Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111828
Revised: July 28, 2025
Accepted: October 23, 2025
Published online: March 9, 2026
Processing time: 239 Days and 16.4 Hours
Functional constipation (FC) is a global burden, and data on its prevalence based on Rome IV diagnostic criteria in young children, particularly after the coro
To determine the prevalence of FC in Thai infants and toddlers using the Rome IV criteria and identify associated factors.
The Thai version of the Rome IV Diagnostic Questionnaire for Pediatric Fun
A total of 1874 guardians participated, yielding a high response rate of 98.1%. The mean age of the children was 20.3 months ± 11.4 months, and 53.5% were male. The prevalence of FC was 16.1% [95% confidence interval (CI), 14.5%-17.8%]. Painful or hard bowel movements (81.5%), large-diameter stools (77.5%), and excessive stool retention (72.2%) were the most common symptoms. Factors independently associated with FC were age > 1 year [adjusted odds ratio (aOR), 3.36; 95%CI: 2.29-4.94], a first-degree relative with a history of constipation (aOR: 2.48; 95%CI: 1.85-3.35), fiber supplementation (aOR: 7.85; 95%CI: 3.56-17.30), and exposure to stressful family events (aOR: 1.41; 95%CI: 1.03-1.92).
FC is prevalent in Thai infants and toddlers. Older age, family history, fiber supplementation, and stress are key factors, highlighting genetic, dietary, and gut-brain influences that need further investigations.
Core Tip: To our knowledge, this nationwide study is the first to report the prevalence of functional constipation (FC) in Thai infants and toddlers using the validated Rome IV Diagnostic Questionnaire. FC affected 16.1% of children, demonstrating strong associations with age > 1 year, family history of constipation, fiber supplementation, and family-related stress. Findings highlight a complex interplay among genetic factors, dietary practices, and gut-brain interactions, particularly in the post-pandemic context while exploring the clinical value of fiber supplementation as a treatment. This study underscores the need for improved awareness and holistic management strategies to improve pediatric gastrointestinal health in Thailand.
- Citation: Chatpermporn K, Ngoenmak T, Charoenwat B, Chanpong A, Sriphongphankul H, Hanpitakpong N, Sintusek P. Functional constipation in Thai infants and toddlers: The role of genetic-gut-brain interaction and fiber supplementation. World J Clin Pediatr 2026; 15(1): 111828
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111828.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111828
Functional constipation (FC) is one of the most prevalent functional gastrointestinal disorders (FGIDs) in young children[1,2]. It is characterized by infrequent, difficult, or painful defecation, often accompanied by stool-withholding behavior, fecal impaction, and incontinence[3]. The diagnosis is clinical and based on the Rome IV criteria, which were introduced in 2016 to standardize the identification of pediatric FGIDs[3].
Globally, the prevalence of pediatric FC varies widely, ranging from 0.5% to 32.2%, depending on diagnostic criteria, study methodologies, and population demographics[4]. Notably, the adoption of the Rome IV criteria has led to an increase in the reported prevalence from 9.5% under Rome III to 14.4%[4,5]. Beyond its high prevalence, FC imposes a significant healthcare burden, often requiring long-term management strategies, such as dietary modifications, lifestyle modification, pharmacological treatments, and toilet training[6,7]. If left untreated, chronic FC can lead to chronic anal fissure, rectal distension, psychological disturbance, reduced quality of life, among other complications.
To promote standardized assessment, the Rome Foundation developed the Rome IV Diagnostic Questionnaire on Pediatric Functional Gastrointestinal Disorders (R4PDQ) as a validated research tool for valuating pediatric FGIDs. Among its tailored instruments, the R4PDQ-toddler: Parent-report form was specifically designed for neonates and toddlers (0-3 years). Previously, our team translated, validated, and assessed the reliability of the Thai version of the R4PDQ-Toddler after securing approval from the Rome Foundation[8,9]. Accordingly, this study aimed to determine the prevalence of FC in Thai children using the Thai validated version of the R4PDQ-Toddler and investigate potential associated factors, such as age, genetic predisposition, demographic characteristics, environmental influences, lifestyle habits, and psychosocial stressors, within a large population-based cohort. By identifying key determinants, this study seeks to promote early recognition and guide targeted interventions for improving pediatric gastrointestinal health.
This study enrolled the guardians of healthy children aged 6-48 months who sought care at the following outpatient clinics in six tertiary hospitals across Thailand: King Chulalongkorn Memorial Hospital, Naresuan University Hospital, Buddhachinaraj Hospital, Srinagarind Hospital, Songklanagarind Hospital, and Burapha University Hospital. A structured questionnaire survey was used, and the study was conducted between August 2023 and September 2024.
The exclusion criteria were as follows: Guardians of children with pre-existing conditions such as mechanical gastrointestinal abnormalities, history of abdominal surgery, thyroid disorders, developmental delays, neurological or spinal abnormalities, genetic or chromosomal disorders, or congenital heart disease.
The study protocol was approved by the Institutional Review Board of Chulalongkorn University (IRB 0041/66, COA 0469/2023), and written informed consent was obtained from all participants before the completion of the questionnaire.
Each study site employed at least one pediatric gastroenterologist and a research assistant. Guardians were given time to complete the questionnaire independently in a face-to-face setting. Research assistants clarify any questions, and pediatric gastroenterologists provide additional guidance, as needed.
The questionnaire was administered in a paper-based format and consisted of three sections:
General information: This included demographic data, participant characteristics, family education, and socioeconomic status.
Bowel movement assessment: The validated Thai version of the R4PDQ-toddler, along with two supplementary sections, was used.
Potential factors associated with FC: This included family history of constipation or atopy, lifestyle factors, and exposure to stressful life events.
Additional items included feeding type during the first 6 months, daily physical activity, media exposure, probiotic use, and fiber supplementation. Stressful life events were recorded separately for the children and parents. The guardians were also asked about their perception of whether their child had experienced constipation, previous physician consultations, and any previous treatments for constipation (Supplementary material).
FC: This was diagnosed based on the fulfillment of at least two criteria from the Rome IV guidelines for either toilet-trained or non-toilet-trained children[3].
Nutritional status: This was classified according to the World Health Organization[10]: Overweight: Weight-for-length/height > 2 SD; Obesity: Weight-for-length/height > 3 SD; Underweight: Weight-for-length/height ≤ 2 SD; Malnutrition: Weight-for-length/height ≤ 3 SD.
Atopy: This encompassed a history of allergic diseases, including atopic dermatitis, food allergy, allergic rhinitis, and asthma diagnosed by healthcare providers.
Fluid intake: Daily consumptions of water, milk, and other beverages were recorded in milliliters, which was converted from the number of glasses consumed per day (1 glass equal to 240 mL). Fluid adequacy was assessed using the guidelines of the National Institute for Health and Care Excellence and was stratified by age[11]: Infants (≤ 1 year): < 960 mL/day = inadequate fluid intake; Young children (> 1 year): < 1200 mL/day = inadequate fluid intake.
Fiber intake: Fruit and vegetable consumption was documented in portion sizes, with adequacy determined based on the guidelines of the United States Department of Agriculture[12]. Infants (< 1 year): < 0.5 cup equivalent of fruit and vegetables/day = inadequate intake; Children (1-3 years): < 1 cup equivalent of fruit and vegetables/day = inadequate intake (one cup is roughly equivalent to the size of an adult's fist or about 3.5 inches in diameter).
Total sleep duration: The total sleep duration, including naps, was recorded in hours. Sleep adequacy was evaluated using recommendations from the American Academy of Sleep Medicine and the American Academy of Pediatrics (AAP)[13]. Infants (≤ 1 year): < 12 hours/day = inadequate sleep; Children (> 1 year): < 10 hours/day = inadequate sleep.
Physical activity: Adequate physical activity is defined as at least 60 minutes per day for children aged > 1 year, in accordance with the Recommendations for Preventive Pediatric Health Care by AAP[14].
Screen time or media exposure: This refers to the amount of time a child spends using digital devices, such as sm
Stressful life events: Stressful events were categorized as follows: Family-related stressors, such as neglect, parental divorce, unemployment, financial difficulties, domestic violence, serious illness, or death of a family member; Environmental stressors include relocation, school transitions, severe illness, or death of a pet; Psychosocial stressors, such as exposure to violent incidents or traumatic events that affect mental well-being.
Pediatric gastroenterologists, assistant nurses, or research assistants collected all the completed questionnaires and electronically entered data at each study site. Data were managed using Research Electronic Data Capture (REDCap), hosted at the Chula Data Management Centre - Faculty of Medicine, Chulalongkorn University. REDCap is a secure, web-based application designed for research data management[15]. Following data entry, all records were systematically reviewed to ensure adherence to the exclusion criteria before analysis.
Continuous variables are summarized as mean (SD). Comparisons between continuous variables were conducted using Student’s t-test, whereas categorical variables were analyzed with the χ2 test. To identify associated factors, univariate and multivariate logistic regression analyses were employed. Any covariate that presented significance < 0.05 (P < 0.05) was subjected to multiple logistic regression analysis to obtain the adjusted odds ratio (aOR), with P < 0.05 indicating a noteworthy association. Statistical analyses were performed using IBM SPSS Statistics for Windows version 23.0 (IBM Corp., Armonk, NY, United States) and Stata version 18 (StataCorp LLC, College Station, TX, United States). A P value of 0.05 indicated significance.
With a response rate of 98.1%, a total of 1952 guardians completed the questionnaire. After applying the exclusion criteria, 78 participants were excluded because their children had thyroid abnormalities (n = 18), congenital heart disease (n = 42), a history of abdominal surgery (n = 3), or genetic/chromosomal disorders (n = 15). Thus, 1874 guardians remained for analysis.
The mean age of the children was 20.3 ± 11.4 months, and 53.5% were male. Most participants resided in central Thailand (41.3%), were > 1 year, and had a normal nutritional status. The majority had mothers as their primary caregivers, with a high educational level and household income.
A total of 302 children fulfilled at least two items from the Rome IV diagnostic criteria for FC for > 1 month. The prevalence of FC was determined to be 16.1% (95%CI: 14.5%-17.8%). Notably, excluding cases where the presence of a large fecal mass in the rectum necessitated physical examination by a doctor, seven children would have otherwise been overlooked.
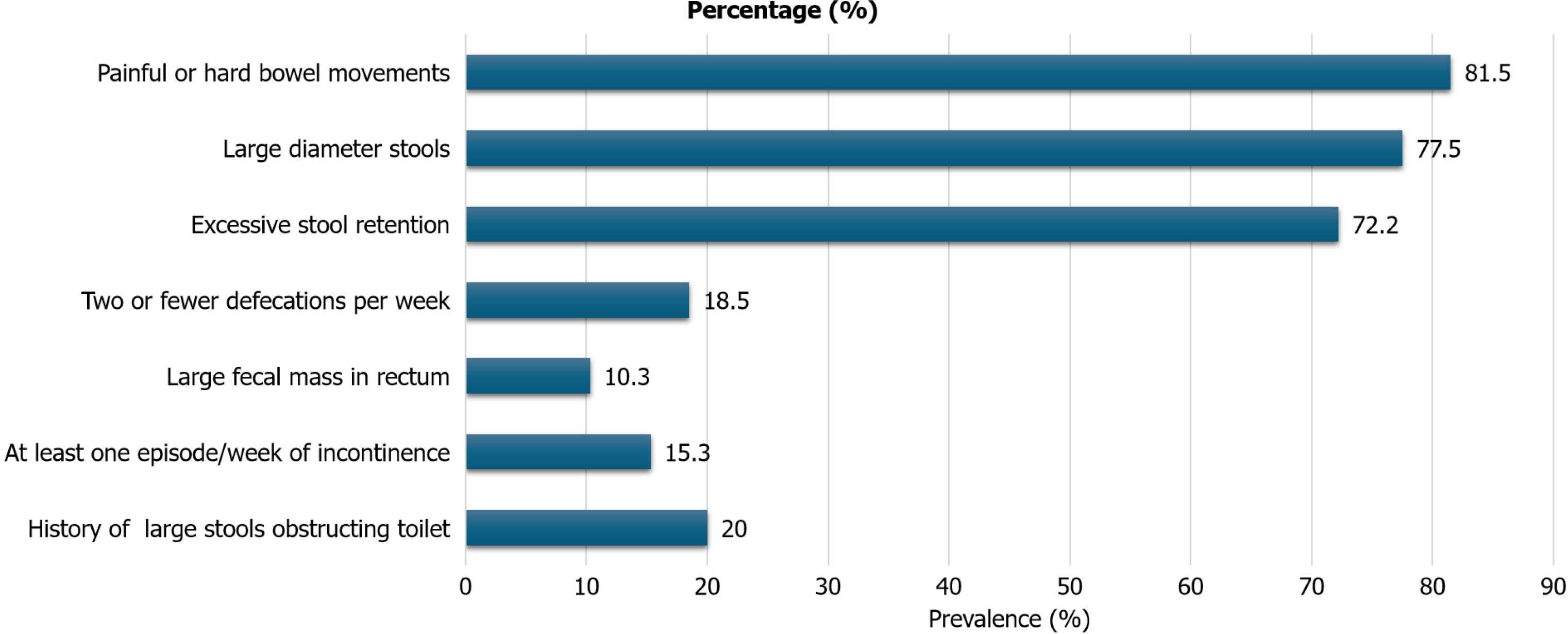
According to the criteria, the presenting symptoms included painful or hard bowel movements (81.5%), large-diameter stools (77.5%), excessive stool retention (72.2%), ≤ 2 defecations per week (18.5%), and the presence of a large fecal mass in the rectum (10.3%). Within the toilet-trained group, a history of large-diameter stools potentially obstructing the toilet (20.0%) and experiencing at least one episode per week of incontinence after acquiring toileting skills (15.3%) were notable findings (Figure 1).
Demographic data and family status: The demographic data demonstrated a diverse distribution across various backgrounds, such area of residence, age, sex, nutritional status, family type, parental education, and family status. Significant differences in older age, family debt, family history of constipation, and child history of atopy were found between the FC and non-FC groups. The FC group had an older age (P < 0.001), more atopic diseases (P = 0.014), more family debt (P = 0.015), and more family members with a history of constipation (P < 0.001) than the non-FC group (Table 1).
| Total (n = 1874) | Constipated (n = 302) | Non-constipated (n = 1572) | P value | |
| Area of residence | 0.206 | |||
| Central | 774 (41.3) | 132 (43.7) | 642 (40.8) | |
| Eastern | 207 (11) | 34 (11.3) | 173 (11) | |
| Northern | 363 (19.4) | 59 (19.5) | 304 (19.3) | |
| Northeastern | 326 (17.4) | 56 (18.5) | 270 (17.2) | |
| Southern | 204 (10.9) | 21 (7) | 183 (11.6) | |
| Male | 1003 (53.5) | 147 (48.7) | 856 (54.5) | 0.065 |
| Age (months) | 20.3 ± 11.4 | 25.6 ± 11.2 | 19.3 ± 11.2 | < 0.001a |
| 6-12 | 550 (29.3) | 32 (10.6) | 518 (33) | < 0.001a |
| ≥ 12 | 1324 (70.7) | 270 (89.4) | 1054 (67) | |
| Nutritional status | 0.161 | |||
| Obese | 24 (1.3) | 8 (2.6) | 16 (1) | |
| Overweight | 89 (4.7) | 12 (4) | 77 (4.9) | |
| Normal | 1671 (89.2) | 265 (87.7) | 1406 (89.4) | |
| Underweight | 82 (4.4) | 15 (5) | 67 (4.3) | |
| Malnutrition | 8 (0.4) | 2 (0.7) | 6 (0.4) | |
| Family number | 4.75 ± 1.74 | 4.84 ± 1.86 | 4.73 ± 1.71 | 0.313 |
| Single family | 904 (48.3) | 154 (51) | 750 (47.7) | 0.3 |
| Main caregiver: Mother | 1190 (63.5) | 206 (68.2) | 984 (62.6) | 0.063 |
| Main caregiver educational level | 0.316 | |||
| Lower than mandatory education (below high school) | 436 (23.3) | 77 (25.5) | 359 (22.8) | |
| At least mandatory education (at least high school) | 1438 (76.7) | 225 (74.5) | 1213 (77.2) | |
| Family status | 0.76 | |||
| Together | 1740 (92.8) | 283 (93.7) | 1457 (92.7) | |
| Separated | 91 (4.9) | 12 (4) | 79 (5) | |
| Divorced | 32 (1.7) | 6 (2) | 26 (1.7) | |
| Widowed | 11 (0.6) | 1 (0.3) | 10 (0.6) | |
| Family income | 0.493 | |||
| Low income (< 30000 bahts/month) | 718 (38.5) | 127 (40.2) | 591 (38.1) | |
| Family debt | 1187 (63.4) | 210 (69.5) | 977 (62.2) | 0.015a |
| First degree relative with a history of constipation | 323 (17.2) | 98 (32.5) | 225 (14.3) | < 0.001a |
| Family history of atopy | 634 (33.8) | 116 (38.4) | 518 (33) | 0.066 |
| Child history of atopy | 220 (11.7) | 48 (15.9) | 172 (10.9) | 0.014a |
Lifestyles and stressful life events: No difference in the exclusive breastfeeding type was noted during the first 6 months of life and adequate fluid intake per age group. However, the non-FC group had a more adequate fiber intake per day than the FC group. In the FC group, children aged < 2 years had more media exposure than those in the non-FC group (P < 0.001), although both groups had adequate sleep duration per day based on age-specific recommendations. Older children with FC tended to have more screen time and lower physical activity levels, although these differences did not reach significance. Additionally, the FC group was more likely to receive probiotics and fiber supplementation (P < 0.001).
The FC group experienced significantly more stressful life events than the non-FC group (P = 0.001). The most common stressors included relocation or change of residence (5.0%), insufficient family income (4.5%), family member’s divorce (3.31%), serious illness or death of a family member (2.4%), and family member’s unemployment (1.6%) (Table 2).
| Total (n = 1874) | Constipated (n = 302) | Non-constipated (n = 1572) | P value | |
| Exclusive breastfeeding for the 1st 6 months | 889 (47.5) | 151 (50) | 738 (47) | 0.467 |
| Adequate fluid intake per age | 1495 (79.8) | 229 (75.8) | 1266 (80.5) | 0.062 |
| Adequate fruit and vegetable consumption per age | 1729 (92.3) | 259 (85.8) | 1470 (93.5) | < 0.001a |
| History of probiotics exposure | 267 (14.2) | 69 (22.8) | 198 (12.4) | < 0.001a |
| History of fiber supplementation | 100 (5.3) | 48 (15.9) | 52 (3.3) | < 0.001a |
| Media exposure (age < 2 years) | 221 (17.8) | 48 (31.8) | 173 (15.8) | < 0.001a |
| Screen time (age > 2 years); > 1 hour | 176 (26.8) | 72 (27.8) | 104 (26.1) | 0.623 |
| Physical activity (age > 1 year) | N = 1326 | N = 271 | N = 1055 | 0.079 |
| No | 147 (11.1) | 21 (7.8) | 126 (11.9) | |
| < 1 hour | 372 (28.1) | 86 (31.8) | 286 (27.11) | |
| > 1 hour | 807 (60.9) | 164 (60.1) | 643 (61.0) | |
| Adequate sleep duration for age | 1794 (95.7) | 292 (96.7) | 1502 (95.5) | 0.369 |
| Stressful life event in family | 302 (16.1) | 66 (21.9) | 213 (13.6) | < 0.001a |
| (1) Family related stressors | ||||
| Neglect | 9 (0.5) | 2 (0.7) | 7 (0.5) | |
| Parental divorce | 39 (2.1) | 10 (3.3) | 29 (1.8) | |
| Unemployment | 29 (1.6) | 10 (3.3) | 19 (1.2) | |
| Financial difficulties | 85 (4.5) | 19 (6.3) | 66 (4.2) | |
| Domestic violence | 8 (0.4) | 2 (0.7) | 6 (0.4) | |
| Serious illness or death of a family member | 44 (2.4) | 8 (2.7) | 36 (2.3) | |
| (2) Environmental stressors | ||||
| Relocation | 94 (5.0) | 20 (6.6) | 74 (4.7) | |
| School transitions | 23 (1.2) | 5 (1.7) | 18 (1.2) | |
| Severe illness or death of pet | 20 (1.1) | 3 (1.0) | 17 (1.1) | |
| (3) Psychological stressors | ||||
| Traumatic events impacted mental well-being | 11 (0.6) | 4 (1.3) | 7 (0.4) | |
| (4) Others | 13 (0.7) | 6 (2.0) | 7 (0.4) |
Factors associated with FC: Multivariate logistic regression analysis identified four independent factors significantly associated with FC: Older age (> 1 year; aOR: 3.36; 95%CI: 2.29-4.94), history of constipation in first-degree relatives (aOR: 2.48; 95%CI: 1.85-3.35), fiber supplementation (aOR: 7.85; 95%CI: 3.56-17.30), and stressful life events in the family (aOR: 1.41; 95%CI: 1.03-1.93) (Table 3).
| Variables | Crude OR (95%CI) | P value | Adjust OR (95%CI) | P value |
| Age > 1 year | 4.15 (2.83-6.07) | < 0.001a | 3.36 (2.29-4.94) | < 0.001a |
| First degree relatives with a history of constipation | 2.68 (2.03-3.54) | < 0.001a | 2.48 (1.85-3.35) | < 0.001a |
| Family debt | 1.31 (1.01-1.70) | 0.039a | 1.30 (0.99-1.72) | 0.067 |
| Child atopic disease | 1.54 (1.09-2.18) | 0.02a | 1.27(0.88-1.84) | 0.193 |
| Inadequate fruit and vegetable consumption | 2.39 (1.64-3.50) | 0.001a | 1.19 (0.89-1.57) | 0.293 |
| Probiotics supplement | ||||
| Past | 1.98 (1.34-2.94) | 0.001a | 1.41 (0.92-2.17) | 0.112 |
| Current | 2.12 (1.39-3.24) | < 0.001a | 1.55 (0.98-2.46) | 0.059 |
| Supplementary fiber | ||||
| Past | 4.49 (2.68-7.50) | < 0.001a | 3.26 (1.89-5.62) | < 0.001a |
| Current | 10.20 (4.83-21.52) | < 0.001a | 7.85 (3.56-17.30) | < 0.001a |
| Stressful life events in family | 1.59 (1.19-2.12) | 0.002a | 1.41 (1.03-1.93) | 0.029a |
This study stated the prevalence of FC in Thai infants and toddlers based on the Rome IV diagnostic criteria and a validated, standardized research tool. Conducted on a nationally representative cohort, the findings reveal a prevalence of 16.1% (95%CI: 14.5%-17.8%). The most common clinical manifestations were painful or hard bowel movements, large stools, and stool-withholding behavior. Notably, the key independent factors associated with FC included older age (> 1 year), FC history in a first-degree relative, significant psychological stress within the family, and fiber supplementation.
To our knowledge, this is the first nationwide study in Thailand utilizing the validated Rome IV criteria, officially authorized by the Rome Foundation, to assess FC prevalence in young children. Previous studies in Thailand using the Rome III criteria reported have varied FC prevalence rates. A study in Bangkok, primarily involving school-aged children, found a prevalence of 25.1%, possibly reflecting the older age group and increased urban stressors[16]. Another study in southern Thailand, focusing predominantly on infants aged < 1 year, reported a much lower prevalence of 1.6%[17]. The methodological differences in the age distribution, regional demographics, and diagnostic criteria may explain these variations. When compared with international studies employing the Rome IV criteria, this study reported a notably higher FC prevalence than those conducted in young children across Asia, Europe, and North America[2,18]. A possible explanation for this disparity is the timing of our survey, as it was conducted after the coronavirus disease 2019 pandemic, a period of heightened stress and lifestyle disruptions[19-22]. Nearly one-fourth of our participants had experienced significant stressful life events, such as financial instability and parental unemployment, which may contribute to FC through the gut-brain axis[23].
FC is a multifactorial disorder influenced by both physiological and psychosocial components. Stool-withholding behavior is a key pathophysiological mechanism, particularly in toddlers and young children, where prior painful defecation, forceful toilet training, or rectal interventions may reinforce avoidance behaviors and perpetuate FC[24]. This study found that age > 1 year is significantly associated with FC. A large cross-sectional study in children aged ≤ 2 years similarly reported a notably lower prevalence of constipation in the first year of life compared with that in the second year[25]. This difference may be due to dietary factors and behavioral changes related to toilet training. Infants generally exhibit softer stools and more frequent bowel movements owing to the easily digestible nature of breast milk, which acts as a natural laxative[26]. In contrast, older children undergoing toilet training may develop stool-withholding behaviors because of fear or anxiety surrounding defecation, resulting in harder stools and a constipation cycle. A study in children aged 18-30 months revealed that stool-toileting refusal occurred in approximately one in five children. This evidence supported this hypothesis of the high prevalence of FC in children aged > 1 year[26,27].
Genetic predisposition is another well-established risk factor for FC. A family history of constipation, particularly among first-degree relatives, has been consistently associated with an increased likelihood of FC in children[28-30]. Although the precise genetic mechanisms remain unclear, shared environmental factors—particularly those related to stress and dietary habits—may also contribute to familial clustering. Investigating this further through genetic studies, particularly identical twin studies, could provide more definitive insights.
Adequate intake of fiber and water was the key factor in preventing FC. Contrary to conventional understanding, no significant association between either inadequate fiber or water intake and FC was found in this study. Interestingly, the FC group was more likely to have received fiber supplementation. This paradoxical finding is likely due to the cross-sectional nature of this study, where fiber supplementation may represent a reactive measure taken by parents after FC onset rather than a risk factor. Additionally, fiber supplementation might be an ineffective treatment for FC; thus, it should not be advocated for FC, as it could delay the initiation of more effective treatments, such as osmotic laxatives and age-appropriate toilet training programs.
Despite accumulating data supporting the gut microbiota diversity in FGIDs, including FC, this study found an insignificant association between probiotic supplementation and FC. A randomized clinical trial demonstrated that the prophylactic use of Lactobacillus reuteri DSM 17938 during the first 3 months of life resulted in a higher mean number of daily evacuations at 3 months of age compared with placebo. However, this finding did not specifically indicate a preventive effect on FC[31]. According to the treatment effect of probiotics, several systematic reviews and meta-analyses in 2017-2024 have examined the efficacy of probiotics in treating pediatric FC. Both a 2017 systematic review and a 2022 Cochrane Review found no significant improvement in treatment success when compared with placebo[32,33]. A recent network meta-analysis from China suggested that only Lactobacillus casei rhamnosus Lcr35, when combined with standard laxatives, may increase bowel movement frequency; however, the overall evidence remains inconclusive[34]. As the present study included both pharmaceutical and nonpharmaceutical probiotic use, specific strains and dosages could not be accurately documented, which might explain why probiotic supplementation does not influence FC in this study. Further nationwide studies on specific strains of probiotics supplementation are warranted.
Disorders of gut-brain interactions are proposed to be the major pathogenesis of FGIDs and are an area of concern that is integrated as a part of treatment for FGIDs. Similar to other FGIDs, studies have identified child stress as a contributing factor to FC[4,30,35]. However, these studies were conducted in children aged 7-16 years. The reported stressors included separation from a close friend, parental job loss, and severe illness within the family[30]. Thus, to our knowledge, this study is the first to evaluate stress in young children, including overall family stress, involve parent and caregiver stress, and found an association. Stress is linked to slow colonic transit, a key factor in FC. Psychological distress can affect the rectal mucosal blood flow through autonomic pathways. These factors, acting via the enteric nervous system, may inhibit colonic motility and prolong transit time[36,37]. Additionally, this study evaluated other factors, such as socioeconomic status and education, and the results did not reveal significant differences in parental educational level, family income, and parental separation, as noted in previous studies[28,30,38].
A key strength of this study is the use of the validated Thai version of the R4PDQ-Toddler, which ensures standardized and reliable data collection. Additionally, the large, multicenter, population-based design enhances the generalizability of the findings to the broader Thai pediatric population.
However, several limitations should be acknowledged. First, the study population was drawn from tertiary hospitals, which may introduce selection bias; although most participants were attending well-child visits for vaccinations. Second, the stress factors were not evaluated using standardized measurements, which could have led to an overestimation or underestimation of their effect. However, our results may help draw attention to the psychological health of children and families in relation to FC. Third, the cross-sectional design precludes causal inferences regarding risk factors, such as fiber supplementation and FC. Thus, more longitudinal studies are needed to establish temporal relationships and further elucidate causality.
The prevalence of FC among Thai young children is higher than that previously reported both within Thailand and in other Asian populations. Independent risk factors include older age, a family history of constipation, and stressful life events. The observed association of fiber supplementation with FC highlights the need for further investigations to determine its clinical implications. Moreover, advocacy for the understanding and knowledge of FC should be promoted among guardians to enable them to seek appropriate treatment for their children.
The authors express their gratitude to Dr. Yuda Chongpison, PhD, for her invaluable assistance with statistical con
| 1. | Steutel NF, Zeevenhooven J, Scarpato E, Vandenplas Y, Tabbers MM, Staiano A, Benninga MA. Prevalence of Functional Gastrointestinal Disorders in European Infants and Toddlers. J Pediatr. 2020;221:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (4)] |
| 2. | Huang Y, Tan SY, Parikh P, Buthmanaban V, Rajindrajith S, Benninga MA. Prevalence of functional gastrointestinal disorders in infants and young children in China. BMC Pediatr. 2021;21:131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 3. | Benninga MA, Faure C, Hyman PE, St James Roberts I, Schechter NL, Nurko S. Childhood Functional Gastrointestinal Disorders: Neonate/Toddler. Gastroenterology. 2016;S0016-5085(16)00182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 406] [Article Influence: 40.6] [Reference Citation Analysis (9)] |
| 4. | Koppen IJN, Vriesman MH, Saps M, Rajindrajith S, Shi X, van Etten-Jamaludin FS, Di Lorenzo C, Benninga MA, Tabbers MM. Prevalence of Functional Defecation Disorders in Children: A Systematic Review and Meta-Analysis. J Pediatr. 2018;198:121-130.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 237] [Article Influence: 29.6] [Reference Citation Analysis (5)] |
| 5. | Tran DL, Sintusek P. Functional constipation in children: What physicians should know. World J Gastroenterol. 2023;29:1261-1288. [PubMed] [DOI] [Full Text] |
| 6. | Silverman AH, Berlin KS, Di Lorenzo C, Nurko S, Kamody RC, Ponnambalam A, Mugie S, Gorges C, Sanghavi R, Sood MR. Measuring Health-Related Quality of Life With the Parental Opinions of Pediatric Constipation Questionnaire. J Pediatr Psychol. 2015;40:814-824. [PubMed] [DOI] [Full Text] |
| 7. | Varni JW, Bendo CB, Nurko S, Shulman RJ, Self MM, Franciosi JP, Saps M, Pohl JF; Pediatric Quality of Life Inventory (PedsQL) Gastrointestinal Symptoms Module Testing Study Consortium. Health-related quality of life in pediatric patients with functional and organic gastrointestinal diseases. J Pediatr. 2015;166:85-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 210] [Article Influence: 19.1] [Reference Citation Analysis (3)] |
| 8. | Sperber AD. Translation and validation of study instruments for cross-cultural research. Gastroenterology. 2004;126:S124-S128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 462] [Cited by in RCA: 478] [Article Influence: 21.7] [Reference Citation Analysis (4)] |
| 9. | Chatpermporn K, Chongpison Y, Ngoenmak T, Treepongkaruna S, Sintusek P. Validity and reliability of the Thai "Rome IV diagnostic questionnaires" for functional gastrointestinal disorders in neonates and toddlers. World J Clin Cases. 2025;13:105022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | World Health Organization. WHO child growth standards: growth velocity based on weight, length and head circumference: methods and development. Geneva: World Health Organization, 2009. |
| 11. | National Institute for Health and Care Excellence. Constipation in children and young people: diagnosis and management, 2010. Available from: https://www.nice.org.uk/guidance/cg99/chapter/Recommendations#diet-and-lifestyle. |
| 12. | Arnold MJ, Harding MC, Conley AT. Dietary Guidelines for Americans 2020-2025: Recommendations from the U.S. Departments of Agriculture and Health and Human Services. Am Fam Physician. 2021;104:533-536. [PubMed] |
| 13. | Paruthi S, Brooks LJ, D'Ambrosio C, Hall WA, Kotagal S, Lloyd RM, Malow BA, Maski K, Nichols C, Quan SF, Rosen CL, Troester MM, Wise MS. Consensus Statement of the American Academy of Sleep Medicine on the Recommended Amount of Sleep for Healthy Children: Methodology and Discussion. J Clin Sleep Med. 2016;12:1549-1561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 539] [Article Influence: 53.9] [Reference Citation Analysis (0)] |
| 14. | Canadian Paediatric Society, Digital Health Task Force, Ottawa, Ontario. Screen time and young children: Promoting health and development in a digital world. Paediatr Child Health. 2017;22:461-477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 166] [Article Influence: 18.4] [Reference Citation Analysis (5)] |
| 15. | Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44307] [Cited by in RCA: 41212] [Article Influence: 2424.2] [Reference Citation Analysis (5)] |
| 16. | Piriyanon P, Intarakhao S, Yuangthong A. The prevalence of constipation in preschool children at The Kindergarten of Thammasat University: a school-based study using Rome III criteria. TMJ. 2018;18:202-209. |
| 17. | Osatakul S, Puetpaiboon A. Use of Rome II versus Rome III criteria for diagnosis of functional constipation in young children. Pediatr Int. 2014;56:83-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Chia LW, Nguyen TVH, Phan VN, Luu TTN, Nguyen GK, Tan SY, Rajindrajith S, Benninga MA. Prevalence and risk factors of functional gastrointestinal disorders in Vietnamese infants and young children. BMC Pediatr. 2022;22:315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 19. | Zeigler Z. COVID-19 Self-quarantine and Weight Gain Risk Factors in Adults. Curr Obes Rep. 2021;10:423-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 20. | Werneck AO, Silva DR, Malta DC, Gomes CS, Souza-Júnior PR, Azevedo LO, Barros MB, Szwarcwald CL. Associations of sedentary behaviours and incidence of unhealthy diet during the COVID-19 quarantine in Brazil. Public Health Nutr. 2021;24:422-426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 21. | Aziz I, Whitehead WE, Palsson OS, Törnblom H, Simrén M. An approach to the diagnosis and management of Rome IV functional disorders of chronic constipation. Expert Rev Gastroenterol Hepatol. 2020;14:39-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 261] [Article Influence: 43.5] [Reference Citation Analysis (1)] |
| 22. | Sharma A, Rao SSC, Kearns K, Orleck KD, Waldman SA. Review article: diagnosis, management and patient perspectives of the spectrum of constipation disorders. Aliment Pharmacol Ther. 2021;53:1250-1267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 23. | Liyanarachchi H, Rajindrajith S, Kuruppu C, Chathurangana P, Ranawaka R, Devanarayana NM, Benninga MA. Association between childhood constipation and exposure to stressful life events: a systematic review. Neurogastroenterol Motil. 2022;34:e14231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 24. | Chogle A, Velasco-Benitez CA, Koppen IJ, Moreno JE, Ramírez Hernández CR, Saps M. A Population-Based Study on the Epidemiology of Functional Gastrointestinal Disorders in Young Children. J Pediatr. 2016;179:139-143.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (3)] |
| 25. | Loening-Baucke V. Prevalence, symptoms and outcome of constipation in infants and toddlers. J Pediatr. 2005;146:359-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 123] [Article Influence: 5.9] [Reference Citation Analysis (3)] |
| 26. | Mulhem E, Khondoker F, Kandiah S. Constipation in Children and Adolescents: Evaluation and Treatment. Am Fam Physician. 2022;105:469-478. [PubMed] |
| 27. | Taubman B. Toilet training and toileting refusal for stool only: a prospective study. Pediatrics. 1997;99:54-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 58] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 28. | Park M, Bang YG, Cho KY. Risk Factors for Functional Constipation in Young Children Attending Daycare Centers. J Korean Med Sci. 2016;31:1262-1265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 29. | Zhou LP, Deng L, Huang GL, Li G, Tang N, Cai WZ. [Epidemiologic study on functional constipation in adolescents]. Zhongguo Ertong Baojian Zazhi. 2010;18:566-568. |
| 30. | Inan M, Aydiner CY, Tokuc B, Aksu B, Ayvaz S, Ayhan S, Ceylan T, Basaran UN. Factors associated with childhood constipation. J Paediatr Child Health. 2007;43:700-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 31. | Indrio F, Di Mauro A, Riezzo G, Civardi E, Intini C, Corvaglia L, Ballardini E, Bisceglia M, Cinquetti M, Brazzoduro E, Del Vecchio A, Tafuri S, Francavilla R. Prophylactic use of a probiotic in the prevention of colic, regurgitation, and functional constipation: a randomized clinical trial. JAMA Pediatr. 2014;168:228-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 147] [Article Influence: 12.3] [Reference Citation Analysis (6)] |
| 32. | Wojtyniak K, Szajewska H. Systematic review: probiotics for functional constipation in children. Eur J Pediatr. 2017;176:1155-1162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 33. | Wallace C, Sinopoulou V, Gordon M, Akobeng AK, Llanos-Chea A, Hungria G, Febo-Rodriguez L, Fifi A, Fernandez Valdes L, Langshaw A, Saps M. Probiotics for treatment of chronic constipation in children. Cochrane Database Syst Rev. 2022;3:CD014257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Yang WC, Zeng BS, Liang CS, Hsu CW, Su KP, Wu YC, Tu YK, Lin PY, Stubbs B, Chen TY, Chen YW, Shiue YL, Zeng BY, Suen MW, Hung CM, Wu MK, Tseng PT. Efficacy and acceptability of different probiotic products plus laxatives for pediatric functional constipation: a network meta-analysis of randomized controlled trials. Eur J Pediatr. 2024;183:3531-3541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 35. | Devanarayana NM, Rajindrajith S. Association between constipation and stressful life events in a cohort of Sri Lankan children and adolescents. J Trop Pediatr. 2010;56:144-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 3.8] [Reference Citation Analysis (3)] |
| 36. | Chan AO, Cheng C, Hui WM, Hu WH, Wong NY, Lam KF, Wong WM, Lai KC, Lam SK, Wong BC. Differing coping mechanisms, stress level and anorectal physiology in patients with functional constipation. World J Gastroenterol. 2005;11:5362-5366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 37. | Towers AL, Burgio KL, Locher JL, Merkel IS, Safaeian M, Wald A. Constipation in the elderly: influence of dietary, psychological, and physiological factors. J Am Geriatr Soc. 1994;42:701-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 99] [Article Influence: 3.1] [Reference Citation Analysis (3)] |
| 38. | Udoh EE, Rajindrajith S, Devanarayana NM, Benninga MA. Prevalence and risk factors for functional constipation in adolescent Nigerians. Arch Dis Child. 2017;102:841-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/