Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.116780
Revised: December 22, 2025
Accepted: January 29, 2026
Published online: March 26, 2026
Processing time: 123 Days and 15.8 Hours
The relationship between trimethylamine N-oxide (TMAO), gut microbiome (GM), and major adverse cardiovascular events (MACE) has gained increasing interest in clinical research. TMAO, a metabolite formed by GM metabolism of dietary nutrients like choline and L-carnitine, has been implicated in the pa
Core Tip: Trimethylamine N-oxide (TMAO) is increasingly proposed as a prognostic biomarker and therapeutic target in cardiovascular disease, yet its clinical signal is strongly shaped by renal function and cardiometabolic comorbidity. We synthesize evidence linking TMAO to risk stratification, including incremental value beyond GRACE in acute coronary syndrome, highlight mechanistic advances from microbiota-targeted trimethylamine-lyase inhibitors, 3,3-dimethyl-1-butanol/fluoromethylcholine, that improve vascular and cardiac phenotypes in preclinical models, and outline a precision-nutrition framework that tailors TMAO-lowering strategies to individual producer metabotypes.
- Citation: Jaimez Alvarado S, Flores Enciso MF, Amedei A, Aguirre García MM. Gut microbiota-derived metabolite trimethylamine N-oxide in major adverse cardiovascular events: Mechanisms, risk assessment, and therapeutic strategies. World J Cardiol 2026; 18(3): 116780
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/116780.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.116780
Patients with a history of cardiovascular diseases (CVD) are at an increased risk of major adverse cardiovascular events (MACE), such as acute myocardial infarction (AMI), stroke, and heart failure (HF). These events are associated with increased hospitalization, high care costs, and finally impaired quality of life[1]. Worldwide, CVD accounts for more than 17.9 million deaths each year, 32% of all deaths with AMI and CVD responsible for approximately 85% of these fatalities[2].
Most CVD can be prevented by addressing environmental and behavioral risk factors, such as smoking, unhealthy eating habits, obesity, physical inactivity, alcoholism, and environmental pollution[2]. These factors directly affect both cardiovascular health and the composition and balance of gut microbiome (GM), suggesting an indirect but significant link between the environment, lifestyle, and cardiovascular risk. In this context, GM has emerged as a key factor in cardiovascular health. Through the production of metabolites such as trimethylamine N-oxide (TMAO), derived from nutrients like choline, L-carnitine, and betaine, elevated TMAO levels have been associated with an increased risk of MACE, positioning it as a potential prognostic biomarker in CVD patients.
GM is an ecosystem of bacteria, fungi, viruses, and parasites residing in the gastrointestinal (GI) tract[3]. Established at birth and evolving throughout life, it maintains a commensal relationship with the host and is a fundamental part of the human body[4]. GM is determined by the host’s diet, environment, and activity patterns[5]. It is also essential for maintaining body homeostasis, modulating the immune system, and influencing its development and physiology. It also plays a relevant role in host metabolism, participating in nutrient metabolism, producing essential vitamins, and removing xenobiotics, among other functions[6]. When all these mechanisms fail, GM dysbiosis occurs, usually caused by an inadequate diet, constant stress, increased inflammatory markers, and the inappropriate use of drugs, mainly antibiotics. These disrupt the body’s homeostatic functions and can play a significant role in the pathophysiology of a plethora of metabolic and CVD[3].
The GM is primarily composed of the commensal bacterial phyla Bacteroidetes, Firmicutes, Actinobacteria, and Proteobac
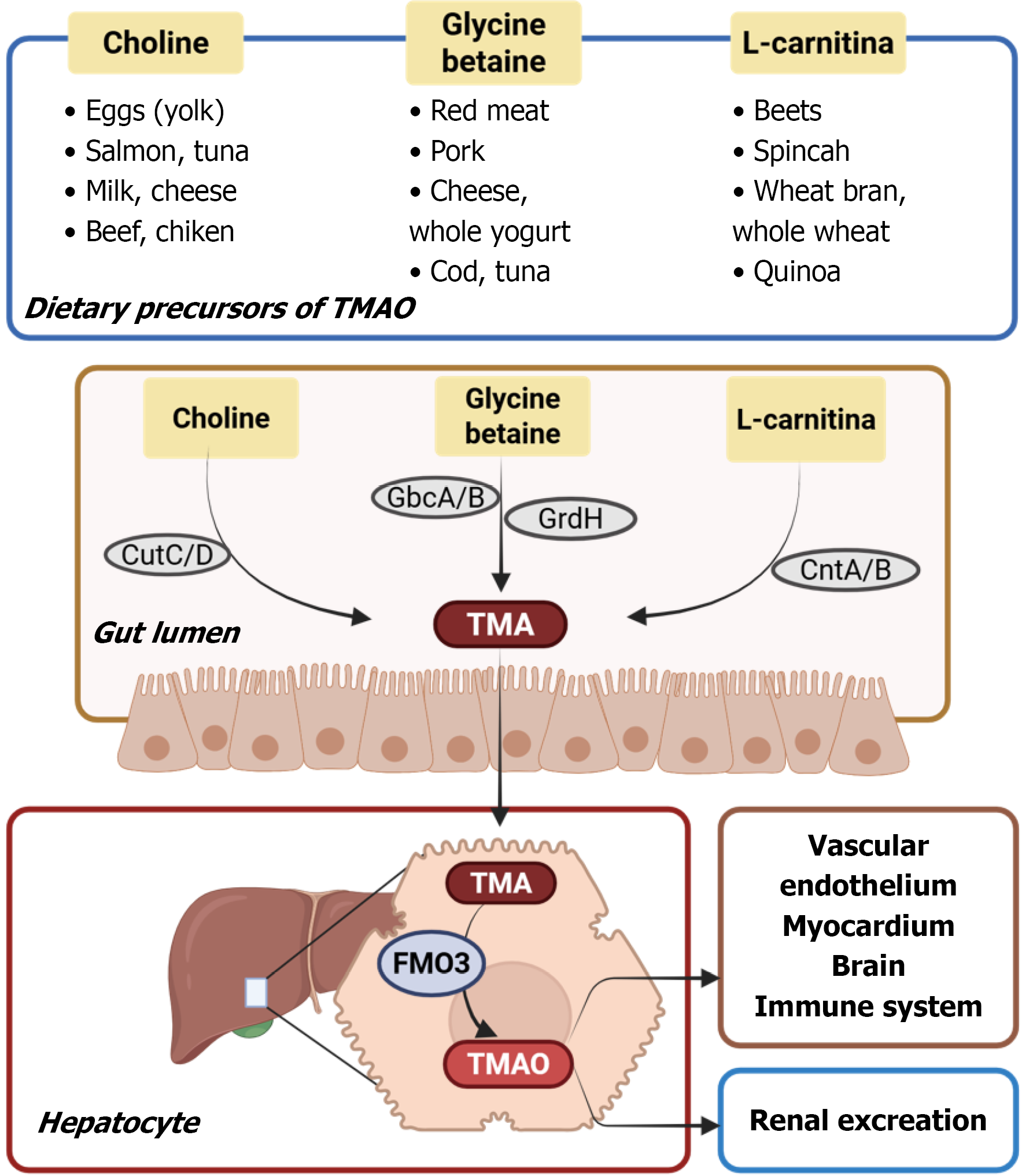
As previously reported, TMAO is a small molecule formed by the TMA oxidation by flavin-containing hepatic monooxygenases[8]. TMA is produced by the GM from dietary precursors, primarily betaine, L-carnitine, and choline, and subsequently crosses the gut lumen and is transported through the portal circulation to the liver. TMA is then converted to TMAO by flavin monooxygenase (FMO). Of the five members of the FMO family, only FMO1 and FMO3 have the ability to oxidize TMA to TMAO, with FMO3 being the major isoform expressed in the human liver. Once produced, TMAO is removed from the systemic circulation primarily through urinary excretion, in its unmodified form, by means of filtration and active secretion mechanisms[9] (Figure 1).
It has been suggested that hepatic TMAO production could increase cardiovascular and mortality risk by promoting the development of atherosclerotic lesions, but at the same time, it has positive effects on metabolic health via increasing glucose metabolism in the muscle and heart[10,11]. Several families of bacteria from the phyla Firmicutes, Proteobacteria, and Actinobacteria have been identified as producing high TMAO concentrations when compared to Bacteroidetes. The most abundant genera have been identified as belonging to the families Clostridiaceae, Lachnospiraceae, and Veillonellaceae, as well as Clostridiales from the phylum Firmicutes.
The CVD patients have been found to have a different GM composition compared to healthy individuals[5], for example, plasma TMAO levels were shown to be associated with Prevotella and Peptococcaceae[12], and a positive corre
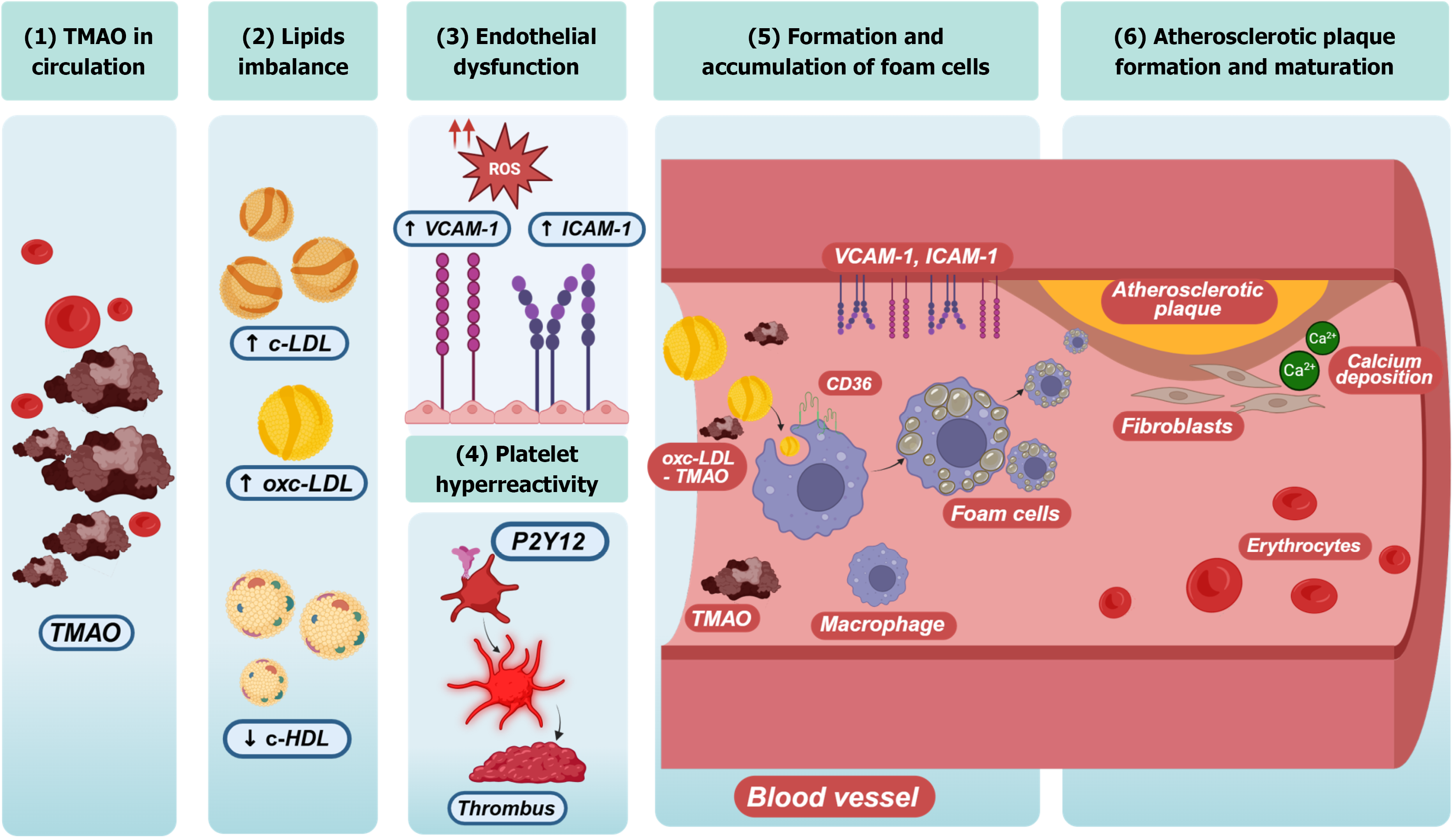
Atherosclerosis is an inflammatory disease triggered by lipid accumulation and the deposition of fatty, fibrous, and calcified material in the innermost layer of large and medium-sized arteries[17]. Atherosclerosis develops through three fundamental processes: (1) The deposition of atherogenic lipids; (2) Proinflammatory conditions; and (3) Endothelial dysfunction. In this context, elevated low-density lipoprotein cholesterol (LDL-C) and reduced high-density lipoprotein cholesterol (HDL-C) levels favor the formation of foam cells from macrophages, a key process in atherogenesis[18]. This process is further amplified by the presence of lipopolysaccharide (LPS), an endotoxin derived from gram-negative bacteria that, upon crossing the altered GI barrier, enters the systemic circulation and activates monocytes and macro
Once formed, foam cells secrete proinflammatory cytokines, mainly interleukin-1 (IL-1), IL-6, tumor necrosis factor alpha (TNF-α), and monocyte chemoattractant protein 1, promoting further monocyte recruitment and perpetuating the inflammatory response[17]. Consequently, vascular smooth muscle cells proliferate, causing intimal thickening. As the process repeats, plaque thickness increases, forming a fibrous cap that, together with programmed leukocyte cell death and microcalcifications, favors the formation of a necrotic core (Figure 2). These plaques, more vulnerable to rupture, can negatively modify blood flow, induce thrombosis and impair or even block blood flow, leading to acute coronary syn
It has been proposed that TMAO is related to the atherosclerosis pathogenesis, activating proinflammatory pathways and making changes that promote endothelial dysfunction by activating signaling cascades. Additionally, evidence shows that TMAO induces mitogen-activated protein kinases, extracellular signal-regulated kinase and nuclear factor kappa B, whose signaling is activated in association with the G-protein beta-gamma subunit dimer, in endothelial cells[11,21]. Moreover, TMAO significantly increases inflammatory markers, including cyclooxygenase 2, IL-6, E-selectin, and intercellular adhesion molecule 1, which recruit leukocytes and induce vascular inflammation[21]. There is an increase in thrombosis through platelet hyperreactivity, which occurs since direct exposure of platelets to TMAO enhances stimulus-dependent platelet activation from multiple agonists through augmented Ca2+ release from intracellular stores[11,22] and atherosclerosis development promoted by the accumulation of cholesterol in macrophages, induced by the expression of scavenger-like receptors, CD36 and scavenger receptor A1[23], and decreased reverse cholesterol transport, as well as de
Growing evidence suggests that GM and oral microbiota (OM) play an essential role in cardiovascular health[24]. TMAO plays a pivotal role in the development of atherosclerosis. As mentioned above, this metabolite not only induces local inflammation that contributes to the progression of CVD, but also alters blood lipid levels, further accelerating this process[25]. A systematic review and meta-analysis by Hassan et al[5] reported that the increased abundance of the genera bacteria Acidaminococcus and Aggregatibacter may unfavorably modulate HDL-C levels, promoting lipid dysregulation and atherogenic risk. This lipid perturbation may contribute to the establishment of advanced arterial obstructions and myocardial ischemia. In addition, elevated TMAO concentrations have been shown to be associated with an in
In this context, CAD represents one of the most common clinical manifestations of the afore-mentioned disorders, characterized by the accumulation of plaque in the heart arteries. If not treated promptly, it can progress to ACS, such as AMI, or even stroke[27]. CAD is highly associated with complex metabolic disorders, including obesity, insulin resis
TMAO has gained relevance as a potential marker associated with CAD progression. In a prospective study conducted in China, patients diagnosed with CAD were found to have significantly higher plasma TMAO levels compared with subjects without the disease[29]. Similarly, another study in CAD patients documented that those with plaque rupture had significantly higher TMAO concentrations compared with those without plaque rupture (8.6 ± 4.8 μmol/L vs 4.2 ± 2.4 μmol/L)[30]. These findings support the hypothesis that circulating TMAO levels may mirror the progression and instability of coronary plaques.
ACS are critical conditions characterized by coronary artery occlusion due to the rupture of atherosclerotic plaques. A portion of the thrombus can break off and block one of the coronary arteries, leading to diminished blood flow and causing AMI[31-33]. TMAO is closely linked to the ACS progression. Furthermore, TMAO concentrations in patients with moderate atherosclerosis were higher than those with mild atherosclerosis, suggesting a strong association between plasma TMAO concentrations and the severity of coronary atherosclerosis[33]. These findings reinforce the concept that TMAO could be a relevant risk factor for severe atherosclerosis and a predictive biomarker for ACS[29,33].
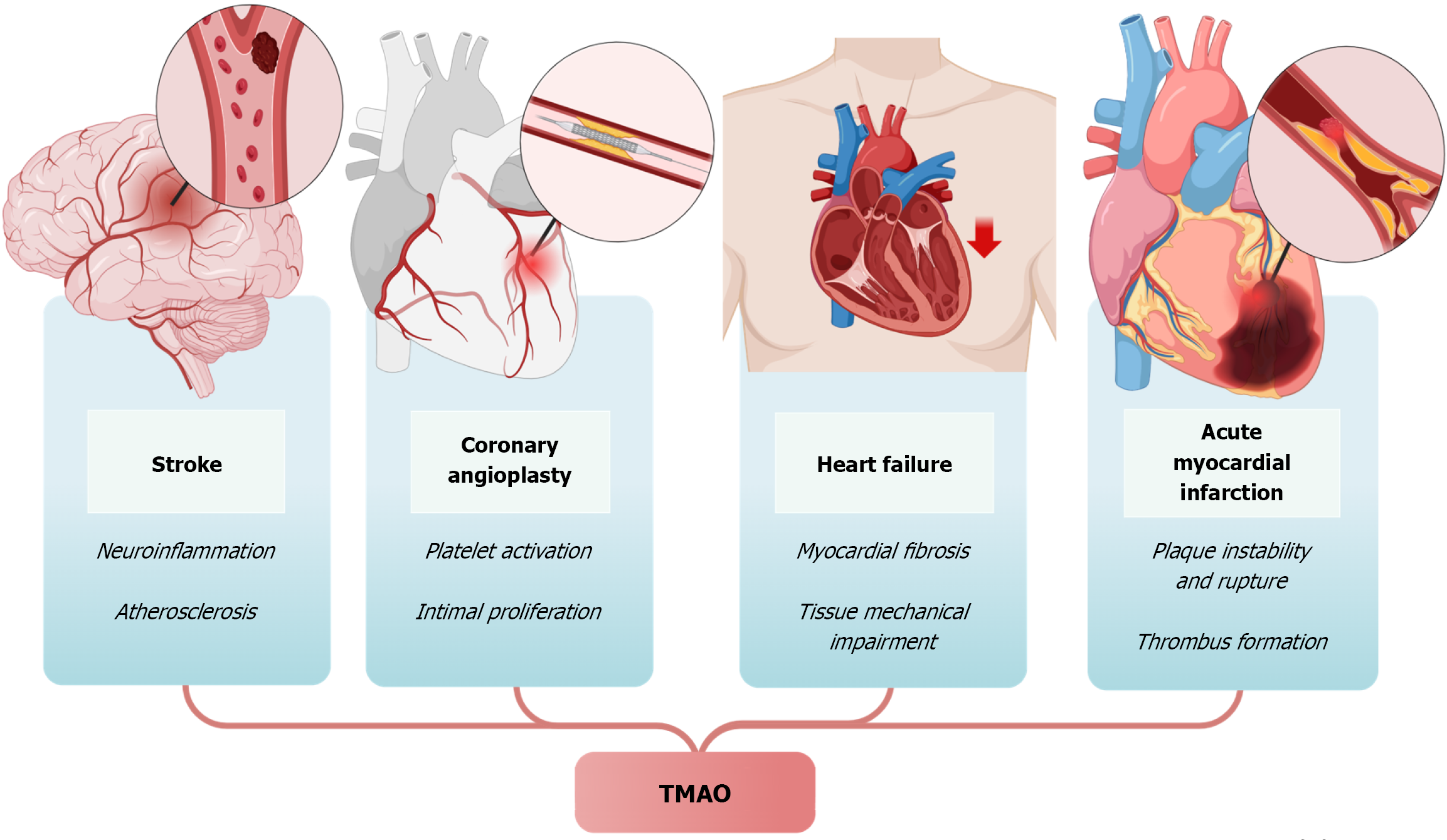
AMI is the leading cause of morbidity and mortality worldwide[34]. It is characterized by clinical evidence of ischemia, along with alterations in cardiac troponin levels. It is classified into ST-segment elevation myocardial infarction (STEMI) and non-STEMI[35]. The AMI pathophysiology involves a network of complex interactions, with atherosclerosis as a principal risk factor and multiple downstream processes, including endothelial dysfunction, vascular inflammation and thrombosis, culminating in intimal thickening and, ultimately, obstruction of coronary blood flow[14,36] (Figure 3). Within this multifactorial framework, the OM has been proposed as an additional contributor to acute coronary in
In AMI patients, GM is altered and characterized by a lower abundance of the phylum Firmicutes and a slightly higher abundance of the phylum Bacteroidetes[34]. Beyond these broad taxonomic shifts, case-control data further suggest that ACS is accompanied by more specific, potentially pathogenic microbial signatures and host-barrier perturbations. One case-control study showed that patients with ACS had a nine-fold increase in gut Gammaproteobacteria, a class that includes gram-negative pathogens, opportunistic bacteria, and Enterobacteriaceae, along with increased gut permeability compared with controls. In the same cohort, compositional differences were also observed at genus/order level, with ACS patients showing higher levels of Prevotella and controls having higher levels of Bacteroidales overall. Notably an elevated Prevotella/Bacteroides ratio has been associated with a high-carbohydrate dietary pattern, which may further contribute to cardiometabolic risk[38]. Similarly, a prospective study, with 211 STEMI patients, documented that patients with plaque rupture had three times as many levels of TMAO compared to those without plaque rupture (3.33 μmol/L vs 1.21 μmol/L)[26]. For another hand, in a study with STEMI patients who presented STEMI, they measured plasma TMAO levels at baseline and again in the chronic phase (10 months later). TMAO levels increased significantly from the acute to the chronic phase of STEMI (median: 5.63-6.76 μmol/L). During a 5.4 year follow up, 17 patients experienced cardiovascular events, and chronic-phase TMAO levels independently predicted subsequent events. These findings underscore the potential prognostic value of chronic phase TMAO for risk stratification after STEMI[39].
Strokes occur when blood flow to certain brain regions is interrupted, causing oxygen and nutrient deprivation and resulting in neurodegeneration[40]. This interruption can be caused by an arterial occlusion, leading to ischemic stroke, the most common form, or by intracranial bleeding, resulting in hemorrhagic stroke, which is generally associated with higher fatality[41]. Beyond vascular and hemostatic mechanisms, growing evidence indicates that stroke pathophysiology is influenced by systemic host-GM interactions. It has been shown that the central nervous system maintains constant interaction with the GM, giving rise to the gut-brain axis, where GM-derived metabolites can modulate brain develop
This compromises the integrity of the blood-brain barrier by altering the expression of tight junction proteins, increasing its permeability, and allowing the passage of immune cells and GM-derived metabolites, such as TMAO and LPS, into the brain. Consequently, GM, gut metabolites, and immune cells translocate from the leaky gut to the infarcted region of the brain through systemic circulation, exacerbating the stroke severity[40] (Figure 3).
TMAO levels have been associated with the incidence and severity of CVD, including infarct size and functional deficit, suggesting that TMAO may serve as a prognostic biomarker and risk stratifier for adverse outcomes including death[40]. Consistent with this broader vascular risk signal, evidence from stroke-focused cohorts has linked baseline TMAO levels to both incident and recurrent cerebrovascular events. Using data from the China Stroke Primary Prevention Trial, one analysis evaluated baseline serum TMAO concentrations and the risk of a first stroke and found that individuals with higher TMAO levels (≥ 1.79 μmol/L) had a 34% higher risk of a first stroke compared with those with lower levels (< 1.79 μmol/L), supporting an association between TMAO and the stroke risk in hypertensive patients[43].
Xue et al[44] conducted a prospective, multicenter study of patients with acute ischemic stroke, participants with high TMAO and choline levels had significantly higher cumulative recurrence rates than those with low TMAO and choline levels. Similarly, two prospective cohort studies involving patients with previous ischemic stroke further reinforced the association between TMAO levels and the risk of subsequent cardiovascular events, mainly AMI, recurrent stroke, and cardiovascular death, suggesting that it might be useful in predicting these outcomes[45]. In conclusion, TMAO has been identified as a significant risk factor in the development and severity of various diseases, including stroke.
HF is a clinical syndrome characterized by a decreased ability of the heart to pump or fill with blood, resulting in inade
HF is a multisystem disorder that affects the heart and circulation but also other systems, including the metabolic system[19]. In this context, the GI tract has acquired relevance as a key organ in the pathogenesis of chronic HF. Mesenteric venous congestion, characteristic of this condition, can increase gut permeability, promoting bacterial translocation and endotoxin release. This process contributes to sustained immune activation, evidenced by increased levels of CD14, TNF-α, proinflammatory cytokines such as IL-1 and IL-6, and adhesion molecules such as intercellular adhesion molecule 1[49-51]. Mechanistically, this GM-hypothesis of HF links hemodynamic derangements to barrier failure and systemic inflammation.
It has been proposed that activation of the sympathetic nervous system, although with a compensatory purpose in HF due to its inotropic and vasoconstrictor effect, may contribute to gut ischemia by reducing splanchnic blood flow[52]. Under these conditions, LPS can cross the compromised gut barrier and enter the systemic circulation, promoting the release of proinflammatory mediators. Sandek et al[19], reported that the gut permeability index, measured by the urinary lactulose/mannitol ratio, has been shown to be significantly higher in patients with chronic HF, mirroring a 35% increase in gut permeability. This barrier dysfunction provides a plausible substrate for GM-host interaction that may amplify systemic inflammation and metabolic perturbations in HF.
GM dysbiosis is increasingly recognized as a contributor to elevated TMAO levels and has been linked to the development and progression of HF[23]. Tang et al[47] proposed that gut congestion and interstitial edema could alter GM composition, thereby enhancing microbial TMA production and downstream TMAO production. Clinically, higher circulating TMAO levels have been associated with adverse cardiac structure and function, including impaired diastolic indices, such as increased left atrial volume, suggesting that its accumulation could affect tissue mechanics, through increased myocardial fibrosis, resulting in cardiac hypertrophy, and increased accompanying diastolic dysfunction[25,53]. These observations support the view that TMAO may act as a risk marker, and potentially a mechanistic mediator of HF development and progression[25].
As previously reported, subjects with a higher abundance of bacteria from the genera Prevotella, Clostridium, Ruminococcus and the family Lachnospiraceae, and a lower presence of bacteria from the phylum Bacteroidota, have been shown to have elevated plasma concentrations of TMAO[54]. The taxonomic patterns support the concept that compositional shifts in GM can translate into measurable differences in circulating microbial metabolites relevant to HF risk. Furthermore, indole-3-propionic acid, mainly produced by Clostridium sporogenes in GI tract, has been identified to regulate mi
In HF patients, especially those with HF with reduced ejection fraction, a decrease in GM diversity has been observed. Luedde et al[56] observed lower levels of Blautia, Collinsella and the Erysipelotrichaceae family, alongside increases in the abundance of Streptococcus and Veillonella has been reported[57]. Complementing these findings, Katsimichas et al[57] reported a higher proportion of Fusobacterium prausnitzii in chronic HF. More recently, Pasini et al[58] identified correlations between the presence of Candida, Campylobacter, Shigella, and Yersinia and HF severity.
In a prospective observational study, TMAO levels were significantly higher in patients with ischemic HF than in those with dilated cardiomyopathy or stable CAD, suggesting that both HF and ischemic etiology may contribute to elevated TMAO levels. Consistent with this clinical gradient, TMAO levels were associated with New York Heart Association functional class, a standard measure of HF severity, as well as congestion related hemodynamics indices, including pul
Beyond cross-sectional severity markers, TMAO has also been linked to prognostic indicators. A significant correlation was observed between TMAO levels and brain-type natriuretic peptide levels, and elevated TMAO was associated with an increased risk of 5-year mortality risk[61]. Moreover, Tang et al[62], reported two prospective cohorts and found that circulating TMAO levels, along with related metabolites such as croto-betaine, betaine, carnitine, and choline, were associated with an increased risk of HF. Supporting a mechanistic link to cardiac structure and function, Tang et al[47] reported elevated TMAO levels, choline and betaine have been associated with higher N-terminal-pro brain-type natriuretic peptide levels and more advanced left ventricular diastolic dysfunction. Summarizing the literature data indicates that elevated TMAO levels are closely related to HF and its etiology, especially in patients with ischemic HF. These higher levels are associated with greater disease severity, as reflected in the New York Heart Association functional classification. Furthermore, elevated TMAO levels correlate with a higher risk of long-term mortality.
MACE represents a group of serious complications, and its incidence rates are closely linked to various risk factors. These factors, categorized as modifiable and non-modifiable, include metabolic, cardiovascular, chronic, and autoimmune conditions[63]. Alterations in GM composition may contribute to the development and complications of CVD through mechanisms such as systemic inflammation, dysbiosis, and metabolite production.
Dietary patterns critically shape GM composition and function, with Western and Mediterranean diets exerting distinct effects on microbial diversity, host-microbe interactions, and finally TMAO metabolism. The Western diet, characterized by a high intake of red meat, saturated fats, refined sugars, and predominantly ultra-processed foods, markedly decreases overall GM diversity, promotes significant weight gain and a pro-inflammatory tone, and is associated with a higher relative abundance of Firmicutes alongside a decrease in Bacteroidetes[64,65]. Consistently, Western diet promotes choline and carnitine catabolism, leading to significantly elevated plasma and urinary TMAO concentrations[66,67].
In contrast, adherence to a Mediterranean-style diet, characterized by lower fat intake and high dietary fiber consump
It has been reported that short-term high-fat feeding rapidly compromises gut barrier integrity and mucosal immunity, while promoting obesity, insulin resistance, and non-alcoholic steatohepatitis, in association with GM dysbiosis characterized by a shift from Bacteroidetes toward Firmicutes[74,75]. Likewise, chronic exposure to a high-fat diet enhances choline catabolism, leading to increased TMA/TMAO production and renal excretion, alongside reduced GM diversity[67,76]. Taken together, these findings suggest that high-fat consumption creates a permissive microbial and immunological environment that facilitates barrier dysfunction and enhanced TMAO production, contributing to adverse metabolic outcomes.
Aging is a gradual decline in homeostasis, functional capacity, leading to increased susceptibility to death[77]. Gut permeability increases with age as a consequence of GM alterations, promoting the translocation of microbial products into the circulation and triggering systemic inflammation, which has been linked to increased mortality[78-81]. Older individuals exhibit distinct GM profiles compared with younger populations; however, evidence is heterogeneous, with some studies reporting a higher abundance of Bacteroidetes and reduced Firmicutes, while others describe decreased Bacteroidetes abundance accompanied by lower overall GM diversity[82-84]. Moreover, advanced age is associated with an enrichment of TMA-producing bacteria carrying the cutC and cntA genes, greater availability of dietary precursors, and elevated circulating TMAO levels, contributing to a persistent inflammatory state and vascular aging[85-87]. In this context, TMAO emerges as a key link between the GM and pathological aging by promoting inflammation, gut barrier dysfunction and endothelial impairment.
Antibiotics and their microbial regulation: Antibiotics can induce GM dysbiosis. In detail, broad-spectrum agents promote excessive inflammation and pathobiont expansion, leading to immune alterations and dysbiotic GM characterized by selective enrichment and widespread taxonomic loss. In contrast, antibiotics, such as metronidazole, appear to preserve anti-inflammatory capacity by enriching Lactobacillus and supporting innate immune responses[88-92]. Alternatively, antibiotic administration markedly decreases plasma TMAO levels, suggesting therapeutic potential; but, TMAO may also promote antibiotic resistance in certain bacteria, thereby limiting therapeutic efficacy[16,93,94]. Therefore, antibiotics exert complex and context-dependent effects on the GM, with broad-spectrum agents driving dysbiosis and immune disruption, while modulating microbial metabolites such as TMAO, underscoring both their therapeutic potential and limitations.
Metformin’s microbial footprint, inconsistent effects on dysbiosis and TMAO: Metformin, a first-line therapy for T2DM, has emerged as a potential modulator of TMAO concentrations but the evidence remains contrasting. While some studies report increased circulating TMAO concentrations alongside improved glucose control, others document that metformin reduces plasma TMAO levels and bacterial TMA production from choline, with or without accompanying changes in GM diversity[95-98]. However, some evidence indicates that metformin does not significantly affect serum TMAO levels or related metabolic and cardiac outcomes[99]. In sum, the metformin impact on TMAO levels and GM composition remains inconsistent. These discrepancies emphasize the context-dependent nature of metformin-GM interactions and the need for further investigation.
Systemic arterial hypertension (SAH) is the most prevalent risk factor for MACE[100,101] and, due to its ability to cause endothelial damage and vascular remodeling, remains one of the leading causes of premature mortality[102,103]. Recent advances suggest that GM composition is closely related to SAH[104-108], and animal models confirm this association[109-111]. High plasma TMAO concentrations increase the risk of SAH developing by 12%[112]. Since diet modulates GM and its metabolites[112,113], dietary and GM variations are the main determinants of circulating TMAO levels[114-116]. TMAO promotes hypertension by enhancing Ang II-induced vasoconstriction through activation of the protein kinase r-like endoplasmic reticulum kinase/reactive oxygen species/calcium/calmodulin-dependent protein kinase II/phospho
T2DM is a relevant cause of AMI and stroke[122]. TMAO is linked to T2DM, promoting insulin resistance in animal models, with elevated levels consistently associated with higher disease incidence, and FMO3 playing a key mechanistic role[123-128]. Moreover, high intake of red meat, eggs, and dairy products is associated with higher plasma TMAO concentrations[129,130] and, independently, a higher T2DM risk[131-133]. This is associated with GM dysbiosis, which not only contributes to T2DM development but also worsens its cardiometabolic complications. In T2DM patients undergoing percutaneous coronary intervention, elevated TMAO levels were associated with a higher risk of MACE and mortality[134], highlighting its prognostic value; however, in the ACCORD trial involving poorly controlled, high-cardiovascular-risk individuals, TMAO did not discriminate between those with and without MACE[135]. Thus, TMAO emerges as a biomarker and potential therapeutic target, although its clinical utility may depend on the patient’s glycemic profile and metabolic control.
TMAO has emerged as a microbiota-derived metabolite closely linked to chronic kidney disease (CKD), defined as abnormalities of kidney structure or function, present for a minimum of 3 months, with implications for health[136]. In this context, patients with CKD exhibit elevated circulating TMAO levels, which are associated with greater renal fibrosis, amplified inflammatory responses, and an increased risk of acute kidney injury-to-CKD progression[137,138]. Moreover, circulating TMAO levels and the TMAO/creatinine ratio increase in a CKD stage-dependent manner, while urinary TMAO levels decline, with the abundance of microbial TMA-producing genes, cutC and cntA, correlating with plasma TMAO and estimated glomerular filtration rate; in parallel, higher TMAO levels are associated with an increased risk of incident CKD and a more rapid annual decline in estimated glomerular filtration rate[139,140].
Dietary habits, aging and certain pharmacological interventions significantly influence GM dysbiosis and circulating TMAO levels. While western diets and high-fat intake promote dysbiosis and TMAO elevation, Mediterranean-style diets and high fiber consumption support a healthier GM and limit TMAO formation. Conditions such as CKD, SAH and T2DM are associated with elevated TMAO, highlighting the critical role of the GM and its metabolites in cardiometabolic risk. These findings emphasize the importance of targeting modifiable factors to prevent metabolic and cardiovascular complications.
CVD prognostic assessment underpins risk stratification and therapeutic decisions but incompletely captures biological heterogeneity and dynamic risk. In this scenario, TMAO has been proposed as a prognostic biomarker and potential causal risk factor through its links to key cardiovascular pathophysiological pathways.
Clinical risk models are tools that integrate clinical and laboratory variables to estimate the outcome probability. In ACS, the GRACE score[141] combines readily available variables to predict the risk of death and AMI. In this scenario, TMAO has been proposed as a complementary biomarker that could add predictive value. Suzuki et al[142] documented that TMAO independently predicted death and recurrent AMI at 2 years and improved 6-month GRACE stratification by down-classifying risk. Similarly, Tan et al[143] reported a prospective cohort of 444 STEMI patients undergoing primary percutaneous coronary intervention; plasma TMAO levels measured before the procedure and predicted 6-month MACE. Of note, adding myeloperoxidase plus TMAO to the GRACE score increased discrimination and improved reclassification, supporting the incremental prognostic value of TMAO for refining GRACE-based risk stratification. In 2017, Li et al[144], reported that higher TMAO levels predicted a markedly increased risk of 30-day and 6-month MACE, and predicted 7-year mortality, consistent with results from a study conducted a year earlier by the same group[145]. Notably, TMAO retained short-term prognostic value even among patients who were troponin T-negative at presentation, suggesting potential utility for early risk stratification before conventional biomarkers become positive.
In complex CAD, the SYNTAX score[146] is used to stratify atherosclerotic disease severity. Senthong et al[147] docu
In primary prevention, the Framingham Risk Score[148,149], and QRISK[150] are used to estimate cardiovascular risk in the general population. Sikora et al[151], in a pilot study in psoriasis, analysis indicated that TMAO was an inde
Translating TMAO from an associative finding into a clinically actionable predictor faces two key challenges: Assay standardization and clarifying the biological meaning of an elevated concentration[153]. First, reported TMAO levels can vary across studies due to differences in sample type, pre-analytical conditions and analytical platforms, thereby limiting cross-study comparability. Second, a critical distinction remains between TMAO as a causal risk factor vs risk biomarker, since observed associations may be influenced by confounding or mediation.
In a 2018 meta-analysis of 11 prospective cohort studies[154], higher TMAO levels were linked to a 23% higher risk of cardiovascular events and a 55% higher risk of all-cause mortality. In addition, a 2017 meta-analysis[155] reported a dose-response relationship, estimating that in CVD populations all-cause mortality risk increases by 7.6% per 10 μmol/L increment in plasma TMAO, consistent with a graded association between TMAO levels and mortality. In 2024, it reported a significant positive association between TMAO and MACE, all-cause mortality, and recurrent AMI in patients with AMI; this finding contrasts with results reported in the PEGASUS-TIMI 54 trial[156]. Other systematic reviews and meta-analyses report broadly concordant association[157,158]. Overall, the meta-analytic literature supports an as
However, more recently, Obeid et al[159], published an umbrella review summarizing evidence linking circulating TMAO levels with cardiovascular, cerebrovascular, and renal outcomes, including mortality. The main finding was that most reviews reported higher TMAO levels among individuals who experienced adverse outcomes. Consistent with this, like Hu et al[160] that patients with ischemic stroke tend to have higher TMAO levels, yet analyses did not support a causal TMAO effect on stroke occurrence. Notably, Obeid et al[159] emphasized that the underlying primary studies were predominantly conducted in clinically ill, high-risk populations with multiple comorbidities, which may confound causal inference and limit “pure” prognostic interpretation[159]. In addition, definitions of “elevated” TMAO varied substantially across studies, as previously reported[161,162], hindering the establishment of clinically meaningful thresholds for high-risk classification. TMAO interpretation in clinical cohorts is complicated by strong dependence on renal clearance[163-165] and cardiometabolic comorbidity like T2DM and MetS[166] which can confound or mediate observed associations. TMAO often reflects underlying cardiorenal risk rather than acting as an independent causal mediator. In summary, the current evidence remains insufficient to rightly determine whether TMAO is a causal mediator, a risk marker, or a reliable standalone prognostic factor, and higher-quality prospective studies and randomized interventions are needed to clarify its causal and predictive roles.
3,3-dimethyl-1-butanol (3,3-DMB), a choline analogue, inhibitor of microbial TMA-lyases that suppresses upstream TMA formation, and improves endothelial function reduced foam cell formation and atherosclerotic lesion development[167]. Brunt et al[168] further showed that 3,3-DMB mitigates diet-induced endothelial dysfunction and is associated with improved exercise tolerance. Chen et al[169] extended these findings to cardiac phenotypes by demonstrating that, lowers TMAO and prevents cardiac dysfunction, inflammation and fibrosis. Wang et al[170] similarly reported attenuation of adverse cardiac remodeling in a HF setting, collectively supporting that microbial TMA inhibition via 3,3-DMB can beneficially modulate atherosclerotic, vascular, and cardiac endpoints across multiple disease models, including CKD-associated endothelial dysfunction[171]. However, there is evidence suggesting limited or missing TMA/TMAO suppres
Halomethylcholines, such as fluoromethylcholine and iodomethylcholine, are mechanism-based, irreversible inhibitors of the microbial cutC/D enzyme, representing promising microbiome-targeted compounds to lower TMAO[173]. These compounds are reported to be substantially more potent than 3,3-DMB in suppressing microbial TMA/TMAO at lower effective concentrations[174]. Future studies should prioritize well-controlled intervention studies that define dose-response relationships and the durability of TMAO suppression, establish safety and off-target consequences, and test whether microbiome-targeted cutC/D inhibition yields consistent benefit across diverse cardiometabolic and vascular phenotypes and in translational settings.
Direct hepatic FMO3 inhibition has mainly been explored as an experimental strategy to modulate the TMA/TMAO axis. Methimazole is a high-affinity FMO3 substrate and widely used tool inhibitor that can competitively suppress[175,176]. Dietary indole-derived compounds can also inhibit FMO activity[177]. Finally, phenylthiourea has been used to suppress clearance of FMO3-dependent substrates/metabolites[178].
It has been shown that regulation of hepatic FMO3 decreases TMA levels and increases circulating TMAO levels, whereas silencing it has the opposite effect, i.e., elevates TMA levels and reduces TMAO levels[179]. This phenomenon has been further evidenced in studies where hepatic FMO3 expression was decreased in LDL-C receptor knockout mice using an antisense oligonucleotide. In these experiments, FMO3 reduction resulted in a decrease in both plasma TMAO levels and atherosclerosis[180]. These findings highlight the FMO3 relevance in regulating TMAO and its role in preventing atherosclerosis, thus highlighting alternative therapeutic pathways for CVD.
Another emerging approach, intermittent hypoxia-hyperoxia exposure (IHHE), consists of alternating short periods of reduced and enriched oxygen in order to induce beneficial physiological adaptations[181,182]. This treatment was initially studied in cardiac rehabilitation and fitness improvement[183], its impact on GM[184] and metabolism has now been explored[185,186]. Evidence on whether IHHE can alter GM or reduce TMAO is still under investigation. A prospective study evaluating IHHE in MetS patients with elevated baseline TMAO levels (≥ 5 μmol/L) showed a significant decrease after the intervention. Furthermore, the reduction in total cholesterol and LDL-C was more notable in subjects with normal TMAO levels at baseline[187]; this suggests that controlled hypoxia could have beneficial cardiometabolic effects, especially in individuals with elevated TMAO. Further studies are required to confirm its effectiveness.
TMAO has been reported to be associated with glucose metabolism and independently predicts the risk of CVD and mortality[154]. In a clinical study in obese subjects at risk of insulin resistance, supplementation with flavonoids from tea and cocoa failed to significantly reduce TMAO levels[188] (Table 1). However, a pilot randomized controlled trial in adults at elevated risk for T2DM. Six weeks of inulin supplementation did not decrease fasting or postprandial plasma TMAO, indicating that simply increasing fermentable fiber is not consistently sufficient to suppress the TMO/TMAO axis[189] (Table 1). In contrast, a clinical trial in prediabetic subjects[190] showed that regular pistachio consumption reduced urinary metabolites related to GM, including TMAO, suggesting a potential beneficial effect on the T2DM risk (Table 1). Similarly, a prospective study showed that a vegan diet[191] significantly reduced TMAO levels, although these levels increased after participants resumed a normal diet (Table 1). These findings support a vegan diet as an effective inter
| Ref. | Intervention | Mechanism | Sample type | Duration | Population | Results |
| Specific diets and foods | ||||||
| Argyridou et al[191], 2021 | Vegan | Reduces dietary precursors | Plasma | 8 weeks | Overweight patients over 18 years of age with untreated HbA1c ≥ 5.7% and ≤ 8% | TMAO decrease |
| Diao et al[211], 2024 | DASH | GM remodeling and concurrent lowering LPS | Plasma | 12 weeks | Overweight and obese, ages 20-55 years | TMAO decrease; Firmicutes/Bacteroidetes ratio decrease |
| Hernández-Alonso et al[190], 2017 | Pistachios (57 g/day) | Modulation of GM related metabolism chronic pistachio intake | Urinary | 15 days | BMI ≤ 35 kg/m2 and glucose levels between 100 and 125 mg/dL, ages 25-65 years | TMAO decrease |
| Haas et al[214], 2022 | Red wine 250 mL/day | Redox homeostasis | Plasma | 3 weeks | CAD, average age 60 years | TMAO (ns) |
| Thomas et al[192], 2022 | Consume three eggs per day (400 mg/day) | Dietary choline (phosphatidylcholine), influences GM-dependent TMA generation | Plasma | 4 weeks | MetS, ages 35-70 years | TMAO (-); antioxidants increase |
| Angiletta et al[188], 2018 | Cocoa drink (28 g) and green tea (1.2 g) | Nutritionally relevant flavonol doses could reduce TMAO production | Plasma | 5 days | Obesity and risk of insulin resistance, ages 25-55 years | TMAO (-) |
| Bergeron et al[222], 2016 | CHO and resistant starches | GM remodeling and CHO diet-GM interaction influence TMAO production, not simply precursor intake | Plasma | 56 days | Men > 20 years and postmenopausal women, BMI ≥ 20 and ≤ 35 kg/m2 | TMAO increase |
| Prebiotics | ||||||
| Baugh et al[189], 2018 | Inulin 10 g/day added to a standardized diet | Modulate GM and thereby reduce microbial TMA production | Plasma | 6 weeks | Sedentary overweight/obese adults elevated risk for T2DM | TMAO (ns); TMA moieties (ns) |
| Probiotics | ||||||
| Malik et al[216], 2018 | Lactobacillus plantarum 299v (20 billion CFU once daily) | Changes in GM derived metabolites (not TMAO) | Plasma | 6 weeks | History of stable CAD, age 40-75 years | TMAO (-); propionate increase; acetate decrease; Lactobacillus in stool increase; endothelial function increase |
| Spasova et al[215], 2024 | Lactobacillus plantarum GLP3 (1 × 109 CFU) | GM modulation (increase in Lactobacillus spp.) with plausible contribution from prebiotic and polyphenol | Plasma | 12 weeks | History of CAD, age 58.5 ± 8.16 years | TMAO decrease |
| Tripolt et al[217], 2015 | Lactobacillus casei Shirota (6.5 × 109 CFU 3 ×/day) | GM modulation could alter microbial TMA-formation | Plasma | 12 weeks | MetS, age 52 ± 11 years | TMAO (ns) |
| Awoyemi et al[224], 2021 | Saccharomyces boulardii 250 mg capsules twice daily and rifaximin 550 mg orally twice daily | GM modulation affects composition and function reducing microbial-linked metabolites (TMAO) | Plasma | 3 months | HFrEF and NYHA functional class II or III, average age 60 years | TMAO (-) |
| Other interventions | ||||||
| Smits et al[200], 2018 | Vegan donor FMT (autologous) | GM replacement strategy to reduce GM conversion of choline/carnitine to TMA | Plasma | 2 weeks | MetS, no history of CVD, aged 21-69 years | TMAO decrease |
| Bestavashvili et al[187], 2023 | IHHE (total 15 sessions) | Hypoxic conditioning antioxidant/anti-inflammatory signaling and improving gut-liver axis | Stool and plasma | 3 weeks | MetS, age 29-74 years | TMAO (ns) |
In a clinical trial, daily egg intake did not increase plasma TMAO levels nor significantly alter GM compared to a choline supplement, although it increased antioxidant levels of lutein and zeaxanthin[192]. Probiotics have emerged as a complementary strategy to modulate GM[193-195] in metabolic and cardiovascular pathologies[196]. Likewise, fecal microbiota transplantation (FMT), studied in different diseases[197-199], showed, in a pilot study with vegan donors for subjects with MetS, partial changes in GM without effect on TMAO levels and vascular inflammation[200] (Table 1). Together, these findings show the complexity of modulating the GM-TMAO axis in MetS. In detail, probiotics require more specific combinations and FMT requires more intensive or repeated strategies to influence cardiometabolic parameters.
Obesity is a global health concern associated with an increased risk of CVD[201,202]. Dietary interventions[203,204] and the adoption of healthy lifestyles[205,206] can modulate GM, offering an effective complementary therapeutic option to mitigate metabolic effects. It has been reported that reducing dietary choline and L-carnitine was associated with improvements in insulin resistance with a low-calorie weight-loss diet. Furthermore, increased TMAO levels were associated with less improvement in those following a high-fat diet[207]. A combined intervention of a low-calorie diet and exercise was assessed to determine whether it could reduce TMAO levels more effectively compared to diet alone in obese women[208]. Similarly, other studies involving comparable populations and interventions found that although TMAO levels do not significantly change, it is suggested that a combined intervention may be useful in decreasing TMAO production[209,210] (Table 1). Approaches that combine exercise and diet show clear metabolic benefits and could be useful in reducing TMAO. There are specialized diets, such as the Dietary Approaches to Stop Hypertension[211] that have shown a significant decrease in TMAO levels and the Firmicutes/Bacteroidetes index, which influences TMAO levels (Table 1). The cardiovascular benefits of the Dietary Approaches to Stop Hypertension diet may have a more favorable impact on microbial metabolites involved in cardiovascular risk.
CAD progresses with high TMAO levels, so reducing this metabolite is a key therapeutic goal. A study evaluated how the consumption of products rich in L-carnitine and phosphatidylcholine influences TMAO levels in CAD patients. Compared with the control group, CAD patients more frequently consumed red meat and dairy products and had higher blood concentrations of this metabolite and a greater abundance of TMA-producing bacteria[212]. Wine consumption has been associated with cardiovascular benefits[213] (Table 1), although no changes in TMAO levels have been observed, changes in GM and an improvement in redox balance were observed[214]. A study in men with a history of atherosclerotic disease evaluated the effect of Lactobacillus plantarum GLP3 supplementation, and patients showed a significant reduction in TMAO levels[215] (Table 1). In contrast, Malik et al[216] reported that supplementation with the probiotic Lactobacillus plantarum 299v improved vascular endothelial function and reduced inflammatory biomarkers, despite no measurable reduction in circulating TMAO. (Table 1). In another hand, supplementation with Lactobacillus casei Shirota produced a non-significant reduction in TMAO (Table 1). The variability in results with probiotics, prebiotics, and other modulators indicates that many formulations have yet to consistently alter this metabolite[217] (Table 1). Reducing TMAO levels in CAD patients is not only possible but also relevant to prevent cardiovascular risk.
The most consistent TMAO lowering occurs when red meat is restricted and protein is shifted toward non-meat sources[218]. Complementary evidence shows that plant-forward substitutions can reduce TMAO over 8 weeks[219]. Argyridou et al[191] suggests rapid responsiveness to vegan diet, with fasting TMAO reductions detectable by week 1 and maintained at week 8. By contrast, fiber supplementation shows context-dependent effects; it showed a potential effect in individuals with lower habitual meat intake[220]. Finally, broader pattern changes such as a 6-month Mediterranean diet not uniformly lower fasting TMAO[221]. Same results reported Bergeron et al[222], evaluated diets varying in resistant starch and carbohydrate contests and concluded that high resistant-starch intake increased plasma TMAO levels.
Interventional evidence is still lacking to demonstrate that lowering TMAO reduces MACE, most clinical studies are not good quality[191]. In PEGASUS-TIMI 54, higher baseline TMAO related to certain outcomes, yet ticagrelor’s efficacy was consistent irrespective of TMAO levels, illustrating that a TMAO risk signal does not automatically translate into a TMAO-dependent treatment effect[156]. Notably, TMAO levels can also increase in contexts not clearly harmful, such as after fish intake and “healthy” diets enriched in marine n-3 fatty acids[13,223], complicating a superficial “lower is better” interpretation. Meanwhile, dietary manipulation can meaningfully shift TMAO levels within weeks[218,219], but these remain surrogate-endpoint demonstrations rather than proof of MACE reduction. Ongoing registered studies targeting the pathway are primarily designed around TMAO modulation, not MACE endpoints, including trials in CKD (ClinicalTrials ID: NCTNCT03152097 and NCT03348592), and mechanistic nutrition designs (ClinicalTrials ID: NCT06518343).
Several strategies to decrease TMAO levels have shown promise. Ranging from dietary modifications and targeted nutrient substitution to GM directed approaches, such as the use of probiotics, prebiotics and more experimental tools such as 3,3-DMB, fluoromethylcholine and FMT. Collectively, these interventions underscore the potential to modulate GM and its metabolite output as a complementary avenue for CVD prevention and management.
Yet, the process of translating findings faces some limitations due to significant variations in dietary intake, kidney function, comorbidities, prescribed medications, and baseline GM. All these factors can greatly affect levels of circulating TMAO, making it difficult to draw causal relationships and compare results across different studies. Thus, upcoming investigations should focus on meticulously controlled and adequately sized human trials that unify sampling methods, pre-examination procedures, and testing systems; incorporate microbial “producer” profiling and functional outputs, such as cutC/D capability, in association with host factors and evaluate if strategies targeting TMAO provide additional predictive value beyond current risk assessment scores and, importantly, lead to enhancements in significant clinical outcomes. Setting a consistent clinical threshold and showing improved outcomes in forward-looking trials will be crucial for transitioning TMAO from merely a strong association to a clinically relevant biomarker and treatment focus.
Risk factors and unhealthy lifestyle choices can disrupt GM balance, negatively affecting cardiovascular health. When dysbiosis occurs, metabolites such as TMAO are generated, which plays a role in the activation of pathophysiological mechanisms. Several studies have shown that elevated TMAO levels are associated with the progression and poor prognosis of ACS, CVA, and HF, due to its ability to cross biological barriers such as the gut and the blood-brain barrier and directly influence cardiac or brain function. The apparent clinical benefit often mirrors implicit assumptions, that lowering TMAO certainly modifies risk, that studies are directly comparable, and that TMAO is an independent causal marker, in reality, context like diet, renal function, comorbidities, methodological heterogeneity, and reliance on surrogate endpoints may account for discordant findings without, by themselves, excluding a biologically relevant role for the TMA/TMAO axis. Future progress will depend on standardized pre-analytical and analytical workflows, functional stratification of GM producer phenotypes, and adequately powered prospective trials designed to test whether TMAO-targeted interventions provide incremental prognostic value and translate into decreases of adjudicated MACE.
The authors would like to thank Dr. Brenda Rosario Sandoval Meza, part of the Medical Translation Area at the Research Division, Faculty of Medicine, Universidad Nacional Autónoma de México for translating and proofreading this paper.
| 1. | Khoury H, Lavoie L, Welner S, Folkerts K. The Burden of Major Adverse Cardiac Events and Antiplatelet Prevention in Patients with Coronary or Peripheral Arterial Disease. Cardiovasc Ther. 2016;34:115-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | World Health Organization. Cardiovascular diseases fact sheet. Jul 31, 2025. [cited 1 Nov 2025]. Available from: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds). |
| 3. | Lau K, Srivatsav V, Rizwan A, Nashed A, Liu R, Shen R, Akhtar M. Bridging the Gap between Gut Microbial Dysbiosis and Cardiovascular Diseases. Nutrients. 2017;9:859. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 143] [Article Influence: 15.9] [Reference Citation Analysis (1)] |
| 4. | Wang J, Zhu N, Su X, Gao Y, Yang R. Gut-Microbiota-Derived Metabolites Maintain Gut and Systemic Immune Homeostasis. Cells. 2023;12:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 445] [Cited by in RCA: 386] [Article Influence: 128.7] [Reference Citation Analysis (1)] |
| 5. | Hassan D, Zahra T, Kanaan G, Khan MU, Mushtaq K, Nashwan AJ, Hamid PF. The Impact of Gut Microbiome Constitution to Reduce Cardiovascular Disease Risk: A Systematic Review and Meta-Analysis. Curr Probl Cardiol. 2023;48:101459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013;11:227-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3064] [Cited by in RCA: 2539] [Article Influence: 195.3] [Reference Citation Analysis (3)] |
| 7. | Yoo JY, Sniffen S, McGill Percy KC, Pallaval VB, Chidipi B. Gut Dysbiosis and Immune System in Atherosclerotic Cardiovascular Disease (ACVD). Microorganisms. 2022;10:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 8. | Papandreou C, Moré M, Bellamine A. Trimethylamine N-Oxide in Relation to Cardiometabolic Health-Cause or Effect? Nutrients. 2020;12:1330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 105] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 9. | Canyelles M, Borràs C, Rotllan N, Tondo M, Escolà-Gil JC, Blanco-Vaca F. Gut Microbiota-Derived TMAO: A Causal Factor Promoting Atherosclerotic Cardiovascular Disease? Int J Mol Sci. 2023;24:1940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 119] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 10. | Ussher JR, Lopaschuk GD, Arduini A. Gut microbiota metabolism of L-carnitine and cardiovascular risk. Atherosclerosis. 2013;231:456-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 146] [Article Influence: 11.2] [Reference Citation Analysis (8)] |
| 11. | Farhangi MA. Gut microbiota-dependent trimethylamine N-oxide and all-cause mortality: Findings from an updated systematic review and meta-analysis. Nutrition. 2020;78:110856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 12. | Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, Britt EB, Fu X, Wu Y, Li L, Smith JD, DiDonato JA, Chen J, Li H, Wu GD, Lewis JD, Warrier M, Brown JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, Hazen SL. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med. 2013;19:576-585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3789] [Cited by in RCA: 3328] [Article Influence: 256.0] [Reference Citation Analysis (7)] |
| 13. | Cho CE, Taesuwan S, Malysheva OV, Bender E, Tulchinsky NF, Yan J, Sutter JL, Caudill MA. Trimethylamine-N-oxide (TMAO) response to animal source foods varies among healthy young men and is influenced by their gut microbiota composition: A randomized controlled trial. Mol Nutr Food Res. 2017;61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 286] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 14. | Renvert S, Pettersson T, Ohlsson O, Persson GR. Bacterial profile and burden of periodontal infection in subjects with a diagnosis of acute coronary syndrome. J Periodontol. 2006;77:1110-1119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Serino M, Blasco-Baque V, Nicolas S, Burcelin R. Far from the eyes, close to the heart: dysbiosis of gut microbiota and cardiovascular consequences. Curr Cardiol Rep. 2014;16:540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 16. | Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med. 2013;368:1575-1584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2881] [Cited by in RCA: 2550] [Article Influence: 196.2] [Reference Citation Analysis (6)] |
| 17. | Schoch L, Alcover S, Padró T, Ben-Aicha S, Mendieta G, Badimon L, Vilahur G. Update of HDL in atherosclerotic cardiovascular disease. Clin Investig Arterioscler. 2023;35:297-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 18. | Nayor M, Brown KJ, Vasan RS. The Molecular Basis of Predicting Atherosclerotic Cardiovascular Disease Risk. Circ Res. 2021;128:287-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 19. | Sandek A, Bauditz J, Swidsinski A, Buhner S, Weber-Eibel J, von Haehling S, Schroedl W, Karhausen T, Doehner W, Rauchhaus M, Poole-Wilson P, Volk HD, Lochs H, Anker SD. Altered intestinal function in patients with chronic heart failure. J Am Coll Cardiol. 2007;50:1561-1569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 499] [Article Influence: 26.3] [Reference Citation Analysis (4)] |
| 20. | Mansour MA. Hyperlipidemia. In: Sidawy AN, Perler BA. Rutherford’s vascular surgery and endovascular therapy. Netherlands: Elsevier, 2023: 143-160. |
| 21. | Seldin MM, Meng Y, Qi H, Zhu W, Wang Z, Hazen SL, Lusis AJ, Shih DM. Trimethylamine N-Oxide Promotes Vascular Inflammation Through Signaling of Mitogen-Activated Protein Kinase and Nuclear Factor-κB. J Am Heart Assoc. 2016;5:e002767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 779] [Cited by in RCA: 730] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 22. | Zhu W, Gregory JC, Org E, Buffa JA, Gupta N, Wang Z, Li L, Fu X, Wu Y, Mehrabian M, Sartor RB, McIntyre TM, Silverstein RL, Tang WHW, DiDonato JA, Brown JM, Lusis AJ, Hazen SL. Gut Microbial Metabolite TMAO Enhances Platelet Hyperreactivity and Thrombosis Risk. Cell. 2016;165:111-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1716] [Cited by in RCA: 1516] [Article Influence: 151.6] [Reference Citation Analysis (4)] |
| 23. | Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4847] [Cited by in RCA: 4279] [Article Influence: 285.3] [Reference Citation Analysis (7)] |
| 24. | Hou K, Wu ZX, Chen XY, Wang JQ, Zhang D, Xiao C, Zhu D, Koya JB, Wei L, Li J, Chen ZS. Microbiota in health and diseases. Signal Transduct Target Ther. 2022;7:135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2537] [Cited by in RCA: 2040] [Article Influence: 510.0] [Reference Citation Analysis (6)] |
| 25. | Moludi J, Saiedi S, Ebrahimi B, Alizadeh M, Khajebishak Y, Ghadimi SS. Probiotics Supplementation on Cardiac Remodeling Following Myocardial Infarction: a Single-Center Double-Blind Clinical Study. J Cardiovasc Transl Res. 2021;14:299-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 26. | Tan Y, Sheng Z, Zhou P, Liu C, Zhao H, Song L, Li J, Zhou J, Chen Y, Wang L, Qian H, Sun Z, Qiao S, Xu B, Gao R, Yan H. Plasma Trimethylamine N-Oxide as a Novel Biomarker for Plaque Rupture in Patients With ST-Segment-Elevation Myocardial Infarction. Circ Cardiovasc Interv. 2019;12:e007281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 27. | American Heart Association. Acute coronary syndrome. Dec 10, 2024. [cited 1 Nov 2025]. Available from: https://www.heart.org/en/health-topics/heart-attack/about-heart-attacks/acute-coronary-syndrome. |
| 28. | Li Y, Zhang D, He Y, Chen C, Song C, Zhao Y, Bai Y, Wang Y, Pu J, Chen J, Yang Y, Dou K. Investigation of novel metabolites potentially involved in the pathogenesis of coronary heart disease using a UHPLC-QTOF/MS-based metabolomics approach. Sci Rep. 2017;7:15357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 29. | Zhong Z, Liu J, Zhang Q, Zhong W, Li B, Li C, Liu Z, Yang M, Zhao P. Targeted metabolomic analysis of plasma metabolites in patients with coronary heart disease in southern China. Medicine (Baltimore). 2019;98:e14309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 30. | Fu Q, Zhao M, Wang D, Hu H, Guo C, Chen W, Li Q, Zheng L, Chen B. Coronary Plaque Characterization Assessed by Optical Coherence Tomography and Plasma Trimethylamine-N-oxide Levels in Patients With Coronary Artery Disease. Am J Cardiol. 2016;118:1311-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 31. | Dean YE, Rouzan SS, Loayza Pintado JJ, Talat NE, Mohamed ARH, Verma S, Anwar Kamdi Z, Gir D, Helmy A, Helmy Z, Afzal A, Mady T, Hazimeh Y, Aiash H. Serum trimethylamine N-oxide levels among coronary artery disease and acute coronary syndrome patients: a systematic review and meta-analysis. Ann Med Surg (Lond). 2023;85:6123-6133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 32. | American Heart Association. Coronary artery disease - coronary heart disease. Jan 10, 2024. [cited 1 Nov 2025]. Available from: https://www.heart.org/en/health-topics/consumer-healthcare/what-is-cardiovascular-disease/coronary-artery-disease. |
| 33. | Kong W, Ma J, Lin Y, Chen W. Positive Association of Plasma Trimethylamine-N-Oxide and Atherosclerosis in Patient with Acute Coronary Syndrome. Cardiovasc Ther. 2022;2022:2484018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Han Y, Gong Z, Sun G, Xu J, Qi C, Sun W, Jiang H, Cao P, Ju H. Dysbiosis of Gut Microbiota in Patients With Acute Myocardial Infarction. Front Microbiol. 2021;12:680101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (3)] |
| 35. | Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD; Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). Circulation. 2018;138:e618-e651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2931] [Cited by in RCA: 2560] [Article Influence: 320.0] [Reference Citation Analysis (2)] |
| 36. | Bentzon JF, Otsuka F, Virmani R, Falk E. Mechanisms of plaque formation and rupture. Circ Res. 2014;114:1852-1866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1980] [Cited by in RCA: 1684] [Article Influence: 140.3] [Reference Citation Analysis (3)] |
| 37. | Hernández-Ruiz P, Escalona Montaño AR, Amezcua-Guerra LM, González-Pacheco H, Niccolai E, Amedei A, Aguirre-García MM. Potential Association of the Oral Microbiome with Trimethylamine N-Oxide Quantification in Mexican Patients with Myocardial Infarction. Mediators Inflamm. 2024;2024:3985731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 38. | Alhmoud T, Kumar A, Lo CC, Al-Sadi R, Clegg S, Alomari I, Zmeili T, Gleasne CD, Mcmurry K, Dichosa AEK, Vuyisich M, Chain PSG, Mishra S, Ma T. Investigating intestinal permeability and gut microbiota roles in acute coronary syndrome patients. Hum Microb J. 2019;13:100059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 39. | Matsuzawa Y, Nakahashi H, Konishi M, Sato R, Kawashima C, Kikuchi S, Akiyama E, Iwahashi N, Maejima N, Okada K, Ebina T, Hibi K, Kosuge M, Ishigami T, Tamura K, Kimura K. Microbiota-derived Trimethylamine N-oxide Predicts Cardiovascular Risk After STEMI. Sci Rep. 2019;9:11647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 40. | Dolkar P, Deyang T, Anand N, Rathipriya AG, Hediyal TA, Chandrasekaran V, Krishnamoorthy NK, Gorantla VR, Bishir M, Rashan L, Chang SL, Sakharkar MK, Yang J, Chidambaram SB. Trimethylamine-N-oxide and cerebral stroke risk: A review. Neurobiol Dis. 2024;192:106423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 41. | American Association of Neurological Surgeons. Cerebrovascular disease. Apr 29, 2024. [cited 3 Jul 2025]. Available from: https://www.aans.org/patients/conditions-treatments/cerebrovascular-disease/. |
| 42. | Lana D, Giovannini MG. The Microbiota-Gut-Brain Axis in Behaviour and Brain Disorders. Int J Mol Sci. 2023;24:8460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 43. | Nie J, Xie L, Zhao BX, Li Y, Qiu B, Zhu F, Li GF, He M, Wang Y, Wang B, Liu S, Zhang H, Guo H, Cai Y, Huo Y, Hou FF, Xu X, Qin X. Serum Trimethylamine N-Oxide Concentration Is Positively Associated With First Stroke in Hypertensive Patients. Stroke. 2018;49:2021-2028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 133] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 44. | Xue J, Xu J, Zhao M, Jin A, Cheng A, Jiang X, Li K, Lin J, Meng X, Li H, Zheng L, Wang Y. Residual Risk of Trimethylamine-N-Oxide and Choline for Stroke Recurrence in Patients With Intensive Secondary Therapy. J Am Heart Assoc. 2022;11:e027265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 45. | Haghikia A, Li XS, Liman TG, Bledau N, Schmidt D, Zimmermann F, Kränkel N, Widera C, Sonnenschein K, Haghikia A, Weissenborn K, Fraccarollo D, Heimesaat MM, Bauersachs J, Wang Z, Zhu W, Bavendiek U, Hazen SL, Endres M, Landmesser U. Gut Microbiota-Dependent Trimethylamine N-Oxide Predicts Risk of Cardiovascular Events in Patients With Stroke and Is Related to Proinflammatory Monocytes. Arterioscler Thromb Vasc Biol. 2018;38:2225-2235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 246] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 46. | Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GMC, Coats AJS. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2023;118:3272-3287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2589] [Cited by in RCA: 2155] [Article Influence: 718.3] [Reference Citation Analysis (1)] |
| 47. | Tang WH, Wang Z, Shrestha K, Borowski AG, Wu Y, Troughton RW, Klein AL, Hazen SL. Intestinal microbiota-dependent phosphatidylcholine metabolites, diastolic dysfunction, and adverse clinical outcomes in chronic systolic heart failure. J Card Fail. 2015;21:91-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 303] [Cited by in RCA: 273] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 48. | Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW; ACC/AHA Joint Committee Members. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1736] [Cited by in RCA: 1858] [Article Influence: 464.5] [Reference Citation Analysis (2)] |
| 49. | Krack A, Sharma R, Figulla HR, Anker SD. The importance of the gastrointestinal system in the pathogenesis of heart failure. Eur Heart J. 2005;26:2368-2374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 209] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 50. | Anker SD, Egerer KR, Volk HD, Kox WJ, Poole-Wilson PA, Coats AJ. Elevated soluble CD14 receptors and altered cytokines in chronic heart failure. Am J Cardiol. 1997;79:1426-1430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 260] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 51. | Deswal A, Petersen NJ, Feldman AM, Young JB, White BG, Mann DL. Cytokines and cytokine receptors in advanced heart failure: an analysis of the cytokine database from the Vesnarinone trial (VEST). Circulation. 2001;103:2055-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 736] [Article Influence: 29.4] [Reference Citation Analysis (3)] |
| 52. | Triposkiadis F, Karayannis G, Giamouzis G, Skoularigis J, Louridas G, Butler J. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications. J Am Coll Cardiol. 2009;54:1747-1762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 704] [Article Influence: 41.4] [Reference Citation Analysis (0)] |
| 53. | Tang WHW, Li DY, Hazen SL. Dietary metabolism, the gut microbiome, and heart failure. Nat Rev Cardiol. 2019;16:137-154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 672] [Cited by in RCA: 586] [Article Influence: 83.7] [Reference Citation Analysis (4)] |
| 54. | Rodrigues A, Gonçalves A, Morais J, Araujo R, Falcão-Pires I. Diet-Induced Microbiome's Impact on Heart Failure: A Double-Edged Sword. Nutrients. 2023;15:1223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 55. | Gesper M, Nonnast ABH, Kumowski N, Stoehr R, Schuett K, Marx N, Kappel BA. Gut-Derived Metabolite Indole-3-Propionic Acid Modulates Mitochondrial Function in Cardiomyocytes and Alters Cardiac Function. Front Med (Lausanne). 2021;8:648259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 56. | Luedde M, Winkler T, Heinsen FA, Rühlemann MC, Spehlmann ME, Bajrovic A, Lieb W, Franke A, Ott SJ, Frey N. Heart failure is associated with depletion of core intestinal microbiota. ESC Heart Fail. 2017;4:282-290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 241] [Article Influence: 26.8] [Reference Citation Analysis (1)] |
| 57. | Katsimichas T, Ohtani T, Motooka D, Tsukamoto Y, Kioka H, Nakamoto K, Konishi S, Chimura M, Sengoku K, Miyawaki H, Sakaguchi T, Okumura R, Theofilis K, Iida T, Takeda K, Nakamura S, Sakata Y. Non-Ischemic Heart Failure With Reduced Ejection Fraction Is Associated With Altered Intestinal Microbiota. Circ J. 2018;82:1640-1650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 58. | Pasini E, Aquilani R, Testa C, Baiardi P, Angioletti S, Boschi F, Verri M, Dioguardi F. Pathogenic Gut Flora in Patients With Chronic Heart Failure. JACC Heart Fail. 2016;4:220-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 341] [Article Influence: 34.1] [Reference Citation Analysis (0)] |
| 59. | Trøseid M, Ueland T, Hov JR, Svardal A, Gregersen I, Dahl CP, Aakhus S, Gude E, Bjørndal B, Halvorsen B, Karlsen TH, Aukrust P, Gullestad L, Berge RK, Yndestad A. Microbiota-dependent metabolite trimethylamine-N-oxide is associated with disease severity and survival of patients with chronic heart failure. J Intern Med. 2015;277:717-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 364] [Article Influence: 33.1] [Reference Citation Analysis (3)] |
| 60. | The Joint Commission. New York Heart Association (NYHA) Classification. Sept 4, 2025. [cited 1 Nov 2025]. Available from: https://manual.jointcommission.org/releases/TJC2025B1/DataElem0439.html. |
| 61. | Tang WH, Wang Z, Fan Y, Levison B, Hazen JE, Donahue LM, Wu Y, Hazen SL. Prognostic value of elevated levels of intestinal microbe-generated metabolite trimethylamine-N-oxide in patients with heart failure: refining the gut hypothesis. J Am Coll Cardiol. 2014;64:1908-1914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 614] [Cited by in RCA: 563] [Article Influence: 46.9] [Reference Citation Analysis (4)] |
| 62. | Tang WHW, Lemaitre RN, Jensen PN, Wang M, Wang Z, Li XS, Nemet I, Lee Y, Lai HTM, de Oliveira Otto MC, Fretts AM, Sotoodehnia N, DiDonato JA, Bäckhed F, Psaty BM, Siscovick DS, Budoff MJ, Mozaffarian D, Hazen SL. Trimethylamine N-Oxide and Related Gut Microbe-Derived Metabolites and Incident Heart Failure Development in Community-Based Populations. Circ Heart Fail. 2024;17:e011569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 63. | López-Tenorio II, Aguilar-Villegas ÓR, Espinoza-Palacios Y, Segura-Real L, Peña-Aparicio B, Amedei A, Aguirre-García MM. Primary Prevention Strategy for Non-Communicable Diseases (NCDs) and Their Risk Factors: The Role of Intestinal Microbiota. Biomedicines. 2024;12:2529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 64. | Turnbaugh PJ, Bäckhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3:213-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2527] [Cited by in RCA: 2209] [Article Influence: 122.7] [Reference Citation Analysis (10)] |
| 65. | Fouesnard M, Zoppi J, Petera M, Le Gleau L, Migné C, Devime F, Durand S, Benani A, Chaffron S, Douard V, Boudry G. Dietary switch to Western diet induces hypothalamic adaptation associated with gut microbiota dysbiosis in rats. Int J Obes (Lond). 2021;45:1271-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 66. | Wang Z, Bergeron N, Levison BS, Li XS, Chiu S, Jia X, Koeth RA, Li L, Wu Y, Tang WHW, Krauss RM, Hazen SL. Impact of chronic dietary red meat, white meat, or non-meat protein on trimethylamine N-oxide metabolism and renal excretion in healthy men and women. Eur Heart J. 2019;40:583-594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 420] [Cited by in RCA: 385] [Article Influence: 55.0] [Reference Citation Analysis (0)] |
| 67. | Yoo W, Zieba JK, Foegeding NJ, Torres TP, Shelton CD, Shealy NG, Byndloss AJ, Cevallos SA, Gertz E, Tiffany CR, Thomas JD, Litvak Y, Nguyen H, Olsan EE, Bennett BJ, Rathmell JC, Major AS, Bäumler AJ, Byndloss MX. High-fat diet-induced colonocyte dysfunction escalates microbiota-derived trimethylamine N-oxide. Science. 2021;373:813-818. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 250] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 68. | Fritsch J, Garces L, Quintero MA, Pignac-Kobinger J, Santander AM, Fernández I, Ban YJ, Kwon D, Phillips MC, Knight K, Mao Q, Santaolalla R, Chen XS, Maruthamuthu M, Solis N, Damas OM, Kerman DH, Deshpande AR, Lewis JE, Chen C, Abreu MT. Low-Fat, High-Fiber Diet Reduces Markers of Inflammation and Dysbiosis and Improves Quality of Life in Patients With Ulcerative Colitis. Clin Gastroenterol Hepatol. 2021;19:1189-1199.e30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 250] [Article Influence: 50.0] [Reference Citation Analysis (3)] |
| 69. | Ghosh TS, Rampelli S, Jeffery IB, Santoro A, Neto M, Capri M, Giampieri E, Jennings A, Candela M, Turroni S, Zoetendal EG, Hermes GDA, Elodie C, Meunier N, Brugere CM, Pujos-Guillot E, Berendsen AM, De Groot LCPGM, Feskins EJM, Kaluza J, Pietruszka B, Bielak MJ, Comte B, Maijo-Ferre M, Nicoletti C, De Vos WM, Fairweather-Tait S, Cassidy A, Brigidi P, Franceschi C, O'Toole PW. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut. 2020;69:1218-1228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 826] [Cited by in RCA: 692] [Article Influence: 115.3] [Reference Citation Analysis (1)] |
| 70. | Desai MS, Seekatz AM, Koropatkin NM, Kamada N, Hickey CA, Wolter M, Pudlo NA, Kitamoto S, Terrapon N, Muller A, Young VB, Henrissat B, Wilmes P, Stappenbeck TS, Núñez G, Martens EC. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell. 2016;167:1339-1353.e21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2431] [Cited by in RCA: 2098] [Article Influence: 209.8] [Reference Citation Analysis (4)] |
| 71. | De Filippis F, Pellegrini N, Vannini L, Jeffery IB, La Storia A, Laghi L, Serrazanetti DI, Di Cagno R, Ferrocino I, Lazzi C, Turroni S, Cocolin L, Brigidi P, Neviani E, Gobbetti M, O'Toole PW, Ercolini D. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. 2016;65:1812-1821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1352] [Cited by in RCA: 1144] [Article Influence: 114.4] [Reference Citation Analysis (3)] |
| 72. | Haas M, Brandl B, Neuhaus K, Wudy S, Kleigrewe K, Hauner H, Skurk T. Effect of dietary fiber on trimethylamine-N-oxide production after beef consumption and on gut microbiota: MEATMARK - a randomized cross-over study. Eur J Clin Nutr. 2025;79:980-990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 73. | Krishnan S, O'Connor LE, Wang Y, Gertz ER, Campbell WW, Bennett BJ. Adopting a Mediterranean-style eating pattern with low, but not moderate, unprocessed, lean red meat intake reduces fasting serum trimethylamine N-oxide (TMAO) in adults who are overweight or obese. Br J Nutr. 2021;128:1-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 74. | Xiong L, Diwakarla S, Chatzis R, Artaiz O, Macowan M, Zhang S, Garnham A, Morgan PK, Mellett NA, Meikle PJ, Lancaster GI, Marsland BJ, Nutt SL, Seillet C. Acute exposure to high-fat diet impairs ILC3 functions and gut homeostasis. Immunity. 2025;58:1185-1200.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 31] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 75. | Hewady S, Manuel CR, Pasquali C, Koya J, Reznik SE. OM-85 attenuates high-fat diet-induced obesity, insulin resistance, gut dysbiosis and nonalcoholic steatohepatitis in a murine model. Biomed Pharmacother. 2024;181:117710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 76. | Szudzik M, Zajdel M, Samborowska E, Perlejewski K, Radkowski M, Ufnal M. High-Fat Diet with Normal Caloric Intake Elevates TMA and TMAO Production and Reduces Microbial Diversity in Rats. Nutrients. 2025;17:2230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 77. | Ghosh TS, Shanahan F, O'Toole PW. The gut microbiome as a modulator of healthy ageing. Nat Rev Gastroenterol Hepatol. 2022;19:565-584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 658] [Cited by in RCA: 553] [Article Influence: 138.3] [Reference Citation Analysis (0)] |
| 78. | Rera M, Clark RI, Walker DW. Intestinal barrier dysfunction links metabolic and inflammatory markers of aging to death in Drosophila. Proc Natl Acad Sci U S A. 2012;109:21528-21533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 587] [Cited by in RCA: 487] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 79. | Conley MN, Wong CP, Duyck KM, Hord N, Ho E, Sharpton TJ. Aging and serum MCP-1 are associated with gut microbiome composition in a murine model. PeerJ. 2016;4:e1854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 80. | Thevaranjan N, Puchta A, Schulz C, Naidoo A, Szamosi JC, Verschoor CP, Loukov D, Schenck LP, Jury J, Foley KP, Schertzer JD, Larché MJ, Davidson DJ, Verdú EF, Surette MG, Bowdish DME. Age-Associated Microbial Dysbiosis Promotes Intestinal Permeability, Systemic Inflammation, and Macrophage Dysfunction. Cell Host Microbe. 2017;21:455-466.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1066] [Cited by in RCA: 944] [Article Influence: 104.9] [Reference Citation Analysis (4)] |
| 81. | Salminen A, Huuskonen J, Ojala J, Kauppinen A, Kaarniranta K, Suuronen T. Activation of innate immunity system during aging: NF-kB signaling is the molecular culprit of inflamm-aging. Ageing Res Rev. 2008;7:83-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 433] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 82. | Claesson MJ, Cusack S, O'Sullivan O, Greene-Diniz R, de Weerd H, Flannery E, Marchesi JR, Falush D, Dinan T, Fitzgerald G, Stanton C, van Sinderen D, O'Connor M, Harnedy N, O'Connor K, Henry C, O'Mahony D, Fitzgerald AP, Shanahan F, Twomey C, Hill C, Ross RP, O'Toole PW. Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc Natl Acad Sci U S A. 2011;108 Suppl 1:4586-4591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1476] [Cited by in RCA: 1274] [Article Influence: 84.9] [Reference Citation Analysis (8)] |
| 83. | Hopkins MJ, Macfarlane GT. Changes in predominant bacterial populations in human faeces with age and with Clostridium difficile infection. J Med Microbiol. 2002;51:448-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 389] [Cited by in RCA: 330] [Article Influence: 13.8] [Reference Citation Analysis (3)] |
| 84. | Woodmansey EJ, McMurdo ME, Macfarlane GT, Macfarlane S. Comparison of compositions and metabolic activities of fecal microbiotas in young adults and in antibiotic-treated and non-antibiotic-treated elderly subjects. Appl Environ Microbiol. 2004;70:6113-6122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 296] [Article Influence: 13.5] [Reference Citation Analysis (3)] |
| 85. | Rath S, Rox K, Kleine Bardenhorst S, Schminke U, Dörr M, Mayerle J, Frost F, Lerch MM, Karch A, Brönstrup M, Pieper DH, Vital M. Higher Trimethylamine-N-Oxide Plasma Levels with Increasing Age Are Mediated by Diet and Trimethylamine-Forming Bacteria. mSystems. 2021;6:e0094521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 86. | Ganapathy T, Yuan J, Ho MY, Wu KK, Hoque MM, Wang B, Li X, Wang K, Wabitsch M, Feng X, Niu Y, Long K, Lian Q, Zhu Y, Cheng KK. Adipocyte FMO3-derived TMAO induces WAT dysfunction and metabolic disorders by promoting inflammasome activation in ageing. Nat Commun. 2025;16:8873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 87. | Ke Y, Li D, Zhao M, Liu C, Liu J, Zeng A, Shi X, Cheng S, Pan B, Zheng L, Hong H. Gut flora-dependent metabolite Trimethylamine-N-oxide accelerates endothelial cell senescence and vascular aging through oxidative stress. Free Radic Biol Med. 2018;116:88-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 219] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 88. | Scott NA, Andrusaite A, Andersen P, Lawson M, Alcon-Giner C, Leclaire C, Caim S, Le Gall G, Shaw T, Connolly JPR, Roe AJ, Wessel H, Bravo-Blas A, Thomson CA, Kästele V, Wang P, Peterson DA, Bancroft A, Li X, Grencis R, Mowat AM, Hall LJ, Travis MA, Milling SWF, Mann ER. Antibiotics induce sustained dysregulation of intestinal T cell immunity by perturbing macrophage homeostasis. Sci Transl Med. 2018;10:eaao4755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 295] [Cited by in RCA: 264] [Article Influence: 33.0] [Reference Citation Analysis (4)] |
| 89. | Strati F, Pujolassos M, Burrello C, Giuffrè MR, Lattanzi G, Caprioli F, Troisi J, Facciotti F. Antibiotic-associated dysbiosis affects the ability of the gut microbiota to control intestinal inflammation upon fecal microbiota transplantation in experimental colitis models. Microbiome. 2021;9:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 96] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 90. | Burrello C, Garavaglia F, Cribiù FM, Ercoli G, Bosari S, Caprioli F, Facciotti F. Short-term Oral Antibiotics Treatment Promotes Inflammatory Activation of Colonic Invariant Natural Killer T and Conventional CD4(+) T Cells. Front Med (Lausanne). 2018;5:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 91. | Jabs S, Biton A, Bécavin C, Nahori MA, Ghozlane A, Pagliuso A, Spanò G, Guérineau V, Touboul D, Giai Gianetto Q, Chaze T, Matondo M, Dillies MA, Cossart P. Impact of the gut microbiota on the m(6)A epitranscriptome of mouse cecum and liver. Nat Commun. 2020;11:1344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 92. | Yang M, Zheng X, Fan J, Cheng W, Yan TM, Lai Y, Zhang N, Lu Y, Qi J, Huo Z, Xu Z, Huang J, Jiao Y, Liu B, Pang R, Zhong X, Huang S, Luo GZ, Lee G, Jobin C, Eren AM, Chang EB, Wei H, Pan T, Wang X. Antibiotic-Induced Gut Microbiota Dysbiosis Modulates Host Transcriptome and m(6)A Epitranscriptome via Bile Acid Metabolism. Adv Sci (Weinh). 2024;11:e2307981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 93. | Hoyles L, Jiménez-Pranteda ML, Chilloux J, Brial F, Myridakis A, Aranias T, Magnan C, Gibson GR, Sanderson JD, Nicholson JK, Gauguier D, McCartney AL, Dumas ME. Metabolic retroconversion of trimethylamine N-oxide and the gut microbiota. Microbiome. 2018;6:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (4)] |
| 94. | Pilarczyk-Zurek M, Sitkiewicz I, Koziel J. The Clinical View on Streptococcus anginosus Group - Opportunistic Pathogens Coming Out of Hiding. Front Microbiol. 2022;13:956677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 95. | Huo T, Cai S, Lu X, Sha Y, Yu M, Li F. Metabonomic study of biochemical changes in the serum of type 2 diabetes mellitus patients after the treatment of metformin hydrochloride. J Pharm Biomed Anal. 2009;49:976-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 85] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 96. | Kuka J, Videja M, Makrecka-Kuka M, Liepins J, Grinberga S, Sevostjanovs E, Vilks K, Liepinsh E, Dambrova M. Metformin decreases bacterial trimethylamine production and trimethylamine N-oxide levels in db/db mice. Sci Rep. 2020;10:14555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 97. | Glaros SB, Mishra SP, Jain S, Davis FS, Gabel SA, Mueller GA, Jarmusch AK, Mabundo L, Courville AB, Walter MF, Walter PJ, Overdahl KE, Yadav H, Chung ST. Systemic and gut microbiome changes with metformin and liraglutide in youth-onset type 2 diabetes: the MIGHTY study. Gut Microbes. 2025;17:2558071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 98. | Su C, Li X, Yang Y, Du Y, Zhang X, Wang L, Hong B. Metformin alleviates choline diet-induced TMAO elevation in C57BL/6J mice by influencing gut-microbiota composition and functionality. Nutr Diabetes. 2021;11:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 99. | Melenovský V, Hošková E, Velebová K, Veleba J, Borlaug BA, Benes J, Kuda O, Čajka T, Segeťová M, Thieme L, Kopecký J Jr, Kopecký J Sr, Pelikánová T, Haluzík M, Hill M, Kahleová H. Metformin Improves Glycemic Control and Postprandial Metabolism and Enhances Postprandial Glucagon-Like Peptide 1 Secretion in Patients With Type 2 Diabetes and Heart Failure: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin Diabetes. 2025;43:23-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 100. | Meador M, Lewis JH, Bay RC, Wall HK, Jackson C. Who Are the Undiagnosed? Disparities in Hypertension Diagnoses in Vulnerable Populations. Fam Community Health. 2020;43:35-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 101. | Eslami A, Naghibi Irvani SS, Ramezankhani A, Fekri N, Asadi K, Azizi F, Hadaegh F. Incidence and associated risk factors for premature death in the Tehran Lipid and Glucose Study cohort, Iran. BMC Public Health. 2019;19:719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 102. | Kjeldsen SE. Hypertension and cardiovascular risk: General aspects. Pharmacol Res. 2018;129:95-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 424] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 103. | Fuchs FD, Whelton PK. High Blood Pressure and Cardiovascular Disease. Hypertension. 2020;75:285-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1587] [Cited by in RCA: 1237] [Article Influence: 206.2] [Reference Citation Analysis (1)] |
| 104. | Li J, Zhao F, Wang Y, Chen J, Tao J, Tian G, Wu S, Liu W, Cui Q, Geng B, Zhang W, Weldon R, Auguste K, Yang L, Liu X, Chen L, Yang X, Zhu B, Cai J. Gut microbiota dysbiosis contributes to the development of hypertension. Microbiome. 2017;5:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1455] [Cited by in RCA: 1253] [Article Influence: 139.2] [Reference Citation Analysis (4)] |
| 105. | Verhaar BJH, Collard D, Prodan A, Levels JHM, Zwinderman AH, Bäckhed F, Vogt L, Peters MJL, Muller M, Nieuwdorp M, van den Born BH. Associations between gut microbiota, faecal short-chain fatty acids, and blood pressure across ethnic groups: the HELIUS study. Eur Heart J. 2020;41:4259-4267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 164] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 106. | Aguirre-García MM, Amedei A, Hernández-Ruiz P, Gómez-García AP, Niccolai E, Moreno-Rodríguez AM, Pinto-Cardoso S, Alviter-Plata A, Escalona-Montaño AR, Ordaz-Robles ER, González-Salazar MDC, Springall Del Villar R, Berrios-Bárcenas EA, Ávila-Vanzzini N. Cytokine and microbiota profiles in obesity-related hypertension patients. Front Cell Infect Microbiol. 2023;13:1325261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 107. | Nakai M, Ribeiro RV, Stevens BR, Gill P, Muralitharan RR, Yiallourou S, Muir J, Carrington M, Head GA, Kaye DM, Marques FZ. Essential Hypertension Is Associated With Changes in Gut Microbial Metabolic Pathways: A Multisite Analysis of Ambulatory Blood Pressure. Hypertension. 2021;78:804-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 108. | Yang T, Santisteban MM, Rodriguez V, Li E, Ahmari N, Carvajal JM, Zadeh M, Gong M, Qi Y, Zubcevic J, Sahay B, Pepine CJ, Raizada MK, Mohamadzadeh M. Gut dysbiosis is linked to hypertension. Hypertension. 2015;65:1331-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1334] [Cited by in RCA: 1141] [Article Influence: 103.7] [Reference Citation Analysis (3)] |
| 109. | Jama HA, Beale A, Shihata WA, Marques FZ. The effect of diet on hypertensive pathology: is there a link via gut microbiota-driven immunometabolism? Cardiovasc Res. 2019;115:1435-1447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 77] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 110. | Jama HA, Kaye DM, Marques FZ. The gut microbiota and blood pressure in experimental models. Curr Opin Nephrol Hypertens. 2019;28:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 111. | Marques FZ, Mackay CR, Kaye DM. Beyond gut feelings: how the gut microbiota regulates blood pressure. Nat Rev Cardiol. 2018;15:20-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 301] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 112. | Han JM, Guo L, Chen XH, Xie Q, Song XY, Ma YL. Relationship between trimethylamine N-oxide and the risk of hypertension in patients with cardiovascular disease: A meta-analysis and dose-response relationship analysis. Medicine (Baltimore). 2024;103:e36784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 113. | Ge X, Zheng L, Zhuang R, Yu P, Xu Z, Liu G, Xi X, Zhou X, Fan H. The Gut Microbial Metabolite Trimethylamine N-Oxide and Hypertension Risk: A Systematic Review and Dose-Response Meta-analysis. Adv Nutr. 2020;11:66-76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 138] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 114. | Schmedes M, Balderas C, Aadland EK, Jacques H, Lavigne C, Graff IE, Eng Ø, Holthe A, Mellgren G, Young JF, Sundekilde UK, Liaset B, Bertram HC. The Effect of Lean-Seafood and Non-Seafood Diets on Fasting and Postprandial Serum Metabolites and Lipid Species: Results from a Randomized Crossover Intervention Study in Healthy Adults. Nutrients. 2018;10:598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 115. | Bielinska K, Radkowski M, Grochowska M, Perlejewski K, Huc T, Jaworska K, Motooka D, Nakamura S, Ufnal M. High salt intake increases plasma trimethylamine N-oxide (TMAO) concentration and produces gut dysbiosis in rats. Nutrition. 2018;54:33-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 116. | Hartiala J, Bennett BJ, Tang WH, Wang Z, Stewart AF, Roberts R, McPherson R, Lusis AJ, Hazen SL, Allayee H; CARDIoGRAM Consortium. Comparative genome-wide association studies in mice and humans for trimethylamine N-oxide, a proatherogenic metabolite of choline and L-carnitine. Arterioscler Thromb Vasc Biol. 2014;34:1307-1313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 117. | Jiang S, Shui Y, Cui Y, Tang C, Wang X, Qiu X, Hu W, Fei L, Li Y, Zhang S, Zhao L, Xu N, Dong F, Ren X, Liu R, Persson PB, Patzak A, Lai EY, Wei Q, Zheng Z. Gut microbiota dependent trimethylamine N-oxide aggravates angiotensin II-induced hypertension. Redox Biol. 2021;46:102115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 201] [Cited by in RCA: 172] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 118. | Xu Q, Wang Y, Xie Y, Zheng J, Guo R, Dai Y, Sun Z, Xing L, Zhang X, Ruan S, Zheng L, Sun Y. Blood Pressure Changes in a Chinese Population Have a Greater Impact on Short-Term Outcomes Rather Than Long-Term Outcomes of Major Adverse Cardiovascular Events. Asia Pac J Public Health. 2021;33:39-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 119. | Yao X, Li N, Heizhati M, Wang Y, Ma Y, Wang R, Zhang D, Luo Q, Hu J, Wang M, Zhu Q. The association between blood pressure control and long-term cardiovascular outcomes in Hypertension coexistent with obstructive sleep apnea. BMC Cardiovasc Disord. 2023;23:574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 120. | Cardoso CRL, Salles GF. Refractory Hypertension and Risks of Adverse Cardiovascular Events and Mortality in Patients With Resistant Hypertension: A Prospective Cohort Study. J Am Heart Assoc. 2020;9:e017634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 121. | Zheng J, Xie Y, Wang Y, Guo R, Dai Y, Sun Z, Xing L, Zhang X, Sun Y, Zheng L. Short- and long-term systolic blood pressure changes have different impacts on major adverse cardiovascular events: Results from a 12.5 years follow-up study. Int J Cardiol. 2020;306:190-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 122. | American Diabetes Association. 11. Microvascular Complications and Foot Care: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020;43:S135-S151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 292] [Article Influence: 48.7] [Reference Citation Analysis (3)] |
| 123. | Gao X, Liu X, Xu J, Xue C, Xue Y, Wang Y. Dietary trimethylamine N-oxide exacerbates impaired glucose tolerance in mice fed a high fat diet. J Biosci Bioeng. 2014;118:476-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 289] [Article Influence: 24.1] [Reference Citation Analysis (4)] |
| 124. | Dambrova M, Latkovskis G, Kuka J, Strele I, Konrade I, Grinberga S, Hartmane D, Pugovics O, Erglis A, Liepinsh E. Diabetes is Associated with Higher Trimethylamine N-oxide Plasma Levels. Exp Clin Endocrinol Diabetes. 2016;124:251-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 184] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 125. | Shan Z, Sun T, Huang H, Chen S, Chen L, Luo C, Yang W, Yang X, Yao P, Cheng J, Hu FB, Liu L. Association between microbiota-dependent metabolite trimethylamine-N-oxide and type 2 diabetes. Am J Clin Nutr. 2017;106:888-894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 126] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 126. | Sanchez-Alcoholado L, Castellano-Castillo D, Jordán-Martínez L, Moreno-Indias I, Cardila-Cruz P, Elena D, Muñoz-Garcia AJ, Queipo-Ortuño MI, Jimenez-Navarro M. Role of Gut Microbiota on Cardio-Metabolic Parameters and Immunity in Coronary Artery Disease Patients with and without Type-2 Diabetes Mellitus. Front Microbiol. 2017;8:1936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 127. | Croyal M, Saulnier PJ, Aguesse A, Gand E, Ragot S, Roussel R, Halimi JM, Ducrocq G, Cariou B, Montaigne D, Wargny M, Krempf M, Hadjadj S. Plasma Trimethylamine N-Oxide and Risk of Cardiovascular Events in Patients With Type 2 Diabetes. J Clin Endocrinol Metab. 2020;105:dgaa188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 128. | Miao J, Ling AV, Manthena PV, Gearing ME, Graham MJ, Crooke RM, Croce KJ, Esquejo RM, Clish CB; Morbid Obesity Study Group, Vicent D, Biddinger SB. Flavin-containing monooxygenase 3 as a potential player in diabetes-associated atherosclerosis. Nat Commun. 2015;6:6498. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 340] [Cited by in RCA: 301] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 129. | Kang JW, Zivkovic AM. Are eggs good again? A precision nutrition perspective on the effects of eggs on cardiovascular risk, taking into account plasma lipid profiles and TMAO. J Nutr Biochem. 2022;100:108906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 130. | Wang Z, Tang WH, Buffa JA, Fu X, Britt EB, Koeth RA, Levison BS, Fan Y, Wu Y, Hazen SL. Prognostic value of choline and betaine depends on intestinal microbiota-generated metabolite trimethylamine-N-oxide. Eur Heart J. 2014;35:904-910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 517] [Cited by in RCA: 467] [Article Influence: 38.9] [Reference Citation Analysis (0)] |
| 131. | Kalagi NA, Thota RN, Stojanovski E, Alburikan KA, Garg ML. Association between Plasma Trimethylamine N-Oxide Levels and Type 2 Diabetes: A Case Control Study. Nutrients. 2022;14:2093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 132. | Li D, Kirsop J, Tang WH. Listening to Our Gut: Contribution of Gut Microbiota and Cardiovascular Risk in Diabetes Pathogenesis. Curr Diab Rep. 2015;15:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 133. | Kim Y, Keogh J, Clifton P. A review of potential metabolic etiologies of the observed association between red meat consumption and development of type 2 diabetes mellitus. Metabolism. 2015;64:768-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 131] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 134. | Ilyas A, Wijayasinghe YS, Khan I, El Samaloty NM, Adnan M, Dar TA, Poddar NK, Singh LR, Sharma H, Khan S. Implications of trimethylamine N-oxide (TMAO) and Betaine in Human Health: Beyond Being Osmoprotective Compounds. Front Mol Biosci. 2022;9:964624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 135. | Cardona A, O'Brien A, Bernier MC, Somogyi A, Wysocki VH, Smart S, He X, Ambrosio G, Hsueh WA, Raman SV. Trimethylamine N-oxide and incident atherosclerotic events in high-risk individuals with diabetes: an ACCORD trial post hoc analysis. BMJ Open Diabetes Res Care. 2019;7:e000718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 136. | Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024;105:S117-S314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3017] [Cited by in RCA: 2748] [Article Influence: 1374.0] [Reference Citation Analysis (9)] |
| 137. | Tang Y, Li Y, Yang X, Lu T, Wang X, Li Z, Liu J, Wang J. Intestinal metabolite TMAO promotes CKD progression by stimulating macrophage M2 polarization through histone H4 lysine 12 lactylation. Cell Death Differ. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 138. | Lee J, Lee J, Kim K, Lee J, Jung Y, Hyeon JS, Seo A, Jin W, Weon B, Shin N, Kim S, Lim CS, Kim YS, Lee JP, Hwang GS, Yang SH. Antibiotic-induced intestinal microbiota depletion can attenuate the acute kidney injury to chronic kidney disease transition via NADPH oxidase 2 and trimethylamine-N-oxide inhibition. Kidney Int. 2024;105:1239-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 139. | Holle J, McParland V, Anandakumar H, Gerritzmann F, Behrens F, Schumacher F, Thumfart J, Eckardt KU, Kleuser B, Bartolomaeus H, Wilck N. Gut dysbiosis contributes to TMAO accumulation in CKD. Nephrol Dial Transplant. 2024;39:1923-1926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 140. | Wang M, Tang WHW, Li XS, de Oliveira Otto MC, Lee Y, Lemaitre RN, Fretts A, Nemet I, Sotoodehnia N, Sitlani CM, Budoff M, DiDonato JA, Wang Z, Bansal N, Shlipak MG, Psaty BM, Siscovick DS, Sarnak MJ, Mozaffarian D, Hazen SL. The Gut Microbial Metabolite Trimethylamine N -oxide, Incident CKD, and Kidney Function Decline. J Am Soc Nephrol. 2024;35:749-760. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 141. | Fox KA, Dabbous OH, Goldberg RJ, Pieper KS, Eagle KA, Van de Werf F, Avezum A, Goodman SG, Flather MD, Anderson FA Jr, Granger CB. Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE). BMJ. 2006;333:1091. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1294] [Cited by in RCA: 1130] [Article Influence: 56.5] [Reference Citation Analysis (0)] |
| 142. | Suzuki T, Heaney LM, Jones DJ, Ng LL. Trimethylamine N-oxide and Risk Stratification after Acute Myocardial Infarction. Clin Chem. 2017;63:420-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 143. | Tan Y, Zhou J, Yang S, Li J, Zhao H, Song L, Yan H. Addition of Plasma Myeloperoxidase and Trimethylamine N-Oxide to the GRACE Score Improves Prediction of Near-Term Major Adverse Cardiovascular Events in Patients With ST-Segment Elevation Myocardial Infarction. Front Pharmacol. 2021;12:632075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 144. | Li XS, Obeid S, Klingenberg R, Gencer B, Mach F, Räber L, Windecker S, Rodondi N, Nanchen D, Muller O, Miranda MX, Matter CM, Wu Y, Li L, Wang Z, Alamri HS, Gogonea V, Chung YM, Tang WH, Hazen SL, Lüscher TF. Gut microbiota-dependent trimethylamine N-oxide in acute coronary syndromes: a prognostic marker for incident cardiovascular events beyond traditional risk factors. Eur Heart J. 2017;38:814-824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 251] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 145. | Senthong V, Wang Z, Li XS, Fan Y, Wu Y, Tang WH, Hazen SL. Intestinal Microbiota-Generated Metabolite Trimethylamine-N-Oxide and 5-Year Mortality Risk in Stable Coronary Artery Disease: The Contributory Role of Intestinal Microbiota in a COURAGE-Like Patient Cohort. J Am Heart Assoc. 2016;5:e002816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 212] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 146. | Yang H, Zhang L, Xu CH. Use of the SYNTAX Score II to predict mortality in interventional cardiology: A systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e14043. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 147. | Senthong V, Li XS, Hudec T, Coughlin J, Wu Y, Levison B, Wang Z, Hazen SL, Tang WH. Plasma Trimethylamine N-Oxide, a Gut Microbe-Generated Phosphatidylcholine Metabolite, Is Associated With Atherosclerotic Burden. J Am Coll Cardiol. 2016;67:2620-2628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 188] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 148. | D'Agostino RB Sr, Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, Kannel WB. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117:743-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6219] [Cited by in RCA: 5522] [Article Influence: 306.8] [Reference Citation Analysis (3)] |
| 149. | Wilson PW, D'Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation. 1998;97:1837-1847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7137] [Cited by in RCA: 6158] [Article Influence: 219.9] [Reference Citation Analysis (6)] |
| 150. | Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. BMJ. 2017;357:j2099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1248] [Cited by in RCA: 1100] [Article Influence: 122.2] [Reference Citation Analysis (4)] |
| 151. | Sikora M, Kiss N, Stec A, Giebultowicz J, Samborowska E, Jazwiec R, Dadlez M, Olszewska M, Rudnicka L. Trimethylamine N-Oxide, a Gut Microbiota-Derived Metabolite, Is Associated with Cardiovascular Risk in Psoriasis: A Cross-Sectional Pilot Study. Dermatol Ther (Heidelb). 2021;11:1277-1289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 152. | Zou X, Xiong L, Guo Z, Li Y, Ning K, Liu X, Xie Y, Peng Y, Liu Y, Cai X, Zhao B, Zhou F, Yu C, Zheng X, Campbell SC, Zhang Z. Trimethylamine-N-oxide (TMAO) and predicted risk of cardiovascular events after partial nephrectomy. Asian J Surg. 2024;47:281-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 153. | Leng X, Wei X, Wang J, Yao X, Zhang M, Sun D, Liang J, Chi L, Cheng Y. Impacts of intestinal microbiota metabolite trimethylamine N-oxide on cardiovascular disease: a bibliometric analysis. Front Microbiol. 2024;15:1491731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 154. | Qi J, You T, Li J, Pan T, Xiang L, Han Y, Zhu L. Circulating trimethylamine N-oxide and the risk of cardiovascular diseases: a systematic review and meta-analysis of 11 prospective cohort studies. J Cell Mol Med. 2018;22:185-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 211] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 155. | Schiattarella GG, Sannino A, Toscano E, Giugliano G, Gargiulo G, Franzone A, Trimarco B, Esposito G, Perrino C. Gut microbe-generated metabolite trimethylamine-N-oxide as cardiovascular risk biomarker: a systematic review and dose-response meta-analysis. Eur Heart J. 2017;38:2948-2956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 437] [Article Influence: 48.6] [Reference Citation Analysis (0)] |
| 156. | Gencer B, Li XS, Gurmu Y, Bonaca MP, Morrow DA, Cohen M, Bhatt DL, Steg PG, Storey RF, Johanson P, Wang Z, Hazen SL, Sabatine MS. Gut Microbiota-Dependent Trimethylamine N-oxide and Cardiovascular Outcomes in Patients With Prior Myocardial Infarction: A Nested Case Control Study From the PEGASUS-TIMI 54 Trial. J Am Heart Assoc. 2020;9:e015331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 157. | Zhou Z, Jin H, Ju H, Sun M, Chen H, Li L. Circulating Trimethylamine-N-Oxide and Risk of All-Cause and Cardiovascular Mortality in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis. Front Med (Lausanne). 2022;9:828343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 158. | Mansuri NM, Mann NK, Rizwan S, Mohamed AE, Elshafey AE, Khadka A, Mosuka EM, Thilakarathne KN, Mohammed L. Role of Gut Microbiome in Cardiovascular Events: A Systematic Review. Cureus. 2022;14:e32465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 159. | Obeid R, Mohr L, White BA, Heine GH, Emrich I, Geisel J, Carter RC. Circulating trimethylamine N-oxide and cardiovascular, cerebral, and renal diseases including mortality: Umbrella review of published systematic reviews and meta-analyses. Nutr Metab Cardiovasc Dis. 2025;35:103908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 160. | Hu X, Ren H, Cao Y. The association between trimethylamine N-oxide levels and ischemic stroke occurrence: a meta-analysis and Mendelian randomization study. BMC Neurol. 2023;23:413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 161. | Zhao S, Tian Y, Wang S, Yang F, Xu J, Qin Z, Liu X, Cao M, Zhao P, Zhang G, Wang Z, Zhang Y, Wang Y, Lin K, Fang S, Wang Z, Han T, Tian M, Yin H, Tian J, Yu B. Prognostic value of gut microbiota-derived metabolites in patients with ST-segment elevation myocardial infarction. Am J Clin Nutr. 2023;117:499-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 162. | Zhou X, Jin M, Liu L, Yu Z, Lu X, Zhang H. Trimethylamine N-oxide and cardiovascular outcomes in patients with chronic heart failure after myocardial infarction. ESC Heart Fail. 2020;7:188-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 163. | Jia J, Dou P, Gao M, Kong X, Li C, Liu Z, Huang T. Assessment of Causal Direction Between Gut Microbiota-Dependent Metabolites and Cardiometabolic Health: A Bidirectional Mendelian Randomization Analysis. Diabetes. 2019;68:1747-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 146] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 164. | Gruppen EG, Garcia E, Connelly MA, Jeyarajah EJ, Otvos JD, Bakker SJL, Dullaart RPF. TMAO is Associated with Mortality: Impact of Modestly Impaired Renal Function. Sci Rep. 2017;7:13781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 118] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 165. | Kim RB, Morse BL, Djurdjev O, Tang M, Muirhead N, Barrett B, Holmes DT, Madore F, Clase CM, Rigatto C, Levin A; CanPREDDICT Investigators. Advanced chronic kidney disease populations have elevated trimethylamine N-oxide levels associated with increased cardiovascular events. Kidney Int. 2016;89:1144-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 149] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 166. | Mueller DM, Allenspach M, Othman A, Saely CH, Muendlein A, Vonbank A, Drexel H, von Eckardstein A. Plasma levels of trimethylamine-N-oxide are confounded by impaired kidney function and poor metabolic control. Atherosclerosis. 2015;243:638-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 183] [Article Influence: 16.6] [Reference Citation Analysis (3)] |
| 167. | Wang Z, Roberts AB, Buffa JA, Levison BS, Zhu W, Org E, Gu X, Huang Y, Zamanian-Daryoush M, Culley MK, DiDonato AJ, Fu X, Hazen JE, Krajcik D, DiDonato JA, Lusis AJ, Hazen SL. Non-lethal Inhibition of Gut Microbial Trimethylamine Production for the Treatment of Atherosclerosis. Cell. 2015;163:1585-1595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1173] [Cited by in RCA: 1040] [Article Influence: 94.5] [Reference Citation Analysis (4)] |
| 168. | Brunt VE, Greenberg NT, Sapinsley ZJ, Casso AG, Richey JJ, VanDongen NS, Gioscia-Ryan RA, Ziemba BP, Neilson AP, Davy KP, Seals DR. Suppression of trimethylamine N-oxide with DMB mitigates vascular dysfunction, exercise intolerance, and frailty associated with a Western-style diet in mice. J Appl Physiol (1985). 2022;133:798-813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 169. | Chen K, Zheng X, Feng M, Li D, Zhang H. Gut Microbiota-Dependent Metabolite Trimethylamine N-Oxide Contributes to Cardiac Dysfunction in Western Diet-Induced Obese Mice. Front Physiol. 2017;8:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 246] [Cited by in RCA: 204] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 170. | Wang G, Kong B, Shuai W, Fu H, Jiang X, Huang H. 3,3-Dimethyl-1-butanol attenuates cardiac remodeling in pressure-overload-induced heart failure mice. J Nutr Biochem. 2020;78:108341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 171. | Li T, Gua C, Wu B, Chen Y. Increased circulating trimethylamine N-oxide contributes to endothelial dysfunction in a rat model of chronic kidney disease. Biochem Biophys Res Commun. 2018;495:2071-2077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 172. | Fechtner S, Allen BE, Chriswell ME, Jubair WK, Robertson CE, Kofonow JN, Frank DN, Holers VM, Kuhn KA. 3,3-dimethyl-1-butanol and its metabolite 3,3-dimethylbutyrate ameliorate collagen-induced arthritis independent of choline trimethylamine lyase activity. Res Sq. 2023;rs.3.rs-3297018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 173. | Pathak P, Helsley RN, Brown AL, Buffa JA, Choucair I, Nemet I, Gogonea CB, Gogonea V, Wang Z, Garcia-Garcia JC, Cai L, Temel R, Sangwan N, Hazen SL, Brown JM. Small molecule inhibition of gut microbial choline trimethylamine lyase activity alters host cholesterol and bile acid metabolism. Am J Physiol Heart Circ Physiol. 2020;318:H1474-H1486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 68] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 174. | Roberts AB, Gu X, Buffa JA, Hurd AG, Wang Z, Zhu W, Gupta N, Skye SM, Cody DB, Levison BS, Barrington WT, Russell MW, Reed JM, Duzan A, Lang JM, Fu X, Li L, Myers AJ, Rachakonda S, DiDonato JA, Brown JM, Gogonea V, Lusis AJ, Garcia-Garcia JC, Hazen SL. Development of a gut microbe-targeted nonlethal therapeutic to inhibit thrombosis potential. Nat Med. 2018;24:1407-1417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 499] [Cited by in RCA: 438] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 175. | Shimizu M, Shiraishi A, Sato A, Nagashima S, Yamazaki H. Potential for drug interactions mediated by polymorphic flavin-containing monooxygenase 3 in human livers. Drug Metab Pharmacokinet. 2015;30:70-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 176. | Störmer E, Roots I, Brockmöller J. Benzydamine N-oxidation as an index reaction reflecting FMO activity in human liver microsomes and impact of FMO3 polymorphisms on enzyme activity. Br J Clin Pharmacol. 2000;50:553-561. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 92] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 177. | Cashman JR, Xiong Y, Lin J, Verhagen H, van Poppel G, van Bladeren PJ, Larsen-Su S, Williams DE. In vitro and in vivo inhibition of human flavin-containing monooxygenase form 3 (FMO3) in the presence of dietary indoles. Biochem Pharmacol. 1999;58:1047-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 71] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 178. | El-Serafi I, Terelius Y, Abedi-Valugerdi M, Naughton S, Saghafian M, Moshfegh A, Mattsson J, Potácová Z, Hassan M. Flavin-containing monooxygenase 3 (FMO3) role in busulphan metabolic pathway. PLoS One. 2017;12:e0187294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 179. | Bennett BJ, de Aguiar Vallim TQ, Wang Z, Shih DM, Meng Y, Gregory J, Allayee H, Lee R, Graham M, Crooke R, Edwards PA, Hazen SL, Lusis AJ. Trimethylamine-N-oxide, a metabolite associated with atherosclerosis, exhibits complex genetic and dietary regulation. Cell Metab. 2013;17:49-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 942] [Cited by in RCA: 842] [Article Influence: 64.8] [Reference Citation Analysis (4)] |
| 180. | Shih DM, Wang Z, Lee R, Meng Y, Che N, Charugundla S, Qi H, Wu J, Pan C, Brown JM, Vallim T, Bennett BJ, Graham M, Hazen SL, Lusis AJ. Flavin containing monooxygenase 3 exerts broad effects on glucose and lipid metabolism and atherosclerosis. J Lipid Res. 2015;56:22-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 289] [Cited by in RCA: 265] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 181. | Behrendt T, Bielitzki R, Behrens M, Herold F, Schega L. Effects of Intermittent Hypoxia-Hyperoxia on Performance- and Health-Related Outcomes in Humans: A Systematic Review. Sports Med Open. 2022;8:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 182. | Burtscher J, Mallet RT, Pialoux V, Millet GP, Burtscher M. Adaptive Responses to Hypoxia and/or Hyperoxia in Humans. Antioxid Redox Signal. 2022;37:887-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 104] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 183. | Glazachev O, Kopylov P, Susta D, Dudnik E, Zagaynaya E. Adaptations following an intermittent hypoxia-hyperoxia training in coronary artery disease patients: a controlled study. Clin Cardiol. 2017;40:370-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 184. | Han N, Pan Z, Liu G, Yang R, Yujing B. Hypoxia: The "Invisible Pusher" of Gut Microbiota. Front Microbiol. 2021;12:690600. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 185. | Uzun AB, Iliescu MG, Stanciu LE, Ionescu EV, Ungur RA, Ciortea VM, Irsay L, Motoașcă I, Popescu MN, Popa FL, Pazara L, Tofolean DE. Effectiveness of Intermittent Hypoxia-Hyperoxia Therapy in Different Pathologies with Possible Metabolic Implications. Metabolites. 2023;13:181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 186. | Bestavashvili A, Glazachev O, Bestavashvili A, Suvorov A, Zhang Y, Zhang X, Rozhkov A, Kuznetsova N, Pavlov C, Glushenkov D, Kopylov P. Intermittent Hypoxic-Hyperoxic Exposures Effects in Patients with Metabolic Syndrome: Correction of Cardiovascular and Metabolic Profile. Biomedicines. 2022;10:566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 187. | Bestavashvili A, Glazachev O, Ibragimova S, Suvorov A, Bestavasvili A, Ibraimov S, Zhang X, Zhang Y, Pavlov C, Syrkina E, Syrkin A, Kopylov P. Impact of Hypoxia-Hyperoxia Exposures on Cardiometabolic Risk Factors and TMAO Levels in Patients with Metabolic Syndrome. Int J Mol Sci. 2023;24:14498. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 188. | Angiletta CJ, Griffin LE, Steele CN, Baer DJ, Novotny JA, Davy KP, Neilson AP. Impact of short-term flavanol supplementation on fasting plasma trimethylamine N-oxide concentrations in obese adults. Food Funct. 2018;9:5350-5361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 189. | Baugh ME, Steele CN, Angiletta CJ, Mitchell CM, Neilson AP, Davy BM, Hulver MW, Davy KP. Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes. Nutrients. 2018;10:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 190. | Hernández-Alonso P, Cañueto D, Giardina S, Salas-Salvadó J, Cañellas N, Correig X, Bulló M. Effect of pistachio consumption on the modulation of urinary gut microbiota-related metabolites in prediabetic subjects. J Nutr Biochem. 2017;45:48-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (2)] |
| 191. | Argyridou S, Davies MJ, Biddle GJH, Bernieh D, Suzuki T, Dawkins NP, Rowlands AV, Khunti K, Smith AC, Yates T. Evaluation of an 8-Week Vegan Diet on Plasma Trimethylamine-N-Oxide and Postchallenge Glucose in Adults with Dysglycemia or Obesity. J Nutr. 2021;151:1844-1853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 192. | Thomas MS, DiBella M, Blesso CN, Malysheva O, Caudill M, Sholola M, Cooperstone JL, Fernandez ML. Comparison between Egg Intake versus Choline Supplementation on Gut Microbiota and Plasma Carotenoids in Subjects with Metabolic Syndrome. Nutrients. 2022;14:1179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 193. | Taslim NA, Yusuf M, Ambari AM, Del Rosario Puling IM, Ibrahim FZ, Hardinsyah H, Kurniawan R, Gunawan WB, Mayulu N, Joseph VFF, Sabrina N, Rizal M, Tallei TE, Kim B, Tsopmo A, Nurkolis F. Anti-Inflammatory, Antioxidant, Metabolic and Gut Microbiota Modulation Activities of Probiotic in Cardiac Remodeling Condition: Evidence from Systematic Study and Meta-Analysis of Randomized Controlled Trials. Probiotics Antimicrob Proteins. 2023;15:1049-1061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 194. | Azad MAK, Sarker M, Li T, Yin J. Probiotic Species in the Modulation of Gut Microbiota: An Overview. Biomed Res Int. 2018;2018:9478630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 598] [Cited by in RCA: 493] [Article Influence: 61.6] [Reference Citation Analysis (1)] |
| 195. | Gan XT, Ettinger G, Huang CX, Burton JP, Haist JV, Rajapurohitam V, Sidaway JE, Martin G, Gloor GB, Swann JR, Reid G, Karmazyn M. Probiotic administration attenuates myocardial hypertrophy and heart failure after myocardial infarction in the rat. Circ Heart Fail. 2014;7:491-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 241] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 196. | Madan S, Mehra MR. Gut dysbiosis and heart failure: navigating the universe within. Eur J Heart Fail. 2020;22:629-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 197. | Dow DE, Seed PC. Clostridium difficile cure with fecal microbiota transplantation in a child with Pompe disease: a case report. J Med Case Rep. 2018;12:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 198. | van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG, Speelman P, Dijkgraaf MG, Keller JJ. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3187] [Cited by in RCA: 2729] [Article Influence: 209.9] [Reference Citation Analysis (6)] |
| 199. | Khanna S, Pardi DS, Kelly CR, Kraft CS, Dhere T, Henn MR, Lombardo MJ, Vulic M, Ohsumi T, Winkler J, Pindar C, McGovern BH, Pomerantz RJ, Aunins JG, Cook DN, Hohmann EL. A Novel Microbiome Therapeutic Increases Gut Microbial Diversity and Prevents Recurrent Clostridium difficile Infection. J Infect Dis. 2016;214:173-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 249] [Article Influence: 24.9] [Reference Citation Analysis (3)] |
| 200. | Smits LP, Kootte RS, Levin E, Prodan A, Fuentes S, Zoetendal EG, Wang Z, Levison BS, Cleophas MCP, Kemper EM, Dallinga-Thie GM, Groen AK, Joosten LAB, Netea MG, Stroes ESG, de Vos WM, Hazen SL, Nieuwdorp M. Effect of Vegan Fecal Microbiota Transplantation on Carnitine- and Choline-Derived Trimethylamine-N-Oxide Production and Vascular Inflammation in Patients With Metabolic Syndrome. J Am Heart Assoc. 2018;7:e008342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 189] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 201. | Riaz H, Khan MS, Siddiqi TJ, Usman MS, Shah N, Goyal A, Khan SS, Mookadam F, Krasuski RA, Ahmed H. Association Between Obesity and Cardiovascular Outcomes: A Systematic Review and Meta-analysis of Mendelian Randomization Studies. JAMA Netw Open. 2018;1:e183788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 272] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 202. | Mohammadian Khonsari N, Khashayar P, Shahrestanaki E, Kelishadi R, Mohammadpoor Nami S, Heidari-Beni M, Esmaeili Abdar Z, Tabatabaei-Malazy O, Qorbani M. Normal Weight Obesity and Cardiometabolic Risk Factors: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne). 2022;13:857930. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 83] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 203. | Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K, Abrouk M, Farahnik B, Nakamura M, Zhu TH, Bhutani T, Liao W. Influence of diet on the gut microbiome and implications for human health. J Transl Med. 2017;15:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2184] [Cited by in RCA: 1768] [Article Influence: 196.4] [Reference Citation Analysis (4)] |
| 204. | Oh KK, Gupta H, Min BH, Ganesan R, Sharma SP, Won SM, Jeong JJ, Lee SB, Cha MG, Kwon GH, Jeong MK, Hyun JY, Eom JA, Park HJ, Yoon SJ, Choi MR, Kim DJ, Suk KT. Elucidation of Prebiotics, Probiotics, Postbiotics, and Target from Gut Microbiota to Alleviate Obesity via Network Pharmacology Study. Cells. 2022;11:2903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 205. | Gil-Hernández E, Ruiz-González C, Rodriguez-Arrastia M, Ropero-Padilla C, Rueda-Ruzafa L, Sánchez-Labraca N, Roman P. Effect of gut microbiota modulation on sleep: a systematic review and meta-analysis of clinical trials. Nutr Rev. 2023;81:1556-1570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 206. | Heianza Y, Sun D, Li X, DiDonato JA, Bray GA, Sacks FM, Qi L. Gut microbiota metabolites, amino acid metabolites and improvements in insulin sensitivity and glucose metabolism: the POUNDS Lost trial. Gut. 2019;68:263-270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 152] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 207. | Battillo DJ, Malin SK. Impact of Caloric Restriction and Exercise on Trimethylamine N-Oxide Metabolism in Women with Obesity. Nutrients. 2023;15:1455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 208. | Erickson ML, Malin SK, Wang Z, Brown JM, Hazen SL, Kirwan JP. Effects of Lifestyle Intervention on Plasma Trimethylamine N-Oxide in Obese Adults. Nutrients. 2019;11:179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 209. | Alzughayyar DN, Weber RM, Husain S, Schoch N, Englert H. Impact of the Healthy Lifestyle Community Program (HLCP-3) on Trimethylamine N-Oxide (TMAO) and Risk Profile Parameters Related to Lifestyle Diseases During the Six Months Following an Intervention Study. Nutrients. 2025;17:298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 210. | Petersen KS, Kris-Etherton PM. Diet Quality Assessment and the Relationship between Diet Quality and Cardiovascular Disease Risk. Nutrients. 2021;13:4305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 143] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 211. | Diao Z, Molludi J, Latef Fateh H, Moradi S. Comparison of the low-calorie DASH diet and a low-calorie diet on serum TMAO concentrations and gut microbiota composition of adults with overweight/obesity: a randomized control trial. Int J Food Sci Nutr. 2024;75:207-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 212. | Rees K, Takeda A, Martin N, Ellis L, Wijesekara D, Vepa A, Das A, Hartley L, Stranges S. Mediterranean-style diet for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2019;3:CD009825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 155] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 213. | Weaver SR, Rendeiro C, McGettrick HM, Philp A, Lucas SJE. Fine wine or sour grapes? A systematic review and meta-analysis of the impact of red wine polyphenols on vascular health. Eur J Nutr. 2021;60:1-28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 214. | Haas EA, Saad MJA, Santos A, Vitulo N, Lemos WJF, Martins AMA, Picossi CRC, Favarato D, Gaspar RS, Magro DO, Libby P, Laurindo FRM, Da Luz PL; WineFlora Study. A red wine intervention does not modify plasma trimethylamine N-oxide but is associated with broad shifts in the plasma metabolome and gut microbiota composition. Am J Clin Nutr. 2022;116:1515-1529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 215. | Spasova N, Somleva D, Krastev B, Tropcheva R, Svinarov D, Kundurzhiev T, Kinova E, Goudev A. Effect of Lactobacillus plantarum supplementation on trimethylamine-N-oxide levels in 30 patients with atherosclerotic cardiovascular disease: A double-blind randomized controlled trial. Folia Med (Plovdiv). 2024;66:682-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 216. | Malik M, Suboc TM, Tyagi S, Salzman N, Wang J, Ying R, Tanner MJ, Kakarla M, Baker JE, Widlansky ME. Lactobacillus plantarum 299v Supplementation Improves Vascular Endothelial Function and Reduces Inflammatory Biomarkers in Men With Stable Coronary Artery Disease. Circ Res. 2018;123:1091-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 166] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 217. | Tripolt NJ, Leber B, Triebl A, Köfeler H, Stadlbauer V, Sourij H. Effect of Lactobacillus casei Shirota supplementation on trimethylamine-N-oxide levels in patients with metabolic syndrome: An open-label, randomized study. Atherosclerosis. 2015;242:141-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 218. | Coutinho-Wolino KS, de F Cardozo LFM, de Oliveira Leal V, Mafra D, Stockler-Pinto MB. Can diet modulate trimethylamine N-oxide (TMAO) production? Eur J Nutr. 2021;60:3567-3584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 219. | Crimarco A, Springfield S, Petlura C, Streaty T, Cunanan K, Lee J, Fielding-Singh P, Carter MM, Topf MA, Wastyk HC, Sonnenburg ED, Sonnenburg JL, Gardner CD. A randomized crossover trial on the effect of plant-based compared with animal-based meat on trimethylamine-N-oxide and cardiovascular disease risk factors in generally healthy adults: Study With Appetizing Plantfood-Meat Eating Alternative Trial (SWAP-MEAT). Am J Clin Nutr. 2020;112:1188-1199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 189] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 220. | DiMattia ZS, Zhao J, Hao F, Koshkin S, Bisanz JE, Patterson AD, Fleming JA, Kris-Etherton PM, Petersen KS. Effect of Varying Quantities of Lean Beef as Part of a Mediterranean-Style Dietary Pattern on Gut Microbiota and Plasma, Fecal, and Urinary Metabolites: A Randomized Crossover Controlled Feeding Trial. J Am Heart Assoc. 2025;14:e041063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 221. | Griffin LE, Djuric Z, Angiletta CJ, Mitchell CM, Baugh ME, Davy KP, Neilson AP. A Mediterranean diet does not alter plasma trimethylamine N-oxide concentrations in healthy adults at risk for colon cancer. Food Funct. 2019;10:2138-2147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (4)] |
| 222. | Bergeron N, Williams PT, Lamendella R, Faghihnia N, Grube A, Li X, Wang Z, Knight R, Jansson JK, Hazen SL, Krauss RM. Diets high in resistant starch increase plasma levels of trimethylamine-N-oxide, a gut microbiome metabolite associated with CVD risk. Br J Nutr. 2016;116:2020-2029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 85] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 223. | Costabile G, Vetrani C, Bozzetto L, Giacco R, Bresciani L, Del Rio D, Vitale M, Della Pepa G, Brighenti F, Riccardi G, Rivellese AA, Annuzzi G. Plasma TMAO increase after healthy diets: results from 2 randomized controlled trials with dietary fish, polyphenols, and whole-grain cereals. Am J Clin Nutr. 2021;114:1342-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 224. | Awoyemi A, Mayerhofer C, Felix AS, Hov JR, Moscavitch SD, Lappegård KT, Hovland A, Halvorsen S, Halvorsen B, Gregersen I, Svardal A, Berge RK, Hansen SH, Götz A, Holm K, Aukrust P, Åkra S, Seljeflot I, Solheim S, Lorenzo A, Gullestad L, Trøseid M, Broch K. Rifaximin or Saccharomyces boulardii in heart failure with reduced ejection fraction: Results from the randomized GutHeart trial. EBioMedicine. 2021;70:103511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |