Core tip: The reverse cholesterol transport pathway played with high-density lipoprotein (HDL) has several potential antiatherogenic properties. Both ATP-binding cassette (ABC) A1 and ABCG1 are lipid transporters and have been involved in mediating cholesterol effluxes from cells in the presence of HDL or apoA-I, and they exerted antiatherosclerotic effects. As a therapeutic approach for increasing HDL cholesterol levels, much focus has been placed on increasing not only HDL cholesterol levels, but also HDL-biological functions. Reconstituted HDL and apoA-I mimetics have significant atheroprotective potential, as it uses lipid transporter-targeting agents, and may prove to be a novel therapeutic tool for atherosclerotic cardiovascular diseases.
INTRODUCTION
High-density lipoprotein (HDL) cholesterol is widely known as “good cholesterol”, because various previous studies have found a negative correlation between the risk of cardiovascular events and serum HDL cholesterol levels[1]. However, this is still controversial whether the association is the cause or just only an ensuing symptom of a general atherosclerotic damage. HDL has several potential for antiatherogenic properties, for instance, cholesterol is transported from peripheral tissues such as the cells in the arterial walls to the liver by HDL particles, where it is used for a composition of lipoproteins and a synthesis of bile acids, steroid hormones, or fat-soluble vitamins[1]. Whereas, low-HDL cholesterolemia is often observed as a characterized component of metabolic syndrome, such as in people who are overweight or obese, those with glucose intolerance or have obvious diabetes, those with hypertriglyceridemia, and those with high blood pressure, each of which conditions contribute to the cause of atherosclerosis[2].
METABOLISM AND THE FUNCTIONS OF HDL
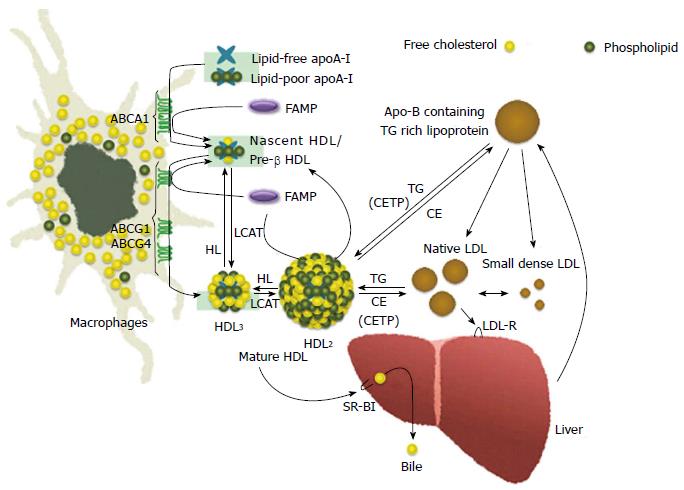
Although HDL is a lipoprotein when isolated by ultracentrifugation has a density in the range of 1.063-1.21 g/mL (HDL2, 1.063 < d < 1.125 g/mL; HDL3, 1.125 < d < 1.21 g/mL), HDL composes a heterogeneous group of particles that differ in density, size, composition of apolipoprotein (apo) or lipid, and electrophoretic mobility[3]. It is possible to separate HDL into two major subfractions on the basis of electro-mobility by electrophoresis; the major subfraction has the same mobility as alpha HDL, whereas the other subfractions migrate similar to pre-beta HDL, in addition the majority of HDL particles in human plasma are alpha HDL, and pre-beta HDL represents only 2%-14% of all apoA-I[4,5] (Figure 1).
Figure 1 Illustration of high-density lipoprotein metabolism and suggested function of fukuoka university ApoA-I mimetic peptide in high-density lipoprotein metabolism.
ABC: ATP-binding cassette transporter; TG: Triglyceride; CE: Cholesteryl ester; CETP: Cholesteryl ester transfer protein; HL: Hepatic lipase; apo: Apolipoprotein; HDL: High-density lipoprotein; FAMP: Fukuoka university ApoA-I mimetic peptide; CETP: CE transfer protein; SR-BI: Scavenger receptor BI; LDL: Llow-density lipoprotein; LCAT: Lecithin cholesterol acyltransferase; LDL-R: Low-density lipoprotein receptor; SR-BI: Scavenger receptor class B, type I; FAMP: Fukuoka University ApoA-I mimetic peptide.
HDL metabolism has the complicated mechanisms in association with several HDL-related genes such as various enzymes and protein, lipids, receptors, or transporters and its synthesis involves a complex pathway. The underlying genetic deficiency in many cases of primary low-HDL cholesterolemia are not clearly understood, however mutations in three pivotal genes as apoA-I, lecithin: cholesterol acyltransferase, and ATP-binding cassette transporter (ABC) A1, are associated with reducing serum HDL cholesterol levels, furthermore some of these genes’ mutations are also closely correlated with an increased risk of premature atherosclerosis and coronary artery disease (CAD)[6].
TANGIER DISEASE, A FAMILIAL HDL DEFICIENCY
Tangier disease (TD) is the most severe form of HDL deficiency, which was first described by Fredrickson et al[7]. The biological hallmarks of TD patients’ plasma are a defect of HDL cholesterol, reduced low-density lipoprotein (LDL) cholesterol levels, and moderate increased triglyceride. The plasma apoA-I concentration in TD is markedly decreased to approximately 1%-3% of normal. TD is a very rare autosomal recessive disorder which is characterized by the almost absence serum apoA-I and HDL cholesterol levels. Furthermore, cholesteryl ester (CE) accumulates in many macrophage enriched tissues, such as tonsils, spleen, liver, lymph nodes, peripheral nerves, thymus, and also arterial walls. Clinical symptoms among homozygotes patients include hepatosplenomegaly, hyperplastic orange-yellow tonsils, corneal opacification, and premature CAD and atheosclerosis in a half of cases as well as relapsing peripheral neuropathy due to CE deposition in macrophages and Schwann cells[7-9].
In 1999, a cause of TD was found in a defect of the ABCA1 (formerly ABC1) gene[1,10,11] that is located on chromosome 9q31. This gene comprises 50 exons that span a region of approximately 149 kb[12,13]. ABCA1 has been identified as an important gene for regulating cellular cholesterol homeostasis and serum HDL cholesterol levels, which is defect in patients with TD. ABCA1 gene mutations cause gene dose-dependent decreases in serum HDL cholesterol levels and a decreased capacity of skin fibroblasts and monocyte-derived macrophages releasing cholesterol in the presence of extracellular apolipoproteins in TD patients and their heterozygous relatives[1,10,11,14,15].
A transmembrane protein, ABC transporter facilitates to carry out the specific substrates across cell membranes in an ATP-dependent manner. ABCA1 is a member of the ABC transporter superfamily comprised 48 human transporters, and the superfamily is divided into seven subfamilies, including from half- to full-transporters, designated ABCA-ABCG. These transporters are integral membrane proteins carrying out various substrates, including lipids, ions, peptides, amino acids, carbohydrates, vitamins, glucuronides, glutathione conjugates, and xenobiotics[16,17]. ABCA1 is expressed in various organs in human, particularly the highest expression levels are existed in the placenta, liver, lung, adrenal glands, and fetal tissues[18].
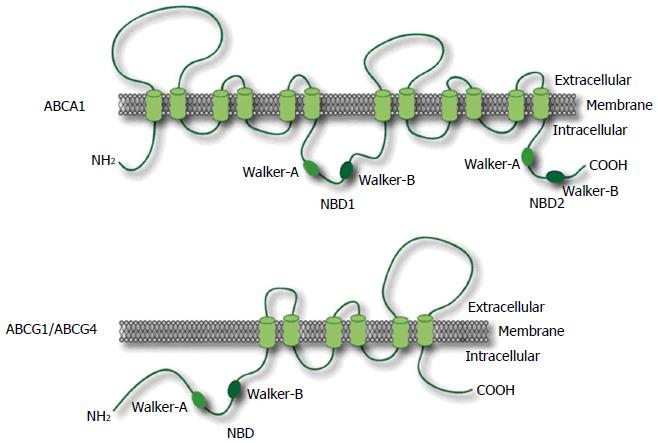
ABC transporter superfamilies are defined by the presence of similar nucleotide binding domains (NBD) to interact with ATP. These domains have two conserved peptide motifs, Walker-A and Walker-B, which are found in many proteins that utilize ATP[16,19] (Figure 2).
Figure 2 Secondary structures of the ATP-binding cassette transporter A1, ATP-binding cassette G1 and ATP-binding cassette G4 transporters.
The ATP-binding cassette transporters (ABC)A1 transporter comprises 2201 amino acids with two transmembrane domains comprising two nucleotide binding domains (NBD-1 and -2) and six transmembrane helices, which contain two conserved peptide motifs, Walker-A and -B. ABCA1 is characterized as two large extracellular loops and N-terminus oriented towards the cytosol. Both ABCG1 and ABCG4 proteins have one transmembrane domain comprising six transmembrane helices and one NBD that contains two conserved peptide motifs, Walker-A and Walker-B.
ABC TRANSPORTER ROLES IN HDL METABOLISM
ABCA1 transporter functions and their relationships with HDL metabolism
ABCA1 proteins transport phospholipids (PLs) and cholesterol from the membranous inner leaflet to the outer leaflet, subsequently lipid-poor or lipid-free apoA-I takes up this transported cholesterol and PLs by ABCA1 to form nascent HDL[20]. ABCA1 is localized at the plasma membrane and intracellular compartments, where it can potentially facilitate lipid transport to either cell surface-bound[21] or internalized apolipoproteins[22].
HDL metabolism is composed of at least three different steps. As the first step, lipid-free or lipid-poor apoA-I removes free cholesterol from peripheral cells through ABCA1 transporter to form nascent-HDL. Second, nascent-HDL has a further lipidation, thereafter it grows to mature-HDL. Third, mature-HDL interacts with other apoB containing lipoproteins, such as intermediate density lipoprotein (IDL) and very-low-density lipoprotein (VLDL). Thus, ABCA1 is indispensable for the nascent-HDL formation, in addition it is also an important and essential molecule for the initial step of the reverse cholesterol transport (RCT).
Cultured blood monocyte-derived (mod)-macrophages from a healthy subject showed an approximately 125% increase in cholesterol efflux mediated lipid-free apoA-I, whereas it did not respond to apoA-I mediated efflux in macrophages from TD patients[23]. Although a lipid-free apoA-I showed an increase the cholesterol efflux mediated by in cultured mod-macrophages from healthy persons, the apoA-I did not elevate cholesterol efflux in mod-macrophages from TD patients. These results indicated that ABCA1 is a key molecule for apoA-I-specific cholesterol efflux pathway, but not basal efflux in macrophages.
Since ABCA1 plays an important role in mediating cholesterol and PL effluxes by lipid-free apoA-I, it is involved in a formation of discoidal HDL precursor, furthermore ABCA1 poorly interacts with HDL2 and HDL3. Patients with TD have extremely low levels of HDL cholesterol and they cannot compose nascent HDL particles due to a genetic defect in ABCA1 gene.
Disrupting the ABCA1 in mice resulted in HDL deficiency and impaired cholesterol transport similar to TD[24,25]. ABCA1 overexpression resulted in increased apoA-I-mediated cholesterol efflux in transgenic mice[26,27]. These results indicate that ABCA1 is an important gene in regulating circulating HDL cholesterol levels and cellular cholesterol homeostasis.
ABCG1 transporter functions and their relationships to HDL metabolism
ABCG1, formerly ABC8 is also a member of the ABC transporter family which has been mapped on chromosome 21q22.3[19,28-32]. ABCG1 is one of half-transporter that contains only one NBD and a transmembrane domain, in contrast to ABCA1[19,31] (Figure 2). Thus, ABCG1 may require a dimeric partner to become active with ABCG1 or ABCG4.
Although ABCA1 promotes cholesterol efflux to lipid-poor or lipid-free apoA-I, it only modestly induces lipid efflux of smaller particles, such as HDL3, and does not promote a cholesterol efflux of the larger HDL2 fraction[33,34]. It has been also shown by Wang et al[35] that ABCG1 and ABCG4 contributed to HDL2- and HDL3-mediated cholesterol effluxes and had an important function related to HDL lipidation[35-37].
Administering a high-cholesterol, high-fat diet to ABCG1 knock-out mice resulted in a large amount of lipid accumulation in macrophages, whereas overexpression of human ABCG1 gene was able to protect a dietary fat-induced lipid accumulation in murine model[38]. Moreover, It was shown by Mauldin et al[39] that reduced function of ABCG1 facilitated foam cell formation in diabetes mice[39]. Transplanting bone marrow from ABCG1-deficient (ABCG1-/-) mice into LDL receptor-deficient mice, a model of familial hypercholesterolemia, produced contrasting effects on the formation of atherosclerotic lesion[40-42]. In contrast to these report, decreased lesion size and formation were observed in the absence of macrophages from ABCG1-deficient mice[41,42], and whole body ABCG1 expression protected against the development of early atherosclerotic plaque[43]. However, it remains unclear that the physiological roles of ABCG1 and its contribution to atherosclerotic progression in humans. In addition, ABC transporters such as ABCG1 and ABCG4, but not ABCA1, are not only responsible for passive and nonspecific efflux pathway but also mature HDL-mediated cholesterol efflux, which are spherical and transport almost all HDL cholesterol[35,37].
ROLES OF ABCG5 AND ABCG8 TRANSPORTER
ABCG5 and ABCG8 are half-transporters as well as ABCG1 that function together as a heterodimer, and mutations in either of these genes can cause sitosterolemia which is a rare autosomal, recessively inherited disorder, characterized by premature atherosclerosis and xanthomas[44-47]. These transporters mediate the sterols efflux including cholesterol and plant sterols from enterocytes return into the intestinal lumen and their excretion into the bile[44,48]. Accordingly, they protect the lipid accumulation in the body and augment RCT system. In animal model, ABCG5 and ABCG8 deficient mice have been shown to reduce a secretion of cholesterol in the bile and elevate sterol absorption[49], on the other hand ABCG5 and ABCG8 genes-overexpressed mice promotes cholesterol secretion in the bile, decreases cholesterol absorption from diet, and increases neutral sterol excretion in the feces[50]. Liver X receptor (LXR) agonists promote the cholesterol efflux by the upregulation of ABCA1 and ABCG1, and also stimulate ABCG5 and ABCG8 which accelerate direct HDL transport of intestine into the lumen, thus these genes also play an important role in the RCT system and their enhancement by LXR agonists prevent an atherosclerotic development[51].
MECHANISMS OF ABCA1 AND ABCG1 GENE REGULATION
ABCA1 gene expression and cellular efflux of cholesterol are enhanced by cholesterol[15,18], oxysterols[52], retinoids[53], and cAMP analogs[15,54]. The ABCA1 gene promoter has been analyzed[13,52]. Both oxysterols and retinoids are ligands for the nuclear transcription factor, LXRα/β and retinoid X receptor-alpha (RXRα), respectively, which have been identified as an enhancer of ABCA1 gene expression[52,53,55,56]. It is present in dimeric form of LXR and RXR as active transcriptional heterodimers that preferentially bind to responsive elements in the ABCA1 gene promoter[13,57]. LXRα/β and RXRα bind to the specific responsive element, called direct repeat 4 (DR4) element within the ABCA1 promoter, which is characterized by two direct hexameric repeats separated by four nucleotides, thereafter they are activated by oxysterols and retinoids[58,59]. ABCA1 transcription are activated to bind either one or both ligands. Treatment with either a ligand of LXRα/β or RXRα enhances cellular ABCA1 expression, furthermore their combination treatment has a marked synergistic effect[60].
Since peroxisome proliferator activating receptor (PPAR)-α and -γ agonists such as fibrates and thiazolidine derivative (TZD) upregulate LXR mRNA expression, the activation of PPARs indirectly enhances a transcription activity of ABCA1 via LXR in cultured cells. In contrast, it is already known the zinc finger protein ZNF202 transcription factor as a major transcriptional repressor for ABCA1. In addition to the factor ZNF202, unsaturated fatty acids, but not saturated one, drastically suppress ABCA1-mediated cholesterol effluxes from macrophages by which they antagonize the binding of specific agonist, oxysterol to LXR[61,62]. Moreover, various transcription factors, such as upstream stimulatory factor (USF)1, USF2, Fra2, and Sp3, also have the potential to repress the ABCA1 transcription[63].
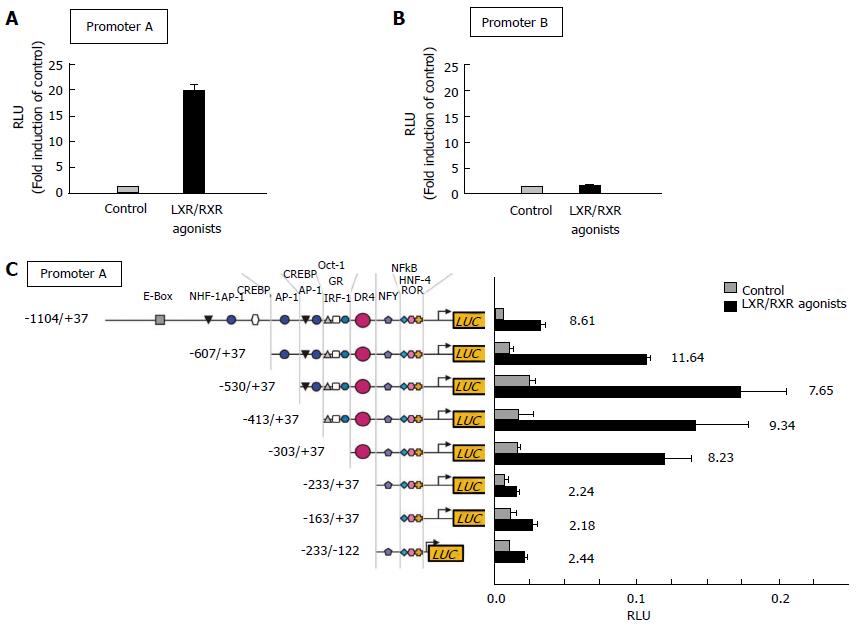
The ABCG1 gene has a promoter upstream of exon 1 and another intron promoter, which encodes several transcripts[64-66]. Our previous study demonstrated that LXR activation drastically increased the ABCG1 promoter activity (Promoter-A) located upstream of exon 1 as well as the ABCA1 gene (Figure 3A). On the other hand, the activity of ABCG1 promoter-B located within intron 4 was not changed by an activation of LXR (Figure 3B)[62]. These results indicate that the gene transcription of exon 5 and subsequent exons might be also regulated, at least in part, by the ABCG1 promoter-A.
Figure 3 Response of liver X receptor and retinoid X receptor agonists to human ATP-binding G1 promoter activities in RAW264 cells.
A: Human wild-type ATP-binding cassette transporter G1 (ABCG1) promoter-A located upstream of exon 1; B: Human wild-type ABCG1 promoter-B located upstream of exon 5; C: ABCG1 promoter (promoter-A; upstream of exon 1) vectors that contain a truncated 5′-region of the ABCG1 gene. After transfection, cells were incubated with or without agonists of LXR [22(R)-hydroxycholesterol, 10 μmol/L] and RXR (9-cis-retinoic acid, 10 μmol/L). Results are expressed as mean ± SD. Graphs modified from the paper by Uehara et al[62]. LXR: Liver X receptor; RXR: Retinoid X receptor; RLU: Relative luciferase units .
Electrophoretic mobility shift assay was done to confirm these findings, and it showed the existence of DNA-binding nuclear receptors on extracted ABCG1 promoter-A having DR4 element. As would be expected from these finding, only the ABCG1 promoter-A contained a DR4 element, but not promoter-B, which is required for binding to LXRα/RXR. In fact, a promoter response to ligands of LXR/RXR was totally abolished in the mutated ABCG1 promoter lacked an active DR4 element[62] (Figure 3C).
ABCG1 SINGLE NUCLEOTIDE POLYMORPHISMS
It remains unclear whether ABCG1 itself contributes to circulating lipid levels, such as HDL cholesterol and arterial plaque regression in humans. There have been only five reports on ABCG1 polymorphisms. Our previous study was the first regarding an ABCG1 polymorphism, which appeared to be a potent functional ABCG1 polymorphism located in the promoter region[67-71]. The ABCG1 promoter -257T>G polymorphism, rs1378577, -394 T/G from the transcription start site (NM_207627.1:c. -394T>G), -134 T/G from exon 1 (NM_207627.1) is a single nucleotide mutation (SNP) on the ABCG1 promoter region upstream of exon 1, which was reported to be a functional promoter with an LXR-responsive element[62,67].
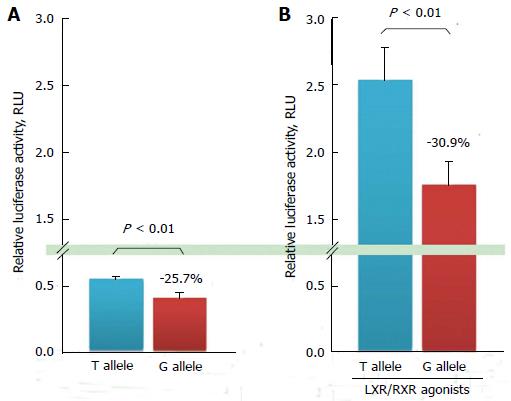
To investigate whether this promoter polymorphism influenced gene transcriptional activity, in vitro luciferase reporter gene assays were performed after transient transfection in cultured cells. In these experiments, the amount of luciferase activity was 25.7% higher in T allelic sequence containing construct than that in G allelic one on ABCG1 promoter-A; these responses were significantly different (Figure 4A). ABCG1 promoter activity induced by LXR and RXR agonists increased by 4.6-fold, and the amount of luciferase produced by the construct containing the T allelic sequence was 30.9% higher than that produced by the construct containing the G allele, which was also significantly different as well as in the absence of LXR/RXR agonists (Figure 4B). The transcription activity in the T allelic sequence was significantly higher than that in the G allelic sequence on ABCG1 promoter-A.
Figure 4 In vitro promoter activity assay for ATP-binding G1 promoter-A.
ATP-binding cassette transporter G1 (ABCG1) promoter construct with a −257T/G mutation, −394 T/G from the transcription start site (NM_207627.1: c.-394T > G) on ABCG1 promoter-A, which is reported to be a functional promoter with an LXR-responsive element. A: ABCG1 transcription activity on a construct that contains the T or G allelic sequence; B: ABCG1 transcription activity induced by 5 μmol/L of T0901317 (T0) and 9-cis-retinoic acid (9cisRA) on constructs that contain the T or G allelic sequence. Results are expressed as mean ± SD. Graphs are modified from the paper by Furuyama et al[67]. ABC: ATP-binding cassette; RXR: Retinoid X receptor; LXR: Liver X receptor.
Furthermore, the ABCG1 promoter showed increased activity via stimulation by LXR and RXR, and a similar genotype-dependent effect on ABCG1 gene transcription under these conditions was identified. These results suggest that the ABCG1 promoter polymorphism might be an isolated regulating factor for ABCG1 gene transcription activity, independent of LXR and RXR.
We genotyped 109 Japanese male CAD patients for the ABCG1 promoter SNP. This polymorphism was associated with CAD severity in Japanese men, but not with changes in lipid levels under fasting conditions in a case control study. Logistic regression analysis showed that there was an interaction between the ABCG1 promoter genotype and CAD severity.
Genotype frequencies were grouped on the basis of whether patients had multi- or single-vessel CAD. The adjusted relative risk associated with the G allele (assuming an additive effect) in a matched-pair analysis was 2.1 for multi-vessel CAD compared with single-vessel CAD and 3.5 for the G/G and T/G genotypes compared with T/T (assuming a dominant effect of the G allele)[67]. These results were consistent with the proposition that the variations for ABCG1 gene might make a contribution to interindividual variability in susceptibility or severity of atherosclerotic changes.
ABCG1 expression levels in atherosclerotic tissues might be lower among those with the G allele and may be associated with a mechanism for an increased incidence of atherosclerosis in these individuals. These results were similar to a previous study by Baldán et al[72] of transgenic mice in whom the ABCG1 gene was deleted[73]. Furthermore, a recent study regarding ABCG1 as a candidate gene with possible important antiatherogenic properties also illustrates the current interest in this transporter.
HDL-TARGETING THERAPY FOR ATHEROSCLEROSIS
Inhibiting scavenger receptor BI (SR-BI), CE transfer protein (CETP) or PL transfer protein, and an activating ABCA1 or apoA-I elevate HDL cholesterol levels. However, it is uncertain whether the effects of these interventions on atherosclerosis are consistence with the results of studies with animal models and inborn human HDL metabolism errors. Although it has not found a such small molecule which strongly promotes apoA-I production, one possible candidate molecule is LXR agonist which increase HDL cholesterol levels via upregulation of ABCA1 and ABCG1 expressions. Unfortunately, previous study has shown that concurrent with an activation of RCT, the agonist induces hypertriglycemia consequent on increasing hepatic VLDL production.
As a therapeutic approach for increasing HDL levels, much research has focused both increasing HDL cholesterol levels and on enhancing HDL biochemical functions. HDL therapies that used injections of reconstituted HDL, apoA-I mimetics, or full-length apoA-I are remarkably effective[74,75]. Nissen et al[75] showed that in humans, intravenous administation of ETC-216, an apoA-I-Milano complexed with phospholipids, produced a significant regression of coronary atherosclerotic plaques as determined by intravascular ultrasound (IVUS). After infusing ETC-216, regression of coronary atherosclerosis was accompanied by reverse remodeling of the external elastic membrane and with no changes in luminal dimensions as assessed by IVUS analyses[76].
Reconstituted HDL (rHDL), a complex of apoA-I or apoA-I mimetics with PL, must be shaped as disc, and it may be a suitable administration in patients with atherosclerotic plaque and TD. ABCA1 plays an important role for apoA-I-mediated cholesterol efflux in macrophages, and thereby is involved in discoidal HDL precursor formation. Mature HDL particles shaped spherical induce cholesterol effluxes by other transporters such as ABCG1 and ABCG4, rather than ABCA1[35]. We previously established a discoidal rHDL, which was a complex of human serum-derived full length of apoA-I with PL, 1-palmitoyl-2-oleoylphosphatidylcholine (POPC)[77]. Interestingly, the apoA-I complex with a PL, a POPC/apoA-I disc, could take up cholesterol from macrophages in both normal subjects and TD patients.
Although studies on the use of apoA-I mimetic peptides (e.g., 4F and L37pA) are underway[78-80], none of these agents are currently available for clinical use. To develop a physiological HDL-generating apoA-I mimetic peptide that functions with ABCA1 transporter, different candidate peptides were synthesized by focusing on the amino acid sequence alignments of human apoA-I interactions with ABCA1. We recently established a novel short apoA-I mimetic peptide that comprised 24 amino acids and without phospholipids Fukuoka University ApoA-I Mimetic Peptide (FAMP), which retained the amphipathic helical structure of the 243-amino acid apoA-I and the ability to associate with lipids[81]. This was shown to enhance HDL function and suppress aortic plaque formation in apoE-knockout mice that were fed a high-fat diet. FAMP markedly increased pre-beta HDL formation as well as increased the overall cholesterol effluxes from peripheral tissues[82].
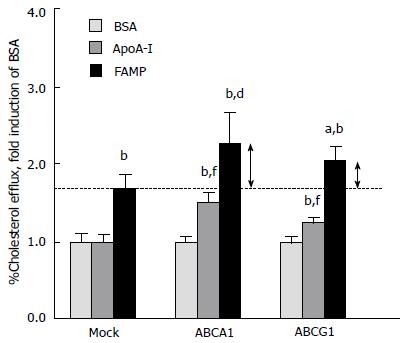
In contrast to apoA-I, FAMP-mediated cholesterol effluxes were not completely abolished under ABCA1-inactivated conditions, such as in cells treated with probucol, an ABCA1 antagonist, and Tangier macrophages. These results suggested that FAMP functioned in removing cholesterol through both the ABCA1 pathway and another specific pathway that must be dependent on ABCG1 transporters (Figure 1). In support of this, COS-7 cells that were transiently transfected with the ABCA1 and ABCG1 genes had significantly increased FAMP-mediated effluxes compared with mock transfection (Figure 5).
Figure 5 Fukuoka University apoA-I mimetic peptide effects on cellular cholesterol effluxes in cells that express ATP-binding A1 and ATP-binding G1.
COS-7 cells were transiently transfected with an empty vector (mock) or with human ATP-binding cassette transporter A1 (ABCA1) and ATP-binding cassette transporter G1 (ABCG1) cDNAs. Cholesterol efflux was determined after incubation with apoA-I or FAMP. Results are expressed as mean ± SD. aP < 0.05 vs FAMP in mock; bP < 0.01 vs BSA; dP < 0.01 vs FAMP in mock; fP < 0.01 vs apoA-I in mock. Graph modified from the paper by Uehara et al[82]. FAMP: Fukuoka University ApoA-I mimetic peptide; BSA: bovine serum albumin; apoA-I: apolipoprotein A-I; FAMP: Fukuoka University ApoA-I Mimetic Peptide..
Injections of HDL apo-A-I mimetics, apoA-I-Milano, and full-length apoA-I are effective both in vitro and in vivo. However, it remains unclear whether apoA-I or its mimetics actually enter atherosclerotic plaque lesions and remove cholesterol. ApoA-I may generate nascent, new HDL and reverse the macrophage foam cell phenotype.
We developed a novel PET tracer that was functionalized with DOTA and labeled with 68Ga to specifically image the status of atherosclerotic plaques. Atherosclerotic plaques and aortic atherosclerotic plaques show high uptake of this tracer, and this novel tracer provides for impressive in vivo imaging of an aortic plaque using PET/CT[83]. HDL-targeting therapy, including FAMP, may have tremendous atheroprotective potential and prove to be a new therapeutic tool for atherosclerotic cardiovascular disease. While most research has focused on the therapeutic use of HDL, an apoA-I mimetic peptide may also contribute to the development of a tool for plaque diagnosis.
CONCLUSION
The RCT pathway has several potential antiatherogenic properties. Both ABCA1 and ABCG1 are lipid transporters on plasma membrane that have been contributed in mediating effluxes of cholesterol and PLs from cells in the presence of lipid-poor or lipid-free apoA-I and HDL. As a therapeutic approach for increasing HDL levels, much research has focused both on increasing HDL cholesterol levels and on enhancing HDL biochemical functions. HDL therapies with reconstituted HDL, apoA-I mimetics, or full-length apoA-I am dramatically effective. In particular, a novel apoA-I mimetic peptide, FAMP, effectively removes cholesterol via specific ABCA1 and other transporters, such as ABCG1. FAMP has an antiatherosclerotic effect by enhancing biological HDL functions without changing circulating HDL cholesterol levels. These HDL-targeting therapies have significant atheroprotective potential, as they are lipid transporter-targeting agents. Thus, HDL-targeting therapy may prove to be a therapeutic tool for atherosclerotic cardiovascular diseases.
P- Reviewer: Albacker T, Biyik I, Corciu AI, Can M, Kato M, Kobza R, Latif N, Prella F S- Editor: Qi Y L- Editor: A E- Editor: Wu HL