Published online Mar 18, 2026. doi: 10.5312/wjo.v17.i3.115155
Revised: November 10, 2025
Accepted: January 5, 2026
Published online: March 18, 2026
Processing time: 158 Days and 7.9 Hours
Despite growing recognition of locomotive syndrome (LS) in the general elderly population, research focusing on its prevalence and clinical implications among elderly lung cancer patients remains limited. Existing studies have predominantly examined nutritional, immune, or systemic functional factors influencing che
To investigate the effect of pre-chemotherapy LS status on short-term adverse outcome events among elderly lung cancer patients.
This study employed convenience sampling to recruit 410 elderly lung cancer patients admitted to the Affiliated Hospital of Jiangnan University from January 2024 to June 2024. Clinical data of the patients were collected, and they were divided into the LS group and the non-LS group according to their LS status before chemotherapy. The patients were followed up for 6 months. Toxicity during chemotherapy and adverse outcomes such as death, incapacitation, and unplanned hospitalization during the follow-up period were recorded.
This study finished data collection on 386 elderly lung cancer patients. Among them, 161 were diagnosed with LS before chemotherapy, while 225 were non-LS patients. After chemotherapy, the incidence of LS was significantly higher in the LS group than in the non-LS group (P < 0.05). During the chemotherapy, the LS group experienced a significantly greater incidence of toxicities when compared to the non-LS group (P < 0.05). The toxicity grading in the LS group was significantly higher than that in the non-LS group (P < 0.05). During the follow-up period, the mortality rate of the LS group was significantly higher than that of the non-LS group (P < 0.05). Moreover, patients in the LS group faced more severe incapacitation and a higher rate of unplanned hospitalizations than those in the non-LS group (P < 0.05).
Pre-chemotherapy LS has become more prevalent among elderly lung cancer patients, and its incidence further increases after chemotherapy. Elderly lung cancer patients with pre-chemotherapy LS experience numerous and severe toxic side effects during chemotherapy. They also face a high risk of death, suffer from severe disability, and have a high rate of unplanned hospitalization during the follow-up period.
Core Tip: Currently, more studies focus on the effects of nutritional, immune, or systemic functional status on chemotherapy toxicity and prognosis in elderly lung cancer patients. In contrast, research on the locomotive syndrome status of elderly lung cancer patients before chemotherapy is relatively scarce. In this study, we intend to investigate the locomotive syndrome status of elderly lung cancer patients before chemotherapy and analyze its impact on short-term adverse events.
- Citation: Liang DH, Feng XR, Li L, Chen Y, Yang YL. Impact of pre-chemotherapy locomotive syndrome status on short-term adverse outcome events in elderly lung cancer patients: A longitudinal study. World J Orthop 2026; 17(3): 115155
- URL: https://www.wjgnet.com/2218-5836/full/v17/i3/115155.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i3.115155
In recent years, the incidence of malignant tumors among the elderly has been rising annually[1]. In 2020, there were approximately 19.29 million new cancer cases globally, with 4.57 million in China. Most of the newly diagnosed patients were aged between 60 and 79[2]. According to global cancer statistics, lung cancer caused 1.796 million deaths that year, accounting for 18.0% of overall cancer-related deaths and ranking as the leading cause of cancer death worldwide[3]. Against the backdrop of health promotion and population aging in China, maintaining and improving the musculoskeletal function (MF) and activities of daily living (ADL) of elderly lung cancer patients, maximizing their independent living time, and reducing the care burden on families and society have become major concerns in contemporary society[4].
The address age-related declines in physical function, the Japanese Orthopedic Association proposed the concept of “locomotive syndrome (LS)”, which emphasizes the residents’ awareness of the importance of MF and ADL[5]. LS is defined as a high-risk state in which an individual’s daily life is restricted due to degenerative dysfunction of one or more parts of the musculoskeletal system[6]. The musculoskeletal system, including bones, joints, intervertebral discs, muscles, and peripheral nerves[7,8]. As LS progresses, it can lead to a cascade of musculoskeletal and neurological disorders - such as osteoporosis and fragility fractures, osteoarthritis, spondylosis, sarcopenia, and neurological disorders-that result in pain, limited joint mobility, postural instability, and physical imbalance in patients, leading to difficulties in standing and walking, reduced MF function, and decreased ADL ability. Eventually, patients may end up in a state where they need others’ care, and the risk of falls and death also rises[5-7].
Unlike sarcopenia or frailty, which focus on isolated physiological or symptomatic domains, LS provides an integrated assessment of overall musculoskeletal health[9,10]. The degeneration of muscles, bones, and other locomotor organs is typically gradual and subclinical, often preceding observable weakness or mobility limitations. According to the Eastern Cooperative Oncology Group Performance Status, 29.7% of tumor patients who moved normally were diagnosed with LS. This indicates that LS may occur even if tumor patients show no symptoms of sarcopenia or weakness[9-11]. Research has shown that LS is reversible. Implementing effective physical interventions at this stage can maintain or even improve an individual’s MF and ADL before irreversible physical impairment sets in[12].
In oncology practice, a substantial proportion of elderly lung-cancer patients are diagnosed at an advanced stage and thus miss the optimal window for surgical resection. Consequently, non-surgical treatment methods such as radio
Despite growing recognition of LS in the general elderly population, research focusing on its prevalence and clinical implications among elderly lung cancer patients remains limited. Existing studies have predominantly examined nutritional, immune, or systemic functional factors influencing chemotherapy tolerance and prognosis, while musculoskeletal health - an equally critical determinant of patient outcomes - has received insufficient attention. Therefore, this study aims to investigate the prevalence and characteristics of LS in elderly lung cancer patients prior to chemotherapy and to explore its impact on short-term adverse events. By addressing this gap, our research seeks to provide new evidence to support early functional assessment and targeted intervention strategies in this vulnerable population.
This was a longitudinal study involving repeated assessments of participants across multiple time points. Participants voluntarily enrolled and provided written informed consent. The study was approved by the Ethics Committee of the Affiliated Hospital of Jiangnan University, No. LS2023101. It was registered in the Chinese Clinical Trial Registry, No. ChiCTR2400079958 on January 17, 2024. The study adhered to the principles of the Declaration of Helsinki.
This study recruited elderly lung cancer patients hospitalized at the Affiliated Hospital of Jiangnan University from January to June 2024, using convenience sampling.
Inclusion criteria: (1) Meet the diagnostic criteria for primary lung cancer in the Clinical Guidelines for the Diagnosis and Treatment of Cancer of the Chinese Medical Association (2024 Edition) and be newly diagnosed[17]; (2) Be in clinical stages II to stages III; (3) Be aged 60 or above; (4) Meet the indications for chemotherapy, have no contraindications to chemotherapy, and agree to receive chemotherapy; (5) Have a Barthel index greater than 60 before chemotherapy; (6) Have basic comprehension and writing skills and be able to communicate and interact normally; and (7) Voluntarily sign an informed consent form and agree to participate in this study.
Exclusion criteria: (1) Have metastatic lung cancer or in combination with malignant tumors of other systems; (2) Have other critical illnesses that prevent cooperation in the survey and data collection; and (3) Have abnormal mental and behavioral manifestations.
To avoid the influence of different chemotherapeutic drugs on adverse reactions, all patients selected in this study received chemotherapy with docetaxel (produced by Jiangsu Hengrui Pharmaceutical Co., Ltd., National Pharmaceutical License No. H20020543, Jiangsu Province, China). On the first day of each chemotherapy cycle, patients were given an intravenous injection of docetaxel at a dose of 75 mg/m2 once a week. One chemotherapy cycle lasted three weeks, and the total treatment course was set at four cycles, totaling 12 weeks. During chemotherapy, patients received a series of symptomatic treatments, including antiemetics, gastric protection, liver protection, and reduction of myelosuppression. To minimize the risk of allergic reactions, patients were required to take dexamethasone orally for three consecutive days before the intravenous injection of docetaxel, at a daily dose of 7.5 mg, divided into two doses. Moreover, throughout the entire chemotherapy period, all patients received health education, nursing care, and counseling guidance from the same medical team.
General information: A self-designed general information questionnaire was used to collect patients’ clinical information, including gender, age, education level, marital status, place of residence, pathology type, and tumor stage.
This study employed the 25-question Geriatric Locomotive Function Scale (GLFS-25) to assess the participants. Developed by Japanese scholars Seichi et al[18], this scale was specifically designed to investigate the LS status of subjects. It was validated by Chinese scholars to be suitable for Chinese elderly oncology patients[19,20]. GLFS-25 consisted of 25 entries across four dimensions: Physical pain (4 entries), ADL (16 entries), social activities (3 entries), and mental status (2 entries). Each entry was scored on a 5-point Likert scale (0-4 points), where higher scores meant poorer motor function. A total score of 16 or above indicated motor dysfunction. The scale had a Cronbach’s a coefficient of 0.93, suggesting good reliability and validity.
Based on GLFS-25 total scores, participants were categorized into two groups: An LS group (scores ≥ 16) and a non-LS group (scores < 16). The presence of LS signifies a fundamentally different baseline risk profile in terms of frailty, musculoskeletal reserve, and functional capacity. This classification allowed us to explore how differences in locomotive function influenced short-term clinical outcomes among elderly lung cancer patients undergoing chemotherapy.
From the start of chemotherapy until 6 months after its completion, patients were followed up by telephone every 2 weeks to monitor the occurrence of adverse outcome events. These events included toxic side effects during chemo
Toxic side effects during chemotherapy: Based on the Common Terminology Criteria for Adverse Events version 5.0 issued by the United States National Cancer Institute, each toxic side effect was categorized into grades 1-5. The identification of toxic side effects was jointly determined by two or more attending physicians, and the number and grades of toxic side effects experienced by patients were recorded. If a patient experienced multiple levels of toxic side effects, the most severe level was used as the final assessment grade[21].
Incapacitation: Incapacitation was defined as a patient’s inability to perform daily activities independently and require assistance from others. The Barthel index was used for assessment, covering 10 items such as eating, bathing, grooming, dressing, bowel control, urination control, toileting, bed and chair transfer, flat floor activities, and going up and down stairs. Each item was scored from 0 point to 15 points, with a total score of 100 points. A score ≤ 40 indicated complete incapacitation, 4-60 indicated moderate incapacitation, and a score > 60 indicated non-incapacitation. Cronbach’s a coefficient of the Barthel index ranged from 0.84 to 0.94, indicating good reliability and validity[22].
Unplanned hospitalization: Unplanned hospitalization refers to hospital admissions not scheduled for chemotherapy, follow-up, or other specific reasons. Same-day admissions and discharges were not recorded as hospitalizations.
The data were analyzed using SPSS 23.0 statistical software. Measurement data following normal distributions were expressed as mean ± SD, and independent samples t-tests were used for comparisons between two groups. Counting data were expressed as relative numbers, and χ2 tests were used for inter-group comparisons. Rank sum tests were used for comparing rank data. Differences were considered statistically significant when P < 0.05.
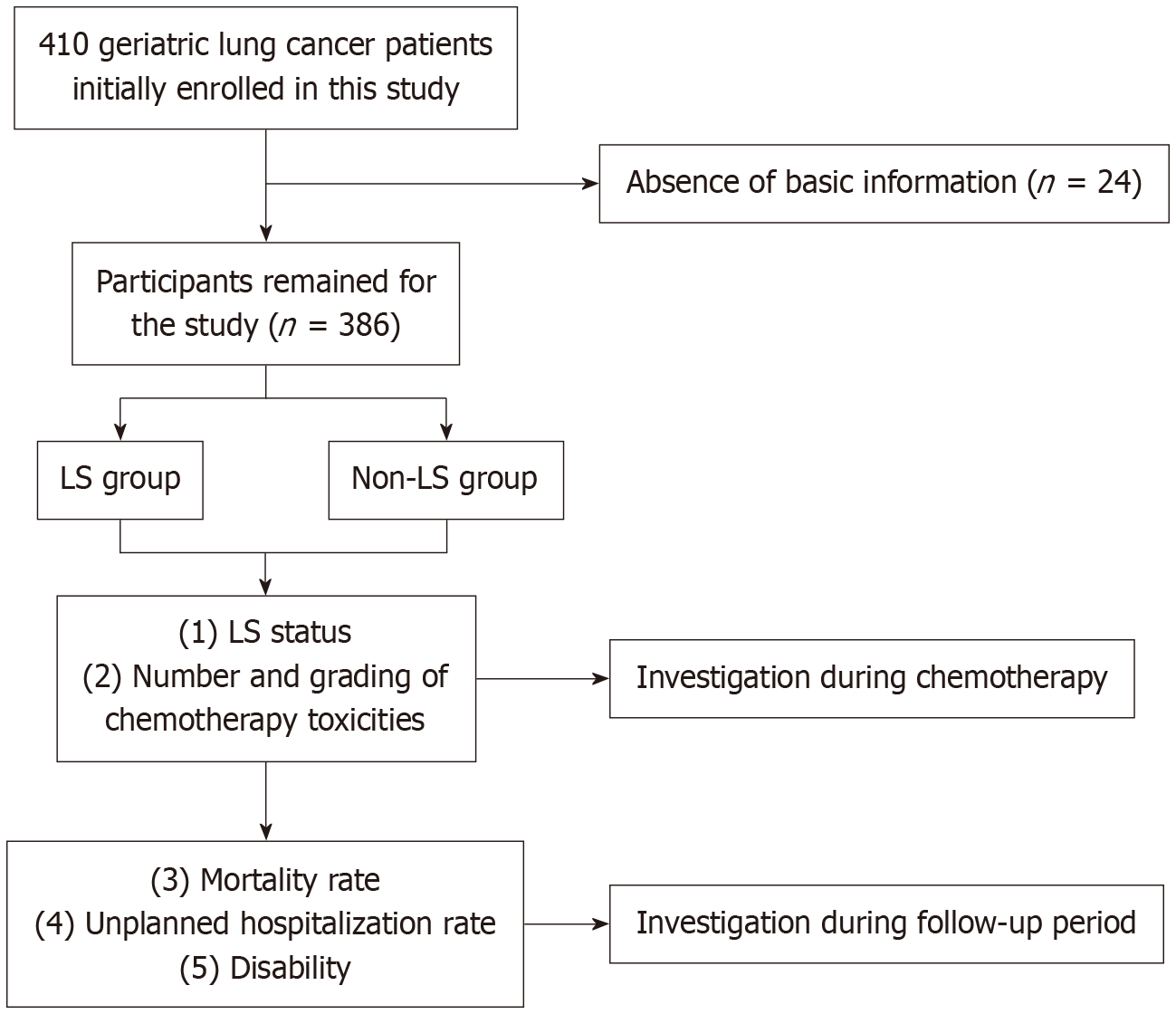
The data of 386 patients were finally completed (Figure 1). Among them, 167 were male, and 219 were female. Patients’ age ranged from 60 years to 76 years, with a mean age of 69.4 ± 7.6 years. In terms of education level, 168 patients had junior high school education or below, and 218 had higher education or above. Regarding living status, 255 patients lived with cohabitants, and 131 lived alone. As for the place of residence, 145 patients lived in rural areas, and 241 lived in towns. For tumor stage, 184 patients were in stage II, and 202 were in stage III. Before chemotherapy, 161 patients had LS, and 225 did not.
The presentation of these baseline characteristics, including sociodemographic factors such as educational level, living status, and residency, serves two primary purposes. First, it allows for an assessment of the comparability between the two groups at baseline. The results demonstrate that there were no statistically significant differences between the LS and non-LS groups in terms of gender, age, education level, living status, residency, pathological type, or tumor stage (all P > 0.05), indicating that the groups were well-balanced in these respects. Second, describing these factors is crucial as they represent potential social determinants of health that may influence outcomes; their presentation here establishes a foundational context for interpreting the subsequent findings and confirms that any observed differences in outcomes are less likely to be confounded by these baseline variables. The only significant difference observed was, as expected, a markedly higher incidence of LS after chemotherapy in the LS group compared to the non-LS group (P < 0.001) (Table 1).
| Item | LS group (n = 161) | Non-LS group (n = 225) | χ2/t/Z | P value |
| Gender (M/F) | 57/104 | 110/115 | 4.783 | 0.058 |
| Age (mean ± SD), years | 69.2 ± 7.2 | 70.1 ± 6.9 | -0.894 | 0.423 |
| Educational level | 1.982 | 0.221 | ||
| Junior high school and below | 79 (48.9) | 89 (39.6) | ||
| High school and above | 82 (51.1) | 136 (60.4) | ||
| Living status | 4.894 | 0.094 | ||
| With cohabitants | 98 (60.9) | 157 (69.8) | ||
| Live alone | 63 (39.1) | 68 (30.2) | ||
| Residency | 2.567 | 0.223 | ||
| Rural | 53 (32.9) | 92 (40.9) | ||
| Towns | 108 (67.1) | 133 (59.1) | ||
| Pathological type | 1.434 | 0.323 | ||
| Adenocarcinoma | 122 (75.8) | 152 (67.6) | ||
| Squamous carcinoma | 39 (24.2) | 73 (32.4) | ||
| Staging of the tumor | 0.468 | 0.398 | ||
| Stage II | 73 (45.3) | 111 (49.3) | ||
| Stage III | 88 (54.7) | 114 (50.7) | ||
| LS after chemotherapy | 78.066 | < 0.001 | ||
| Yes | 161 (100) | 87 (38.7) | ||
| No | 0 (0) | 138 (61.3) | ||
Data on the number and severity ofchemotherapy-related toxicities are presented in Table 2 with two primaryobjectives. First, it serves to quantify the comparative burden of treatmenttoxicity between patients with and without pre-existing LS. Second, it allowsfor an investigation into whether LS is associated with diminishedphysiological reserve, thereby manifesting as reduced tolerance to cytotoxictherapy. Our analysis confirmed that patients in the LS group suffered from asignificantly higher number and more severe gradings of toxic side effects thanthose in the non-LS group (all P < 0.05). This result substantiatesthe role of LS as a key risk factor for increased treatment toxicity, highlighting the need for tailored supportive care strategies for thisvulnerable subgroup.
| Group | n | Toxic side effects (mean ± SD) | Grading of toxic side effects | ||||
| Grade 1 | Grade 2 | Grade 3 | Grade 4 | Grade 5 | |||
| LS group | 161 | 16.1 ± 3.8 | 27 (16.7) | 36 (22.4) | 38 (23.6) | 36 (22.4) | 24 (14.9) |
| Non-LS group | 225 | 11.3 ± 4.1 | 52 (23.1) | 68 (30.3) | 75 (33.3) | 20 (8.9) | 10 (4.4) |
| t/Z | 14.127 | 3.234 | |||||
| P value | < 0.001 | 0.006 | |||||
During the follow-up period, 17 patients (10.6%) in the LS group and 7 patients (3.3%) in the non-LS group passed away. The mortality rate in the LS group was significantly higher than that in the non-LS group (χ2 = 5.456, P = 0.028).
During the follow-up period, the LS group experienced more severe incapacitation and a higher unplanned hospitalization rate than the non-LS group. The difference was statistically significant (P < 0.05) (Table 3).
| Group | n | Incapacitation | Unplanned hospitalization rate | ||
| Complete incapacitation | Moderate incapacitation | Non-incapacitation | |||
| LS group | 144 | 22 (15.3) | 39 (26.8) | 83 (57.9) | 54 (37.2) |
| Non-LS group | 218 | 7 (3.4) | 24 (10.8) | 187 (85.8) | 22 (10.1) |
| Z/χ2 | -5.454 | 20.113 | |||
| P value | < 0.001 | < 0.001 | |||
In this study, we investigated 386 elderly lung cancer patients. The results indicated that the incidence of LS in these patients before chemotherapy was 41.7% (161/386), and it significantly increased to 64.2% (248/386) after chemotherapy. A further comparison revealed that the post-chemotherapy LS incidence in the LS group was significantly higher than that in the non-LS group (100% vs 38.7%). This suggested that LS status was prevalent in elderly lung cancer patients before chemotherapy, and chemotherapy might further elevate the LS incidence. A study noted that the LS incidence among the elderly in Japanese communities was 11.6%, which was lower than our study results[23]. This difference might be attributed to the high catabolism of tumors, which puts the patient’s body in a state of microinflammation. Additionally, a series of symptoms that lead to a decrease in the patient’s physical function can affect the motor function of the musculoskeletal system, ultimately increasing the LS incidence[11]. Another previous study found that the LS incidence in elderly cancer patients was 55.7%[19]. In this study, the pre-chemotherapy LS incidence in elderly lung cancer patients was lower than that in elderly tumor patients, but the post-chemotherapy LS incidence was higher. This could be because LS is a dynamic process that changes with the environment, stress, disease, and treatment. Chemotherapeutic drugs usually have low selectivity. While killing tumor cells, they can damage normal organs or tissues, causing myelosuppression, toxic side effects, etc. Patients are prone to symptoms like fatigue, gastrointestinal reactions, sleep disorders, and emotional abnormalities[24]. Under the dual effects of chemotherapy stress and disease-related con
The detrimental impacts of LS on tumor patients are extensive and multifaceted, establishing it as a critical marker of patient vulnerability. Specifically, it can lead to a heightened risk of mortality, a higher incidence of postoperative complications, an exacerbation of radiotherapy-induced toxicity, and a diminished tolerance to treatment[11]. The findings of our longitudinal study provide robust evidence supporting this view, particularly in the context of che
Expanding beyond treatment toxicity, this study also showed that during the follow-up period, the mortality rate and unplanned hospitalization rate in the LS group were significantly higher, and there were more incapacitating conditions in the LS group than in the non-LS group. This consistent pattern across multiple short-term adverse outcomes suggested that elderly lung cancer patients with pre-chemotherapy LS were at a significantly higher short-term risk of these adverse events. The interplay between toxicities, functional decline, and hospitalization creates a vicious cycle: Chemotherapy-induced side effects can precipitate a decline in functional status, which in turn increases susceptibility to infections and other complications that lead to unplanned hospitalizations, thereby elevating mortality risk.
Studies have confirmed that LS worsens fatigue and sleep disorders in elderly cancer patients, which are key components of the incapacitation spectrum[16,25]. However, there is a lack of analysis on the association between LS and pre-chemotherapy adverse outcomes like death, incapacitation, and unplanned hospitalization in lung cancer patients. Consequently, we recommend LS assessment for cancer patients undergoing or about to start chemotherapy, and implementing intervention measures before and/or during chemotherapy to prevent, slow, or even reverse the pro
This study has some limitations. Firstly, as a single-center study with a relatively limited sample size, the findings may be subject to selection bias and may not fully represent the broader population of patients with similar characteristics. Future multicenter studies with larger and more diverse cohorts are needed to enhance the generalizability of the results. Second, the relatively short observation period limited the ability to assess long-term outcomes and the sustained effects of the intervention. Extended follow-up studies would help clarify the long-term prognostic implications and durability of the observed effects. In addition, potential confounding factors, such as variations in patients’ comorbidities, treatment adherence, and lifestyle behaviors, may have influenced the outcomes despite efforts to control for them statistically. Future research incorporating longitudinal designs and more comprehensive covariate control is warranted to further validate these findings.
In summary, pre-chemotherapy LS is increasingly prevalent among elderly lung cancer patients, and its incidence rises further after chemotherapy. Elderly lung cancer patients with pre-chemotherapy LS experience numerous and severe toxic side effects during chemotherapy. They also face a high risk of death, severe incapacitation, and a high rate of unplanned hospitalization during follow-up. Building on the findings of this study, which established a link between pre-chemotherapy LS and short-term adverse outcomes in elderly lung cancer patients, several critical avenues for future research are proposed. First, interventional studies are warranted to investigate whether targeted pre-habilitation programs - focusing on improving MF, balance, and nutritional status - can effectively mitigate the risk of chemotherapy toxicity, functional decline, and unplanned hospitalization in this vulnerable population. Second, future work should aim to develop and validate a comprehensive risk prediction model that integrates LS severity with other geriatric assessment parameters and tumor-specific factors to better stratify patients and personalize treatment strategies. Lastly, longitudinal studies with extended follow-up are needed to elucidate the long-term impact of LS on overall survival, quality of life, and sustained disability beyond the immediate treatment period.
We also appreciate all participants for their time and effort.
| 1. | Wu JW, Zhang J. [A new system for tumor evaluation and decision-making in elderly patients with cancer]. Zhongguo Zhongliu Linchuang. 2022;49:449-454. [DOI] [Full Text] |
| 2. | Qiu H, Cao S, Xu R. Cancer incidence, mortality, and burden in China: a time-trend analysis and comparison with the United States and United Kingdom based on the global epidemiological data released in 2020. Cancer Commun (Lond). 2021;41:1037-1048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 852] [Cited by in RCA: 719] [Article Influence: 143.8] [Reference Citation Analysis (0)] |
| 3. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69214] [Article Influence: 13842.8] [Reference Citation Analysis (18)] |
| 4. | Chen X, Giles J, Yao Y, Yip W, Meng Q, Berkman L, Chen H, Chen X, Feng J, Feng Z, Glinskaya E, Gong J, Hu P, Kan H, Lei X, Liu X, Steptoe A, Wang G, Wang H, Wang H, Wang X, Wang Y, Yang L, Zhang L, Zhang Q, Wu J, Wu Z, Strauss J, Smith J, Zhao Y. The path to healthy ageing in China: a Peking University-Lancet Commission. Lancet. 2022;400:1967-2006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 589] [Article Influence: 147.3] [Reference Citation Analysis (0)] |
| 5. | Hirano K, Imagama S, Hasegawa Y, Ito Z, Muramoto A, Ishiguro N. The influence of locomotive syndrome on health-related quality of life in a community-living population. Mod Rheumatol. 2013;23:939-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 6. | Akahane M, Maeyashiki A, Tanaka Y, Imamura T. The impact of musculoskeletal diseases on the presence of locomotive syndrome. Mod Rheumatol. 2019;29:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 7. | Nishizawa A, Katsuhira J, Watanabe M, Fujii T, Yoshimoto T, Oka H, Matsudaira K. Relationship between the locomotive syndrome and kinetic and kinematic parameters during static standing and level walking. Gait Posture. 2022;93:146-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 8. | Hirano K, Imagama S, Hasegawa Y, Wakao N, Muramoto A, Ishiguro N. Effect of back muscle strength and sagittal spinal imbalance on locomotive syndrome in Japanese men. Orthopedics. 2012;35:e1073-e1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Tokida R, Ikegami S, Takahashi J, Ido Y, Sato A, Sakai N, Horiuchi H, Kato H. Association between musculoskeletal function deterioration and locomotive syndrome in the general elderly population: a Japanese cohort survey randomly sampled from a basic resident registry. BMC Musculoskelet Disord. 2020;21:431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, Schneider SM, Sieber CC, Topinkova E, Vandewoude M, Visser M, Zamboni M; Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48:601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2270] [Cited by in RCA: 1799] [Article Influence: 257.0] [Reference Citation Analysis (0)] |
| 11. | Hirahata M, Imanishi J, Fujinuma W, Abe S, Inui T, Ogata N, Iimuro S, Fujita R, Sato K, Tokizaki T, Matsuyama T, Kawano H. Cancer may accelerate locomotive syndrome and deteriorate quality of life: a single-centre cross-sectional study of locomotive syndrome in cancer patients. Int J Clin Oncol. 2023;28:603-609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 12. | Yurube T, Ito M, Takeoka T, Watanabe N, Inaoka H, Kakutani K, Kuroda R, Nishida K. Possible Improvement of the Sagittal Spinopelvic Alignment and Balance through "Locomotion Training" Exercises in Patients with "Locomotive Syndrome": A Literature Review. Adv Orthop. 2019;2019:6496901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 13. | Villalobos M, Siegle A, Hagelskamp L, Jung C, Thomas M. Communication along Milestones in Lung Cancer Patients with Advanced Disease. Oncol Res Treat. 2019;42:41-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Li Y, Yan B, He S. Advances and challenges in the treatment of lung cancer. Biomed Pharmacother. 2023;169:115891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 284] [Reference Citation Analysis (0)] |
| 15. | Islam KM, Anggondowati T, Deviany PE, Ryan JE, Fetrick A, Bagenda D, Copur MS, Tolentino A, Vaziri I, McKean HA, Dunder S, Gray JE, Huang C, Ganti AK. Patient preferences of chemotherapy treatment options and tolerance of chemotherapy side effects in advanced stage lung cancer. BMC Cancer. 2019;19:835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 16. | Yang MY, Chen Y, Su H, Lv Y, Yang YL. The prevalence and correlation of cancer-related fatigue and locomotive syndrome in geriatric cancer patients. PLoS One. 2025;20:e0319511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 17. | Oncology Society of Chinese Medical Association. [Chinese Medical Association guideline for clinical diagnosis and treatment of lung cancer (2024 edition)]. Zhonghua Zhong Liu Za Zhi. 2024;46:805-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 18. | Seichi A, Hoshino Y, Doi T, Akai M, Tobimatsu Y, Iwaya T. Development of a screening tool for risk of locomotive syndrome in the elderly: the 25-question Geriatric Locomotive Function Scale. J Orthop Sci. 2012;17:163-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 393] [Cited by in RCA: 319] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 19. | Yang YL, Su H, Lu H, Yu H, Wang J, Zhou YQ, Li L, Chen Y. Current status and risk determinants of locomotive syndrome in geriatric cancer survivors in China-a single-center cross-sectional survey. Front Public Health. 2024;12:1421280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Yang YL, Wang HH, Su H, Lu H, Yu H, Wang J, Zhou YQ, Li L, Chen Y. Reliability and validity tests of the Chinese version of the Geriatric Locomotive Function Scale (GLFS-25) in tumor survivors. Heliyon. 2024;10:e29604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 21. | Basch E, Becker C, Rogak LJ, Schrag D, Reeve BB, Spears P, Smith ML, Gounder MM, Mahoney MR, Schwartz GK, Bennett AV, Mendoza TR, Cleeland CS, Sloan JA, Bruner DW, Schwab G, Atkinson TM, Thanarajasingam G, Bertagnolli MM, Dueck AC. Composite grading algorithm for the National Cancer Institute's Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Clin Trials. 2021;18:104-114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 155] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 22. | Dos Santos Barros V, Bassi-Dibai D, Guedes CLR, Morais DN, Coutinho SM, de Oliveira Simões G, Mendes LP, da Cunha Leal P, Dibai-Filho AV. Barthel Index is a valid and reliable tool to measure the functional independence of cancer patients in palliative care. BMC Palliat Care. 2022;21:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 23. | Encho H, Uchida K, Nakamura J, Mizuta S, Akisue T, Kowa H, Ono R. Association between locomotive syndrome and anemia among community-dwelling older adults. Geriatr Gerontol Int. 2023;23:426-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 24. | Kawano H, Hirahata M, Imanishi J. Locomotive syndrome in cancer patients: a new role of orthopaedic surgeons as a part of comprehensive cancer care. Int J Clin Oncol. 2022;27:1233-1237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 25. | Yang YL, Su H, Lu H, Wang J, Zhou YQ, Li L, Yu H, Lv Y, Chen Y. The effect of locomotive syndrome on the trajectory of sleep disturbance in geriatric oncology inpatients. Front Oncol. 2025;15:1440747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |