©The Author(s) 2025.
World J Orthop. Mar 18, 2025; 16(3): 104451
Published online Mar 18, 2025. doi: 10.5312/wjo.v16.i3.104451
Published online Mar 18, 2025. doi: 10.5312/wjo.v16.i3.104451
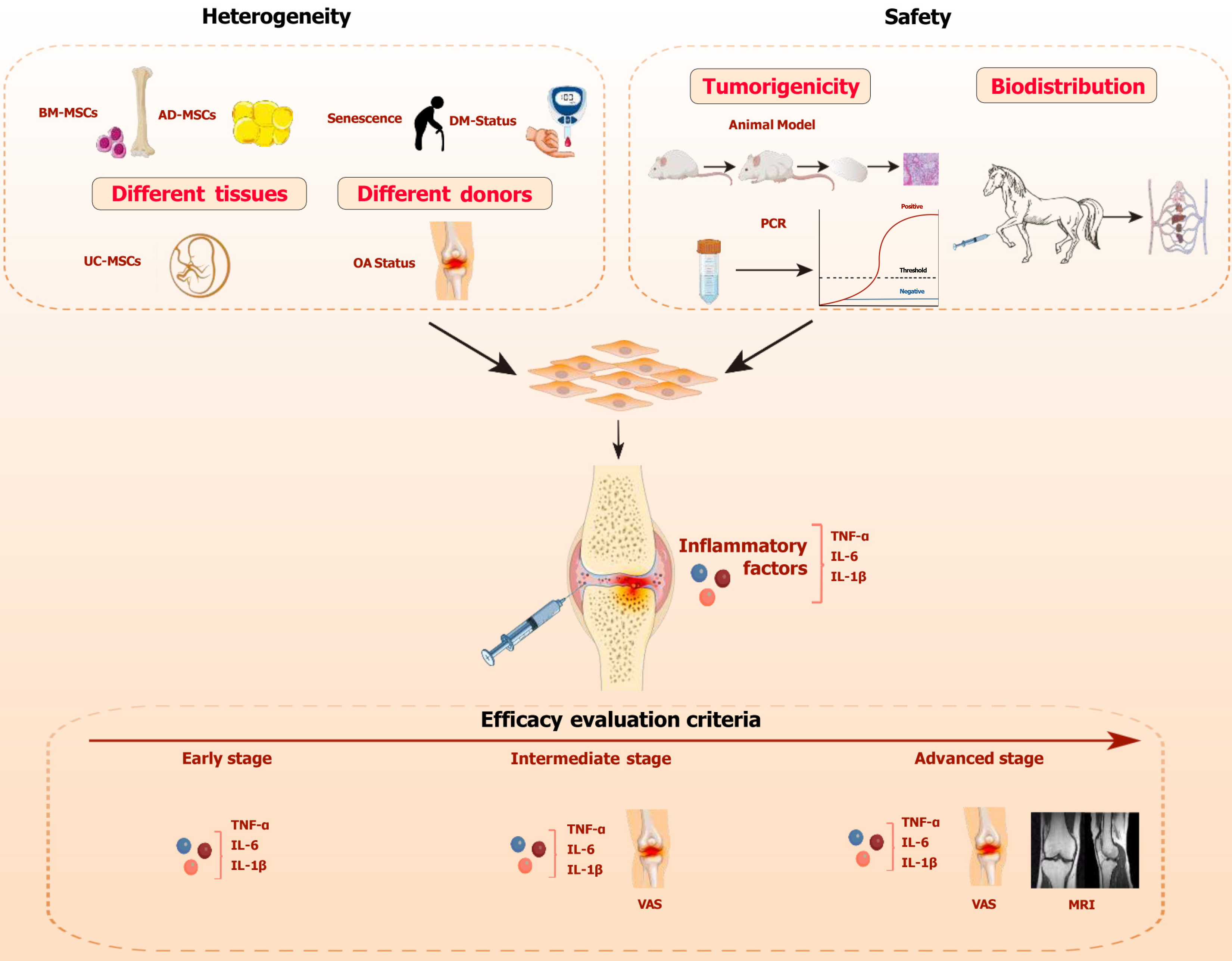
Figure 1 The key considerations for mesenchymal stem cell therapy in the treatment of osteoarthritis.
The dashed box highlights three key factors to consider: The heterogeneity of mesenchymal stem cell (MSCs), the safety of MSCs, and the standards for evaluating their efficacy. Heterogeneity includes tissue heterogeneity and donor heterogeneity. Safety encompasses tumorigenicity and biodistribution. The horizontal red arrow indicates the progression timeline of osteoarthritis, with different efficacy evaluation standards applied at each stage. MSC: Mesenchymal stem cell; OA: Osteoarthritis; BM-MSC: Bone-marrow-mesenchymal stem cell; AD-MSC: Adipose-tissue-derived mesenchymal stem cell; UC-MSC: Umbilical-cord-derived mesenchymal stem cell; IL: Interleukin; TNF-α: Tumor necrosis factor-alpha; VAS: Visual analog scale; MRI: Magnetic resonance imaging.
- Citation: Wang YC, Cheng JB, Feng ML. Requirements for standardizing the assessment of mesenchymal stem cell therapy and its effects on osteoarthritis. World J Orthop 2025; 16(3): 104451
- URL: https://www.wjgnet.com/2218-5836/full/v16/i3/104451.htm
- DOI: https://dx.doi.org/10.5312/wjo.v16.i3.104451