Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111501
Revised: August 7, 2025
Accepted: November 14, 2025
Published online: March 9, 2026
Processing time: 248 Days and 12.4 Hours
Cesarean section (C-section) delivery rates are rising globally, raising concern about their potential long-term health consequences on offspring. Emerging evidence suggests that C-section, by bypassing the physiological and microbial exposures of vaginal birth, may adversely affect neonatal gastrointestinal (GI) development and immune regulation.
To evaluate the short- and long-term gastrointestinal outcomes in infants born via C-section, compared to those born vaginally.
A systematic literature search was conducted across PubMed, Scopus, and Web of Science for studies published from inception to 2024. Studies were included if they reported gastrointestinal outcomes among C-section-born infants and included a vaginal delivery comparison group. Outcomes of interest included short-term disorders (infantile colic, gastroesophageal reflux, constipation, dyschezia, and functional gastrointestinal disorders) and long-term conditions (inflammatory bowel disease, food allergies including cow's milk protein allergy, celiac disease, and microbiota dysbiosis). Data extraction and synthesis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.
C-section -born infants demonstrated a higher risk of several short-term GI disturbances, including infantile colic, reflux, and constipation. These outcomes were consistently associated with early gut microbiota dysbiosis, characterized by reduced microbial diversity and delayed colonization by Bifidobacteria and Bacteroides. Long-term associations with C-section included a modest increase in the risk of Crohn’s disease, cow’s milk protein allergy, and food sensitization, while the links with ulcerative colitis and celiac disease were inconsistent. Breastfeeding, probiotic/synbiotic supplementation, and limited antibiotic exposure emerged as protective modifiable factors.
C-section delivery is associated with a higher risk of adverse gastrointestinal outcomes in infancy and childhood, driven in part by early-life gut microbial disruption. Preventive strategies targeting microbial restoration and immune modulation, particularly breastfeeding and probiotic interventions, may help mitigate these risks. Clinicians should be aware of these associations to guide personalized care and parental counseling, particularly in elective C-section deliveries.
Core Tip: Cesarean section (C-section) delivery is associated with altered neonatal gut microbiota and a higher risk of both short- and long-term gastrointestinal (GI) disorders. This systematic review summarizes evidence linking C-section to infantile colic, gastroesophageal reflux, constipation, food allergies, inflammatory bowel disease, and celiac disease. The review highlights gut dysbiosis as a central mechanism and identifies modifiable factors—including breastfeeding, probiotic use, and reduced antibiotic exposure—that can mitigate risks. Understanding these associations enables clinicians to provide personalized postnatal care and early interventions that may prevent chronic GI outcomes in C-section-born infants.
- Citation: Al-Beltagi M, Alzayani S, Saeed NK, Bediwy AS, Prabu Kumar A, Bediwy HA, Elbeltagi R. Gastrointestinal consequences of cesarean section birth: A systematic review of short- and long-term effects in infancy and beyond. World J Clin Pediatr 2026; 15(1): 111501
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111501.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111501
Cesarean section (C-section) has become one of the most commonly performed surgical procedures worldwide, with global rates steadily rising in both high-income and low- to middle-income countries[1]. While C-sections are often lifesaving and medically necessary, an increasing proportion are elective or performed without a clear medical indication. This shift has prompted renewed interest in the potential health consequences associated with non-vaginal delivery, particularly in the neonatal and early developmental stages[2].
One of the most critical areas influenced by the mode of delivery is the gastrointestinal (GI) system. During vaginal delivery, neonates are naturally exposed to maternal vaginal and fecal microbiota, which play a fundamental role in the initial colonization of the infant gut[3]. In contrast, infants delivered via C-section often experience a markedly different microbial trajectory, characterized by delayed colonization, reduced microbial diversity, and dominance of skin- and environment-associated microbes[4]. Early-life microbial dysbiosis has been linked to a range of both short-term and long-term GI outcomes[5]. In the immediate postnatal period, C-section-delivered infants demonstrate higher rates of functional GI symptoms such as infantile colic, feeding intolerance, and gastroesophageal reflux[6]. Over the longer term, emerging evidence suggests a potential link between C-section and increased risk of functional GI disorders (FGIDs), food allergies, and even inflammatory bowel disease (IBD)[7]. These effects are thought to be mediated not only by altered microbial colonization but also by the downstream effects on gut immune development, mucosal integrity, and metabolic signaling[8].
Early research, preceding the last two decades, laid the groundwork for our current understanding. For instance, pioneering studies in the late 20th century first proposed that the mode of delivery could significantly shape the infant's immunological and microbial development, setting the stage for the 'hygiene hypothesis' and subsequent large-scale investigations[9]. Despite growing interest, the evidence base remains fragmented, with significant heterogeneity in study designs, outcome definitions, and follow-up durations. Some studies highlight breastfeeding and probiotic interventions as potential modifiers of risk, while others point to antibiotic exposure or lack of labor as additional contributors[10].
This systematic review aims to critically evaluate and synthesize the available literature on the GI effects associated with C-section delivery, focusing on both short-term symptoms and long-term disease outcomes in infancy and early childhood. By consolidating the evidence, this review aims to clarify the extent of GI impact, identify modifiable risk factors, and inform pediatric healthcare strategies for optimizing gut health in infants delivered by C-section.
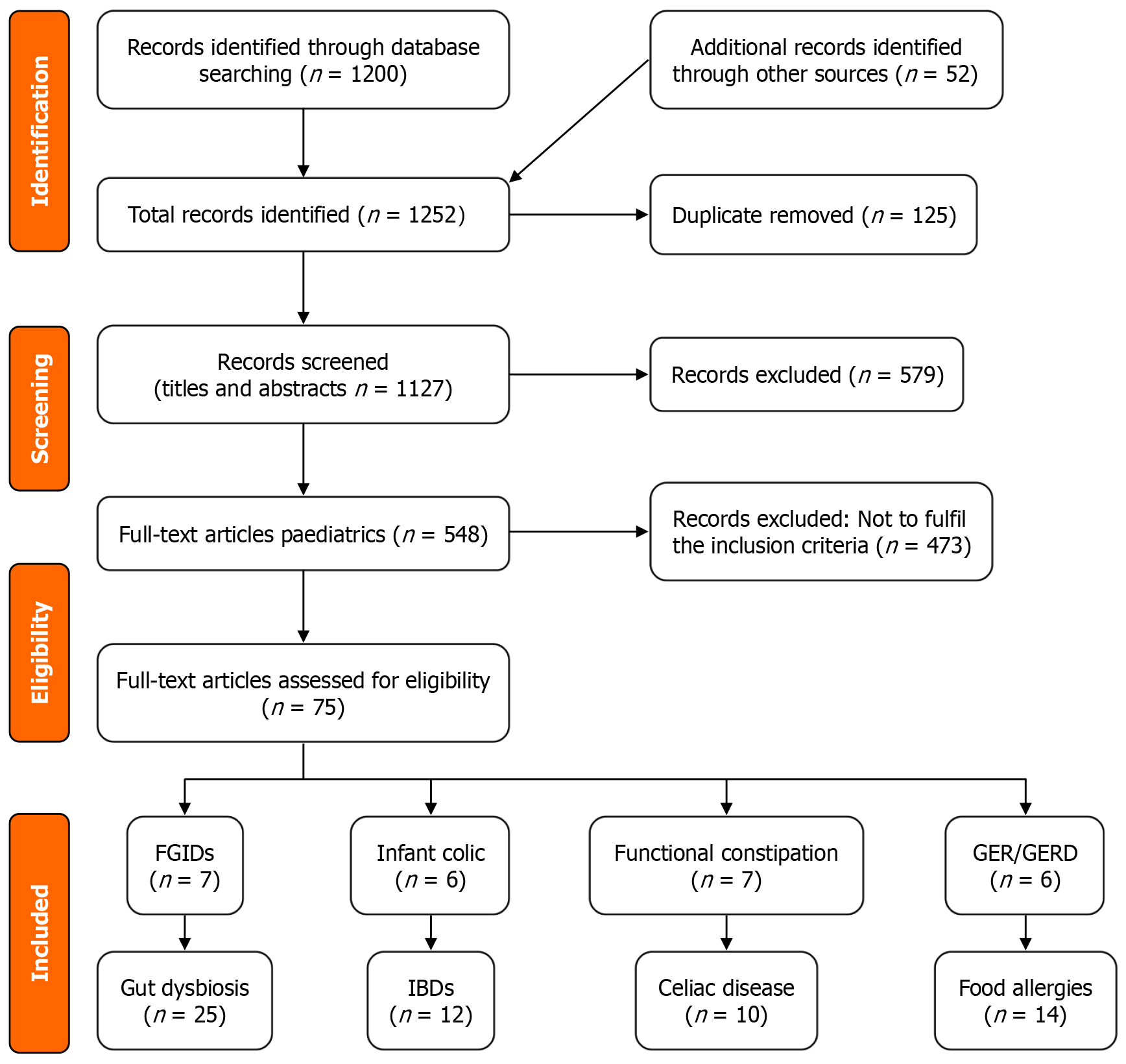
This systematic review was designed and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. The protocol for this review has been prospectively registered with the International Prospective Register of Systematic Reviews (Registration ID: CRD420251073280).
This review aims to investigate the relationship between C-section delivery and GI outcomes in infants. The review is guided by the Population, Intervention, Comparison, Outcome framework (Table 1), where the Population (P) includes infants and young children born via C-section; the Intervention or Exposure (I) is delivery by C-section (elective or emergency); the Comparison (C) group includes infants delivered vaginally; and the Outcomes (O) include both short- and long-term GI effects, such as infantile colic, constipation, gastroesophageal reflux, altered gut microbiota (dysbiosis), FGIDs, food allergies, and IBD.
| PICO element | Description |
| Research question | What are the short- and long-term gastrointestinal effects in infants born via C-section compared to those born via vaginal delivery? |
| Population (P) | Infants born via C-section (elective or emergency), from term or preterm gestations, across various geographic and ethnic backgrounds |
| Intervention (I) | Birth by C-section (elective or emergency) |
| Comparison (C) | Infants born via vaginal delivery (with or without labor) |
| Outcome (O) | Short- and long-term gastrointestinal outcomes, including gut dysbiosis, functional gastrointestinal disorders, infantile colic, constipation, gastroesophageal reflux, inflammatory bowel disease, food allergies (including cow’s milk protein allergy), and coeliac disease |
| Study design | Systematic review of observational studies (cohort, case-control, cross-sectional), randomized controlled trials, and relevant systematic reviews/meta-analyses |
| Other | Inclusion: Human studies, studies reporting delivery mode and gastrointestinal outcomes, studies with clearly defined outcome measures, and English language. Exclusion: Case reports, animal studies, conference abstracts without full data, studies not distinguishing C-section from vaginal delivery or lacking gastrointestinal outcomes |
Studies were eligible for inclusion if they met the following criteria: (1) Involve human infants or children delivered by C-section (elective or emergency), with or without a comparator group delivered vaginally; (2) Reported on one or more GI outcomes—either short-term (e.g., colic, constipation, gastroesophageal reflux, gut dysbiosis) or long-term (e.g., food allergies, FGIDs, IBD); (3) Were original research articles, including randomized controlled trials (RCTs), cohort studies, or case-control studies; and (4) Were published in English within the past 20 years. However, while the primary focus was on studies from the past 20 years, landmark studies from prior periods that provided foundational insights into microbiota colonization or long-term GI outcomes were also considered. Exclusion criteria included animal studies, narrative reviews, editorials, case reports, conference abstracts, and studies without a clearly defined delivery mode or GI outcome. Studies for which full-text access was unavailable were also excluded.
A comprehensive literature search was performed across multiple databases, including PubMed (MEDLINE), EMBASE, Scopus, Web of Science, and the Cochrane Library. The search used both controlled vocabulary (e.g., MeSH terms) and free-text keywords. Terms included combinations such as: “Cesarean section” OR “C-section” OR “Cesarean delivery”; “vaginal delivery”; “infant” OR “newborn”; “gastrointestinal” OR “colic” OR “constipation” OR “reflux” OR “dysbiosis” OR “gut microbiota” OR “inflammatory bowel disease” OR “functional gastrointestinal disorder” OR “food allergy”. Reference lists of all included articles and relevant reviews were manually searched to identify additional eligible studies. All references were managed using citation software (e.g., EndNote or Zotero), and duplicates will be removed before screening.
Two reviewers were independently screened to review the titles and abstracts of all retrieved articles. Studies deemed potentially eligible underwent a full-text review based on the inclusion criteria. Discrepancies between reviewers were resolved through discussion and consensus, with the involvement of a third reviewer if necessary. A PRISMA 2020 flow diagram was used to document the selection process (Figure 1).
Data was extracted independently by two reviewers using a standardized data extraction form. Information collected included: Study design, author(s), year of publication, country, sample size, population characteristics (e.g., gestational age, feeding method, sex), delivery mode (elective or emergency Cesarean vs. vaginal), GI outcomes, follow-up duration, and key results.
To support more granular analysis, modifying variables were extracted where available. These included: Type of C-section (elective vs emergency); Timing (pre-labor vs during labor); Feeding type (exclusive breastfeeding, formula, or mixed feeding); Antibiotic exposure (maternal intrapartum or neonatal); Probiotic or synbiotic supplementation during infancy.
Two independent reviewers assessed the quality of included studies. Observational studies were evaluated using the Newcastle-Ottawa Scale (NOS), which assesses the domains of selection, comparability, and outcome/exposure. Randomized controlled trials were assessed using the Cochrane Risk of Bias 2.0 (RoB 2) tool. Discrepancies were resolved by discussion or consultation with a third reviewer.
A narrative synthesis was conducted to summarize findings from all eligible studies, stratified into short-term and long-term GI outcomes. When appropriate, and where studies were sufficiently homogeneous in design and outcome mea
Subgroup analyses were conducted to examine the potential modifying effects of: Type of C-section (elective vs emergency); Feeding practices (exclusive breastfeeding vs formula); Perinatal antibiotic exposure; Use of probiotic or synbiotic interventions. Sensitivity analyses were also conducted to assess the robustness of pooled results by excluding studies with a high risk of bias or extreme outcomes.
Multiple studies have shown that infants born via C-section exhibit a higher incidence of FGIDs compared to those delivered vaginally (Table 2). For instance, Salvatore et al[11] demonstrated that C-section was associated with a 1.23-fold increased risk of infantile colic (P = 0.003), a 1.20-fold increase in dyschezia (P = 0.053), and a 1.14-fold overall increase in the risk of having at least one FGID (P < 0.001). Similarly, Bi et al[12] reported that 73.4% of preterm infants experienced at least one FGID, with C-section being a significant contributing factor. The link between C-section and GI symptoms was further strengthened by findings of altered gut microbiota, with lower levels of protective species, such as Bifidobacteria and Lactobacilli, and higher colonization by opportunistic Proteobacteria, especially Escherichia coli and Klebsiella[13].
| Ref. | Sample size | FGID evaluated | C-section association | Statistical significance | Risk of bias |
| Salvatore et al[11], 2019 | 934 | Colic, regurgitation, dyschezia, constipation | ↑ Colic (RR: 1.23), ↑ dyschezia (RR: 1.20), ↑ any FGID (RR: 1.14) | Significant (P < 0.01 for all) | Low (well-powered, defined FGIDs, adjusted) |
| Bi et al[12], 2023 | 988 (preterm) | Colic, regurgitation, dyschezia, constipation | 73.4% had ≥ 1 FGID; C-section strongly associated | Significant (P < 0.001) | Moderate (preterm bias, but strong stats) |
| Pantazi et al[13], 2025 | 134 | FGID + gut microbiota profile | C-section → ↑ dysbiosis, ↓ Bifidobacterium, ↑ E. coli | Significant (P < 0.05 for key microbes) | Moderate (small sample, integrated analysis) |
| Ziętek et al[14], 2024 | 82 | Colic, regurgitation, constipation | ↑ Regurgitation in C-section-born at 3-6 months | Significant (P = 0.0434) | High (small cohort, limited follow-up) |
| Bekem et al[15], 2021 | 213 | Dyschezia, regurgitation | ↑ Dyschezia (C-section: 83.3%), ↑ regurgitation (C-section: 88.9%) | Significant (P = 0.006 and 0.035) | Moderate (retrospective design) |
| Velasco-Benitez et al[16], 2020 | 1497 | School-age FGIDs (longitudinal) | No significant C-section association in older children | Non-significant (P > 0.05) | Low (large cohort, age-stratified, well-controlled) |
The impact of C-section on early-life GI health is often compounded by associated modifiers, including prematurity, formula feeding, neonatal antibiotic exposure, and maternal factors (e.g., postpartum depression). In addition, longitudinal cohort studies have found that GI symptoms, such as regurgitation and dyschezia, were significantly more common in C-section infants at various time points up to 12 months of age[14,15].
Despite some contradictory evidence (e.g., Velasco-Benitez et al[16]), the weight of the literature supports that C-section plays a notable role in the early development of FGIDs, primarily through microbiome disruption and altered immunological signaling. These short-term GI effects, although transient in some cases, may contribute to discomfort, increased healthcare visits, and prolonged reliance on formula feeding or probiotics.
Infant colic: Six studies were analyzed, encompassing a range of designs (prospective cohorts, case-control studies, and cross-sectional studies) from diverse settings (Table 3). These studies consistently demonstrated that C-section delivery is associated with an increased incidence of infantile colic and other FGIDs, especially in the first few months of life. Additional modifiers such as feeding type, antibiotic exposure, prematurity, and maternal psychological status were also evaluated.
| Ref. | Design & number | C-section vs VD colic prevalence | Main modifiers evaluated | Key findings | Risk of bias |
| Sommermeyer et al[17], 2022 | Cross-sectional, 195 infants | Higher fecal calprotectin in C-section colic | Type of feeding, birth mode | C-section infants had higher calprotectin & lower diagnostic accuracy for IC | Moderate (small sample, limited confounder control) |
| Salvatore et al[11], 2019 | Prospective cohort, 934 infants | C-section: RR 1.23 for IC | Preterm, antibiotics, formula feeding | Strong C-section association with IC; antibiotics further increased the risk | Low (good sample, adjusted) |
| Bi et al[12], 2023 | Retrospective, 988 (preterm) | Higher FGIDs (incl. IC) in C-section | Breastfeeding, smoking, hospitalization, and antibiotics | C-section is significantly associated with IC (P < 0.001) | Moderate (preterm only; confounders considered) |
| Ziętek et al[14], 2024 | Prospective, 82 infants | ↑ Colic at 3 months; ↑ regurgitation in C-section | Antibiotics, feeding pattern | GI disorders more frequent in C-section group | High (small size, short follow-up) |
| Akman et al[18], 2006 | Prospective, 78 infants | IC in 64.7% of C-section infants | Maternal depression, attachment style | C-section, depression, and insecure attachment linked with IC | High (tiny cohort, potential bias) |
| Bekem et al[15], 2021 | Cross-sectional, 213 infants | Infant dyschezia higher in C-section (P = 0.006) | Breastfeeding, maternal QoL, depression | IC more common in depressed mothers; C-section linked to GI symptoms | Moderate (retrospective; subjective outcomes) |
Sommermeyer et al[17] demonstrated that infants delivered by C-section exhibited significantly higher fecal calprotectin levels—an inflammatory marker—particularly in those experiencing colicky symptoms (≥ 100 μg/g). Interestingly, elevated calprotectin was also observed in C-section-born infants without colic, likely reflecting early gut dysbiosis caused by the absence of vaginal microbial exposure. Moreover, the combination of fecal calprotectin levels and delivery mode improved diagnostic accuracy for colic in vaginally born infants (97.8%), but was less predictive for those born via C-section (76.5%). Salvatore et al[11] reported that infantile colic affected 47.4% of infants overall, with a significantly higher incidence in infants delivered by C-section (relative risk: 1.23; 95%CI: 1.07-1.40; P = 0.003). The study also identified other contributing factors to colic, including preterm birth, neonatal antibiotic exposure, exclusive formula feeding, and prolonged hospital stays, all of which commonly co-occur with C-section deliveries.
In a large retrospective study of 988 preterm infants, Bi et al[12] found that 45.4% of the cohort had infantile colic and that C-section was a significant risk factor for all forms of FGIDs, including colic (P < 0.001). This association underscores the compounded vulnerability of preterm, C-section-delivered infants. Ziętek et al[14] investigated GI symptoms in relation to delivery mode. They noted that while most symptoms did not differ significantly by birth mode, regurgitation was notably more frequent in C-section infants (P = 0.0434). Colic was observed to peak at around 3 months of age. It was statistically linked to prior antibiotic exposure and microbiota imbalance, both of which are more prevalent in C-section-delivered infants.
Earlier, Akman et al[18] reported a colic prevalence of 21.7% in their sample, with 64.7% of colicky infants delivered by C-section compared to 59% among non-colicky infants. Although the difference was modest, maternal psychosocial factors such as depression and insecure attachment styles were significantly correlated with the presence of colic, suggesting an interaction between biological and environmental influences. Finally, Bekem et al[15] observed a significantly higher incidence of infantile dyschezia in C-section infants (83.3%, P = 0.006), with a non-significant trend indicating a higher occurrence of colic in infants born to mothers with postpartum depression (P = 0.057). While not directly confirming a causal link, the findings support the broader hypothesis that C-section delivery—especially when accompanied by maternal psychological stress—may predispose infants to GI dysregulation. From the previous studies, we have strong evidence supporting a link between C-section and an increased risk of infant colic, particularly when compounded by other risk factors such as lack of breastfeeding, neonatal antibiotics, and maternal depression (Table 4). These associations may be mediated by altered gut microbiota, increased gut inflammation (e.g., elevated fecal calprotectin levels), and psychosocial stressors.
| Modifier | Effect on infant colic |
| Cesarean section delivery | Consistently associated with a higher risk of colic across most studies |
| Neonatal antibiotic use | Associated with increased gut dysbiosis, leading to higher colic risk |
| Feeding type | Formula feeding and a lack of exclusive breastfeeding increased the risk |
| Maternal psychological status | Maternal postpartum depression and insecure attachment increased colic incidence |
| Prematurity | Associated with a higher prevalence of IC and other FGIDs |
| Probiotics | Mixed results—some studies suggest probiotics may worsen symptoms like constipation |
Constipation: Multiple studies in literature have evaluated the relationship between C-section and the development of functional constipation (FC) in infancy and early childhood (Table 5). The findings, while somewhat heterogeneous, generally indicate a mild to moderate association between C-section birth and an increased risk of constipation, especially when compounded by additional factors like preterm birth, formula feeding, or antibiotic exposure.
| Ref. | Sample size | Age evaluated | Prevalence of FC | C-section vs VD comparison | Significance | Modifiers | Risk of bias |
| Yoshida et al[6], 2018 | 83019 | 1 year | 1.37% | No significant difference (AOR = 0.94) | No | Breastfeeding, GA, BW | Low (large national dataset; adjusted) |
| Nakamura et al[19], 2021 | 71878 | 3 years | 12.3% | Higher in C-section (13.1% vs 12.1%; AOR = 1.064) | Yes | Breastfeeding, solid food, maternal BMI | Low (population-based; confounders addressed) |
| Bi et al[12], 2023 | 988 (preterm) | 0-12 months | 19.2% | Strong C-section association (67.1% vs 40.3%) | Yes | Antibiotics, FF, maternal smoking | Moderate (limited to preterms; good stats) |
| Ziętek et al[14], 2024 | 82 | 3, 6, 12 months | Not isolated | C-section is linked to regurgitation only | No | Probiotics ↑ risk (P = 0.045) | High (small sample; limited power) |
| Salvatore et al[11], 2019 | 934 | 0-12 months | 26.6% | C-section linked to dyschezia, not FC | No | Preterm, antibiotics, FF | Low (good design and adjustment) |
| Neves et al[20], 2023 | 135 | 12 months | 49.6% | C-section infants had more FC, but not significant | No | Feeding method (PLW) | Moderate (small sample; not powered) |
| Hierink et al[21], 2024 | 2643 (mothers) | Adults | 24.6% maternal constipation | More common in VD mothers | Yes | Perineal trauma | Moderate (maternal outcome; subjective reporting) |
One of the largest studies, the Japan Environment and Children's Study, showed no significant association between C-section and constipation at age 1 year[6], but a subsequent follow-up at 3 years[19] revealed a statistically significant but modest increase in FC among C-section-delivered children (13.1% vs 12.1%; adjusted OR = 1.064). This finding suggests that the GI effects of C-section may manifest more clearly beyond infancy, potentially due to persistent alterations in gut microbiota maturation or immune signaling. A strong and early association between C-section and FC was demonstrated by Bi et al[12] in a cohort of 988 preterm infants, where 19.2% developed constipation within the first year, and C-section was a significant risk factor (P < 0.001). This link was compounded by other known risk factors such as neonatal antibiotic exposure, formula feeding, and maternal smoking, highlighting a multifactorial pathway leading to FGIDs in vulnerable populations.
Other studies, such as those by Salvatore et al[11] and Ziętek et al[14], did not find a direct significant relationship between C-section and constipation but did report related FGIDs—such as regurgitation and dyschezia—more frequently among C-section-born infants. Ziętek et al[14] further noted an unexpected finding: Infants given probiotics showed a statistically significant increase in constipation at 3 months of age, suggesting a need to better understand strain-specific effects of probiotics in early life. While Neves et al[20] reported a high overall prevalence of constipation (49.6%) in 12-month-old infants, C-section was not a statistically significant factor, though a higher proportion of C-section infants were constipated. Their results suggest that complementary feeding methods (e.g., baby-led vs parent-led weaning) may contribute more prominently to constipation development than delivery mode alone.
Interestingly, a different angle was explored by Hierink et al[21], who studied maternal outcomes and found that vaginal delivery was associated with a higher prevalence of maternal constipation, likely due to pelvic floor trauma. While this does not directly relate to infant GI outcomes, it supports the idea that mode of delivery can differentially influence GI health across the lifespan. Taken together, these findings suggest that while C-section may be a moderate independent risk factor for functional constipation in infants and toddlers, its impact is most pronounced in populations exposed to additional modifiers such as preterm birth, formula feeding, lack of exclusive breastfeeding, and perinatal antibiotic exposure (Table 6). The variability in results also reflects potential differences in outcome definitions, follow-up periods, and cultural or healthcare-related feeding practices.
| Modifier | Effect on constipation | Ref. | Notes |
| Feeding type | Formula feeding associated with higher risk; exclusive breastfeeding appears protective | Bi et al[12], 2023; Salvatore et al[11], 2019; Yoshida et al[6], 2018 | Formula-fed C-section infants are more prone to delayed gut transit and reduced microbiota diversity |
| Antibiotic exposure | Increased constipation risk via microbiota disruption (in C-section-born, especially) | Bi et al[11], 2023; Salvatore et al[10], 2019 | Antibiotics delay colonization by beneficial bacteria such as Bifidobacteria |
| Prematurity | Preterm C-section infants have a higher risk of functional GI disorders, including constipation | Bi et al[12], 2023; Salvatore et al[11], 2019 | May reflect an immature enteric nervous system and altered motility |
| Probiotic use | Unexpectedly associated with increased constipation at 3 months | Ziętek et al[14], 2024 | Possibly due to strain-specific effects; more research needed |
| Complementary feeding practices | No significant effect overall, but trends show PLW may slightly increase risk | Neves et al[20], 2023 | Cultural and dietary factors may influence stool patterns |
| Maternal depression/smoking | Associated with increased GI symptoms and constipation in C-section-born infants | Bi et al[12], 2023; Bekem et al[15], 2021 | May affect breastfeeding success and infant regulatory behavior |
| Age of evaluation | C-section-related constipation was not apparent at 1 year but was observed at 3 years | Yoshida et al[6], 2018; Nakamura et al[19], 2021 | Suggests delayed or progressive impact of delivery mode on GI function |
Gastroesophageal reflux: Multiple studies included in this review provide converging evidence that C-section delivery is associated with an increased risk of gastroesophageal reflux (GER) and its pathological form, gastroesophageal reflux disease (GERD), during infancy. The evidence points to a multifactorial origin linking C-section birth to reflux symptoms, primarily mediated by microbiome dysbiosis, immaturity of the GI system, neonatal interventions, and maternal factors such as psychiatric comorbidities.
In a large population-based cohort study by Dahlen et al[22], infants born via C-section were significantly more likely to be diagnosed with GER/GERD (37.2%) compared to vaginally delivered infants (28.4%). Notably, infants admitted with GER/GERD also had a higher incidence of comorbidities such as feeding difficulties, excessive crying, and sleep disorders. Multivariate analysis confirmed C-section as a significant risk factor for GER/GERD (AOR: 1.13; 95%CI: 1.08-1.78), alongside maternal psychiatric illness (AOR: 4.68), early gestation, male sex, and neonatal intensive care units (NICU) admission. Pantazi et al[13] analyzed gut microbiota profiles in infants. They found that those with GER had a significant reduction in acidifying flora (e.g., Lactobacillus, Bifidobacterium, Enterococcus) and increased levels of Escherichia coli and Klebsiella, particularly in infants born by C-section. This microbial pattern, strongly correlated with dysbiosis, likely contributes to GER pathophysiology.
Salvatore et al[11] showed that 40% of infants had regurgitation during their first year, with C-section being a moderate risk factor. Bi et al[12] further corroborated this by demonstrating that among preterm infants, C-section significantly increased the odds of GER. Infants with low birth weight, prolonged hospitalization, antibiotic exposure, and absence of breastfeeding were especially vulnerable. Ziętek et al[14] observed that regurgitation was significantly more common in C-section-born infants (P = 0.0434), peaking at three months of age, often coexisting with colic and constipation. Formula feeding and antibiotic use were highlighted as critical modifiers. Interestingly, Guo et al[23] proposed an interventional approach to mitigate GER risk by manually evacuating gastric contents immediately after C-section delivery, reporting no cases of GER or aspiration in their cohort.
The studies highlight several modifiable risk factors for GERD in C-section-born infants, such as gestational age and birth weight, feeding pattern, neonatal antibiotic exposure, NICU admission and medicalization, and possible maternal factors. Prematurity and low birth weight magnify vulnerability to reflux. NICU Admission and medicalization are more frequent in C-section-born infants and are correlated with reflux symptoms. Exclusive formula feeding and lack of early breastfeeding are also associated with increased reflux risk. Neonatal antibiotic exposure can disrupt gut microbiota and is consistently linked with higher GER incidence. Maternal psychiatric illness (especially anxiety and depression) and primiparity are strongly associated with infant GER/GERD diagnoses.
Across all included studies, C-section delivery emerges as a consistent risk factor for early-onset gastroesophageal reflux in infants. Pathogenesis appears to be multifactorial, involving immune immaturity, disturbed microbiota ac
| Ref. | Population | Key findings | C-section association | Modifiable factors | Risk of bias |
| Dahlen et al[22], 2018 | 869188 infants | 1.1% diagnosed with GER/GERD; higher in C-section births | AOR: 1.13 (P < 0.001) | Maternal psychiatric illness, prematurity, NICU stay | Low (large national dataset, adjusted for multiple confounders) |
| Pantazi et al[13], 2025 | 134 infants | Reduced Lactobacillus and Bifidobacterium in GER infants; ↑ E. coli | GER strongly linked with C-section and dysbiosis | Artificial feeding, C-section, antibiotic use | Moderate (small cohort, microbial focus, controlled analysis) |
| Ziętek et al[14], 2024 | 82-term infants | Higher regurgitation in C-section group (P = 0.0434) | Significant increase in GER symptoms | Formula feeding, antibiotic use | High (small sample size, limited adjustment) |
| Salvatore et al[11], 2019 | 934 infants | 40% had regurgitation in year one; C-section increased FGID risk | Associated with dyschezia and diarrhea | Exclusive formula, prolonged hospital stays | Low (well-designed, prospective, controlled) |
| Bi et al[12], 2023 | 988 preterm infants | 41.6% had GER; C-section among strongest risk factors | Yes (χ2 = 33.13, P < 0.001) | Lack of breastfeeding, antibiotic/probiotic use | Moderate (preterm focus, retrospective, good confounder control) |
| Guo et al[23], 2002 | 496 neonates (C-section only) | GER/aspiration prevented with manual intervention | None reported | Early postnatal intervention | High (no comparison group, limited external validity) |
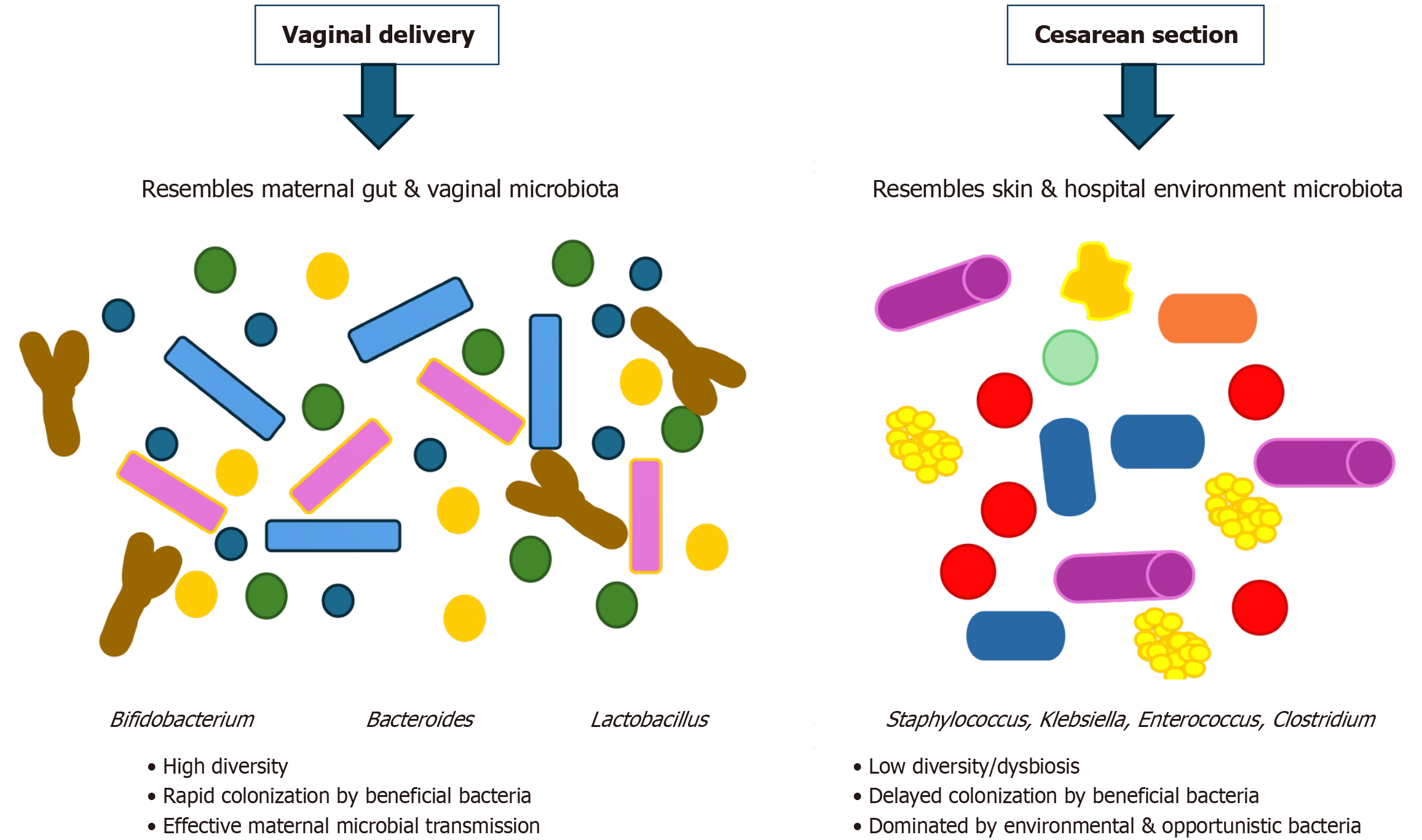
Gut dysbiosis: Infants born via C-section experience distinct microbial colonization patterns compared to those born vaginally, often characterized by lower microbial diversity, a reduced abundance of Bifidobacterium and Bacteroides, and an increased prevalence of opportunistic pathogens, such as Enterobacteriaceae. A consistent finding across the included studies is that C-section delivery significantly alters the early development of the infant gut microbiota compared to vaginal delivery. Infants born via vaginal delivery are typically colonized with beneficial maternal vaginal and intestinal microbes such as Bacteroides, Lactobacillus, and Bifidobacterium. In contrast, C-section-born infants exhibit delayed microbial succession and reduced diversity, often characterized by colonization with environmental and hospital-associated organisms, including Klebsiella, Staphylococcus, Clostridium, and Enterococcus (Tables 8 and 9).
| Feature | Vaginal delivery | C-section |
| Initial colonizers | Lactobacillus, Bifidobacterium, Bacteroides | Enterococcus, Staphylococcus, Klebsiella |
| Microbial diversity | High | Reduced |
| Commensal dominance | Yes | Delayed or absent |
| Opportunistic pathogen presence | Low | High (esp. hospital-acquired) |
| Persistence of differences | Normalize by 6-12 months | May persist > 12 months |
| Transmission from mother | Effective vertical transmission | Severely reduced; environmental transmission |
| Modifiable by breastfeeding | Enhanced commensal colonization | Partially restores microbial balance |
| Effect of Intrapartum Antibiotics | Reduces vertical transmission in VD | Present in all C-section deliveries |
| Racial/geographic variation | Present (e.g., Japanese, Korean, United Kingdom cohorts) | Yes, with consistent patterns across populations |
| Corrective interventions | Less needed | FMT, probiotics, breastfeeding are critical |
| Ref. | Country/population | Key findings | Persistence of difference | Modifiable factors examined | Risk of bias |
| Nagpal et al[27], 2017 | Japan/Healthy full-term infants | VD enriched with Lactobacillus, Bacteroides; C-section infants had reduced colonization | Seen up to 3-6 months | Not specified | Moderate (small sample, unclear confounder control) |
| Kim et al[25], 2021 | Korea/National cohort | C-section associated with lower Bifidobacteria, delayed microbiota maturation | Persisted into infancy (12 months) | Breastfeeding | Low (large cohort, confounders adjusted) |
| Reyman et al[28], 2019 | Netherlands/n = 117 | C-section infants had low species richness, delayed Bacteroides | Up to 6 months | Antibiotics, feeding method | Moderate (small sample, well-defined methods) |
| Shao et al[24], 2019 | United Kingdom/Baby Biome Study | C-section disrupted maternal strain transmission; ↑ Klebsiella, Enterococcus | Persistent at 1 year | Antibiotics, feeding, maternal FMT | Low (large, multicenter, robust metagenomics) |
| Mitchell et al[29], 2020 | United States/Israel/Multiethnic cohort | Bacteroides absent in C-section infants for weeks; antibiotic-sensitive | 6-12 weeks | Antibiotics, breastfeeding | Moderate (multi-site, antibiotic exposure varied) |
| Bäckhed et al[26], 2015 | Sweden/n = 98 | VD infants dominated by Bifidobacterium, Bacteroides; C-section → ↑ Clostridium | Up to 3-4 months | Breastfeeding | Moderate (well-designed but small sample) |
| Dos Santos et al[33], 2023 | Canada/National cohort | C-section disrupted diversity; ↑ Proteobacteria in early life | Up to 12 months | Hospital stay, feeding type | Low (population-wide registry, controlled) |
| Dominguez-Bello et al[34], 2010 | United States/Pilot cohort | VD infants resembled vaginal flora; C-section resembled skin/hospital flora | Neonatal period | FMT, vaginal seeding | High (pilot study, small sample, proof-of-concept only) |
Shao et al[24] from the UK Baby Biome Study found that C-section delivery disrupted the transmission of maternal microbial strains and led to persistent colonization by potentially pathogenic species up to one year of age. Similarly, Kim et al[25] observed lower abundances of Bifidobacteria and Lachnospiraceae in C-section infants in a Korean cohort, with delayed microbiota maturation persisting throughout infancy. In Sweden, Bäckhed et al[26] noted that infants delivered vaginally had earlier colonization with Bacteroides and Bifidobacterium. In contrast, C-section infants exhibited higher levels of Clostridium species for up to four months.
Other studies further supported this trend. Nagpal et al[27] in Japan reported that vaginal delivery infants were enriched with Lactobacillus and Bacteroides, both of which were nearly absent in C-section infants during the first 3-6 months. In the Netherlands, Reyman et al[28] found that C-section infants had significantly lower species richness and delayed colonization by Bacteroides, with notable differences observed up to six months after birth. Mitchell et al[29] observed in a multiethnic cohort from the United States and Israel that Bacteroides was absent for several weeks post-C-section and that these effects were amplified by antibiotic exposure. The microbial differences between delivery modes are not merely taxonomic; they have functional consequences for infant health. Bifidobacterium and Bacteroides species, which are abundant in vaginally delivered infants, are crucial for digesting human milk oligosaccharides, producing short-chain fatty acids that nourish gut cells, and promoting an anti-inflammatory environment. They play a key role in promoting immune tolerance and protecting gut barrier function[30]. Their depletion in C-section-born infants has been linked to increased susceptibility to food allergies and inflammatory bowel diseases (IBDs). Their scarcity in C-section-born infants can impair gut barrier function and immune education[31]. Conversely, the higher prevalence of op
Geographic and racial variations also influenced microbial profiles. For example, the Japanese and Korean cohorts emphasized the dominant role of Lactobacillus, whereas Western studies more frequently focused on Bacteroides depletion[25,27]. Despite these variations, the overall direction of the findings remained consistent: C-section delivery interrupts the natural maternal-to-infant microbial transmission and promotes a dysbiotic gut environment in early life. Notably, several studies examined modifiable factors. Breastfeeding has been repeatedly shown to partially restore the beneficial microbiota in C-section infants, particularly promoting Bifidobacterium dominance[25,26,29,33]. Intrapartum antibiotic exposure, which is widespread in C-section deliveries, further impairs microbiota development[24,28,33]. Additionally, maternal fecal microbiota transplantation and vaginal seeding were investigated as emerging interventions to mimic the microbial transfer that occurs during vaginal birth[24,34]. Collectively, these studies confirm that C-section has a mea
Multiple studies have evaluated various interventions, including vaginal seeding, synbiotics, probiotics, and maternal microbial transfer, to restore microbiota composition and diversity in infants born via C-section (Table 10). Studies by Dominguez-Bello et al[34] and Wilson et al[36] demonstrated partial restoration of microbiota in C-section-born infants when exposed to maternal vaginal fluids. While the oral administration of maternal vaginal microbes showed minimal effects on gut composition, traditional swabbing techniques resulted in a closer resemblance to vaginally born infants, especially at skin and oral sites[37]. Mueller et al[38], added that vaginal seeding enhances maternal microbial engra
| Ref. | Intervention | Microbiota composition | Microbial diversity | Duration of effect | Modifiable factors | Population | Risk of bias |
| Song et al[50], 2021 | Vaginal seeding (swab) | Aligned with vaginally born | Improved | Up to 12 months | Maternal microbiota | United States, Hispanic/Latino | Moderate (small cohort, no randomization) |
| Wilson et al[36], 2021 | Oral vaginal seeding | No significant effect | No change | Up to 3 months | Administration method | New Zealand | High (pilot nature, small sample, no control) |
| Mueller et al[38], 2023 | Vaginal seeding (RCT) | ↑ Maternal strains | ↓ Alpha-diversity | 1 month | Delivery timing | United States | Low (RCT, good design) |
| Lagkouvardos et al[37], 2023 | Synbiotic formula (L. fermentum + GOS) | ↑ Bifidobacterium, ↓ Blautia | ↑ at 4 months | Up to 24 months | Baseline microbiota | European | Low (long follow-up, intervention control) |
| Chua et al[39], 2017 | Synbiotic (scGOS/LcFOS + B. breve) | ↑ Bifidobacteria, ↓ Enterobacteriaceae | Improved by week 8 | 12 weeks post | Feeding method | Multinational (Asia) | Low (RCT, multinational) |
| Wang et al[40], 2025 | Synbiotic (B. breve + scGOS/LcFOS) | ↑ Parabacteroides, Bacteroides | ↑ Diversity | Up to 12 months | Feeding, birth mode | China | Low (RCT design, stratified by C-section) |
| Garcia Rodenas et al[41], 2016 | L. reuteri-enriched formula | Modulated toward VD profile | ↑ Diversity | 4 months | Strain selection | Greece | Moderate (no blinding stated) |
| Yang et al[42], 2021 | Probiotics (varied dose) | ↑ Bifidobacterium, Lactobacillus | ↑ To VD levels | 28 days | Dosage | China | Moderate (dose variability, controlled) |
| Gong et al[43], 2023 | Probiotics (B. longum, L. acidophilus) | ↑ Faecalibacterium, ↓ Klebsiella | Improved | 42 days | Antibiotic exposure | China | Moderate (strain-specific findings) |
| Hurkala et al[44], 2020 | Probiotics (B. breve, L. rhamnosus) | ↑ LAB, ↓ pathogens | Significant | 30 days | Early start post-C-section | Poland | Moderate (observational, small sample) |
| Akagawa et al[45], 2019 | Breastfeeding vs formula | Restoration by 1 month | Equalized | 1 month | Feeding type | Japan | Moderate (observational, controlled) |
| Wu et al[46], 2023 | Breastfeeding in C-section infants | ↑ SCFA metabolism stability | Enhanced | 3 months | Feeding type | China | Low (well-controlled feeding study) |
| Korpela et al[47], 2018 | Probiotic mixture (4 strains) | Corrected dysbiosis in C-section infants | Improved | 3 months | Breastfeeding | Finland | Low (strong probiotic trial, adjusted) |
| Bellomo et al[48], 2024 | B. bifidum only | ↑ Bacteroides, ↓ Shigella, ↑ α-diversity | Sustained ↑ | 12 months | Feeding mode | Italy | Low (well-controlled, single strain tested) |
| Yang et al[49], 2025 | Bovine colostrum vs fortifier | Minor changes | No significant change | 1 month | Fortifier type | Denmark | Moderate (neutral result, no microbiome endpoint) |
| Dominguez-Bello et al[51], 2016 | Vaginal swabbing | Partial restoration (Bacteroides) | Mild increase | 30 days | Swabbing technique | United States, multiethnic | High (pilot nature, small number) |
Clinical trials consistently showed that synbiotic formulas—especially those containing Bifidobacterium breve M-16V and oligosaccharides—promote early Bifidobacterium colonization, lower fecal pH, and emulate gut profiles of breastfed or vaginally delivered infants[39-41]. These effects persisted up to 12 months in some studies. Synbiotics were particularly effective in increasing microbial diversity in infants born via C-section. Several studies have highlighted that targeted probiotic supplementation—specifically, Lactobacillus reuteri, Bifidobacterium bifidum, Bifidobacterium breve, and Lactobacillus rhamnosus—can partially modulate the gut microbiota, increase beneficial bacterial populations, and decrease pathogens such as Klebsiella and Clostridium. However, efficacy varied based on strain and dosage[42-45]. Feeding method (breastfeeding vs formula)[46-49], racial background, hospital practices (e.g., probiotic use), colostrum use[50], and maternal factors (e.g., antibiotic use or probiotic use)[36] significantly modified the efficacy of the intervention. Racial and geo
Synbiotics combining prebiotics and specific Bifidobacterium strains showed the most sustained and significant effects on microbiota restoration and diversity, particularly in C-section-born infants. Vaginal seeding showed partial res
Chronic inflammatory GI conditions in childhood: Numerous large-scale, population-based studies across different countries have examined the link between C-section and the long-term risk of IBD, including Crohn’s disease (CD) and ulcerative colitis (UC) (Table 10). The findings are mixed, with some studies reporting a modest but statistically sig
Studies that support an increased risk of IBDs following C-section: Several large-scale studies have reported a positive association between C-section and the long-term risk of developing IBD, particularly CD (Table 11). Bager et al[52] analyzed a Danish cohort of 2.1 million individuals with up to 35 years of follow-up and found that C-section was associated with a moderate increase in IBD risk before the age of 15 (incidence rate ratio = 1.29; 95%CI: 1.11-1.49), independent of family history. Similarly, Andersen et al[53] examined nearly 2.7 million births in Denmark over 40 years. They reported an increased risk of IBD and other chronic inflammatory diseases in C-section-born individuals [hazard ratio (HR) > 1.0], with both elective and emergency C-sections posing similar levels of risk. Hellsing et al[54] from Sweden, in a cohort of over one million individuals, specifically linked C-section to an elevated risk of CD (adjusted HR = 1.14; 95%CI: 1.02-1.27), although no such association was found with UC. In Israel, Zamstein et al[55] studied breech births and reported that C-section was associated with a significantly higher risk of IBD-related hospitalization (adjusted HR = 3.18; 95%CI: 1.47-6.87), suggesting that the context of delivery may influence long-term outcomes. Malmborg et al[56] also found a modest association in Sweden between C-section and pediatric CD, especially in males (OR = 1.25), with a stronger link noted in elective C-section cases (OR = 1.36). Supporting this trend, Ponsonby et al[57] in Australia observed that elective C-sections were associated with an increased risk of CD, part of a broader rise in incidence that is not fully explained by other perinatal factors. Collectively, these findings suggest that C-section—particularly when elective—may modestly elevate the risk of developing CD later in life.
| Ref. | Country | Sample size | Follow-up duration | Association with IBD | Key notes/modifiable factors | Risk of bias |
| Bager et al[52], 2012 | Denmark | 2.1 million | Up to 35 years | ↑ IBD (IRR: 1.29 for < 15 years) | Adjusted for family history | Low (large population, long-term, adjusted) |
| Andersen et al[53], 2020 | Denmark | 2.7 million | Up to 40 years | ↑ IBD and autoimmune diseases | Risk with both elective/emergency C-section | Low (excellent registry data, confounder control) |
| Hellsing et al[54], 2022 | Sweden | 1.1 million | 17 years | ↑ Crohn’s (HR: 1.14) | No effect on UC | Low (well-stratified outcomes, good adjustment) |
| Zamstein et al[55], 2022 | Israel | 7337 breech births | 18 years | ↑ IBD hospitalizations (aHR: 3.18) | Breech delivery as key variable | Moderate (small subgroup, well-controlled) |
| Malmborg et al[56], 2012 | Sweden | > 16000 | Not specified | ↑ Pediatric Crohn's (boys, OR: 1.25) | Elective C-section more strongly associated | Moderate (good design, duration unclear) |
| Ponsonby et al[57], 2009 | Australia | Not specified | Up to 16 years | ↑ Crohn's disease with elective C-section | Trend noted over time | Moderate (ecological component, unclear sample) |
| Bernstein et al[58], 2016 | Canada | 12159 | Approximately 20 years | No association | Controlled for siblings, urban/rural | Low (sibling matched, good design) |
| Burnett et al[59], 2020 | Canada | 262729 | Approximately 25 years | No association | Two independent cohorts analyzed | Low (large sample, replication) |
| Burgess et al[60], 2022 | Scotland | 2 million | 16 years | No association | Controlled for feeding, GA, mode | Low (robust dataset, adjusted) |
| Sonntag et al[61], 2007 | Germany | 1859 | Retrospective | No association | Preterm birth more influential | Moderate (small sample, retrospective) |
| Soullane et al[62], 2021 | Canada | 934873 | 7.4 years | No IBD association | Pediatric cohort focus | Low (strong national data, pediatric angle) |
Studies that support no increased risk of IBDs following C-section: On the other hands, several high-quality studies have reported no significant association between C-section delivery and the risk of developing IBD. Bernstein et al[58] conducted a matched cohort study involving over 12000 individuals in Canada. They found that C-section was not linked to IBD risk, even after adjusting for potential confounders and performing sibling comparisons. Similarly, Burnett et al[59] in Nova Scotia analyzed two large retrospective cohorts and reported no meaningful connection between C-section and the development of IBD during childhood or young adulthood. In Scotland, Burgess et al[60] conducted a population-based birth cohort study. They found no association between C-section and pediatric IBD, even after adjusting for important variables such as infant feeding method and prematurity. Likewise, Sonntag et al[61] in Germany identified that while preterm birth and early disease exposure were significant contributors to IBD risk, C-section delivery itself was not an independent factor. Supporting these findings, Soullane et al[62] analyzed a Canadian cohort of more than 900000 children and concluded that C-section was not significantly associated with autoimmune diseases, including IBD [HR = 1.15; 95% confidence interval (CI): 0.88-1.49]. Collectively, these studies suggest that C-section alone may not be a decisive factor in the development of IBD, particularly when other early-life influences are taken into account.
A synthesis of the available evidence reveals a consistent pattern of modestly increased risk for CD among children delivered by C-section, particularly when the procedure is elective and in male infants. This association has been reported in large-scale studies from Denmark[52,53], Sweden[54,56], Israel[55], and Australia[62]. In contrast, no strong or consistent link has been found between C-section and UC, which supports the hypothesis that CD may be more influenced by early-life microbial disruptions. The contradictory findings regarding the relationship between C-section and CD likely reflect a complex interplay of regional and population-specific factors. While several large cohort studies from Nordic countries and Israel[52-56] have reported a modest but significant increase in CD risk among C-section-born individuals, equally robust studies from Canada, Germany, and the United Kingdom have not found such associations[57-61]. These discrepancies may be attributed to differences in genetic susceptibility to IBD, baseline gut microbiota composition, and diagnostic practices. Additionally, variations in healthcare systems and clinical protocols—such as the frequency of elective vs emergency C-sections, intrapartum antibiotic use, and breastfeeding initiation rates—can act as confounding variables.
Geographic and racial variations appear to play a significant role in shaping these outcomes. Positive associations between C-section and IBD, especially CD, have been predominantly reported in Nordic and Israeli populations[47-51]. Meanwhile, studies from Canada and the United Kingdom tend to report null findings[58-60,62]. These discrepancies may stem from differences in obstetric practices (e.g., frequency of elective C-section), postnatal care, baseline microbiota composition, genetic susceptibility, and broader healthcare system factors. The length of follow-up also significantly influences findings. Studies with longer follow-up durations (≥ 20 years)[52-55] are more likely to detect cases of late-onset IBD that may otherwise be missed in shorter-term analyses[62].
Several modifiable factors have been identified that may interact with delivery mode to influence the risk of IBD. These include the type of C-section (elective vs emergency), with elective procedures generally posing a slightly higher risk. Infant feeding practices—particularly the absence of breastfeeding and reliance on formula—can alter the composition of the microbiota and affect immune development. Additionally, antibiotic exposure, both perinatal and during infancy, along with early-life infections, may confound or amplify the risk of developing IBD. Therefore, while the association between C-section and long-term risk of IBD remains inconclusive, it is biologically plausible and supported by several high-quality epidemiological studies. The link appears stronger for CD than for ulcerative colitis, likely due to microbiota-mediated mechanisms. However, the heterogeneity across studies in terms of findings, populations, and methodology suggests that C-section should not be considered an isolated risk factor but rather part of a complex, multifactorial pathway involving early immune programming and microbial colonization.
Celiac disease risk and C-section: Several large-scale retrospective and prospective studies from diverse geographic and ethnic populations have investigated the relationship between C-section and the development of celiac disease (Table 12). Some studies have reported a modest increase in celiac disease risk following C-section—particularly elective C-section without labor—while others have found no significant association. In a large Italian multicenter study, C-section was observed in 40% of children diagnosed with celiac disease, higher than the general population, and emergency C-section was associated with earlier celiac disease onset[63]. In Sweden, Mårild et al[64] found a significant association with elective C-section (aOR = 1.15), but not emergency C-section, supporting a microbiota-mediated mechanism. In contrast, large-scale registry studies from Norway and Denmark did not find a significant association[65,66]. Similarly, The Environmental Determinants for Diabetes in the Young (TEDDY) study, which involved HLA-positive children from the United States, Sweden, Finland, and Germany, initially showed a protective effect; however, after adjusting for confoun
| Ref. | Country | Sample size | Follow-up | Association with CD | Notable modifiers | Risk of bias |
| Andersen et al[53], 2020 | Denmark | 2.7 million births | 40 years | ↑ Risk of celiac disease and other inflammatory diseases | Parental autoimmune disease | Low (large national cohort, robust adjustments) |
| Soullane et al[62], 2021 | Canada | 934873 children | 7.4 years | No increased celiac disease risk (HR = 0.86) | Hospitalization data | Low (large cohort, good registry linkage) |
| Iorfida et al[63], 2024 | Italy | 3259 celiac disease patients | 40 years | ↑ C-section rate in celiac disease group; earlier onset in emergency C-section | Emergency vs elective C-section | Moderate (case-control; retrospective risk) |
| Mårild et al[64], 2012 | Sweden | 11749 cases; 53887 controls | 35 years | ↑ Risk with elective C-section (aOR = 1.15) | SGA status, labor exposure | Low (matched controls, well-powered) |
| Dydensborg Sander et al[65], 2018 | Denmark & Norway | Approximately 1.6 million births | 15-18 years | No significant association | Country-level consistency | Low (multinational, controlled) |
| Emilsson et al[66], 2015 | Norway | 114500 children | 8-10 years | No significant association | Maternal celiac disease, type 1 DM, gluten timing | Low (adjusted for strong confounders) |
| TEDDY Study (Koletzko et al[67], 2018) | United States, Europe | 6087 HLA-positive children | 5-10 years | No significant association after adjustments | Genetics, breastfeeding, country | Low (high-quality longitudinal cohort) |
| Tanpowpong et al[68], 2023 | United States | 44539 mother-child pairs | > 6 years | ↑ Risk in C-section without labor (aHR = 1.56) | Labor status at delivery | Moderate (good follow-up, stratified labor data) |
| Decker et al[69], 2010 | Germany | 1950 children | Retrospective | ↑ Celiac disease in C-section-born (OR = 1.8) | Breastfeeding, postnatal complications | Moderate (retrospective, smaller size) |
| Bielik et al[70], 2024 | Slovakia | 1226 (534 celiac disease patients) | Cross-sectional | No C-section-celiac disease link; breastfeeding protective | Antibiotics, family history | High (cross-sectional design, recall bias) |
Tanpowpong et al[68] reported a borderline significant risk of celiac disease (adjusted HR: 1.39, P = 0.06) in C-section births, with a higher risk when delivery occurred without labor (adjusted HR = 1.56, P = 0.046). Andersen et al[53] confirmed that C-section increased the long-term risk of several autoimmune diseases, including celiac disease, over a 40-year follow-up. However, studies from Germany and Slovakia echoed mixed results. The former reported a significantly higher celiac disease prevalence in C-section-born children (OR = 1.8). At the same time, the latter found no significant effect of C-section on celiac disease risk but confirmed the roles of antibiotics and lack of breastfeeding[69,70]. A Ca
The collective evidence suggests that C-section may slightly increase the risk of celiac disease, particularly when elective and without labor, likely due to altered neonatal microbiota colonization. However, the association is not consistent across populations, and the effect size is generally modest or borderline. Geographic variation plays a role—positive associations were more common in Italian, Swedish, German, and United States populations, while large cohorts from Canada, Denmark, and Norway reported no significant link. This discrepancy may stem from differences in clinical practices, genetics, microbiome diversity, and diagnostic strategies.
Key modifiable factors identified include Elective vs Emergency C-section, Absence of labor, Breastfeeding, and Antibiotic exposure. Elective C-section consistently shows stronger associations. There is also a higher celiac disease risk in children born via C-section without labor, while breastfeeding has a protective effect in multiple studies[66,71]. Additionally, early and frequent antibiotic use may increase the risk of celiac disease. Therefore, while some studies demonstrate a modest association between C-section—especially elective C-section without labor—and increased celiac disease risk, other robust datasets report no such link. The current body of evidence does not justify categorizing C-section as an independent risk factor for celiac disease. Instead, a C-section may act as one of several early-life exposures that influence immune and gut development. Future research should continue to explore how perinatal microbial modulation, feeding practices, and genetic predisposition interact in shaping long-term autoimmune outcomes like celiac disease.
A growing body of literature has explored the long-term risk of developing food allergies—including cow’s milk protein allergy (CMPA)—following C-section delivery (Table 13). Several studies suggest that C-sections may increase the risk of food allergies, particularly when elective and not accompanied by labor, likely due to alterations in early gut microbiota colonization and immune system maturation. For example, Papathoma et al[71] from Greece reported that C-section was associated with a more than threefold increase in food allergy risk (OR: 3.15), especially when combined with a parental history of atopy. Similarly, Eggesbø et al[72] in Norway found a sevenfold increase in food allergy risk in C-section-born children of allergic mothers. Mitselou et al[73] conducted a large Swedish cohort study involving over one million children and found a modest but significant association between C-section and food allergy (HR: 1.21). Supporting these findings, Pyrhönen et al[74] from Finland observed that elective C-section doubled the risk of food allergies in children of non-atopic mothers (RR: 2.41). At the same time, a quasi-experimental study by Polos and Fletcher in the United States found a consistent association between C-sections and food allergies across different racial and ethnic groups[75].
| Ref. | Country | Sample size | Follow-up | Association with food allergy | Modifiable factors | Risk of bias |
| Papathoma et al[71], 2016 | Greece | 459 | 3 years | ↑ C-section risk (OR: 3.15); higher with parental atopy | Atopic dermatitis, gestational age | Moderate (small sample, good confounder adjustment) |
| Eggesbø et al[72], 2003 | Norway | 2803 | 2.5 years | ↑ Risk in allergic mothers (OR: 7.0) | Antibiotics not significant | Low (strong prospective design, adjusted) |
| Mitselou et al[73], 2018 | Sweden | 1086378 | 13 years | HR: 1.21 for C-section | LGA, low Apgar score | Low (large population-based cohort) |
| Pyrhönen et al[74], 2022 | Finland | 5564 | 12 years | RR: 2.41 for elective C-section in non-atopic mothers | Sibling atopy modifies risk | Moderate (targeted subgroup, adjusted analysis) |
| Polos and Fletcher[75], 2019 | United States | 150000+ | Cross-sectional | ↑ Food allergy across racial groups | Race, C-section rate, birth cohort effects | Moderate (large dataset, cross-sectional limitations) |
| Currell et al[76], 2022 | Australia | 5276 | 12 months | No association with C-section (aOR: Approximately 1.0) | Labor, feeding, siblings not modifying | Low (clear methodology and control) |
| Chua et al[77], 2025 | Taiwan | > 1 million | Approximately 14 years | No significant food allergy risk (aHR: 1.13) | ↑ Asthma, eczema, obesity | Low (nationwide cohort, adjusted) |
| Tamai et al[78], 2025 | Japan | 2114 | 9 years | No significant association (aRR: 1.1; CI: 0.7-1.7) | Adjusted for child & parental variables | Low (population-based, adjusted with GEE) |
| Adeyeye et al[79], 2019 | United States | Approximately 2900 | 3 years | ↑ Risk with emergency C-section (RR: 3.02) | Breastfeeding (partial mediation) | Moderate (good adjustment, moderate size) |
| Yang M et al[80], 2019 | China | 6768 | 12 months | ↑ C-section and formula feeding in CMPA group | High tolerance after 1 year (77%) | Moderate (well-documented, short-term) |
| Gil et al[81], 2017 | Spain | 211 cases + controls | Retrospective | C-section + formula (FFH) ↑ risk (OR: 11.82) | FFH and breastfeeding key risk factors | Moderate (case-control, smaller scale) |
| Metsälä et al[82], 2010 | Finland | 16237 CMPA cases | 2 years | C-section (OR: 1.18); high maternal age ↑ risk | Smoking & low SES ↓ risk | Low (large birth cohort, adjusted) |
| Toro Monjaraz et al[83], 2015 | Mexico | 101 CMPA, 90 controls | Retrospective | No C-section effect; antibiotics ↑ risk | Breastfeeding duration ↓ risk | High (small sample, retrospective) |
| Kuitunen et al[84], 2009 | Finland | 1018 | 5 years | Probiotics ↓ allergy in C-section-born infants | Perinatal probiotic supplementation | Moderate (well-executed trial, modest size) |
However, not all studies confirmed this relationship. Currell et al[76] in Australia, Chua et al[77] in Taiwan, and Tamai et al[78] in Japan found no significant link between C-section and the development of food allergies, highlighting the potential influence of environmental and genetic modifiers. For example, Chua et al’s study[77] noted a higher risk of asthma and eczema, but not food allergy specifically, in C-section-delivered infants. Other studies have suggested that only specific subtypes of C-section (e.g., emergency) or coexisting factors, such as antibiotic exposure, might contribute to an increased allergy risk. For instance, Adeyeye et al[79] in the United States found that emergency C-section, but not elective C-section, was associated with a higher risk of food allergy (RR: 3.02), partially mediated by reduced breastfeeding.
Regarding CMPA, several studies have provided valuable insights. Yang et al[80] in China observed significantly higher rates of C-section and formula feeding among CMPA infants. Gil et al[81] in Spain and Metsälä et al[82] in Finland confirmed that C-section combined with early formula feeding significantly increased the risk of immunoglobulin E (IgE)-mediated CMPA, with odds ratios as high as 11.82 when formula was introduced in the hospital setting. In contrast, a study by Toro Monjaraz et al[83] in Mexico did not find a direct association between C-section and CMPA. Still, it emphasized the importance of antibiotic use and breastfeeding duration in modifying the risk of allergy. Notably, Kuitunen et al[84] demonstrated that probiotic supplementation in C-section-born infants reduced the incidence of IgE-associated allergy (OR: 0.47), indicating a promising preventive strategy.
Taken together, these findings suggest that C-section—especially elective procedures without labor—may modestly increase the risk of food allergies, including CMPA. The strength of this association varies across studies, possibly due to differences in genetic predisposition, race, feeding practices, and healthcare systems. Modifiable factors, such as early formula introduction, the absence of breastfeeding, and inadequate microbial exposure, play a critical role in determining the magnitude of allergy risk. While some population-based studies report no significant association[76,77], the overall evidence supports a biologically plausible link between C-section and food sensitization, reinforcing the importance of early-life nutritional and microbial interventions in C-section-born infants.
Irritable bowel syndrome: There is no evidence to support an increased incidence of irritable bowel syndrome (IBS) in the offspring of women delivered by C-section. However, C-section delivery has been associated with a higher rate of acute gastroenteritis admission in children[85]. It has been reported that a history of acute gastroenteritis in children is linked to an increased risk of IBS[86].
A total of 75 studies were included in this review, encompassing observational cohorts, case-control studies, randomized controlled trials, cross-sectional designs, and systematic reviews (Table 14). Risk of bias was evaluated using established tools appropriate to each study type, including the NOS for observational studies, the Cochrane RoB 2.0 for randomized trials, and AMSTAR 2 for systematic reviews. Overall, 58.6% (n = 44) of the included studies were assessed as having a low risk of bias, 29.3% (n = 22) had moderate risk, and 12.0% (n = 9) demonstrated a high risk of bias—primarily due to issues such as lack of control groups, self-reported outcomes, or unclear confounding adjustment. The higher-risk studies were predominantly cross-sectional in nature. Findings were interpreted with these quality assessments in mind, and greater weight was given to evidence from high-quality longitudinal and interventional studies.
| Study domain | No. of studies | Risk of bias assessment | Tools used |
| Observational studies (cohort, case-control) | 54 | 37 low risk, 14 moderate, 3 high | Newcastle-Ottawa scale |
| Randomized controlled trials | 6 | 5 low risk, 1 some concerns | RoB 2.0 tool |
| Cross-sectional studies | 12 | 7 moderate risk, 5 high risk | AXIS appraisal tool (when applicable) |
| Systematic reviews/meta-analyses | 3 | 2 low risk, 1 Moderate | AMSTAR 2 |
This systematic review explored the short- and long-term GI consequences of C-section delivery, with a focus on FGIDs, microbiota development, and immune-mediated GI conditions such as IBD, coeliac disease, and food allergies, including CMPA. Our results confirm that C-section delivery is associated with an increased risk of multiple GI disturbances, primarily mediated by early-life disruption of the gut microbiota and modulated by environmental, nutritional, and perinatal factors.
Our findings strongly support the association between C-section delivery and the increased risk of short-term FGIDs such as infantile colic, regurgitation, functional constipation, and infantile dyschezia. Multiple studies have reported that C-section-born infants exhibit higher levels of fecal calprotectin, a marker of intestinal inflammation, and altered patterns of crying and GI discomfort[17,87]. Colic, in particular, peaked at around three months of age and was significantly associated with microbial imbalance and reduced exposure to maternal flora due to surgical delivery[88].
Constipation and dyschezia were also more frequent in C-section-born infants, often co-occurring with maternal depression or early formula feeding[6]. These associations underscore the significance of maternal-infant bonding, hormonal priming (e.g., oxytocin release during vaginal birth), and microbial transmission—all of which are frequently compromised or absent in C-section deliveries[89]. The findings align with Rome IV-based epidemiological research that confirms a higher burden of FGIDs in C-section-born neonates[12]. However, inconsistencies in the prevalence of FGIDs across studies may also stem from variations in diagnostic tools (e.g., parental report vs Rome IV criteria), feeding practices, and inclusion of subtypes (e.g., dyschezia vs colic). Therefore, standardization in outcome measures is essential for future research to enhance comparability and validity across cohorts.
Across nearly all short- and long-term outcomes, gut dysbiosis was a consistent theme. Vaginal delivery is known to expose the neonate to maternal vaginal and fecal microbiota, which play a critical role in priming the neonatal immune system and establishing a beneficial microbial environment[90]. In contrast, C-section-born infants—especially those delivered electively—are colonized by hospital-acquired and skin-associated microbes, with delayed acquisition of core genera such as Bacteroides, Bifidobacterium, and Lactobacillus[91]. This microbial delay was shown to persist for up to 12 months, with downstream effects on mucosal immunity, metabolism, and epithelial barrier function[92].
Importantly, interventions such as breastfeeding, synbiotics (e.g., Bifidobacterium breve M-16V plus scGOS/LcFOS), and maternal fecal microbiota transplantation have demonstrated the potential to partially restore microbial diversity and composition[93]. These strategies were more effective than probiotics alone, particularly in promoting Bifidobacteria colonization and reducing pathogen load (Clostridium difficile, Klebsiella)[94]. The persistence and impact of dysbiosis varied across populations, with racial and geographic factors (e.g., dietary fiber intake, urban vs rural settings) playing a modifying role[95]. Additionally, studies with longer follow-up periods demonstrated that microbial immaturity or deviation in C-section-born infants may persist up to the first or even second year of life. However, whether this delay leads to permanent immune or GI dysfunction remains debated, especially as microbial diversity tends to normalize by 2-3 years in some populations.
The long-term impact of C-section delivery on GI health is complex and variably expressed. Regarding IBD, several Nordic and Israeli cohort studies reported a modest increase in CD risk in C-section-born individuals, particularly males and those born via elective procedures[52]. In contrast, UC showed no consistent association with delivery mode, suggesting that CD may be more susceptible to early-life microbial disruption[54]. However, other high-powered studies from Canada and the United Kingdom found no link between C-section and IBD, indicating a potential influence of the healthcare system, diagnostic criteria, and population genetics[96]. Geographical and racial diversity also likely contributes to these mixed results. For instance, positive associations were more common in Scandinavian and Israeli studies, while Canadian and United Kingdom data more frequently showed null results. These differences could reflect disparities in C-section indication (elective vs. emergency), antibiotic policies, feeding practices, or even genetic susceptibility to immune-mediated diseases.
With coeliac disease, the results were similarly mixed. Several studies have demonstrated a higher incidence of CD in C-section-born children, particularly in those undergoing elective C-section and without labor[68]. In contrast, others, including the TEDDY cohort and large Canadian registries, have reported no significant association[67]. These findings suggest that although C-section may modestly contribute to CD risk in susceptible individuals, it is unlikely to be a sole or major etiological factor.
Food allergies, including CMPA, presented a more consistent pattern. Several studies have found that C-section delivery—particularly when combined with early formula feeding—increases the risk of IgE-mediated food allergies[97]. Elective C-section without labor and lack of breastfeeding emerged as key modifiable risk factors[74]. Children born via C-section were more likely to develop CMPA, with increased sensitivity to cow’s milk proteins and prolonged allergy symptoms, although tolerance often developed after infancy[98]. Probiotic supplementation, particularly during the perinatal period, has been shown to significantly reduce the risk of allergic sensitization in infants born by C-section, highlighting its preventive potential[99]. Nevertheless, variability persists due to differing criteria for allergy diagnosis, including reliance on hospital discharge data, parent-reported symptoms, or food challenge tests. Future studies should strive for harmonized protocols and more precise delineation between IgE and non-IgE-mediated allergies.
Several modifiable and mediating factors were consistently identified throughout this review that may help mitigate the GI risks associated with C-section delivery. Among these, breastfeeding stands out as one of the most protective interventions. Exclusive breastfeeding for at least four to six months has been shown to promote the development of a beneficial gut microbiota, reduce the risk of dysbiosis, and offer significant immunomodulatory effects that can protect against allergies and FGIDs such as colic[100]. In addition to breastfeeding, the use of probiotics and synbiotics has shown promise in restoring microbial balance, particularly in infants born via C-section[101]. Synbiotic formulations—combining prebiotics with specific probiotic strains—were more effective than probiotics alone in enhancing microbial diversity and reducing the incidence and severity of FGID symptoms[31].
Another important modifiable factor is the judicious use of perinatal antibiotics. While antibiotics are often necessary for infection prevention during C-section, minimizing unnecessary exposure can help preserve the integrity of the neonatal gut microbiota and reduce inflammatory responses associated with microbial disruption[102]. Finally, although early skin-to-skin contact and rooming-in practices do not directly influence the microbiota, they are beneficial for enhancing maternal-infant bonding, supporting the release of oxytocin, and facilitating neonatal adaptation[103]. These practices also promote the early initiation of breastfeeding, indirectly supporting microbial and immunological resilience in newborns[104].
It is also important to note that several additional mediators—including maternal atopy, labor exposure, parity, hospital feeding protocols, and NICU admission—can shape GI outcomes. These variables may explain discordant results in seemingly similar populations and highlight the need for multivariate modeling in future studies. Together, these modifiable factors offer practical, evidence-based strategies for reducing the GI-related risks associated with C-section and improving long-term health outcomes in affected infants. In addition, elective vs emergency C-section, labor exposure, maternal atopy, and hospital feeding protocols all played roles in shaping GI outcomes, suggesting that the risk is not inherent to C-section alone but to a constellation of perinatal factors[105].
This systematic review has several limitations that should be acknowledged. First, the included studies varied widely in design, methodology, and quality. While some were large prospective cohorts with long-term follow-up, others were retrospective or cross-sectional, introducing potential recall bias, misclassification, and confounding. Second, diagnostic criteria for GI outcomes such as infantile colic, constipation, or food allergies were not standardized across studies, limiting comparability and increasing heterogeneity in reported findings. Third, the composition and function of the gut microbiota were assessed using different technologies (e.g., culture-based methods, 16S rRNA sequencing, metagenomics), which can yield differing levels of taxonomic resolution and may influence the interpretation of microbial diversity and composition. Fourth, most studies were observational in nature and could not definitively establish causality between C-section delivery and subsequent GI outcomes. Additionally, several studies did not adequately control for important confounding factors such as feeding type, maternal antibiotic exposure, socioeconomic status, or environmental influences—factors known to influence both microbiota development and GI health.
Another limitation relates to geographic and ethnic variability. Many studies have been conducted in high-income countries, primarily in Europe and North America, which may limit their generalizability to low- and middle-income settings where C-section practices, microbial exposures, and healthcare access differ substantially. It is crucial to interpret the association between C-section and GI outcomes within a multifactorial context. While C-section represents a significant early-life event, it is not a standalone determinant of future GI health. Its impact often overlaps with other factors such as perinatal antibiotic use, absence of labor, early feeding practices, genetic predisposition, environmental exposures, early-life infections, and maternal health, all of which collectively shape the infant’s gut development and long-term disease risk. Therefore, C-section should be considered an initiating factor that may increase susceptibility rather than a direct cause. These confounding variables were variably addressed across studies, and future research should utilize multivariate models to better isolate the independent contribution of delivery mode. Finally, publication bias may have favored studies reporting positive associations between C-section and GI outcomes, potentially overestimating the true magnitude of risk. Despite these limitations, the synthesis of available evidence provides meaningful insights into the complex and potentially modifiable relationship between mode of delivery and GI health in infancy and later life.
This review’s strength lies in its comprehensive, multidimensional approach to evaluating C-section-related GI outcomes, with attention to microbial, nutritional, and immunological perspectives. Future research should prioritize RCTs assessing synbiotic and microbial interventions, particularly in high-risk populations born via C-section. Additionally, exploring gene-microbiota interactions, maternal diet, and metabolomic profiling may offer further insight into personalized risk prediction and management.
This systematic review highlights the multifaceted GI consequences associated with C-section delivery, encompassing both short-term and long-term outcomes. Infants born via C-section are at a higher risk for early FGIDs such as colic, regurgitation, dyschezia, and constipation, primarily due to altered microbial colonization and disrupted exposure to maternal hormones and vaginal microbiota. Long-term consequences, including increased susceptibility to CD, food allergies—particularly CMPA—and possibly celiac disease, have been observed in several large-scale studies. However, findings varied by geography, population characteristics, and methodology. Central to many of these outcomes is the disruption of the gut microbiota, with C-section-born infants showing delayed colonization by beneficial bacteria such as Bacteroides and Bifidobacteria. These changes may persist into infancy and are linked with impaired mucosal immunity and metabolic regulation. Nevertheless, this risk is not deterministic. Modifiable factors such as exclusive breastfeeding, early skin-to-skin contact, reduced antibiotic exposure, and the use of targeted nutritional interventions—especially synbiotics—can mitigate many of these adverse effects. While C-section should not be regarded as an inherently harmful mode of delivery, its potential impact on GI development must be acknowledged and addressed through evidence-based postnatal care. Future research should prioritize harmonized outcome definitions, long-term follow-up, and microbiome-integrated analyses to understand better and manage the implications of delivery mode on GI and immune health.
| 1. | Betran AP, Ye J, Moller AB, Souza JP, Zhang J. Trends and projections of caesarean section rates: global and regional estimates. BMJ Glob Health. 2021;6:e005671. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1116] [Cited by in RCA: 894] [Article Influence: 178.8] [Reference Citation Analysis (0)] |
| 2. | Sandall J, Tribe RM, Avery L, Mola G, Visser GH, Homer CS, Gibbons D, Kelly NM, Kennedy HP, Kidanto H, Taylor P, Temmerman M. Short-term and long-term effects of caesarean section on the health of women and children. Lancet. 2018;392:1349-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1025] [Cited by in RCA: 833] [Article Influence: 104.1] [Reference Citation Analysis (3)] |
| 3. | Lai C, Huang L, Wang Y, Huang C, Luo Y, Qin X, Zeng J. Effect of different delivery modes on intestinal microbiota and immune function of neonates. Sci Rep. 2024;14:17452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 4. | Catassi G, Mateo SG, Occhionero AS, Esposito C, Giorgio V, Aloi M, Gasbarrini A, Cammarota G, Ianiro G. The importance of gut microbiome in the perinatal period. Eur J Pediatr. 2024;183:5085-5101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 5. | Hrncir T. Gut Microbiota Dysbiosis: Triggers, Consequences, Diagnostic and Therapeutic Options. Microorganisms. 2022;10:578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 249] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 6. | Yoshida T, Matsumura K, Tsuchida A, Hamazaki K, Inadera H; Japan Environment and Children’s Study Group. Association between cesarean section and constipation in infants: the Japan Environment and Children's Study (JECS). BMC Res Notes. 2018;11:882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | Decker E, Hornef M, Stockinger S. Cesarean delivery is associated with celiac disease but not inflammatory bowel disease in children. Gut Microbes. 2011;2:91-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 8. | Kim S, Ndwandwe C, Devotta H, Kareem L, Yao L, O'Mahony L. Role of the microbiome in regulation of the immune system. Allergol Int. 2025;74:187-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 9. | Apostol AC, Jensen KDC, Beaudin AE. Training the Fetal Immune System Through Maternal Inflammation-A Layered Hygiene Hypothesis. Front Immunol. 2020;11:123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 10. | Dai DLY, Petersen C, Hoskinson C, Del Bel KL, Becker AB, Moraes TJ, Mandhane PJ, Finlay BB, Simons E, Kozyrskyj AL, Patrick DM, Subbarao P, Bode L, Azad MB, Turvey SE. Breastfeeding enrichment of B. longum subsp. infantis mitigates the effect of antibiotics on the microbiota and childhood asthma risk. Med. 2023;4:92-112.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 11. | Salvatore S, Baldassarre ME, Di Mauro A, Laforgia N, Tafuri S, Bianchi FP, Dattoli E, Morando L, Pensabene L, Meneghin F, Dilillo D, Mancini V, Talarico V, Tandoi F, Zuccotti G, Agosti M. Neonatal Antibiotics and Prematurity Are Associated with an Increased Risk of Functional Gastrointestinal Disorders in the First Year of Life. J Pediatr. 2019;212:44-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (6)] |
| 12. | Bi D, Jiang H, Yang K, Guan T, Hou L, Shu G. Neonatal risk factors for functional gastrointestinal disorders in preterm infants in the first year of life. Turk J Pediatr. 2023;65:919-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Pantazi AC, Mihai CM, Lupu A, Balasa AL, Chisnoiu T, Mihai L, Frecus CE, Ungureanu A, Chirila SI, Nori W, Lupu VV, Stoicescu RM, Baciu G, Cambrea SC. Gut Microbiota Profile and Functional Gastrointestinal Disorders in Infants: A Longitudinal Study. Nutrients. 2025;17:701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 14. | Ziętek M, Szczuko M, Machałowski T. Gastrointestinal Disorders and Atopic Dermatitis in Infants in the First Year of Life According to ROME IV Criteria-A Possible Association with the Mode of Delivery and Early Life Nutrition. J Clin Med. 2024;13:927. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Bekem Ö, Günay İ, Çelik F, Apa H. Interaction of functional gastrointestinal disorders with postpartum conditions related to mother and baby. Turk J Pediatr. 2021;63:461-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Velasco-Benitez CA, Axelrod CH, Gutierrez S, Saps M. The Relationship Between Prematurity, Method of Delivery, and Functional Gastrointestinal Disorders in Children. J Pediatr Gastroenterol Nutr. 2020;70:e37-e40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (6)] |
| 17. | Sommermeyer H, Bernatek M, Pszczola M, Krauss H, Piatek J. Supporting the diagnosis of infantile colic by a point of care measurement of fecal calprotectin. Front Pediatr. 2022;10:978545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Akman I, Kusçu K, Ozdemir N, Yurdakul Z, Solakoglu M, Orhan L, Karabekiroglu A, Ozek E. Mothers' postpartum psychological adjustment and infantile colic. Arch Dis Child. 2006;91:417-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 79] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Nakamura M, Matsumura K, Ohnuma Y, Yoshida T, Tsuchida A, Hamazaki K, Inadera H; Japan Environment and Children’s Study Group. Association of cesarean birth with prevalence of functional constipation in toddlers at 3 years of age: results from the Japan Environment and Children's Study (JECS). BMC Pediatr. 2021;21:419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 20. | Neves RO, Nunes LM, Silveira LO, Lima MR, Moreira PR, Bernardi JR. Functional constipation symptoms and complementary feeding methods: A randomized clinical trial. An Pediatr (Engl Ed). 2023;98:267-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Hierink GM, Brinkman LAM, Malmberg GGA, van Eijndhoven HWF, Trzpis M, Broens PMA; LIOMOC-study group. Association of Constipation with Modes of Delivery: A Retrospective Questionnaire-based Study. Int Urogynecol J. 2024;35:1477-1485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 22. | Dahlen HG, Foster JP, Psaila K, Spence K, Badawi N, Fowler C, Schmied V, Thornton C. Gastro-oesophageal reflux: a mixed methods study of infants admitted to hospital in the first 12 months following birth in NSW (2000-2011). BMC Pediatr. 2018;18:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (7)] |
| 23. | Guo HP, Li H, Guo J, Li YW. [Prevention of gastroesophageal reflux and aspiration in neonates by holding with hands immediately after delivery by cesarean section]. Di Yi Jun Yi Da Xue Xue Bao. 2002;22:337-338. [PubMed] |
| 24. | Shao Y, Forster SC, Tsaliki E, Vervier K, Strang A, Simpson N, Kumar N, Stares MD, Rodger A, Brocklehurst P, Field N, Lawley TD. Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature. 2019;574:117-121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 624] [Cited by in RCA: 731] [Article Influence: 104.4] [Reference Citation Analysis (0)] |
| 25. | Kim G, Bae J, Kim MJ, Kwon H, Park G, Kim SJ, Choe YH, Kim J, Park SH, Choe BH, Shin H, Kang B. Delayed Establishment of Gut Microbiota in Infants Delivered by Cesarean Section. Front Microbiol. 2020;11:2099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 26. | Bäckhed F, Roswall J, Peng Y, Feng Q, Jia H, Kovatcheva-Datchary P, Li Y, Xia Y, Xie H, Zhong H, Khan MT, Zhang J, Li J, Xiao L, Al-Aama J, Zhang D, Lee YS, Kotowska D, Colding C, Tremaroli V, Yin Y, Bergman S, Xu X, Madsen L, Kristiansen K, Dahlgren J, Wang J. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell Host Microbe. 2015;17:690-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1477] [Cited by in RCA: 2120] [Article Influence: 192.7] [Reference Citation Analysis (0)] |
| 27. | Nagpal R, Tsuji H, Takahashi T, Nomoto K, Kawashima K, Nagata S, Yamashiro Y. Gut dysbiosis following C-section instigates higher colonisation of toxigenic Clostridium perfringens in infants. Benef Microbes. 2017;8:353-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 28. | Reyman M, van Houten MA, van Baarle D, Bosch AATM, Man WH, Chu MLJN, Arp K, Watson RL, Sanders EAM, Fuentes S, Bogaert D. Impact of delivery mode-associated gut microbiota dynamics on health in the first year of life. Nat Commun. 2019;10:4997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 318] [Cited by in RCA: 279] [Article Influence: 39.9] [Reference Citation Analysis (4)] |
| 29. | Mitchell CM, Mazzoni C, Hogstrom L, Bryant A, Bergerat A, Cher A, Pochan S, Herman P, Carrigan M, Sharp K, Huttenhower C, Lander ES, Vlamakis H, Xavier RJ, Yassour M. Delivery Mode Affects Stability of Early Infant Gut Microbiota. Cell Rep Med. 2020;1:100156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 152] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 30. | Zhang C, Li L, Jin B, Xu X, Zuo X, Li Y, Li Z. The Effects of Delivery Mode on the Gut Microbiota and Health: State of Art. Front Microbiol. 2021;12:724449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 74] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 31. | Inchingolo F, Inchingolo AD, Palumbo I, Trilli I, Guglielmo M, Mancini A, Palermo A, Inchingolo AM, Dipalma G. The Impact of Cesarean Section Delivery on Intestinal Microbiota: Mechanisms, Consequences, and Perspectives-A Systematic Review. Int J Mol Sci. 2024;25:1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 32. | Ronan V, Yeasin R, Claud EC. Childhood Development and the Microbiome-The Intestinal Microbiota in Maintenance of Health and Development of Disease During Childhood Development. Gastroenterology. 2021;160:495-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 184] [Article Influence: 36.8] [Reference Citation Analysis (1)] |
| 33. | Dos Santos SJ, Shukla I, Hill JE, Money DM; Maternal Microbiome Legacy Project Team. Birth Mode Does Not Determine the Presence of Shared Bacterial Strains between the Maternal Vaginal Microbiome and the Infant Stool Microbiome. Microbiol Spectr. 2023;11:e0061423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 34. | Dominguez-Bello MG, Costello EK, Contreras M, Magris M, Hidalgo G, Fierer N, Knight R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci USA. 2010;107:11971-11975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3937] [Cited by in RCA: 3267] [Article Influence: 204.2] [Reference Citation Analysis (4)] |
| 35. | Björkstén B, Sepp E, Julge K, Voor T, Mikelsaar M. Allergy development and the intestinal microflora during the first year of life. J Allergy Clin Immunol. 2001;108:516-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 845] [Cited by in RCA: 777] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 36. | Wilson BC, Butler ÉM, Grigg CP, Derraik JGB, Chiavaroli V, Walker N, Thampi S, Creagh C, Reynolds AJ, Vatanen T, O'Sullivan JM, Cutfield WS. Oral administration of maternal vaginal microbes at birth to restore gut microbiome development in infants born by caesarean section: A pilot randomised placebo-controlled trial. EBioMedicine. 2021;69:103443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 37. | Lagkouvardos I, Intze E, Schaubeck M, Rooney JP, Hecht C, Piloquet H, Clavel T. Early life gut microbiota profiles linked to synbiotic formula effects: a randomized clinical trial in European infants. Am J Clin Nutr. 2023;117:326-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 38. | Mueller NT, Differding MK, Sun H, Wang J, Levy S, Deopujari V, Appel LJ, Blaser MJ, Kundu T, Shah AA, Dominguez Bello MG, Hourigan SK. Maternal Bacterial Engraftment in Multiple Body Sites of Cesarean Section Born Neonates after Vaginal Seeding-a Randomized Controlled Trial. mBio. 2023;14:e0049123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 39. | Chua MC, Ben-Amor K, Lay C, Neo AGE, Chiang WC, Rao R, Chew C, Chaithongwongwatthana S, Khemapech N, Knol J, Chongsrisawat V. Effect of Synbiotic on the Gut Microbiota of Cesarean Delivered Infants: A Randomized, Double-blind, Multicenter Study. J Pediatr Gastroenterol Nutr. 2017;65:102-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 78] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 40. | Wang Y, Wopereis H, Kakourou A, Liu M, Wu J, Li Z, Zhang L, Tan M, Low JSY, Liu M, Roeselers G, Knol J, Cai W. Restoration of gut microbiota with a specific synbiotic-containing infant formula in healthy Chinese infants born by cesarean section. Eur J Clin Nutr. 2025;79:567-575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 41. | Garcia Rodenas CL, Lepage M, Ngom-Bru C, Fotiou A, Papagaroufalis K, Berger B. Effect of Formula Containing Lactobacillus reuteri DSM 17938 on Fecal Microbiota of Infants Born by Cesarean-Section. J Pediatr Gastroenterol Nutr. 2016;63:681-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | Yang W, Tian L, Luo J, Yu J. Ongoing Supplementation of Probiotics to Cesarean-Born Neonates during the First Month of Life may Impact the Gut Microbial. Am J Perinatol. 2021;38:1181-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 43. | Gong Y, Zhong H, Wang J, Wang X, Huang L, Zou Y, Qin H, Yang R. Effect of Probiotic Supplementation on the Gut Microbiota Composition of Infants Delivered by Cesarean Section: An Exploratory, Randomized, Open-label, Parallel-controlled Trial. Curr Microbiol. 2023;80:341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 44. | Hurkala J, Lauterbach R, Radziszewska R, Strus M, Heczko P. Effect of a Short-Time Probiotic Supplementation on the Abundance of the Main Constituents of the Gut Microbiota of Term Newborns Delivered by Cesarean Section-A Randomized, Prospective, Controlled Clinical Trial. Nutrients. 2020;12:3128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 45. | Akagawa S, Tsuji S, Onuma C, Akagawa Y, Yamaguchi T, Yamagishi M, Yamanouchi S, Kimata T, Sekiya SI, Ohashi A, Hashiyada M, Akane A, Kaneko K. Effect of Delivery Mode and Nutrition on Gut Microbiota in Neonates. Ann Nutr Metab. 2019;74:132-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 46. | Wu S, Ren L, Li J, Shen X, Zhou Q, Miao Z, Jia W, He F, Cheng R. Breastfeeding might partially contribute to gut microbiota construction and stabilization of propionate metabolism in cesarean-section infants. Eur J Nutr. 2023;62:615-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 47. | Korpela K, Salonen A, Vepsäläinen O, Suomalainen M, Kolmeder C, Varjosalo M, Miettinen S, Kukkonen K, Savilahti E, Kuitunen M, de Vos WM. Probiotic supplementation restores normal microbiota composition and function in antibiotic-treated and in caesarean-born infants. Microbiome. 2018;6:182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 176] [Article Influence: 22.0] [Reference Citation Analysis (4)] |
| 48. | Bellomo AR, Rotondi G, Rago P, Bloise S, Di Ruzza L, Zingoni A, Di Valerio S, Valzano E, Di Pierro F, Cazzaniga M, Bertuccioli A, Guasti L, Zerbinati N, Lubrano R. RETRACTED: Effect of Bifidobacterium bifidum Supplementation in Newborns Born from Cesarean Section on Atopy, Respiratory Tract Infections, and Dyspeptic Syndromes: A Multicenter, Randomized, and Controlled Clinical Trial. Microorganisms. 2024;12:1093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 49. | Yang L, Hui Y, Sangild PT, Kot WP, Aunsholt L, Zachariassen G, Jiang P-P, Nielsen DS. Gut microbiota development in very preterm infants following fortification of human milk. mSystems. 2025;10:e0091624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 50. | Song SJ, Wang J, Martino C, Jiang L, Thompson WK, Shenhav L, McDonald D, Marotz C, Harris PR, Hernandez CD, Henderson N, Ackley E, Nardella D, Gillihan C, Montacuti V, Schweizer W, Jay M, Combellick J, Sun H, Garcia-Mantrana I, Gil Raga F, Collado MC, Rivera-Viñas JI, Campos-Rivera M, Ruiz-Calderon JF, Knight R, Dominguez-Bello MG. Naturalization of the microbiota developmental trajectory of Cesarean-born neonates after vaginal seeding. Med. 2021;2:951-964.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 51. | Dominguez-Bello MG, De Jesus-Laboy KM, Shen N, Cox LM, Amir A, Gonzalez A, Bokulich NA, Song SJ, Hoashi M, Rivera-Vinas JI, Mendez K, Knight R, Clemente JC. Partial restoration of the microbiota of cesarean-born infants via vaginal microbial transfer. Nat Med. 2016;22:250-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 556] [Cited by in RCA: 654] [Article Influence: 65.4] [Reference Citation Analysis (0)] |
| 52. | Bager P, Simonsen J, Nielsen NM, Frisch M. Cesarean section and offspring's risk of inflammatory bowel disease: a national cohort study. Inflamm Bowel Dis. 2012;18:857-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 130] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 53. | Andersen V, Möller S, Jensen PB, Møller FT, Green A. Caesarean Delivery and Risk of Chronic Inflammatory Diseases (Inflammatory Bowel Disease, Rheumatoid Arthritis, Coeliac Disease, and Diabetes Mellitus): A Population Based Registry Study of 2,699,479 Births in Denmark During 1973-2016. Clin Epidemiol. 2020;12:287-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 54. | Hellsing C, Örtqvist AK, Hagel E, Mesas-Burgos C, Gustafsson UO, Granström AL. Delivery mode and risk of gastrointestinal disease in the offspring. Acta Obstet Gynecol Scand. 2022;101:1146-1152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 55. | Zamstein O, Glusman Bendersky A, Sheiner E, Landau D, Levy A. Association Between Mode of Delivery of the Breech Fetus and Hospitalizations Due to Inflammatory Bowel Disease During Childhood. J Clin Gastroenterol. 2022;56:e161-e165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 56. | Malmborg P, Bahmanyar S, Grahnquist L, Hildebrand H, Montgomery S. Cesarean section and the risk of pediatric Crohn's disease. Inflamm Bowel Dis. 2012;18:703-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 57. | Ponsonby AL, Catto-Smith AG, Pezic A, Dupuis S, Halliday J, Cameron D, Morley R, Carlin J, Dwyer T. Association between early-life factors and risk of child-onset Crohn's disease among Victorian children born 1983-1998: a birth cohort study. Inflamm Bowel Dis. 2009;15:858-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 58. | Bernstein CN, Banerjee A, Targownik LE, Singh H, Ghia JE, Burchill C, Chateau D, Roos LL. Cesarean Section Delivery Is Not a Risk Factor for Development of Inflammatory Bowel Disease: A Population-based Analysis. Clin Gastroenterol Hepatol. 2016;14:50-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 59. | Burnett D, Brown MM, Otley A, Kuhle S. The Association Between Caesarean Section and Inflammatory Bowel Disease in Childhood and Young Adulthood: Findings From 2 Retrospective Cohort Studies. J Pediatr Gastroenterol Nutr. 2020;71:e84-e89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 60. | Burgess CJ, Schnier C, Wood R, Henderson P, Wilson DC. Prematurity, Delivery Method, and Infant Feeding Type Are Not Associated with Paediatric-onset Inflammatory Bowel Disease Risk: A Scottish Retrospective Birth Cohort Study. J Crohns Colitis. 2022;16:1235-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 61. | Sonntag B, Stolze B, Heinecke A, Luegering A, Heidemann J, Lebiedz P, Rijcken E, Kiesel L, Domschke W, Kucharzik T, Maaser C. Preterm birth but not mode of delivery is associated with an increased risk of developing inflammatory bowel disease later in life. Inflamm Bowel Dis. 2007;13:1385-1390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 62. | Soullane S, Henderson M, Kang H, Luu TM, Lee GE, Auger N. Cesarean delivery and risk of hospitalization for autoimmune disorders before 14 years of age. Eur J Pediatr. 2021;180:3359-3366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 63. | Iorfida D, Valitutti F, Vestri A, D'Adamo G, Passaro T, Crocco M, Malerba F, Monzani A, Rabbone I, Pensabene L, Giancotti L, Graziano F, Citrano M, Ferretti F, Trovato CM, Pacenza C, Iasevoli M, Banzato C, Lubrano R, Montuori M; “Food-induced disease” study group of SIGENP. Prevalence of delivery mode in an Italian nationwide cohort with celiac disease: a SIGENP multicenter retrospective study (the CD-deliver-IT). Ital J Pediatr. 2024;50:129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 64. | Mårild K, Stephansson O, Montgomery S, Murray JA, Ludvigsson JF. Pregnancy outcome and risk of celiac disease in offspring: a nationwide case-control study. Gastroenterology. 2012;142:39-45.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 161] [Article Influence: 11.5] [Reference Citation Analysis (4)] |
| 65. | Dydensborg Sander S, Hansen AV, Størdal K, Andersen AN, Murray JA, Husby S. Mode of delivery is not associated with celiac disease. Clin Epidemiol. 2018;10:323-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 66. | Emilsson L, Magnus MC, Størdal K. Perinatal risk factors for development of celiac disease in children, based on the prospective Norwegian Mother and Child Cohort Study. Clin Gastroenterol Hepatol. 2015;13:921-927. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 67. | Koletzko S, Lee HS, Beyerlein A, Aronsson CA, Hummel M, Liu E, Simell V, Kurppa K, Lernmark Å, Hagopian W, Rewers M, She JX, Simell O, Toppari J, Ziegler AG, Krischer J, Agardh D; TEDDY Study Group. Cesarean Section on the Risk of Celiac Disease in the Offspring: The Teddy Study. J Pediatr Gastroenterol Nutr. 2018;66:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 68. | Tanpowpong P, Li S, Espinola JA, Santos LC, James KE, Powe CE, Camargo CA Jr. Pregnancy- and birth-related risk factors for the development of childhood celiac disease. Acta Paediatr. 2023;112:1029-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 69. | Decker E, Engelmann G, Findeisen A, Gerner P, Laass M, Ney D, Posovszky C, Hoy L, Hornef MW. Cesarean delivery is associated with celiac disease but not inflammatory bowel disease in children. Pediatrics. 2010;125:e1433-e1440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 182] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 70. | Bielik M, Selvek M, Suchánková M, Shawkatová I. A case-control epidemiological survey on potential risk factors for celiac disease. Cent Eur J Public Health. 2024;32:119-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 71. | Papathoma E, Triga M, Fouzas S, Dimitriou G. Cesarean section delivery and development of food allergy and atopic dermatitis in early childhood. Pediatr Allergy Immunol. 2016;27:419-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 72. | Eggesbø M, Botten G, Stigum H, Nafstad P, Magnus P. Is delivery by cesarean section a risk factor for food allergy? J Allergy Clin Immunol. 2003;112:420-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 157] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 73. | Mitselou N, Hallberg J, Stephansson O, Almqvist C, Melén E, Ludvigsson JF. Cesarean delivery, preterm birth, and risk of food allergy: Nationwide Swedish cohort study of more than 1 million children. J Allergy Clin Immunol. 2018;142:1510-1514.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 74. | Pyrhönen K, Kulmala P. Delivery mode and the incidence of atopic sensitization and food allergy in a Finnish child population. Pediatr Allergy Immunol. 2022;33:e13584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 75. | Polos J, Fletcher J. Caesarean section and children's health: A quasi-experimental design. Popul Stud (Camb). 2019;73:353-368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 76. | Currell A, Koplin JJ, Lowe AJ, Perrett KP, Ponsonby AL, Tang MLK, Dharmage SC, Peters RL. Mode of Birth Is Not Associated With Food Allergy Risk in Infants. J Allergy Clin Immunol Pract. 2022;10:2135-2143.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 77. | Chua WC, Chen YL, Yen CF, Chen HL. Long-term health outcomes of children born by cesarean section: A nationwide population-based retrospective cohort study in Taiwan. J Formos Med Assoc. 2025;124:1034-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 78. | Tamai K, Matsumoto N, Mitsui T, Masuyama H, Yorifuji T. Association between cesarean delivery and childhood allergic diseases in a longitudinal population-based birth cohort from Japan. Sci Rep. 2025;15:19206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 79. | Adeyeye TE, Yeung EH, McLain AC, Lin S, Lawrence DA, Bell EM. Wheeze and Food Allergies in Children Born via Cesarean Delivery: The Upstate KIDS Study. Am J Epidemiol. 2019;188:355-362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 80. | Yang M, Tan M, Wu J, Chen Z, Long X, Zeng Y, Cai H, Zhang Y, Geng L, Xiao Y, Ke H, Liu Y, Rong L, Fu S, Wang H, Wang Y, Li X, Chen P, Li K, Xie J, Chen H, Li H, Wang H, Li DY, Gong S. Prevalence, Characteristics, and Outcome of Cow's Milk Protein Allergy in Chinese Infants: A Population-Based Survey. JPEN J Parenter Enteral Nutr. 2019;43:803-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 81. | Gil F, Amezqueta A, Martinez D, Aznal E, Etayo V, Durá T, Sánchez-Valverde F. Association between Caesarean Delivery and Isolated Doses of Formula Feeding in Cow Milk Allergy. Int Arch Allergy Immunol. 2017;173:147-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 82. | Metsälä J, Lundqvist A, Kaila M, Gissler M, Klaukka T, Virtanen SM. Maternal and perinatal characteristics and the risk of cow's milk allergy in infants up to 2 years of age: a case-control study nested in the Finnish population. Am J Epidemiol. 2010;171:1310-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 83. | Toro Monjaraz EM, Ramírez Mayans JA, Cervantes Bustamante R, Gómez Morales E, Molina Rosales A, Montijo Barrios E, Zárate Mondragón F, Cadena León J, Cazares Méndez M, López-Ugalde M. Perinatal factors associated with the development of cow's milk protein allergy. Rev Gastroenterol Mex. 2015;80:27-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 84. | Kuitunen M, Kukkonen K, Juntunen-Backman K, Korpela R, Poussa T, Tuure T, Haahtela T, Savilahti E. Probiotics prevent IgE-associated allergy until age 5 years in cesarean-delivered children but not in the total cohort. J Allergy Clin Immunol. 2009;123:335-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 282] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 85. | Bentley JP, Simpson JM, Bowen JR, Morris JM, Roberts CL, Nassar N. Gestational age, mode of birth and breastmilk feeding all influence acute early childhood gastroenteritis: a record-linkage cohort study. BMC Pediatr. 2016;16:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 86. | Thabane M, Simunovic M, Akhtar-Danesh N, Garg AX, Clark WF, Collins SM, Salvadori M, Marshall JK. An outbreak of acute bacterial gastroenteritis is associated with an increased incidence of irritable bowel syndrome in children. Am J Gastroenterol. 2010;105:933-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (3)] |
| 87. | Lee YM, Min CY, Choi YJ, Jeong SJ. Delivery and feeding mode affects fecal calprotectin levels in infants <7months old. Early Hum Dev. 2017;108:45-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 88. | Rhoads JM, Collins J, Fatheree NY, Hashmi SS, Taylor CM, Luo M, Hoang TK, Gleason WA, Van Arsdall MR, Navarro F, Liu Y. Infant Colic Represents Gut Inflammation and Dysbiosis. J Pediatr. 2018;203:55-61.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (4)] |
| 89. | Döblin S, Seefeld L, Weise V, Kopp M, Knappe S, Asselmann E, Martini J, Garthus-Niegel S. The impact of mode of delivery on parent-infant-bonding and the mediating role of birth experience: a comparison of mothers and fathers within the longitudinal cohort study DREAM. BMC Pregnancy Childbirth. 2023;23:285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 90. | Xie J, Tang C, Hong S, Xin Y, Zhang J, Lin Y, Mao L, Xiao Y, Wu Q, Zhang X, Shen H. Maternal vaginal fluids play a major role in the colonization of the neonatal intestinal microbiota. Front Cell Infect Microbiol. 2023;13:1065884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 91. | Hoang DM, Levy EI, Vandenplas Y. The impact of Caesarean section on the infant gut microbiome. Acta Paediatr. 2021;110:60-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 92. | Milani C, Duranti S, Bottacini F, Casey E, Turroni F, Mahony J, Belzer C, Delgado Palacio S, Arboleya Montes S, Mancabelli L, Lugli GA, Rodriguez JM, Bode L, de Vos W, Gueimonde M, Margolles A, van Sinderen D, Ventura M. The First Microbial Colonizers of the Human Gut: Composition, Activities, and Health Implications of the Infant Gut Microbiota. Microbiol Mol Biol Rev. 2017;81:e00036-e00017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1561] [Cited by in RCA: 1281] [Article Influence: 142.3] [Reference Citation Analysis (9)] |
| 93. | Sáez-Fuertes L, Kapravelou G, Grases-Pintó B, Massot-Cladera M, Bernabeu M, Knipping K, Garssen J, Bourdet-Sicard R, Castell M, Rodríguez-Lagunas MJ, Collado MC, Pérez-Cano FJ. Impact of maternal Bifidobacterium breve M-16V and scGOS/lcFOS supplementation during pregnancy and lactation on the maternal immune system and milk composition. Front Immunol. 2024;15:1418594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 94. | Phavichitr N, Wang S, Chomto S, Tantibhaedhyangkul R, Kakourou A, Intarakhao S, Jongpiputvanich S; COLOR Study Group, Roeselers G, Knol J. Impact of synbiotics on gut microbiota during early life: a randomized, double-blind study. Sci Rep. 2021;11:3534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 95. | Sreenikitha K, Das S, Andrighetti T, Sudhakar P. Comparative study of the gut microbiomes between Western and Indigenous cultures - Implications for health and disease. Microbe. 2025;7:100310. [DOI] [Full Text] |
| 96. | Frias Gomes C, Narula N, Morão B, Nicola P, Cravo M, Torres J. Mode of Delivery Does Not Affect the Risk of Inflammatory Bowel Disease. Dig Dis Sci. 2021;66:398-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 97. | Koplin J, Allen K, Gurrin L, Osborne N, Tang ML, Dharmage S. Is caesarean delivery associated with sensitization to food allergens and IgE-mediated food allergy: a systematic review. Pediatr Allergy Immunol. 2008;19:682-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 71] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 98. | Khalil A, Abdel Gawad M, Mokhtar N. Risk factors of delayed tolerance in children with cow milk protein allergy attending gastroenterology clinic in Alexandria University Children’s Hospital. Alex J Pediatr. 2020;33:55. [DOI] [Full Text] |
| 99. | Sestito S, D'Auria E, Baldassarre ME, Salvatore S, Tallarico V, Stefanelli E, Tarsitano F, Concolino D, Pensabene L. The Role of Prebiotics and Probiotics in Prevention of Allergic Diseases in Infants. Front Pediatr. 2020;8:583946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 100. | Valverde-Molina J, García-Marcos L. Microbiome and Asthma: Microbial Dysbiosis and the Origins, Phenotypes, Persistence, and Severity of Asthma. Nutrients. 2023;15:486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 101. | Ngoc APT, Zahoor A, Kim DG, Yang SH. Using Synbiotics as a Therapy to Protect Mental Health in Alzheimer's Disease. J Microbiol Biotechnol. 2024;34:1739-1747. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 102. | Nakandalage R, Guan LL, Malmuthuge N. Microbial Interventions to Improve Neonatal Gut Health. Microorganisms. 2023;11:1328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 103. | Altit G, Hamilton D, O'Brien K. Skin-to-skin care (SSC) for term and preterm infants. Paediatr Child Health. 2024;29:238-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 104. | Notarbartolo V, Giuffrè M, Montante C, Corsello G, Carta M. Composition of Human Breast Milk Microbiota and Its Role in Children's Health. Pediatr Gastroenterol Hepatol Nutr. 2022;25:194-210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 105. | Benzouina S, Boubkraoui Mel-M, Mrabet M, Chahid N, Kharbach A, El-Hassani A, Barkat A. Fetal outcome in emergency versus elective cesarean sections at Souissi Maternity Hospital, Rabat, Morocco. Pan Afr Med J. 2016;23:197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/