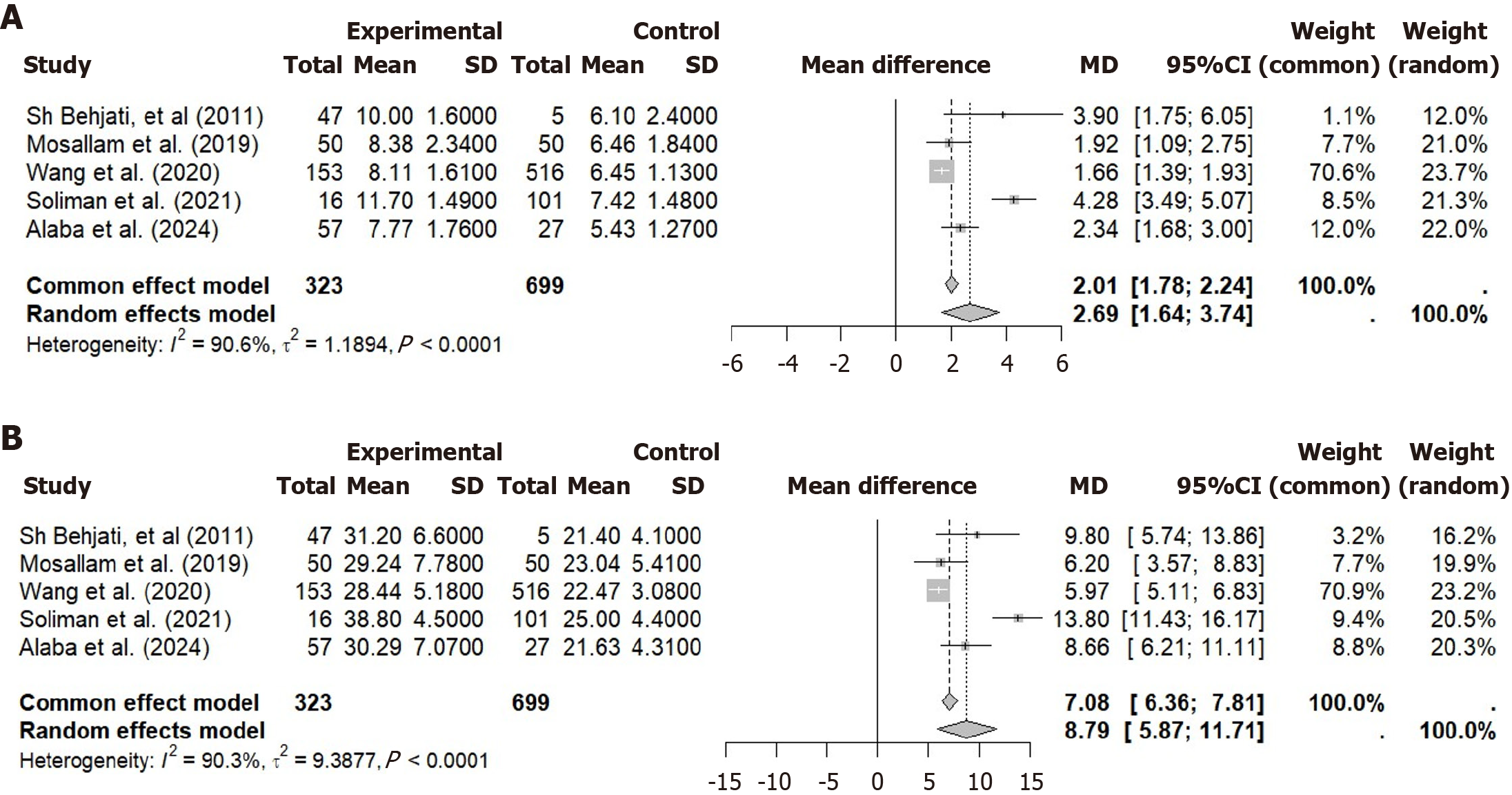
The forest plots for mean difference (MD) between the experimental (B/A ratio or TSB) and control groups are presented below (Figure 2)[8,10-13].
For the B/A ratio, the MD ranged from 1.92 to 4.28 across studies, with a cumulative MD of 2.01 (95%CI: 1.78-2.24) in the common effect model, while the random effects model yields a MD of 2.69 (95%CI: 1.64-3.74). This indicates a moderate effect size in predicting BIND. High heterogeneity (I2 = 90.6%, Tau2 = 1.1894) is observed, indicating variability in effect sizes between studies.
For TSB, the MD ranged from 5.97 to 13.80 across studies, with a cumulative MD of 7.08 (95%CI: 6.36-7.81) in the common effect model, while the random effects model yields a MD of 8.79 (95%CI: 5.87-11.71), indicating a strong association with the risk of BIND. High heterogeneity (I2 = 90.3%, Tau2 = 9.3877) is observed, suggesting variability in effect sizes between studies.
Subsequently, the standardized MD (SMD) were calculated using the data from the forest plots, accounting for variability in standard deviations across studies.
The SMD for the B/A ratio was found to be 1.71 (95%CI: 1.00-2.41, P < 0.0001), which suggests a significant association with the likelihood of BIND.
The SMD for TSB was similarly high at 1.68 (95%CI: 1.05-2.31, P < 0.0001), suggesting that TSB is also a reliable predictor of BIND.
To compare the two biomarkers, a mixed-effects meta-regression was conducted (Tables 1, 2, and 3). When considering biomarker type (TSB vs B/A) as a moderator, no statistically significant difference was observed in predictive accuracy [QM (df = 1) = 0.0022; P = 0.9628). The regression coefficient for TSB was -0.02 (95%CI: -1.02 to 0.98), indicating that both biomarkers are equally effective in identifying neonates vulnerable to BIND.
| Predictor | Standardized mean difference | 95%CI | P value | Interpretation |
| B/A ratio (reference) | 1.71 | 1.00-2.41 | < 0.0001 | Strong, significant predictor of bilirubin-induced neurologic dysfunction |
| Total serum bilirubin (vs B/A) | -0.02 | -1.02 to 0.98 | 0.9628 | No significant difference from B/A ratio |
| Term | Value |
| Intercept | 1.6828 (P < 0.0001) |
| Variable bilirubin/albumin | +0.0238 (P = 0.9628) |
| Parameter | Value |
| Number of studies | 5 |
| Total comparisons | 10 |
| Heterogeneity (I2) | 91.7% |
| Tau2 (residual variance) | 0.5618 |
| R2 (variance explained by biomarker type) | 0% |
| Test for moderator (QM) | QM = 0.0022, P = 0.9628 (df = 1) |
Although the studies consistently showed similar effect directions, there remained high residual heterogeneity (I2 = 91.7%), and biomarker type accounted for none of the variability between studies (R2 = 0%). This implies that additional factors – such as the study context, diagnostic standards, or population traits – may significantly influence the observed differences.
In conclusion, both TSB and B/A ratio demonstrated similarly strong correlations with BIND risk. Given their comparable effect sizes and the absence of a significant difference, either biomarker can serve as a dependable clinical indicator based on resource availability and context of the patient.
In this systematic review and meta-analysis of neonates ≥ 35 weeks, we found that the predictive ability of the B/A ratio for acute BIND was comparable to that of TSB. Both markers showed strong associations with neurologic outcomes, and the pooled effect sizes did not differ significantly between B/A and TSB. In other words, neither metric clearly outperformed the other in predicting BIND. Consistent with this finding, the largest prior study by Iskander et al[7] reported that B/A offered no net improvement over TSB alone. Likewise, Ardakani et al[8] observed essentially identical sensitivity of 100% for their chosen TSB and B/A cut-offs and only modest specificity differences.
Our pooled analysis, however, revealed substantial statistical heterogeneity (I2) high across studies. This likely reflects variation in study designs, patient populations, outcome definitions, and laboratory methods. Our results are largely in line with the existing literature. Several single-centre cohorts have identified elevated B/A ratios in infants with bilirubin encephalopathy and found significant adjusted associations between higher B/A and BIND, study done by Wang et al[11] demonstrated an adjusted odds ratio of 1.23 per unit increase in B/A. However, most studies including our meta-analysis suggest that TSB alone already captures the majority of predictive information. In other words, any additional signal from dividing by albumin appears modestly useful[12]. The high heterogeneity we observed underscores that context matters as some cohorts may have different thresholds for albumin, co-morbidities, or definitions of encephalopathy, which can influence the apparent value of B/A.
Biologically, however, the rationale for a B/A index remains plausible. The B/A ratio can be viewed as a crude measure of an infant’s binding reserve because when bilirubin levels approach albumin capacity, even small bilirubin increments yield large increases in free fraction[16]. As noted, only unbound bilirubin crosses into the brain, and albumin binding is saturable. Thus, for a given TSB, lower albumin, higher B/A, should translate into more free bilirubin and neurotoxicity risk[17]. In practice, our finding of similar effect sizes for B/A and TSB suggests that most infants who reach neurotoxic TSB thresholds also tend to have low enough albumin that the B/A ratio tracks along. In other words, TSB remains a robust marker of risk in most clinical scenarios. It is possible that the B/A ratio might have greater incremental value in specific subgroups including infants with sepsis or hemolysis, but our pooled results do not strongly support a universal advantage.
Clinically, these findings have several implications. The AAP guidelines, by focusing on TSB and known risk factors, remain generally valid for risk stratification. Our analysis suggests that simply adding a B/A threshold would not dramatically change risk classification in most infants. Indeed, one recent study found that albumin < 3.0 g/dL was seen in around 27% of late-preterm and term infants but infrequently altered treatment decisions in practice. In well-resourced settings, routine TSB monitoring and use of risk charts likely capture most high-risk infants, and B/A offers little incremental gain[17].
However, there may be settings where the B/A ratio could add value. In resource-limited regions where kernicterus remains endemic, a readily obtainable ratio of two inexpensive measurements might help identify infants close to neurotoxic thresholds[18]. For now, though, our evidence does not indicate that practice guidelines should immediately incorporate B/A ratios as a mandatory metric. Instead, clinicians should continue to consider albumin as one of multiple risk modifiers. It is notable that the 2022 AAP revisions are expected to safely reduce unnecessary treatment[13,19]; any future guidance on B/A would need to demonstrate that it further improves outcomes or decision-making without undue complexity.
Although 4 of the 5 included studies were prospective in design, one retrospective study contributed over 65% of the total sample size. This likely contributed to the high statistical heterogeneity (I2) observed and complicates the pooling and interpretation of results.
Diagnoses of acute BIND were based on differing criteria. While some studies employed standardized neurological assessments or bilirubin encephalopathy scales, others relied on surrogate markers such as auditory dysfunction. The absence of a uniform diagnostic gold standard introduces variability and may affect the comparative predictive performance of B/A ratio and TSB.
As most included studies were observational, they are prone to confounding. For example, infants with hemolysis or prematurity often have both elevated bilirubin and reduced albumin, making it difficult to isolate the independent predictive value of the B/A ratio. Additionally, potential publication bias cannot be ruled out, particularly if smaller negative studies were not published.
Several studies had a modest number of BIND cases, limiting statistical power. This is a common challenge in studying rare outcomes such as acute bilirubin neurotoxicity and constrains the precision of pooled estimates.
Future research should address these limitations. In particular, integrating B/A ratio into predictive tools or risk calculators may offer added value if validated in larger, high-quality prospective cohorts. The development of affordable, point-of-care assays for albumin or B/A could also improve risk stratification in resource-limited settings. Until such evidence becomes available, TSB remains the most accessible and widely validated biomarker for guiding the management of neonatal hyperbilirubinemia.
| 1. | Olusanya BO, Ogunlesi TA, Slusher TM. Why is kernicterus still a major cause of death and disability in low-income and middle-income countries? Arch Dis Child. 2014;99:1117-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 2. | Olusanya BO, Kaplan M, Hansen TWR. Neonatal hyperbilirubinaemia: a global perspective. Lancet Child Adolesc Health. 2018;2:610-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 194] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 3. | Sgro M, Campbell D, Shah V. Incidence and causes of severe neonatal hyperbilirubinemia in Canada. CMAJ. 2006;175:587-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 184] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 4. | Erdeve O. Management of neonatal jaundice in low-income and middle-income countries. BMJ Paediatr Open. 2020;4:e000845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Daood MJ, McDonagh AF, Watchko JF. Calculated free bilirubin levels and neurotoxicity. J Perinatol. 2009;29 Suppl 1:S14-S19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Brites D, Silva RFM. Bilirubin neurotoxicity: a narrative review on long lasting, insidious, and dangerous effects. Pediatr Med. 2021;4:34-34. [DOI] [Full Text] |
| 7. | Iskander I, Gamaleldin R, El Houchi S, El Shenawy A, Seoud I, El Gharbawi N, Abou-Youssef H, Aravkin A, Wennberg RP. Serum bilirubin and bilirubin/albumin ratio as predictors of bilirubin encephalopathy. Pediatrics. 2014;134:e1330-e1339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 8. | Ardakani SB, Dana VG, Ziaee V, Ashtiani MT, Djavid GE, Alijani M. Bilirubin/Albumin Ratio for Predicting Acute Bilirubin-induced Neurologic Dysfunction. Iran J Pediatr. 2011;21:28-32. [PubMed] |
| 9. | Ruangkit C, Rojsirikulchai N, Keesukphan S, Emrat K, Kongurai P, Swatesutipun B, Rattanamalee R, Soonsawad S. Serum Albumin Assessment in Neonatal Jaundice: Impact on Phototherapy Decision-Making. Hosp Pediatr. 2025;15:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Mosallam D, Said RN, Abd Elsamad MA, Abdelfatah NM. Use of serum bilirubin/albumin ratio for early prediction of bilirubin induced neurological dysfunction. Egypt Pediatr Assoc Gaz. 2019;67:11. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Wang Y, Sheng G, Shi L, Cheng X. Increased serum total bilirubin-albumin ratio was associated with bilirubin encephalopathy in neonates. Biosci Rep. 2020;40:BSR20192152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 12. | Soliman RM, Iskander IF, Elmazzahy EA, Abdellatif MAK. Can bilirubin/albumin ratio predict neurodevelopmental outcome in severe neonatal hyperbilirubinemia? A 3-month follow up study. Egypt Pediatr Assoc Gaz. 2021;69:4. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 13. | Alaba EO, Dedeke IOF, Agbo DD. Utilizing the Bilirubin-Albumin Ratio as a Predictive Marker for Bilirubin-Induced Neurologic Dysfunction: A Comparative Analysis in Two Referral Hospitals in Abeokuta. J Adv Med Med Res. 2024;36:125-134. [DOI] [Full Text] |
| 14. | Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557-560. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 15. | Cohen JF, Deeks JJ, Hooft L, Salameh JP, Korevaar DA, Gatsonis C, Hopewell S, Hunt HA, Hyde CJ, Leeflang MM, Macaskill P, McGrath TA, Moher D, Reitsma JB, Rutjes AWS, Takwoingi Y, Tonelli M, Whiting P, Willis BH, Thombs B, Bossuyt PM, McInnes MDF. Preferred reporting items for journal and conference abstracts of systematic reviews and meta-analyses of diagnostic test accuracy studies (PRISMA-DTA for Abstracts): checklist, explanation, and elaboration. BMJ. 2021;372:n265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 16. | Hulzebos CV, van Imhoff DE, Bos AF, Ahlfors CE, Verkade HJ, Dijk PH. Usefulness of the bilirubin/albumin ratio for predicting bilirubin-induced neurotoxicity in premature infants. Arch Dis Child Fetal Neonatal Ed. 2008;93:F384-F388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 17. | Kumar S, Agrawal K, Kumar S, Chaturvedi A. Risk Stratification of Term Neonates for Development of Significant Hyperbilirubinemia by Cord Serum Bilirubin and Albumin Ratio. Med J Dr DY Patil Vidyapeeth. 2024;17:600-604. [DOI] [Full Text] |
| 18. | Radmacher PG, Groves FD, Owa JA, Ofovwe GE, Amuabunos EA, Olusanya BO, Slusher TM. A modified Bilirubin-induced neurologic dysfunction (BIND-M) algorithm is useful in evaluating severity of jaundice in a resource-limited setting. BMC Pediatr. 2015;15:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 19. | Patel E, Potey GG. A study on Bilirubin / Albumin Ratio in Prediction of Bilirubin Induced Neural Dysfunction in Neonatal Hyperbilirubinemia. Int J Biol Med Res. 2014;5:4554-4555. |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/