Published online Aug 26, 2015. doi: 10.4331/wjbc.v6.i3.231
Peer-review started: April 28, 2015
First decision: May 14, 2015
Revised: May 30, 2015
Accepted: July 21, 2015
Article in press: July 23, 2015
Published online: August 26, 2015
Processing time: 121 Days and 11.1 Hours
Approximately 80% of breast cancers (BC) are estrogen receptor (ER)-positive and thus endocrine therapy (ET) should be considered complementary to surgery in the majority of patients. The advantages of oophorectomy, adrenalectomy and hypophysectomy in women with advanced BC have been demonstrated many years ago, and currently ET consist of (1) ovarian function suppression (OFS), usually obtained using gonadotropin-releasing hormone agonists (GnRHa); (2) selective estrogen receptor modulators or down-regulators (SERMs or SERDs); and (3) aromatase inhibitors (AIs), or a combination of two or more drugs. For patients aged less than 50 years and ER+ BC, there is no conclusive evidence that the combination of OFS and SERMs (i.e., tamoxifen) or chemotherapy is superior to OFS alone. Tamoxifen users exhibit a reduced risk of BC, both invasive and in situ, especially during the first 5 years of therapy, and extending the treatment to 10 years further reduced the risk of recurrences. SERDs (i.e., fulvestrant) are especially useful in the neoadjuvant treatment of advanced BC, alone or in combination with either cytotoxic agents or AIs. There are two types of AIs: type I are permanent steroidal inhibitors of aromatase, while type II are reversible nonsteroidal inhibitors. Several studies demonstrated the superiority of the third-generation AIs (i.e., anastrozole and letrozole) compared with tamoxifen, and adjuvant therapy with AIs reduces the recurrence risk especially in patients with advanced BC. Unfortunately, some cancers are or became ET-resistant, and thus other drugs have been suggested in combination with SERMs or AIs, including cyclin-dependent kinase 4/6 inhibitors (palbociclib) and mammalian target of rapamycin (mTOR) inhibitors, such as everolimus. Further studies are required to confirm their real usefulness.
Core tip: In women with breast cancer (BC), the two major endocrine therapies (ET) available are directed at blocking estrogen receptors (ERs) on cancer cells (selective estrogen receptor modulators), or against the key enzyme in the biosynthesis of estrogens (aromatase inhibitors). The new knowledge about molecular mechanisms regulating tumor progression, suggested the existence of a strict relationship between ERs, intracellular signaling pathways, and other growth factors receptor, which justify the escape of a portion of patients from the inhibitory effects of ET. To restore endocrine sensitivity of BC cells, molecular-target therapies are giving encouraging results, but further studies are required to confirm their real usefulness.
- Citation: Lumachi F, Santeufemia DA, Basso SM. Current medical treatment of estrogen receptor-positive breast cancer. World J Biol Chem 2015; 6(3): 231-239
- URL: https://www.wjgnet.com/1949-8454/full/v6/i3/231.htm
- DOI: https://dx.doi.org/10.4331/wjbc.v6.i3.231
Core tip: In women with breast cancer (BC), the two major endocrine therapies (ET) available are directed at blocking estrogen receptors (ERs) on cancer cells (selective estrogen receptor modulators), or against the key enzyme in the biosynthesis of estrogens (aromatase inhibitors). The new knowledge about molecular mechanisms regulating tumor progression, suggested the existence of a strict relationship between ERs, intracellular signaling pathways, and other growth factors receptor, which justify the escape of a portion of patients from the inhibitory effects of ET. To restore endocrine sensitivity of BC cells, molecular-target therapies are giving encouraging results, but further studies are required to confirm their real usefulness.
Breast cancer (BC) is the most common female cancer worldwide, and the second cause of cancer death in women[1]. In the United States, the expected number of new BC cases in 2015 is approximately 231800, accounting for 29% of all cancers, with an annual change based on incidence of +0.3%[2]. In the European Union, BC seems to have a more favorable trend, with a 10.2% fall from 2009[3].
Estrogen receptors (ERs) are nuclear proteins regulating the expression of specific genes, and approximately 80% of BCs are estrogen receptor (ER)-positive, of which 65% are also progesterone receptor (PR)-positive[4]. Thus, endocrine therapy (ET) should be considered complementary to surgery in the majority of patients, inducing tumor remission and providing consistent clinical benefit. ET has represented the standard adjuvant treatment for ER+ tumors since the 1970s, although the advantages of oophorectomy, adrenalectomy, and hypophysectomy in women with advanced BC had already been demonstrated many years before[5]. ET for BC currently consist of (1) ovarian function suppression (OFS); (2) selective estrogen receptor modulators (SERMs); (3) selective estrogen receptor down-regulators (SERDs); and (4) aromatase inhibitors (AIs), or a combination of two or more drugs. The two major strategies are directed at blocking ERs of cancer cells (SERMs) or against the key enzyme in the biosynthesis of estrogens (AIs).
For decades, in premenopausal women with operable BC in whom ovaries are the main source of estrogen and progesterone, surgical oophorectomy has represented an effective adjuvant therapy. Ovary irradiation was rarely performed for this purpose. Recently, it has been shown that the timing of oophorectomy (i.e., luteal phase of the menstrual cycle) does not affect disease-free survival (DFS) and overall survival (OS) of patients with BC[6]. OFS can be also obtained using gonadotropin-releasing hormone agonists (GnRHa), which are generally considered drugs with reversible actions. Although in patients with BC undergoing simultaneously GnRHa administration and chemotherapy (CT) ovarian function is rarely preserved, this treatment has largely replaced surgery[7,8].
A historic large overview of randomized trials testing the usefulness of ovarian ablation in patients with ER+ early BC, named EBCTCG (Early Breast Cancer Trialists’ Collaborative Group) study, demonstrated that OFS significantly increases survival in women aged below 50 years and that these benefits are independent of axillary nodal status, compared to untreated patients[9]. The advantages of adding a GnRHa to SERMS or CT in terms of reduced recurrence and deaths rate was confirmed in other studies[10,11]. More recently, it has been shown that women who underwent adjuvant GnRHa monotherapy or standard CT regimen had similar DFS and OS, while a combination of GnRHa plus CT or SERMs significantly improved DFS and OS in comparison to CT alone[12]. Further studies suggested that, in women with ER+ early BC, adjuvant treatment with OFS plus SERMs (tamoxifen) does not lead to any advantage, while the combination of OFS and AIs (exemestane) significantly reduces recurrences compared to the other regimen[13,14].
In premenopausal women previously treated with SERMs (tamoxifen), extended therapy with GnRHa and an AI may lead to substantial adverse effects[15]. In conclusion, for patients aged less than 50 years, there is no conclusive evidence that the combination of OFS and SERMs or CT is superior to OFS alone[16].
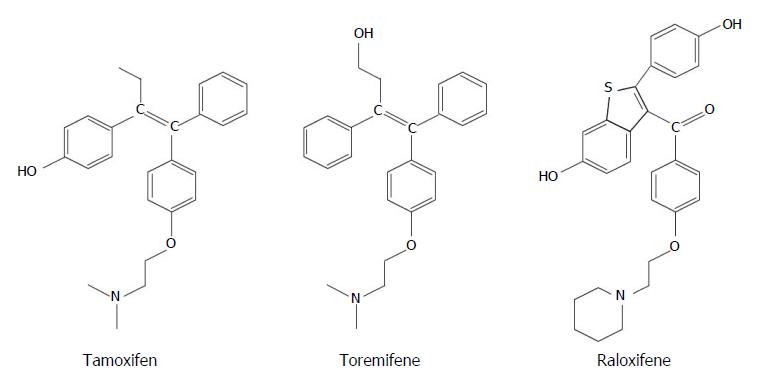
Tamoxifen (TAM) is the drug most extensively used when an ET is required, both in pre- and postmenopausal women, and is still considered the SERM of choice[7]. TAM (Figure 1A) was initially studied as anti-fertility drug, demonstrating soon favorable effects in patients with ER+ BC, as well as on bone tissue and cardiovascular system[17]. TAM acts as a partial nonsteroidal agonist in some tissues, such as liver, uterus and bone, but is a competitive receptor inhibitor in the breast and brain[18]. In these tissues, TAM selectively blocks signaling at the level of ERs, also inhibiting the proliferation of ductal cells[19]. TAM is hydroxylated by the cytochrome P450 enzyme system into 4-hydroxy tamoxifen (4HT), and further metabolized in the liver, thanks to a CYP2D6-mediated oxidation, to endoxifen (4-hydroxy-N-desmethyl-tamoxifen)[20]. Endoxifen and 4HT are the main active metabolites of TAM, exhibit similar ability in binding to ERs, and are more potent than TAM in suppressing estrogen-dependent cancer cells[7]. The relationship between CYP2D6, endoxifen or 4HT serum levels and the response to TAM therapy has long been studied, but the hypothesized role of CYP2D6 genotype as prognostic factor affecting recurrence in patients with BC was not confirmed[21,22]. However, CYP2D6 inhibitors, such as antipsychotic drugs, selective serotonin reuptake inhibitors (SSRIs), and some cardiac medications have the potential to affect response to SERM therapy[23,24].
TAM significantly benefits only women with ER+ BC, and ASCO (American Society of Clinical Oncology) guideline recommendations consider a cut-off of 1% for immunohistochemical testing of ERs in BC cells[25]. A number of studies and meta-analyses confirm its usefulness. According to the 2011 EBCTCG study, the reduction in mortality, expressed as reduction in annual odds of death, is approximately 20% after 5-years of TAM adjuvant therapy, and the 15-year recurrence rate ranges from 4.6% to 17.4%, according to age[16,26]. The long-term mortality among TAM users was 23.9% compared with 33.1% of the control group, with limited age-related side effects, such as increased risk of thromboembolic disease (> 55 years) and endometrial cancer (> 45 years)[26,27]. Extending the treatment to 10 years, the risk of recurrence and overall mortality are further slightly reduced, and thus the 2014 ASCO guidelines recommend the use of TAM for up to 10 years in pre- or perimenopausal women[28,29].
With the aim of limiting the incidence of adverse effects of TAM, eventually improving its effectiveness, other SERMs have been proposed, such as toremifene and raloxifene, both approved by the US Food and Drugs Administration (FDA). Toremifene (Figure 1B) is similar to TAM in clinical activity, efficacy and tolerability, and is cross-resistant to TAM. It causes less vascular and endometrial adverse effects than TAM, but has less favorable actions on bone and lipids levels[30]. Also in patients with metastatic BC, the results of TAM and toremifene therapy did not differ significantly, and the latter represents a reliable alternative to TAM in menopausal women[31]. Raloxifene (Figure 1C) is another nonsteroidal SERM causing less toxicity compared to TAM, especially used for the prevention of osteoporosis in postmenopausal women[32]. However, as preventive therapy, raloxifene has been shown to be less effective than TAM, because it reduces the risk of cancer by 36% compared to 50% of the group treated with TAM[33].
In conclusion, SERMs users exhibit a reduced risk of BC, both invasive and in situ, especially during the first 5 years of therapy, ranging from 53% to 79% according to age and menopausal status[34].
Selective estrogen receptor down-regulators have different pharmacologic characteristics, biochemical structure, and molecular activity with respect of SERM, causing down-regulation and degradation of ERs, and inhibiting proliferation of estrogen-dependent BC cells[35]. SERDs are pure ER antagonist, blocks ERs activity and accelerate their degradation, thus exhibiting exclusively anti-estrogen effects[36]. Fulvestrant, the only SERD approved by FDA to treat patients with BC, has 100-fold the affinity of TAM for ER, in lack of adverse effect on endometrial ERs[37,38]. It is useful especially in patients with advancer BC and as second-line therapy in TAM-resistant tumors[36]. Fulvestrant sensitizes ER-negative BC to chemotherapy, showing a synergistic action with various cytotoxic agents, such as docetaxel[39,40]. In the treatment of patients with metastatic ER+ BC, the combination of fulvestrant plus an AI (anastrozole) was superior to anastrozole alone, and fulvestrant as single-drug therapy represents a reasonable alternative to AIs[41,42]. In the neoadjuvant setting, the use of fulvestrant is controversial, because of the slow response rate and possible non-response. In a phase II randomized study, tumor response rates were 20.6% and 22.9%, according to the dosage of the drug[43].
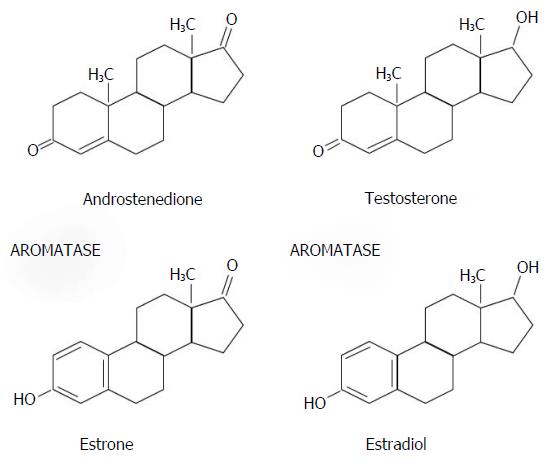
The human CYP19 single-copy gene, located on chromosome 15q21.2, encodes the microsomal enzyme aromatase [cytochrome P450 19 (CYP19)], which catalyzes the conversion of androgens, such as testosterone and androstenedione, to estrogens, such as 17-β-estradiol and estrone[44] (Figure 2).
Aromatase activity is expressed especially in the ovary (premenopausal women), testis, placenta, brain, bone and adipose tissue (postmenopausal women)[45]. AIs block aromatase enzyme activity, safely reducing circulating estrogen levels only in postmenopausal patients. AIs are ineffective in premenopausal women with functionally active ovary in whom they increase gonadotropin secretion, and therefore estrogen production[46]. The combination of AIs and OFS, using a GnRHa to block the pituitary, is usually an effective strategy. According to the conclusions of the STAGE study, 70.4% of postmenopausal patients with early BC treated with an AI (anastrozole) plus GnRHa (goserelin) obtained complete or partial response to therapy, compared with 50.5% of the control group treated with anastrozole plus TAM[47]. Another study, did not found any difference in DFS between premenopausal women with ER+ early BC treated with TAM alone and those treated with anastrozole alone, who presented a slightly increased risk of death[48]. A correlation between elevated body mass index and reduced effect of AIs has been reported in some studies[49].
According to the chemical structure and mechanism of action, there are two types of AIs: type I are permanent steroidal inhibitors of aromatase, while type II are reversible nonsteroidal inhibitors (Table 1).
| Type | Chemical structure | Action | First generation | Second generation | Third generation |
| I | Steroidal | Permanent | Testolactone | Formestane | Examestane |
| II | Nonsteroidal | Reversible | Aminoglutethimide | Fadrozole | Anastrozole |
| Letrozole |
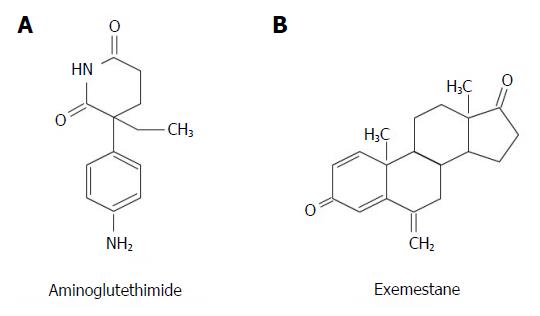
Aminoglutethimide, initially used as anti-epileptic drug, is the oldest nonsteroidal AI (Figure 3A). It inhibits also cytochrome P450scc, blocking the conversion of cholesterol to pregnenolone, and thus decreases the production of several steroids, including cortisol[17]. The subsequent activation of the hypothalamic-pituitary-adrenal axis, causes hypersecretion of adrenocorticotropic hormone (ACTH) leading to the need of increasing the dosage of the drug, and requires corticosteroids supplementation. Due to these side effects, aminoglutethimide has been replaced by better-tolerated and more effective AIs[7].
Exemestane (Figure 3B) is a potent irreversible steroidal AI that acts as a false substrate for aromatase and suppresses selectively estrogens at any site[50]. In some studies, exemestane plus OFS reduced the risk of recurrences in premenopausal women with ER+ early BC, while TAM plus OFS did not provided benefit[51]. In a group of postmenopausal patients at slightly increased risk of cancer according to the Gail model who underwent exemestane therapy, the annual incidence of BC was 0.33% compared to 0.77% of control group[52].
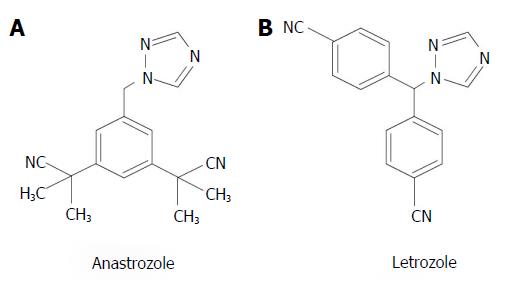
Anastrozole and letrozole (Figure 4) are two nonstedroidal third-generation AIs with similar potency and characteristics. Because their block of ERs is reversible, a continuous drug administration is required. An old comparison between anastrozole and letrozole as second-line therapy in advanced BC, reported an overall response rate of 12% vs 19%, respectively[53]. In postmenopausal women with advanced BC, letrozole was more effective than fadrazole, a second-generation type II nonsteroidal AI with reversible action[54]. The more recent ACOSOG (American College of Surgeons Oncology Group) study compared exemestane, anastrozole, and letrozole as neoadjuvant therapy of women with advanced BC, and reported that the clinical response rate was 62.9%, 69.1%, and 74.8%, respectively[55]. Several meta-analysis underline the superiority of the third-generation AI (but not first- and second-generation AIs) compared with TAM, confirming that adjuvant therapy with AIs reduces the recurrence risk in patients with advanced BC, but does not affect mortality rate[56,57].
The new knowledge about molecular mechanisms regulating tumor progression, suggested the existence of a strict relationship between ERs, intracellular signaling pathways, and other growth factors receptor, which justify the escape of a portion of patients from the inhibitory effects of ET[58]. To restore endocrine sensitivity of ER+ cancer cells, several other drugs have been studied, including cyclin-dependent kinase (CDK) 4/6 inhibitors (palbociclib), epigenetic modulators that inhibit histone deacetylase (HDAC), and other signal pathway inhibitors[59]. Experimentally, combined HDAC inhibition and TAM reverse ET resistance, inducing apoptosis of TAM-resistant BC cells, and in women with advanced ER+ BC and disease progression or recurrence the progression-free survival (PFS) can be reduced adding palbociclib to letrozole[60,61]. The phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) pathway and the serine/threonine kinase mammalian target of rapamycin (mTOR) are part of a signaling network regulating several cell processes, including growth, proliferation and survival of cancer cells[62]. The addition of an mTOR inhibitor (everolimus) to examestane in patients with ER+ BC and advanced disease refractory to both SERMs and AIs, can improve the progression-free survival (PFS) compared to everolimus alone[52]. A recent meta-analysis showed that exemestane and everolimus in combination was more efficacious than fulvestrant alone in patients with relapsed BC previously treated with nonsteroidal AIs[63]. Unfortunately, in women with ER+ BC, the addition of an anti-vascular endothelial growth factor (bevacizumab) to fulvestrant or letrozole did not change the resistance of the tumor to ET, and failed to improve PFS or OS[64].
As in part already reported above, anti-estrogen therapy plays a major role in the prolonged remission in women with metastatic and advanced ER+ BC. Several studies showed that the inhibition of the aromatase enzyme system with third-generation AIs, such as exemestane, anastrozole and letrozole, is associated with better survival compared to SERMs[56]. In terms of both PFS and OS, there are also advantages of treating patients with AIs compared to other ET, and letrozole seems to be the drug of choice[65,66]. The adverse effects observed using letrozole or anastrozole are similar than that observed with TAM, and the time-to-progression was better[67]. The combination anastrozole and fulvestrant in metastatic ER+ BC was superior to anastrozole alone[41].
All endocrine therapies, in particular AIs, synergize their effects with several molecular target drugs, such as everolimus and palbociclib[58,60]. It has also been shown that exemestane plus everolimus were more efficacious than everolimus alone and fulvestrant alone in women with advanced BC and progressive disease[52,63]. The GINECO (Groupe d’Investigateurs Nationaux pour les Etudes des Cancers de l’Ovaire) study showed that the short-term clinical benefit rates obtained with TAM plus everolimus vs TAM alone were 61% and 42%, respectively, and that the time to progression increased from 4.5 to 8.6 mo[68]. However, according to the conclusion of the BOLERO-2 (Breast Cancer Trials of OraL EveROlimus-2) study, the decision of prescribing AIs and mTOR inhibitors in combination should be individualized, taking into account the characteristics of single patient and considering risks and benefits of the treatment[69]. Both CDK and HDAC inhibitors, such as palbociclib and etinostat, in combination with ET, can be useful in patients with advanced ER+ BC as third-line therapy[70].
Unfortunately, both TAM and AIs use is associated with several adverse events (AEs) and distressing symptoms, such as hot flushes, weight gain, sexual dysfunctions, decreased bone mineral density (BMD), osteoporosis and pathologic fractures, or musculoskeletal symptoms, depending on the drug used and the duration of treatment. These effects may be severe enough to cause non-compliance due to cessation of use in a significant proportion of patients. Most of the AEs is related to the estrogen deprivation[7]. According to a systematic review, TAM was associated with more serious AEs in comparison with AIs, while exemestane caused more frequently musculoskeletal symptoms and hot flushes, but in some trials no differences in AEs between SERMs and AIs was found[67]. Another meta-analysis showed that patients treated with AIs had increased risk of gastrointestinal symptoms, such as nausea, vomiting and diarrhea, while the incidence of thromboembolic events and vaginal bleeding were decreased compared to other ETs[66]. In several studies, AIs use was associated with a higher risk of arthralgia and decreased BMD compared to TAM, and the incidence of discontinuation ranges from approximately 12% to 14%[16]. Effects on cognitive function related to the AIs use have also been reported, similar to the so-called “chemotherapy fog” observed following prolonged cancer CT[71]. However, toxicity is related to individual treatment compliance and depends on the way in which the patient describes and reports the symptoms, and they usually resolve with discontinuation of treatment[16]. Careful patient information is recommended before starting any treatment with ETs, especially in premenopausal women.
In patients with BC and ER+ tumors, ET has several advantages, including prolonging DFS and OS, and reducing the risk of recurrence, both in patients with early BC and in those with advanced or relapsed disease. In premenopausal women, OFS and TAM has similar efficacy and there is no evidence that a regimen based on OFS, SERMs, or AIs in combination can be the strategy of choice[7,16]. In postmenopausal women, AIs are more effective than SERMs in reducing relapse of the disease, and extending the TAM administration for more than 5 year represents a useful strategy. In patients with advanced BC, molecular-target therapies are giving encouraging results, but further studies are required to confirm their real usefulness and a careful analysis of cost-benefits is also required.
| 1. | The American Cancer Society. What are the key statistics about breast cancer? 2015. Available from: http://www.cancer.org/cancer/breastcancer/detailedguide/breast-cancer-key-statistics. |
| 2. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8821] [Cited by in RCA: 9723] [Article Influence: 883.9] [Reference Citation Analysis (1)] |
| 3. | Malvezzi M, Bertuccio P, Rosso T, Rota M, Levi F, La Vecchia C, Negri E. European cancer mortality predictions for the year 2015: does lung cancer have the highest death rate in EU women? Ann Oncol. 2015;26:779-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 197] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 4. | National Cancer Institute. Hormone Therapy for Breast Cancer. 2012. Available from: http://www.cancer.gov/cancertopics/types/breast/breast-hormone-therapy-fact-sheet. |
| 5. | Love RR, Philips J. Oophorectomy for breast cancer: history revisited. J Natl Cancer Inst. 2002;94:1433-1434. [PubMed] |
| 6. | Love RR, Laudico AV, Van Dinh N, Allred DC, Uy GB, Quang le H, Salvador JD, Siguan SS, Mirasol-Lumague MR, Tung ND. Timing of adjuvant surgical oophorectomy in the menstrual cycle and disease-free and overall survival in premenopausal women with operable breast cancer. J Natl Cancer Inst. 2015;107:djv064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Lumachi F, Brunello A, Maruzzo M, Basso U, Basso SM. Treatment of estrogen receptor-positive breast cancer. Curr Med Chem. 2013;20:596-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 202] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 8. | Vitek WS, Shayne M, Hoeger K, Han Y, Messing S, Fung C. Gonadotropin-releasing hormone agonists for the preservation of ovarian function among women with breast cancer who did not use tamoxifen after chemotherapy: a systematic review and meta-analysis. Fertil Steril. 2014;102:808-815.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Early Breast Cancer Trialists’ Collaborative Group. Ovarian ablation in early breast cancer: overview of the randomised trials. Early Breast Cancer Trialists’ Collaborative Group. Lancet. 1996;348:1189-1196. [PubMed] |
| 10. | Davidson NE, O’Neill AM, Vukov AM, Osborne CK, Martino S, White DR, Abeloff MD. Chemoendocrine therapy for premenopausal women with axillary lymph node-positive, steroid hormone receptor-positive breast cancer: results from INT 0101 (E5188). J Clin Oncol. 2005;23:5973-5982. [PubMed] |
| 11. | Cuzick J, Ambroisine L, Davidson N, Jakesz R, Kaufmann M, Regan M, Sainsbury R. Use of luteinising-hormone-releasing hormone agonists as adjuvant treatment in premenopausal patients with hormone-receptor-positive breast cancer: a meta-analysis of individual patient data from randomised adjuvant trials. Lancet. 2007;369:1711-1723. [PubMed] |
| 12. | Goel S, Sharma R, Hamilton A, Beith J. LHRH agonists for adjuvant therapy of early breast cancer in premenopausal women. Cochrane Database Syst Rev. 2009;CD004562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Pagani O, Regan MM, Walley BA, Fleming GF, Colleoni M, Láng I, Gomez HL, Tondini C, Burstein HJ, Perez EA. Adjuvant exemestane with ovarian suppression in premenopausal breast cancer. N Engl J Med. 2014;371:107-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 504] [Cited by in RCA: 538] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 14. | Figg WD, Cook K, Clarke R. Aromatase inhibitor plus ovarian suppression as adjuvant therapy in premenopausal women with breast cancer. Cancer Biol Ther. 2014;15:1586-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Ruddy KJ, DeSantis SD, Barry W, Guo H, Block CC, Borges V, Winer EP, Partridge AH. Extended therapy with letrozole and ovarian suppression in premenopausal patients with breast cancer after tamoxifen. Clin Breast Cancer. 2014;14:413-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Schiavon G, Smith IE. Status of adjuvant endocrine therapy for breast cancer. Breast Cancer Res. 2014;16:206. [PubMed] |
| 17. | Budzar AU. Endocrine Therapies in Breast Cancer. New York: Oxford University Press 2007; 29-36. |
| 18. | Powles TJ, Ashley S, Tidy A, Smith IE, Dowsett M. Twenty-year follow-up of the Royal Marsden randomized, double-blinded tamoxifen breast cancer prevention trial. J Natl Cancer Inst. 2007;99:283-290. [PubMed] |
| 19. | Osborne CK, Zhao H, Fuqua SA. Selective estrogen receptor modulators: structure, function, and clinical use. J Clin Oncol. 2000;18:3172-3186. [PubMed] |
| 20. | Wu X, Hawse JR, Subramaniam M, Goetz MP, Ingle JN, Spelsberg TC. The tamoxifen metabolite, endoxifen, is a potent antiestrogen that targets estrogen receptor alpha for degradation in breast cancer cells. Cancer Res. 2009;69:1722-1727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 174] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 21. | Regan MM, Leyland-Jones B, Bouzyk M, Pagani O, Tang W, Kammler R, Dell’orto P, Biasi MO, Thürlimann B, Lyng MB. CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the breast international group 1-98 trial. J Natl Cancer Inst. 2012;104:441-451. [PubMed] [DOI] [Full Text] |
| 22. | Rae JM, Drury S, Hayes DF, Stearns V, Thibert JN, Haynes BP, Salter J, Sestak I, Cuzick J, Dowsett M. CYP2D6 and UGT2B7 genotype and risk of recurrence in tamoxifen-treated breast cancer patients. J Natl Cancer Inst. 2012;104:452-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 192] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 23. | Johnson MD, Zuo H, Lee KH, Trebley JP, Rae JM, Weatherman RV, Desta Z, Flockhart DA, Skaar TC. Pharmacological characterization of 4-hydroxy-N-desmethyl tamoxifen, a novel active metabolite of tamoxifen. Breast Cancer Res Treat. 2004;85:151-159. [PubMed] |
| 24. | Aebi S, Davidson T, Gruber G, Cardoso F. Primary breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2011;22 Suppl 6:vi12-vi24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 25. | Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010;28:2784-2795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2535] [Cited by in RCA: 2425] [Article Influence: 151.6] [Reference Citation Analysis (0)] |
| 26. | Davies C, Godwin J, Gray R, Clarke M, Cutter D, Darby S, McGale P, Pan HC, Taylor C, Wang YC. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378:771-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2515] [Cited by in RCA: 2221] [Article Influence: 148.1] [Reference Citation Analysis (0)] |
| 27. | Freedman OC, Fletcher GG, Gandhi S, Mates M, Dent SF, Trudeau ME, Eisen A. Adjuvant endocrine therapy for early breast cancer: a systematic review of the evidence for the 2014 Cancer Care Ontario systemic therapy guideline. Curr Oncol. 2015;22:S95-S113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Davies C, Pan H, Godwin J, Gray R, Arriagada R, Raina V, Abraham M, Medeiros Alencar VH, Badran A, Bonfill X. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1677] [Cited by in RCA: 1435] [Article Influence: 110.4] [Reference Citation Analysis (0)] |
| 29. | Burstein HJ, Temin S, Anderson H, Buchholz TA, Davidson NE, Gelmon KE, Giordano SH, Hudis CA, Rowden D, Solky AJ. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: american society of clinical oncology clinical practice guideline focused update. J Clin Oncol. 2014;32:2255-2269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 523] [Cited by in RCA: 560] [Article Influence: 46.7] [Reference Citation Analysis (0)] |
| 30. | Mustonen MV, Pyrhönen S, Kellokumpu-Lehtinen PL. Toremifene in the treatment of breast cancer. World J Clin Oncol. 2014;5:393-405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 21] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 31. | Vogel CL, Johnston MA, Capers C, Braccia D. Toremifene for breast cancer: a review of 20 years of data. Clin Breast Cancer. 2014;14:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 32. | Recker RR, Kendler D, Recknor CP, Rooney TW, Lewiecki EM, Utian WH, Cauley JA, Lorraine J, Qu Y, Kulkarni PM. Comparative effects of raloxifene and alendronate on fracture outcomes in postmenopausal women with low bone mass. Bone. 2007;40:843-851. [PubMed] |
| 33. | Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN, Bevers TB, Fehrenbacher L, Pajon ER, Wade JL. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: Preventing breast cancer. Cancer Prev Res (Phila). 2010;3:696-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 538] [Cited by in RCA: 439] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 34. | Vogel VG. Role of hormones in cancer prevention. Am Soc Clin Oncol Educ Book. 2014;34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 35. | Yeh WL, Shioda K, Coser KR, Rivizzigno D, McSweeney KR, Shioda T. Fulvestrant-induced cell death and proteasomal degradation of estrogen receptor α protein in MCF-7 cells require the CSK c-Src tyrosine kinase. PLoS One. 2013;8:e60889. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 65] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 36. | Lumachi F, Luisetto G, Basso SM, Basso U, Brunello A, Camozzi V. Endocrine therapy of breast cancer. Curr Med Chem. 2011;18:513-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 37. | Wardell SE, Marks JR, McDonnell DP. The turnover of estrogen receptor α by the selective estrogen receptor degrader (SERD) fulvestrant is a saturable process that is not required for antagonist efficacy. Biochem Pharmacol. 2011;82:122-130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 113] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 38. | Puhalla S, Bhattacharya S, Davidson NE. Hormonal therapy in breast cancer: a model disease for the personalization of cancer care. Mol Oncol. 2012;6:222-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 39. | Ikeda H, Taira N, Nogami T, Shien K, Okada M, Shien T, Doihara H, Miyoshi S. Combination treatment with fulvestrant and various cytotoxic agents (doxorubicin, paclitaxel, docetaxel, vinorelbine, and 5-fluorouracil) has a synergistic effect in estrogen receptor-positive breast cancer. Cancer Sci. 2011;102:2038-2042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 40. | Jiang D, Huang Y, Han N, Xu M, Xu L, Zhou L, Wang S, Fan W. Fulvestrant, a selective estrogen receptor down-regulator, sensitizes estrogen receptor negative breast tumors to chemotherapy. Cancer Lett. 2014;346:292-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 41. | Mehta RS, Barlow WE, Albain KS, Vandenberg TA, Dakhil SR, Tirumali NR, Lew DL, Hayes DF, Gralow JR, Livingston RB. Combination anastrozole and fulvestrant in metastatic breast cancer. N Engl J Med. 2012;367:435-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 283] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 42. | Gong DD, Man CF, Xu J, Fan Y. Fulvestrant 250 mg versus anastrozole 1 mg in the treatment of advanced breast cancer: a meta-analysis of randomized controlled trials. Asian Pac J Cancer Prev. 2014;15:2095-2100. [PubMed] |
| 43. | Kuter I, Gee JM, Hegg R, Singer CF, Badwe RA, Lowe ES, Emeribe UA, Anderson E, Sapunar F, Finlay P. Dose-dependent change in biomarkers during neoadjuvant endocrine therapy with fulvestrant: results from NEWEST, a randomized Phase II study. Breast Cancer Res Treat. 2012;133:237-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 44. | Ma CX, Adjei AA, Salavaggione OE, Coronel J, Pelleymounter L, Wang L, Eckloff BW, Schaid D, Wieben ED, Adjei AA. Human aromatase: gene resequencing and functional genomics. Cancer Res. 2005;65:11071-11082. [PubMed] |
| 45. | Miller WR. Biological rationale for endocrine therapy in breast cancer. Best Pract Res Clin Endocrinol Metab. 2004;18:1-32. [PubMed] |
| 46. | Miller WR; Breastcancer. org. Topic: Hormonal Therapy 2015; Available from: http://www.breastcancer.org/research-news/ovary-suppression-w-aromasin-better-than-w-tamoxifen. |
| 47. | Masuda N, Sagara Y, Kinoshita T, Iwata H, Nakamura S, Yanagita Y, Nishimura R, Iwase H, Kamigaki S, Takei H. Neoadjuvant anastrozole versus tamoxifen in patients receiving goserelin for premenopausal breast cancer (STAGE): a double-blind, randomised phase 3 trial. Lancet Oncol. 2012;13:345-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 138] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 48. | Gnant M, Mlineritsch B, Stoeger H, Luschin-Ebengreuth G, Knauer M, Moik M, Jakesz R, Seifert M, Taucher S, Bjelic-Radisic V. Zoledronic acid combined with adjuvant endocrine therapy of tamoxifen versus anastrozol plus ovarian function suppression in premenopausal early breast cancer: final analysis of the Austrian Breast and Colorectal Cancer Study Group Trial 12. Ann Oncol. 2015;26:313-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 222] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 49. | Pfeiler G, Königsberg R, Fesl C, Mlineritsch B, Stoeger H, Singer CF, Pöstlberger S, Steger GG, Seifert M, Dubsky P. Impact of body mass index on the efficacy of endocrine therapy in premenopausal patients with breast cancer: an analysis of the prospective ABCSG-12 trial. J Clin Oncol. 2011;29:2653-2659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 182] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 50. | Deeks ED, Scott LJ. Exemestane: a review of its use in postmenopausal women with breast cancer. Drugs. 2009;69:889-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 51. | Francis PA, Regan MM, Fleming GF, Láng I, Ciruelos E, Bellet M, Bonnefoi HR, Climent MA, Da Prada GA, Burstein HJ. Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med. 2015;372:436-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 465] [Cited by in RCA: 520] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 52. | Pritchard KI, Burris HA, Ito Y, Rugo HS, Dakhil S, Hortobagyi GN, Campone M, Csöszi T, Baselga J, Puttawibul P. Safety and efficacy of everolimus with exemestane vs. exemestane alone in elderly patients with HER2-negative, hormone receptor-positive breast cancer in BOLERO-2. Clin Breast Cancer. 2013;13:421-432.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 92] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 53. | Rose C, Vtoraya O, Pluzanska A, Davidson N, Gershanovich M, Thomas R, Johnson S, Caicedo JJ, Gervasio H, Manikhas G. An open randomised trial of second-line endocrine therapy in advanced breast cancer. comparison of the aromatase inhibitors letrozole and anastrozole. Eur J Cancer. 2003;39:2318-2327. [PubMed] |
| 54. | Tominaga T, Adachi I, Sasaki Y, Tabei T, Ikeda T, Takatsuka Y, Toi M, Suwa T, Ohashi Y. Double-blind randomised trial comparing the non-steroidal aromatase inhibitors letrozole and fadrozole in postmenopausal women with advanced breast cancer. Ann Oncol. 2003;14:62-70. [PubMed] |
| 55. | Ellis MJ, Suman VJ, Hoog J, Lin L, Snider J, Prat A, Parker JS, Luo J, DeSchryver K, Allred DC. Randomized phase II neoadjuvant comparison between letrozole, anastrozole, and exemestane for postmenopausal women with estrogen receptor-rich stage 2 to 3 breast cancer: clinical and biomarker outcomes and predictive value of the baseline PAM50-based intrinsic subtype--ACOSOG Z1031. J Clin Oncol. 2011;29:2342-2349. [PubMed] |
| 56. | Mauri D, Pavlidis N, Polyzos NP, Ioannidis JP. Survival with aromatase inhibitors and inactivators versus standard hormonal therapy in advanced breast cancer: meta-analysis. J Natl Cancer Inst. 2006;98:1285-1291. [PubMed] |
| 57. | Dowsett M, Cuzick J, Ingle J, Coates A, Forbes J, Bliss J, Buyse M, Baum M, Buzdar A, Colleoni M. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28:509-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 567] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 58. | Jerusalem G, Bachelot T, Barrios C, Neven P, Di Leo A, Janni W, de Boer R. A new era of improving progression-free survival with dual blockade in postmenopausal HR(+), HER2(-) advanced breast cancer. Cancer Treat Rev. 2015;41:94-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 59. | Saji S, Kimura-Tsuchiya R. Combination of molecular-targeted drugs with endocrine therapy for hormone-resistant breast cancer. Int J Clin Oncol. 2015;20:268-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 60. | Finn RS, Crown JP, Lang I, Boer K, Bondarenko IM, Kulyk SO, Ettl J, Patel R, Pinter T, Schmidt M. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16:25-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1200] [Cited by in RCA: 1477] [Article Influence: 123.1] [Reference Citation Analysis (0)] |
| 61. | Raha P, Thomas S, Thurn KT, Park J, Munster PN. Combined histone deacetylase inhibition and tamoxifen induces apoptosis in tamoxifen-resistant breast cancer models, by reversing Bcl-2 overexpression. Breast Cancer Res. 2015;17:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 62. | Liu P, Cheng H, Roberts TM, Zhao JJ. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov. 2009;8:627-644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2286] [Cited by in RCA: 2165] [Article Influence: 127.4] [Reference Citation Analysis (0)] |
| 63. | Bachelot T, McCool R, Duffy S, Glanville J, Varley D, Fleetwood K, Zhang J, Jerusalem G. Comparative efficacy of everolimus plus exemestane versus fulvestrant for hormone-receptor-positive advanced breast cancer following progression/recurrence after endocrine therapy: a network meta-analysis. Breast Cancer Res Treat. 2014;143:125-133. [PubMed] |
| 64. | Martín M, Loibl S, von Minckwitz G, Morales S, Martinez N, Guerrero A, Anton A, Aktas B, Schoenegg W, Muñoz M. Phase III trial evaluating the addition of bevacizumab to endocrine therapy as first-line treatment for advanced breast cancer: the letrozole/fulvestrant and avastin (LEA) study. J Clin Oncol. 2015;33:1045-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 100] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 65. | Gibson LJ, Dawson C, Lawrence DH, Bliss JM. Aromatase inhibitors for treatment of advanced breast cancer in postmenopausal women. Cochrane Database Syst Rev. 2007;24:CD003370. [PubMed] |
| 66. | Gibson L, Lawrence D, Dawson C, Bliss J. Aromatase inhibitors for treatment of advanced breast cancer in postmenopausal women. Cochrane Database Syst Rev. 2009;CD003370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3850] [Cited by in RCA: 2758] [Article Influence: 919.3] [Reference Citation Analysis (0)] |
| 67. | Riemsma R, Forbes CA, Kessels A, Lykopoulos K, Amonkar MM, Rea DW, Kleijnen J. Systematic review of aromatase inhibitors in the first-line treatment for hormone sensitive advanced or metastatic breast cancer. Breast Cancer Res Treat. 2010;123:9-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 68. | Bachelot T, Bourgier C, Cropet C, Ray-Coquard I, Ferrero JM, Freyer G, Abadie-Lacourtoisie S, Eymard JC, Debled M, Spaëth D. Randomized phase II trial of everolimus in combination with tamoxifen in patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative metastatic breast cancer with prior exposure to aromatase inhibitors: a GINECO study. J Clin Oncol. 2012;30:2718-2724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 510] [Cited by in RCA: 533] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 69. | Piccart M, Hortobagyi GN, Campone M, Pritchard KI, Lebrun F, Ito Y, Noguchi S, Perez A, Rugo HS, Deleu I. Everolimus plus exemestane for hormone-receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: overall survival results from BOLERO-2†. Ann Oncol. 2014;25:2357-2362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 446] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 70. | Akin S, Babacan T, Sarici F, Altundag K. A novel targeted therapy in breast cancer: cyclin dependent kinase inhibitors. J BUON. 2014;19:42-46. [PubMed] |
| 71. | Dent SF, Gaspo R, Kissner M, Pritchard KI. Aromatase inhibitor therapy: toxicities and management strategies in the treatment of postmenopausal women with hormone-sensitive early breast cancer. Breast Cancer Res Treat. 2011;126:295-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Langdon S, Olsha O, Park Y S- Editor: Ji FF L- Editor: A E- Editor: Wang CH