Published online Mar 5, 2026. doi: 10.4331/wjbc.v17.i1.117645
Revised: January 15, 2026
Accepted: February 2, 2026
Published online: March 5, 2026
Processing time: 83 Days and 15 Hours
The diagnosis of immunoglobulin G4 (IgG4)-related autoimmune neuropathies relies on a combination of clinical evaluation, imaging, and biological analyses, including serum and cerebrospinal fluid assessments. Several IgG4 autoantibodies have been described in these disorders, including muscle-specific kinase IgG4, leucine-rich glioma-inactivated 1 IgG4, nodo-paranodal IgG4, Ig-like domain-containing protein 5, anti-dipeptidyl-peptidase-like protein-6 antibodies, and contactin-associated protein-like 2 IgG4. Accurate identification of these autoan
Core Tip: Human leukocyte antigen (HLA) class II molecules present peptides derived from neuronal and glial membrane proteins to CD4+ T cells, particularly follicular helper T cells and interleukin (IL)-10-producing regulatory subsets. In the IL-10-rich milieu associated with chronic antigen exposure, B cells undergo immunoglobulin G4 (IgG4) class switching and differentiate into high-affinity plasma cells and memory cells, thereby driving the production of pathogenic IgG4 neuro-autoantibodies. These antibodies disrupt protein-protein interactions rather than inducing classical inflammatory responses. IgG4 neuro-autoantibodies are mainly detected in serum and cerebrospinal fluid using cell-based assays, although these assays are labor-intensive and require specialized cell culture and transfection facilities. HLA typing may have clinical relevance in patients with intrathecal IgG4 antibodies and may help explain specific phenotypic associations in anti-Ig-like domain-containing protein 5 disease, including the increased frequency of sleep and bulbar manifestations. Distinct HLA associations also underlie the divergent clinical profiles observed in leucine-rich glioma-inactivated 1 and contactin-associated protein-like 2 antibody diseases.
- Citation: Bouayad A. Clinical utility of human leukocyte antigen genotyping and immunoglobulin G4 autoantibody testing in autoimmune neurological diseases: A focused minireview. World J Biol Chem 2026; 17(1): 117645
- URL: https://www.wjgnet.com/1949-8454/full/v17/i1/117645.htm
- DOI: https://dx.doi.org/10.4331/wjbc.v17.i1.117645
Autoimmune neuropathies arise from a breakdown of immunological tolerance to components of the central nervous system (CNS) and the peripheral nervous system (PNS), including myelin, Schwann cells, axons, and motor or autonomic ganglionic neurons[1-3]. In a subset of these disorders, compelling evidence supports antibody-mediated autoimmunity driven by immunoglobulin G4 (IgG4) autoantibodies that target extracellular neuronal and glial antigens[4,5]. IgG4 is the least abundant IgG subclass in serum, accounting for approximately 1%-4% of total circulating IgG[6]. Notably, patients with IgG4-mediated autoimmune neuropathies typically lack the characteristic features of IgG4-related disease (IgG4-RD), such as marked serum IgG4 elevation, tissue fibrosis, and infiltration by IgG4-positive plasma cells[4,7]. The detection of increased IgG4 levels and IgG4 oligoclonal bands in cerebrospinal fluid (CSF) may reflect blood-brain barrier dysfunction or intrathecal immune activation[8,9]. However, these markers lack sufficient specificity and sensitivity as diagnostic markers for IgG4-mediated autoimmune neurological disorders, where diagnosis primarily relies on detecting antigen-specific autoantibodies rather than on CSF IgG4 patterns[10]. Therefore, diagnosis of IgG4-mediated autoimmune neurological disorders indeed relies more heavily on detecting specific pathogenic IgG4 autoantibodies rather than CSF IgG4 patterns.
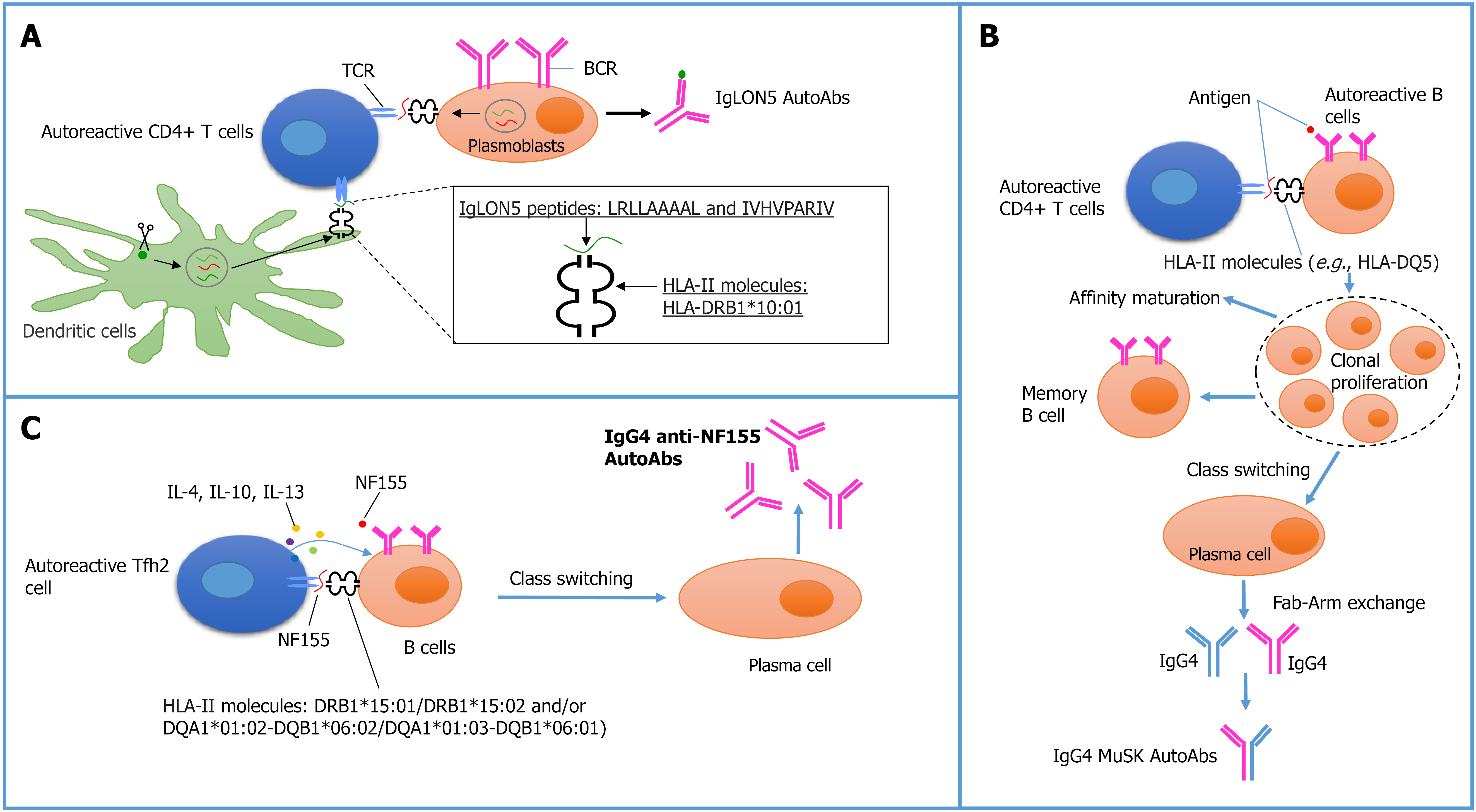
Pathogenic IgG4 autoantibodies have been identified against muscle-specific kinase (MuSK)[11,12], leucine-rich glioma-inactivated 1 (LGI1)[13], contactin-1 (CNTN1)[14], neurofascin-155 (NF155)[15], contactin-associated protein 1 (Caspr1)[16], contactin-associated protein-like 2 (CASPR2)[17], Ig-like domain-containing protein 5 (IgLON5)[18], and dipeptidyl-peptidase-like protein-6 (DPPX)[19,20] in various autoimmune neuropathies. Both human leukocyte antigen (HLA) class II loci[21-24] and environmental factors, including infections and alterations in the gut microbiota[20,25], have been implicated in the pathogenesis of these disorders. However, the mechanisms underlying these associations remain unclear. HLA class II molecules typically bind peptides derived from extracellular neuronal and glial antigens and present them to follicular helper T (Tfh) cells, which play a central role in promoting IgG4 autoantibody responses[26,27]. Several studies have demonstrated that dysregulated Tfh cell activity, particularly within the Tfh2 subset, promotes plasmablast differentiation and IgG4 class switching in IgG4-RD[28,29]. Nevertheless, the direct contribution of autoreactive B cells to the pathogenesis of IgG4-mediated autoimmune neuropathies remains poorly defined and requires further investigation.
Detection of IgG4 autoantibodies has become central to the diagnosis and stratification of neurological disorders, particularly in patients refractory to standard anti-inflammatory therapies, including intravenous immunoglobulin (IVIg), immunosuppressants, and plasmapheresis[5]. While neural autoantibody testing does not replace comprehensive evaluation, including clinical, imaging, electrophysiological, and CSF assessments, it can, when properly applied and interpreted, significantly enhance diagnostic precision in autoimmune neurological disorders[30].
In light of the rapidly advancing understanding of autoantibody-mediated mechanisms in neurological disease, this minireview summarizes current knowledge regarding the role of HLA alleles in regulating IgG4 autoantibody pro
Myasthenia gravis (MG) is a heterogeneous disorder of the neuromuscular junction. Approximately 7% of patients with acetylcholine receptor-negative MG have IgG4 autoantibodies targeting MuSK, a postsynaptic protein. These antibodies reflect defects in both central and peripheral B-cell tolerance, allowing autoreactive B cells to persist and differentiate into plasma cells[31]. The circulating B-cell repertoire in MuSK MG is markedly distorted[32]. It originates from an abnormal naive B-cell pool characterized by biased immunoglobulin heavy chain (IGHV) gene usage, marked by reduced IGHV3 and increased IGHV1 and IGHV4 expression, shaped by defective tolerance checkpoints, and subsequently expands into the memory compartment after activation[32]. IgG4 antibodies can undergo fragment antigen-binding (Fab) arm exchange, leading to bispecific antibodies[32]. The pivotal importance of class-switched, high-affinity IgG4 autoantibodies in the pathophysiology of MuSK MG implies cooperation between autoreactive B cells and CD4+ T lymphocytes[11]. Activation of autoreactive CD4+ T cells necessitates recognition of neural-specific peptides presented by HLA class II molecules to T-cell receptors, thereby implicating HLA class II alleles as key susceptibility factors (Figure 1). This hypothesis is supported by genetic studies across diverse ethnic populations, which have identified specific HLA-DRB1 and DQB1 genes as risk factors for the development of IgG4 autoantibodies. The higher IgG4 titres of MuSK auto
IgLON5 is a neuronal cell adhesion molecule that plays key roles in cell adhesion and signaling through interactions with cytoskeletal proteins. Antibodies targeting IgLON5 are predominantly of the IgG4 subclass, as demonstrated by immunostaining of rat hippocampal tissue[38]. These autoantibodies constitute the immunological hallmark of the disease and are typically detected in both serum and CSF[39].
Anti-IgLON5 antibodies have been significantly associated with the HLA-DRB1*10:01 allele[40,41]. HLA class II alleles may present IgLON5-derived peptides with specific binding affinities that enhance antibody production, or present them in distinct temporal or contextual settings, resulting in stronger antibody responses (Figure 1). In silico predictions identified two IgLON5 peptides (LRLLAAAAL and IVHVPARIV) potentially presented by HLA-DRB1 molecules, particularly HLA-DRB1*10:01, to T cells, thereby initiating or sustaining the autoimmune response in anti-IgLON5 disease[23]. Although the helper T cell and the B cell must interact with parts of the same antigen, they do not need to recognize the same epitope, a phenomenon known as linked recognition[42]. Furthermore, anti-IgLON5 disease shows a strong association with specific HLA-DQ alleles, particularly DQB1*05:01-DQA1*01:01, DQB1*05:01-DQA1*01:05, and DQB1*05:03-DQA1*01:04, implicating HLA-DQ-mediated antigen presentation and T-cell activation as key mechanisms in the initiation of autoimmunity[41,43,44]. HLA-DQB1*05 subtypes may mediate risk more directly than DRB1*10:01 due to LD[43]. Patients with the HLA-DRB1*10:01 allele showed increased serum anti-IgLON5 IgG levels[39]. Interestingly, Koneczny et al[45] demonstrated that patients carrying the HLA-DRB1*10:01 allele may exhibit enhanced antibody production within the brain and spinal cord (intrathecal synthesis) in anti-IgLON5 disease. Therefore, HLA typing may be clinically relevant in patients with intrathecal IgG4 anti-IgLON5 antibodies. Individuals lacking CSF antibodies are more likely to develop a progressive supranuclear palsy-like phenotype and less frequently carry the HLA-DRB1*10:01- DQB1*05:01 haplotype[23]. This rare haplotype, present in less than 3% of the general population, therefore supports the diagnosis of anti-IgLON5 disease[23].
Anti-IgLON5 disease is characterized by progressive brainstem dysfunction, gait instability, sleep disturbances, cognitive decline, and movement disorders[40]. Sleep abnormalities in this condition typically include parasomnias, stridors, and obstructive apnea[46]. The HLA-DRB1*10:01 allele may explain associations with certain clinical features of anti-IgLON5 disease, such as a higher frequency of sleep and bulbar symptoms[23]. Unlike most other autoimmune disorders, anti-IgLON5 disease predominantly affects older individuals, with onset before the age of 50 being uncommon[18]. Moreover, patients carrying specific HLA-DQ risk alleles tend to develop the disease later in life[43].
Expression of CASPR2 and LGI1 occurs in both the CNS and PNS, including the dorsal root ganglia and peripheral nerves. Anti-LGI1 is one of the common neuronal-surface autoantibodies in autoimmune encephalitis affecting adults over 40 years of age, with an incidence of 0.8 per million per year in the Dutch population[47]. Patients with anti-LGI1 and anti-CASPR2 antibodies exhibit marked clinical heterogeneity. Genome-wide and replication studies have shown a strong association between anti-LGI1 encephalitis and the HLA-DRB1*07:01-DQA1*02:01-DQB1*02:02 haplotype[48]. Similarly, anti-LGI1 encephalitis has been linked to the DRB1*07:01-DQB1*02:02 haplotype in an independent cohort[24]. In addition, another study, besides replicating the above-mentioned HLA association with LGI1-mediated encephalitis, described an additional association between the HLA-DRB1*11:01-DQA1*05:01-DQB1*03:01 haplotype and anti-CASPR2 encephalitis[22]. Antibodies against LGI1 are most commonly linked to epilepsy and limbic encephalitis, whereas antibodies against CASPR2 are associated with Morvan syndrome, neuropathic pain, and neuromyotonia[5]. These differences in the clinical heterogeneity of patients with anti-LGI1 and CASPR2 antibodies may depend on the HLA genotype[44]. Recent evidence suggests that HLA genetics may help stratify clinical subtypes of CASPR2 autoimmunity, with HLA-DRB1*11:01 allele enriched specifically among patients with limbic encephalitis[44,49,50]. In silico analyses revealed distinct CASPR2-derived peptides with a high likelihood of presentation by the overrepresented HLA-DQA1*05:01-DQB1*03:01 heterodimer[22].
Together, these data indicate that distinct HLA associations underlie the clinical divergence of LGI1- and CASPR2-antibody diseases: HLA-DRB1*07:01 is linked to LGI1 encephalitis, whereas HLA-DRB1*11:01 specifically enriches CNS forms of CASPR2 autoimmunity, highlighting allele-specific immune pathways that drive phenotype variation.
IgG4 anti-NF155 antibodies are predominantly detected in a subset of chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) known as autoimmune nodopathy[51]. These autoantibodies target the glial isoform of NF155, which is expressed in the paranodal loops of Schwann cells[27,52]. The prevalence of these autoantibodies ranges from 1% to 21% among CIDP patients, depending on the population studied and the detection method used[27]. Anti-NF155-associated autoimmune nodopathy primarily affects young men and is characterized by cerebellar-like tremor, distal motor involvement, and ataxia[53-55]. Patients often exhibit a poor response to IVIg therapy[56].
The mechanisms underlying the production of IgG4 anti-NF155 antibodies remain incompletely understood. Their presence in serum reflects impaired central and peripheral B-cell tolerance, allowing the persistence of autoreactive memory B cells and plasma cells that continuously secrete pathogenic antibodies[2] (Figure 1). Recent single-cell analyses suggest that impaired development of naïve B cells in NF155-mediated autoimmune nodopathy may result from defective BCR signaling and dysregulated CD4+ T cell function[57]. Circulating autoreactive B cells are detectable in patients with antibodies targeting nodal and paranodal proteins, including CNTN1, CASPR1, and most prominently NF155[2]. Several studies have also reported associations between anti-NF155 IgG4 autoantibodies and HLA class II alleles, specifically DRB1*15 and the DRB1*15-DQB1*06 haplotype, across different ethnic populations[58-60]. These findings suggest that cooperation between autoreactive B cells and CD4+ T lymphocytes may drive the generation of IgG4 anti-NF155 antibodies (Figure 1). Presentation of NF155 peptides by HLA class II molecules, particularly DRB1*15:01, DRB1*15:02, DQA1*01:02-DQB1*06:02, and DQA1*01:03-DQB1*06:01, activates naïve T cells and promotes Tfh2/Th1 cell-differentiation (Figure 1)[27]. Tfh2 lymphocytes promote IgG4 class-switch recombination through interleukin (IL)-4, IL-13, and IL-10 secretion[27]. The NF155 epitope with the highest affinity for presentation by HLA class II molecules corresponds to the peptide spanning amino acids 336-344[59].
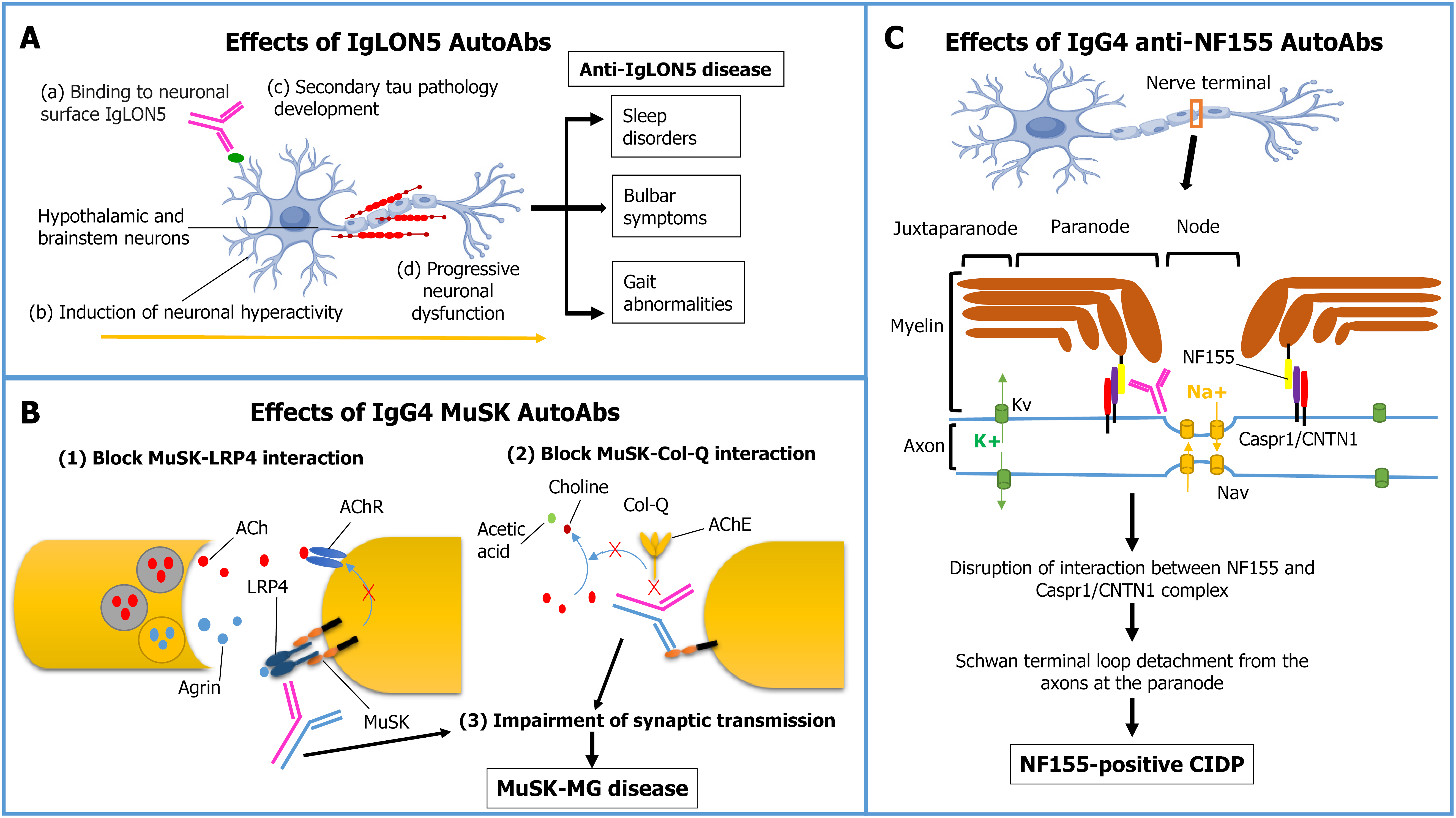
IgG4 autoantibodies do not activate the complement system or engage activating Fcγ receptors on immune cells. IgG4 neuro-autoantibodies cause autoimmune neurological diseases primarily through functional disruption of protein-protein interactions, rather than complement activation or classical inflammation[61]. Their pathophysiological effects fall into three main categories (Figure 2):
Autoantibodies targeting trans-synaptic protein complexes impair synaptic transmission through distinct but interconnected mechanisms. Anti-MuSK antibodies interfere with the MuSK-low-density lipoprotein receptor-related protein 4 (LRP4) interaction, preventing agrin-mediated clustering of acetylcholine receptors at the neuromuscular junction[62-64]. This disruption leads to disorganized postsynaptic architecture and reduced responsiveness to acetylcholine[64,65]. Similarly, anti-LGI1 antibodies disrupt the LGI1-ADAM22/ADAM23 complex, impairing postsynaptic anchoring of AMPA receptors and altering presynaptic Kv1 potassium channel function, thereby contributing to limbic encephalitis[66,67]. Finally, anti-CASPR2 antibodies inhibit the interaction between Caspr2 and contactin-2[17], leading to aberrant Kv1 channel distribution and neuronal hyperexcitability[68], which clinically manifests as Morvan’s syndrome or peripheral nerve hyperexcitability.
IgG4 anti-NF155 antibodies target neurofascin-155 in nerve terminals and roots where the blood-nerve barrier is absent or permeable[69]. These antibodies prevent the interaction between glial NF155 and the axonal contactin-1/Caspr1 complex that normally forms a septate-like junction anchoring myelin loops to the axon[26]. This binding leads to the disassembly of the paranodal junctions and disruption of the paranodal architecture[26]. The disruption leads to loss of ion channel segregation, paranode dismantling, and Schwann cell terminal loop detachment from axons[70]. The pathogenic effect is characterized by reduced Nfasc155 protein levels and impaired formation of the paranodal complex in neonatal animals[69].
Anti-IgLON5 antibodies bind persistently to neuronal cell-surface IgLON5, predominantly expressed in the hypothalamic and brainstem regions, triggering acute neuronal hyperactivity[38]. Prolonged neuronal dysfunction may subsequently lead to tau hyperphosphorylation and accumulation as a downstream neurodegenerative process, particularly in hypothalamic and brainstem neurons, resulting in the characteristic sleep, bulbar, and motor dysfunctions of anti-IgLON5 disease[71-73].
The detection of anti-MuSK antibodies primarily relies on cell-based immunofluorescence assays (CBA-IFA), which are considered viable alternatives to the traditional radioimmunoprecipitation assay (RIA) due to their commercial availability, high sensitivity, specificity, and ability to detect conformationally preserved epitopes[74,75]. Fixed CBA-IFA can serve as a first-step diagnostic test, whereas live CBA-IFA is useful for confirming serum-negative cases identified by RIA and fixed CBA-IFA[76]. However, these assays are time-consuming because they require cell culture facilities and the use of transfected or genetically engineered cell lines. Enzyme-linked immunosorbent assay (ELISA) can be used for the quantitative measurement of MuSK-IgG in patient serum[77]. This immunoassay offers several advantages, including wide accessibility, low cost, simple laboratory procedures, and high specificity, making it easier to implement in routine clinical laboratories, especially when RIA or cell-based assays (CBA) is not available[75]. The use of IgG subclass-specific secondary antibodies in CBA allows identification of IgG4 predominance, which is diagnostically significant in MuSK-MG[78]. Anti-MuSK antibodies primarily recognize the Ig-like domain 1 of the MuSK epitope[12]. Functional assays, including the solid-phase binding assay and the tyrosine phosphorylation assay, are essential for demonstrating the pathogenic activity of MuSK autoantibodies. The solid-phase binding assay directly assesses the inhibition of the MuSK-Lrp4 interaction by IgG4 antibodies, while the tyrosine phosphorylation assay evaluates the blockade of agrin-induced MuSK phosphorylation[12].
IgLON5 autoantibodies are usually detected in serum and CSF using both CBA and immunohistochemistry (IHC)[71]. This approach provides optimal diagnostic sensitivity and specificity[79]. The CBA, employing indirect immunofluorescence on HEK293 cells expressing full-length human IgLON5, provides high specificity and enables subclass analysis, confirming the predominance of IgG4 antibodies[80]. IgLON5 antibodies recognize non-glycosylated epitopes in the Ig-like domain 2 of IgLON5 antigen[80]. IHC on rat hippocampal sections provides confirmatory diagnostic evidence by demonstrating neuronal surface binding of patient antibodies[18]. IgG4 binds to IgLON5 but does not induce internalization, as it is functionally monovalent[80].
The detection of IgG4 subclass of LGI1 and CASPR2 autoantibodies primarily relies on live CBA-IFA[81]. Commercial CBAs offer a rapid, specific, and reproducible diagnostic tool, with strong inter-observer agreement[82,83]. However, these assays have some limitations, including false-negative results and reduced sensitivity for certain autoantibodies, particularly in CSF samples[79,84]. Most studies indicate that LGI1 and CASPR2 antibodies are generally more readily detected in serum than in CSF[13,47,81,85]. Nevertheless, serum antibody positivity may not always correlate with clinical outcomes, underscoring the importance of interpreting results in the context of clinical presentation[86]. IHC on rodent brain tissue may be used as a screening test, showing characteristic hippocampal staining patterns, but it lacks subclass specificity[87].
Detection of anti-NF155 antibodies requires assays that maintain the native conformation structure of the glial isoform. CBA using HEK293 or COS7 cells expressing human NF155 are considered the most reliable diagnostic tools[88]. These assays enable IgG subclass determination and reduce cross-reactivity with other neurofascin isoforms, including NF140 and NF186[89]. Immunofluorescence using teased nerve fibers or paranodal regions of peripheral nerve tissue can confirm antibody localization but should be interpreted in conjunction with CBA results. ELISA can be used as a confirmatory method to quantify antibody titers and identify IgG subclasses[53,60]. Anti-NF155 antibody titers are informative for tracking disease progression, with studies demonstrating their association with clinical severity scores and serum neurofilament levels[60].
First-line treatment of IgG4-mediated neurological autoimmune diseases primarily consists of glucocorticoids (GCs), IVIg, and plasma exchange (PE)[90]. Short courses of high-dose GCs represent the most commonly used first-line acute therapy for MuSK-MG and anti-LGI1/anti-CASPR2 autoimmune encephalitis, often producing rapid initial clinical improvement[91,92]. However, their effectiveness is limited by significant adverse effects, including weight gain, hypertension, hyperglycaemia, and opportunistic infections, as well as by the development of refractory disease in a subset of patients[93].
IVIg has proven to be an effective anti-inflammatory and immunomodulatory therapy in anti-LGI1/anti-CASPR2 autoimmune encephalitis[92] and in DPPX antibody-mediated disease[94]. IVIg is frequently administered in combination with GCs as part of first-line treatment[95]. However, IVIg appears to be less effective in anti-NF155 autoimmune nodopathy[53,60] and in MuSK-MG[91]. This limited efficacy likely reflects the fact that IVIg targets inflammatory mechanisms largely irrelevant to IgG4 pathology and cannot efficiently neutralize or eliminate IgG4 autoantibodies[96].
PE is typically used following GCs and IVIg[90]. Patients receiving first-line immunotherapy combining PE with GCs, with or without IVIg, demonstrate higher recovery rates compared with those receiving regimens without GCs[90]. PE is a well-established and effective treatment for myasthenic exacerbations and crises[91] and appears to be more effective than IVIg in MuSK-MG[97]. PE is also effective in treating LGI1- and CASPR2-associated encephalitis[98].
Second-line treatment of IgG4-mediated neurological autoimmune diseases primarily consists of anti-CD20 agents and cyclophosphamide (CYC), although oral antimetabolite agents such as mycophenolate mofetil and azathioprine may also be considered[90,95,96]. The central role of autoreactive B cells and their cooperation with Tfh cells in driving IgG4 autoantibody production further supports the use of B-cell-targeted therapies in cases of refractory or relapsing disease. Rituximab (RTX) suppresses short-lived plasma cells and their CD20+ precursors involved in IgG4 production, leading to marked reductions in autoantibody titers across several IgG4 autoantibody-mediated neurological disorders, including MuSK-MG, CIDP with anti-NF155 antibodies, and autoimmune encephalitis associated with antibodies targeting the Ranvier paranode and juxtaparanode[99-101]. Clinical guidelines recommend early use of RTX when initial standard immunosuppressive therapy fails to achieve rapid remission[102]. Although the initial cost of RTX is high, this may be partially offset by reductions in long-term treatment costs. Notably, quantitative measurement of anti-NF155 antibody titers is useful for monitoring disease activity in autoimmune nodopathy and may help guide the duration and intensity of RTX therapy[60]. HLA typing may eventually enable risk stratification and personalized treatment strategies based on genetic susceptibility profiles. CYC is primarily used as a second-line immunosuppressive agent in severe or refractory autoimmune encephalitis, including anti-LGI1, anti-CASPR2, and DPPX-associated diseases[95,103,104]. However, CYC is used less frequently than RTX in MuSK-MG and anti-NF155 autoimmune nodopathy[105,106].
Third-line therapies for IgG4-mediated neurological autoimmune diseases include plasma cell-depleting agents and cytokine-targeting treatments[90]. Daratumumab, a monoclonal antibody targeting CD38, can deplete autoreactive long-lived plasma cells and has been shown to reduce anti-CASPR2 antibody levels in the CSF of patients unresponsive to RTX, thereby overcoming the limitations of therapies that target only CD20+ B cells[107]. Tocilizumab, a monoclonal antibody targeting the IL-6 receptor, has demonstrated clinical benefit in anti-LGI1- and CASPR2-associated autoimmune encephalitis, improving clinical outcomes and reducing antibody levels even in cases resistant to first-line therapy and RTX[108,109].
Recognizing IgG4-mediated neurological disorders as a distinct disease spectrum separate from systemic IgG4-related disease enhances diagnostic precision and facilitates the development of rational, targeted therapeutic strategies for these increasingly recognized conditions. These disorders are characterized by distinct pathophysiological mechanisms involving functional disruption of protein-protein interactions rather than classical inflammation; the absence of typical systemic features, such as fibrosis and marked serum IgG4 elevation; the presence of diverse IgG4 autoantibodies targeting antigens in the central and peripheral nervous systems; and specific HLA associations.
Integrating HLA class II typing with precise IgG4 autoantibody detection represents an emerging paradigm for the diagnosis and management of these disorders. However, many current assays require specialized cell-culture facilities and are time-consuming, limiting their availability in routine clinical laboratories. Commercial cell-based assays also show variable performance, particularly in CSF samples, which may contribute to inconsistent results between laboratories and hinder standardization. Future advances should therefore prioritize harmonizing detection methods, developing rapid point-of-care testing, and combining genetic and serological markers to guide personalized immunotherapies tailored to the distinct pathophysiological mechanisms of these increasingly recognized diseases. Additionally, future applications of liquid chromatography-tandem mass spectrometry technology may enable the dissection of the molecular architecture and glycosylation of IgG4 autoantibodies, unlocking novel diagnostic and prognostic biomarkers for autoimmune neurological disorders.
Current evidence for the treatment of IgG4-mediated neurological disorders remains limited, relying primarily on case reports, case series, and a small number of controlled studies. Well-designed studies are needed to establish evidence-based therapeutic strategies.
| 1. | Yang L, Smith RJP, Scolding NJ, Rice CM. IgG4-related disease in the nervous system. Pract Neurol. 2025;25:217-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 2. | Rohrbacher S, Seefried S, Hartmannsberger B, Annabelle R, Appeltshauser L, Arlt FA, Brämer D, Dresel C, Dorst J, Elmas Z, Franke C, Geis C, Högen T, Krause S, Marziniak M, Mäurer M, Prüss H, Schoeberl F, Schrank B, Steen C, Teichtinger H, Thieme A, Wessely L, Zernecke A, Sommer C, Doppler K. Different Patterns of Autoantibody Secretion by Peripheral Blood Mononuclear Cells in Autoimmune Nodopathies. Neurol Neuroimmunol Neuroinflamm. 2024;11:e200295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 3. | Dalakas MC. Pathogenesis of immune-mediated neuropathies. Biochim Biophys Acta. 2015;1852:658-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 84] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 4. | Endmayr V, Tunc C, Ergin L, De Rosa A, Weng R, Wagner L, Yu TY, Fichtenbaum A, Perkmann T, Haslacher H, Kozakowski N, Schwaiger C, Ricken G, Hametner S, Klotz S, Dutra LA, Lechner C, de Simoni D, Poppert KN, Müller GJ, Pirker S, Pirker W, Angelovski A, Valach M, Maestri M, Guida M, Ricciardi R, Frommlet F, Sieghart D, Pinter M, Kircher K, Artacker G, Höftberger R, Koneczny I. Anti-Neuronal IgG4 Autoimmune Diseases and IgG4-Related Diseases May Not Be Part of the Same Spectrum: A Comparative Study. Front Immunol. 2021;12:785247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 5. | Dalakas MC. Autoimmune Neurological Disorders with IgG4 Antibodies: a Distinct Disease Spectrum with Unique IgG4 Functions Responding to Anti-B Cell Therapies. Neurotherapeutics. 2022;19:741-752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 6. | Schur PH. IgG subclasses--a review. Ann Allergy. 1987;58:89-96, 99. [PubMed] |
| 7. | Ng YZJ, Bowen S, Phillips J, Rajasekaran M, Lu SS, Taylor BV. Neurological manifestations of immunoglobulin G(4) related disease: a systematic review of 393 cases. J Neurol Neurosurg Psychiatry. 2025;96:1109-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 8. | Trbojevic-Cepe M. Detection of Oligoclonal Ig Bands: Clinical Significance and Trends in Methodological Improvement. EJIFCC. 2004;15:86-94. [PubMed] |
| 9. | Bouayad A, El Oumri AA. Immunoglobulin G4 biomarkers and pathogenesis in immunoglobulin G4-related spinal pachymeningitis. World J Clin Cases. 2025;13:105331. [PubMed] [DOI] [Full Text] |
| 10. | Min YG, Ko BK, Han HJ, Kim M, Lee DH, Kim SW, Sung JJ, Shin HY. Intrathecal Antibody Synthesis in Autoimmune Nodopathy. J Peripher Nerv Syst. 2025;30:e70057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 11. | Fichtner ML, Vieni C, Redler RL, Kolich L, Jiang R, Takata K, Stathopoulos P, Suarez PA, Nowak RJ, Burden SJ, Ekiert DC, O'Connor KC. Affinity maturation is required for pathogenic monovalent IgG4 autoantibody development in myasthenia gravis. J Exp Med. 2020;217:e20200513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 12. | Huijbers MG, Zhang W, Klooster R, Niks EH, Friese MB, Straasheijm KR, Thijssen PE, Vrolijk H, Plomp JJ, Vogels P, Losen M, Van der Maarel SM, Burden SJ, Verschuuren JJ. MuSK IgG4 autoantibodies cause myasthenia gravis by inhibiting binding between MuSK and Lrp4. Proc Natl Acad Sci U S A. 2013;110:20783-20788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 264] [Cited by in RCA: 211] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 13. | Gadoth A, Pittock SJ, Dubey D, McKeon A, Britton JW, Schmeling JE, Smith A, Kotsenas AL, Watson RE, Lachance DH, Flanagan EP, Lennon VA, Klein CJ. Expanded phenotypes and outcomes among 256 LGI1/CASPR2-IgG-positive patients. Ann Neurol. 2017;82:79-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 262] [Article Influence: 29.1] [Reference Citation Analysis (1)] |
| 14. | Miura Y, Devaux JJ, Fukami Y, Manso C, Belghazi M, Wong AH, Yuki N; CNTN1-CIDP Study Group. Contactin 1 IgG4 associates to chronic inflammatory demyelinating polyneuropathy with sensory ataxia. Brain. 2015;138:1484-1491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 160] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 15. | Shelly S, Klein CJ, Dyck PJB, Paul P, Mauermann ML, Berini SE, Howe B, Fryer JP, Basal E, Bakri HM, Laughlin RS, McKeon A, Pittock SJ, Mills J, Dubey D. Neurofascin-155 Immunoglobulin Subtypes: Clinicopathologic Associations and Neurologic Outcomes. Neurology. 2021;97:e2392-e2403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 16. | Cortese A, Lombardi R, Briani C, Callegari I, Benedetti L, Manganelli F, Luigetti M, Ferrari S, Clerici AM, Marfia GA, Rigamonti A, Carpo M, Fazio R, Corbo M, Mazzeo A, Giannini F, Cosentino G, Zardini E, Currò R, Gastaldi M, Vegezzi E, Alfonsi E, Berardinelli A, Kouton L, Manso C, Giannotta C, Doneddu P, Dacci P, Piccolo L, Ruiz M, Salvalaggio A, De Michelis C, Spina E, Topa A, Bisogni G, Romano A, Mariotto S, Mataluni G, Cerri F, Stancanelli C, Sabatelli M, Schenone A, Marchioni E, Lauria G, Nobile-Orazio E, Devaux J, Franciotta D. Antibodies to neurofascin, contactin-1, and contactin-associated protein 1 in CIDP: Clinical relevance of IgG isotype. Neurol Neuroimmunol Neuroinflamm. 2020;7:e639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 142] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 17. | Patterson KR, Dalmau J, Lancaster E. Mechanisms of Caspr2 antibodies in autoimmune encephalitis and neuromyotonia. Ann Neurol. 2018;83:40-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 89] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 18. | Graus F, Sabater L, Gaig C, Gelpi E, Iranzo A, Dalmau JO, Santamaria J. Anti-IgLON5 Disease 10 Years Later: What We Know and What We Do Not Know. Neurol Neuroimmunol Neuroinflamm. 2025;12:e200353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 19. | Boronat A, Gelfand JM, Gresa-Arribas N, Jeong HY, Walsh M, Roberts K, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R, Graus F, Rudy B, Dalmau J. Encephalitis and antibodies to dipeptidyl-peptidase-like protein-6, a subunit of Kv4.2 potassium channels. Ann Neurol. 2013;73:120-128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 327] [Cited by in RCA: 252] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 20. | Hara M, Ariño H, Petit-Pedrol M, Sabater L, Titulaer MJ, Martinez-Hernandez E, Schreurs MW, Rosenfeld MR, Graus F, Dalmau J. DPPX antibody-associated encephalitis: Main syndrome and antibody effects. Neurology. 2017;88:1340-1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 162] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 21. | Alahgholi-Hajibehzad M, Yilmaz V, Gülsen-Parman Y, Aysal F, Oflazer P, Deymeer F, Saruhan-Direskeneli G. Association of HLA-DRB1∗14, -DRB1∗16 and -DQB1∗05 with MuSK-myasthenia gravis in patients from Turkey. Hum Immunol. 2013;74:1633-1635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 22. | Binks S, Varley J, Lee W, Makuch M, Elliott K, Gelfand JM, Jacob S, Leite MI, Maddison P, Chen M, Geschwind MD, Grant E, Sen A, Waters P, McCormack M, Cavalleri GL, Barnardo M, Knight JC, Irani SR. Distinct HLA associations of LGI1 and CASPR2-antibody diseases. Brain. 2018;141:2263-2271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 132] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 23. | Gaig C, Ercilla G, Daura X, Ezquerra M, Fernández-Santiago R, Palou E, Sabater L, Höftberger R, Heidbreder A, Högl B, Iranzo A, Santamaria J, Dalmau J, Graus F. HLA and microtubule-associated protein tau H1 haplotype associations in anti-IgLON5 disease. Neurol Neuroimmunol Neuroinflamm. 2019;6:e605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 80] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 24. | Kim TJ, Lee ST, Moon J, Sunwoo JS, Byun JI, Lim JA, Shin YW, Jun JS, Lee HS, Lee WJ, Yang AR, Choi Y, Park KI, Jung KH, Jung KY, Kim M, Lee SK, Chu K. Anti-LGI1 encephalitis is associated with unique HLA subtypes. Ann Neurol. 2017;81:183-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 145] [Article Influence: 16.1] [Reference Citation Analysis (1)] |
| 25. | Deng X, Gong X, Zhou D, Hong Z. Perturbations in gut microbiota composition in patients with autoimmune neurological diseases: a systematic review and meta-analysis. Front Immunol. 2025;16:1513599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 26. | Dziadkowiak E, Waliszewska-Prosół M, Nowakowska-Kotas M, Budrewicz S, Koszewicz Z, Koszewicz M. Pathophysiology of the Different Clinical Phenotypes of Chronic Inflammatory Demyelinating Polyradiculoneuropathy (CIDP). Int J Mol Sci. 2021;23:179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 27. | Kira JI. Anti-Neurofascin 155 Antibody-Positive Chronic Inflammatory Demyelinating Polyneuropathy/Combined Central and Peripheral Demyelination: Strategies for Diagnosis and Treatment Based on the Disease Mechanism. Front Neurol. 2021;12:665136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 28. | Akiyama M, Yasuoka H, Yamaoka K, Suzuki K, Kaneko Y, Kondo H, Kassai Y, Koga K, Miyazaki T, Morita R, Yoshimura A, Takeuchi T. Enhanced IgG4 production by follicular helper 2 T cells and the involvement of follicular helper 1 T cells in the pathogenesis of IgG4-related disease. Arthritis Res Ther. 2016;18:167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 147] [Article Influence: 14.7] [Reference Citation Analysis (3)] |
| 29. | Xu J, Zhai J, Zhao J. Pathogenic roles of follicular helper T cells in IgG4-related disease and implications for potential therapy. Front Immunol. 2024;15:1413860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 30. | Dalmau J, Graus F. Diagnostic criteria for autoimmune encephalitis: utility and pitfalls for antibody-negative disease. Lancet Neurol. 2023;22:529-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 120] [Article Influence: 40.0] [Reference Citation Analysis (1)] |
| 31. | Lee JY, Stathopoulos P, Gupta S, Bannock JM, Barohn RJ, Cotzomi E, Dimachkie MM, Jacobson L, Lee CS, Morbach H, Querol L, Shan JL, Vander Heiden JA, Waters P, Vincent A, Nowak RJ, O'Connor KC. Compromised fidelity of B-cell tolerance checkpoints in AChR and MuSK myasthenia gravis. Ann Clin Transl Neurol. 2016;3:443-454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 32. | Stathopoulos P, Kumar A, Heiden JAV, Pascual-Goñi E, Nowak RJ, O'Connor KC. Mechanisms underlying B cell immune dysregulation and autoantibody production in MuSK myasthenia gravis. Ann N Y Acad Sci. 2018;1412:154-165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 33. | Çebi M, Durmuş H, Yılmaz V, Yentür SP, Aysal F, Oflazer P, Parman Y, Deymeer F, Saruhan-Direskeneli G. Relation of HLA-DRB1 to IgG4 autoantibody and cytokine production in muscle-specific tyrosine kinase myasthenia gravis (MuSK-MG). Clin Exp Immunol. 2019;197:214-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 34. | Niks EH, van Leeuwen Y, Leite MI, Dekker FW, Wintzen AR, Wirtz PW, Vincent A, van Tol MJ, Jol-van der Zijde CM, Verschuuren JJ. Clinical fluctuations in MuSK myasthenia gravis are related to antigen-specific IgG4 instead of IgG1. J Neuroimmunol. 2008;195:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 35. | Niks EH, Kuks JB, Roep BO, Haasnoot GW, Verduijn W, Ballieux BE, De Baets MH, Vincent A, Verschuuren JJ. Strong association of MuSK antibody-positive myasthenia gravis and HLA-DR14-DQ5. Neurology. 2006;66:1772-1774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 36. | Hong Y, Li HF, Romi F, Skeie GO, Gilhus NE. HLA and MuSK-positive myasthenia gravis: A systemic review and meta-analysis. Acta Neurol Scand. 2018;138:219-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 37. | Marino M, Maiuri MT, Di Sante G, Scuderi F, La Carpia F, Trakas N, Provenzano C, Zisimopoulou P, Ria F, Tzartos SJ, Evoli A, Bartoccioni E. T cell repertoire in DQ5-positive MuSK-positive myasthenia gravis patients. J Autoimmun. 2014;52:113-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 38. | Landa J, Gaig C, Plagumà J, Saiz A, Antonell A, Sanchez-Valle R, Dalmau J, Graus F, Sabater L. Effects of IgLON5 Antibodies on Neuronal Cytoskeleton: A Link between Autoimmunity and Neurodegeneration. Ann Neurol. 2020;88:1023-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 87] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 39. | Grüter T, Möllers FE, Tietz A, Dargvainiene J, Melzer N, Heidbreder A, Strippel C, Kraft A, Höftberger R, Schöberl F, Thaler FS, Wickel J, Chung HY, Seifert F, Tschernatsch M, Nagel M, Lewerenz J, Jarius S, Wildemann BC, de Azevedo L, Heidenreich F, Heusgen R, Hofstadt-Van Oy U, Linsa A, Maaß JJ, Menge T, Ringelstein M, Pedrosa DJ, Schill J, Seifert-Held T, Seitz C, Tonner S, Urbanek C, Zittel S, Markewitz R, Korporal-Kuhnke M, Schmitter T, Finke C, Brüggemann N, Bien CI, Kleiter I, Gold R, Wandinger KP, Kuhlenbäumer G, Leypoldt F, Ayzenberg I; German Network for Research on Autoimmune Encephalitis (GENERATE). Clinical, serological and genetic predictors of response to immunotherapy in anti-IgLON5 disease. Brain. 2023;146:600-611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 95] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 40. | Gaig C, Graus F, Compta Y, Högl B, Bataller L, Brüggemann N, Giordana C, Heidbreder A, Kotschet K, Lewerenz J, Macher S, Martí MJ, Montojo T, Pérez-Pérez J, Puertas I, Seitz C, Simabukuro M, Téllez N, Wandinger KP, Iranzo A, Ercilla G, Sabater L, Santamaría J, Dalmau J. Clinical manifestations of the anti-IgLON5 disease. Neurology. 2017;88:1736-1743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 306] [Article Influence: 34.0] [Reference Citation Analysis (1)] |
| 41. | Andersen J, Jeffrey B, Varikatt W, Rodriguez M, Lin MW, Brown DA. IgLON5-IgG: Innocent Bystander or Perpetrator? Int J Mol Sci. 2024;25:7956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 42. | Snow EC, Noelle RJ, Uhr JW, Vitetta ES. Activation of antigen-enriched B cells. II. Role of linked recognition in B cell proliferation to thymus-dependent antigens. J Immunol. 1983;130:614-618. [PubMed] |
| 43. | Yogeshwar SM, Muñiz-Castrillo S, Sabater L, Peris-Sempere V, Mallajosyula V, Luo G, Yan H, Yu E, Zhang J, Lin L, Fagundes Bueno F, Ji X, Picard G, Rogemond V, Pinto AL, Heidbreder A, Höftberger R, Graus F, Dalmau J, Santamaria J, Iranzo A, Schreiner B, Giannoccaro MP, Liguori R, Shimohata T, Kimura A, Ono Y, Binks S, Mariotto S, Dinoto A, Bonello M, Hartmann CJ, Tambasco N, Nigro P, Prüss H, McKeon A, Davis MM, Irani SR, Honnorat J, Gaig C, Finke C, Mignot E. HLA-DQB1*05 subtypes and not DRB1*10:01 mediates risk in anti-IgLON5 disease. Brain. 2024;147:2579-2592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (1)] |
| 44. | Muñiz-Castrillo S, Honnorat J. Genetic predisposition to autoimmune encephalitis and paraneoplastic neurological syndromes. Curr Opin Neurol. 2024;37:329-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 45. | Koneczny I, Macher S, Hutterer M, Seifert-Held T, Berger-Sieczkowski E, Blaabjerg M, Breu M, Dreyhaupt J, Dutra LA, Erdler M, Fae I, Fischer G, Frommlet F, Heidbreder A, Högl B, Klose V, Klotz S, Liendl H, Nissen MS, Rahimi J, Reinecke R, Ricken G, Stefani A, Süße M, Teive HAG, Weis S, Berger T, Sabater L, Gaig C, Lewerenz J, Höftberger R. HLA dependency and possible clinical relevance of intrathecally synthesized anti-IgLON5 IgG4 in anti-IgLON5 disease. Front Immunol. 2024;15:1376456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 46. | Gaig C, Iranzo A, Cajochen C, Vilaseca I, Embid C, Dalmau J, Graus F, Santamaria J. Characterization of the sleep disorder of anti-IgLON5 disease. Sleep. 2019;42:zsz133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 47. | van Sonderen A, Thijs RD, Coenders EC, Jiskoot LC, Sanchez E, de Bruijn MA, van Coevorden-Hameete MH, Wirtz PW, Schreurs MW, Sillevis Smitt PA, Titulaer MJ. Anti-LGI1 encephalitis: Clinical syndrome and long-term follow-up. Neurology. 2016;87:1449-1456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 570] [Cited by in RCA: 461] [Article Influence: 46.1] [Reference Citation Analysis (1)] |
| 48. | Mueller SH, Färber A, Prüss H, Melzer N, Golombeck KS, Kümpfel T, Thaler F, Elisak M, Lewerenz J, Kaufmann M, Sühs KW, Ringelstein M, Kellinghaus C, Bien CG, Kraft A, Zettl UK, Ehrlich S, Handreka R, Rostásy K, Then Bergh F, Faiss JH, Lieb W, Franke A, Kuhlenbäumer G, Wandinger KP, Leypoldt F; German Network for Research on Autoimmune Encephalitis (GENERATE). Genetic predisposition in anti-LGI1 and anti-NMDA receptor encephalitis. Ann Neurol. 2018;83:863-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 113] [Article Influence: 14.1] [Reference Citation Analysis (1)] |
| 49. | Muñiz-Castrillo S, Joubert B, Elsensohn MH, Pinto AL, Saint-Martin M, Vogrig A, Picard G, Rogemond V, Dubois V, Tamouza R, Maucort-Boulch D, Honnorat J. Anti-CASPR2 clinical phenotypes correlate with HLA and immunological features. J Neurol Neurosurg Psychiatry. 2020;91:1076-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 50. | Binks S, Irani SR. Combining clinical and molecular heterogeneity within CASPR2-antibody mediated diseases: towards the underlying disease biology. J Neurol Neurosurg Psychiatry. 2020;91:1033-1034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 51. | Wang L, Pan J, Meng H, Yang Z, Zeng L, Liu J. Anti-NF155/NF186 IgG4 Antibody Positive Autoimmune Nodopathy. Brain Sci. 2022;12:1587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 52. | Querol L, Dalakas MC. The Discovery of Autoimmune Nodopathies and the Impact of IgG4 Antibodies in Autoimmune Neurology. Neurol Neuroimmunol Neuroinflamm. 2025;12:e200365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 53. | Querol L, Nogales-Gadea G, Rojas-Garcia R, Diaz-Manera J, Pardo J, Ortega-Moreno A, Sedano MJ, Gallardo E, Berciano J, Blesa R, Dalmau J, Illa I. Neurofascin IgG4 antibodies in CIDP associate with disabling tremor and poor response to IVIg. Neurology. 2014;82:879-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 283] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 54. | Ogata H, Yamasaki R, Hiwatashi A, Oka N, Kawamura N, Matsuse D, Kuwahara M, Suzuki H, Kusunoki S, Fujimoto Y, Ikezoe K, Kishida H, Tanaka F, Matsushita T, Murai H, Kira J. Characterization of IgG4 anti-neurofascin 155 antibody-positive polyneuropathy. Ann Clin Transl Neurol. 2015;2:960-971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 163] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 55. | Devaux JJ, Miura Y, Fukami Y, Inoue T, Manso C, Belghazi M, Sekiguchi K, Kokubun N, Ichikawa H, Wong AH, Yuki N. Neurofascin-155 IgG4 in chronic inflammatory demyelinating polyneuropathy. Neurology. 2016;86:800-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 220] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 56. | Zhang X, Zheng P, Devaux JJ, Wang Y, Liu C, Li J, Guo S, Song Y, Wang Q, Feng X, Wang Y. Chronic inflammatory demyelinating polyneuropathy with anti-NF155 IgG4 in China. J Neuroimmunol. 2019;337:577074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 57. | Roy B, Obaid AH, Wang Z, Kovacs KG, Ohashi S, Kalayci FNC, Joo D, Masi G, Coppola C, Das S, Hernandez AL, Martin-Aguilar L, Lleixà C, Nowak RJ, Querol L, O'Connor KC. B Cell Tolerance and BCR Signaling Dysregulation in NF155-Mediated Autoimmune Nodopathies. bioRxiv. 2025;2025.08.10.669569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 58. | Martinez-Martinez L, Lleixà MC, Boera-Carnicero G, Cortese A, Devaux J, Siles A, Rajabally Y, Martinez-Piñeiro A, Carvajal A, Pardo J, Delmont E, Attarian S, Diaz-Manera J, Callegari I, Marchioni E, Franciotta D, Benedetti L, Lauria G, de la Calle Martin O, Juárez C, Illa I, Querol L. Anti-NF155 chronic inflammatory demyelinating polyradiculoneuropathy strongly associates to HLA-DRB15. J Neuroinflammation. 2017;14:224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 59. | Ogata H, Isobe N, Zhang X, Yamasaki R, Fujii T, Machida A, Morimoto N, Kaida K, Masuda T, Ando Y, Kuwahara M, Kusunoki S, Nakamura Y, Matsushita T, Kira JI. Unique HLA haplotype associations in IgG4 anti-neurofascin 155 antibody-positive chronic inflammatory demyelinating polyneuropathy. J Neuroimmunol. 2020;339:577139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 60. | Martín-Aguilar L, Lleixà C, Pascual-Goñi E, Caballero-Ávila M, Martínez-Martínez L, Díaz-Manera J, Rojas-García R, Cortés-Vicente E, Turon-Sans J, de Luna N, Suárez-Calvet X, Gallardo E, Rajabally Y, Scotton S, Jacobs BC, Baars A, Cortese A, Vegezzi E, Höftberger R, Zimprich F, Roesler C, Nobile-Orazio E, Liberatore G, Hiew FL, Martínez-Piñeiro A, Carvajal A, Piñar-Morales R, Usón-Martín M, Albertí O, López-Pérez MÁ, Márquez F, Pardo-Fernández J, Muñoz-Delgado L, Cabrera-Serrano M, Ortiz N, Bartolomé M, Duman Ö, Bril V, Segura-Chávez D, Pitarokoili K, Steen C, Illa I, Querol L. Clinical and Laboratory Features in Anti-NF155 Autoimmune Nodopathy. Neurol Neuroimmunol Neuroinflamm. 2022;9:e1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 61. | Koneczny I, Yilmaz V, Lazaridis K, Tzartos J, Lenz TL, Tzartos S, Tüzün E, Leypoldt F. Common Denominators in the Immunobiology of IgG4 Autoimmune Diseases: What Do Glomerulonephritis, Pemphigus Vulgaris, Myasthenia Gravis, Thrombotic Thrombocytopenic Purpura and Autoimmune Encephalitis Have in Common? Front Immunol. 2020;11:605214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 62. | Yan M, Liu Z, Fei E, Chen W, Lai X, Luo B, Chen P, Jing H, Pan JX, Rivner MH, Xiong WC, Mei L. Induction of Anti-agrin Antibodies Causes Myasthenia Gravis in Mice. Neuroscience. 2018;373:113-121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 63. | Yu Z, Zhang M, Jing H, Chen P, Cao R, Pan J, Luo B, Yu Y, Quarles BM, Xiong W, Rivner MH, Mei L. Characterization of LRP4/Agrin Antibodies From a Patient With Myasthenia Gravis. Neurology. 2021;97:e975-e987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 64. | Mori S, Kubo S, Akiyoshi T, Yamada S, Miyazaki T, Hotta H, Desaki J, Kishi M, Konishi T, Nishino Y, Miyazawa A, Maruyama N, Shigemoto K. Antibodies against muscle-specific kinase impair both presynaptic and postsynaptic functions in a murine model of myasthenia gravis. Am J Pathol. 2012;180:798-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 89] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 65. | Dziadkowiak E, Baczyńska D, Waliszewska-Prosół M. MuSK Myasthenia Gravis-Potential Pathomechanisms and Treatment Directed against Specific Targets. Cells. 2024;13:556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 66. | Petit-Pedrol M, Sell J, Planagumà J, Mannara F, Radosevic M, Haselmann H, Ceanga M, Sabater L, Spatola M, Soto D, Gasull X, Dalmau J, Geis C. LGI1 antibodies alter Kv1.1 and AMPA receptors changing synaptic excitability, plasticity and memory. Brain. 2018;141:3144-3159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 96] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 67. | Fels E, Muñiz-Castrillo S, Vogrig A, Joubert B, Honnorat J, Pascual O. Role of LGI1 protein in synaptic transmission: From physiology to pathology. Neurobiol Dis. 2021;160:105537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 68. | Habib M, Wiessler AL, Fischer P, Niesner M, Selcho M, Abrante L, Werner C, Sodmann A, Koch M, Zare A, Prüss H, Dargvainiene J, Lewerenz J, Handreka R, Körtvelyessy P, Reinhold D, Thaler FS, Pitarokoili K, Kittel RJ, Briese M, Sendtner M, Rittner H, Leypoldt F, Sommer C, Blum R, Doppler K, Villmann C. Neuropathic Pain and Distinct CASPR2 Autoantibody IgG Subclasses Drive Neuronal Hyperexcitability. Neurol Neuroimmunol Neuroinflamm. 2025;12:e200423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 69. | Manso C, Querol L, Lleixà C, Poncelet M, Mekaouche M, Vallat JM, Illa I, Devaux JJ. Anti-Neurofascin-155 IgG4 antibodies prevent paranodal complex formation in vivo. J Clin Invest. 2019;129:2222-2236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 70. | Pillai AM, Thaxton C, Pribisko AL, Cheng JG, Dupree JL, Bhat MA. Spatiotemporal ablation of myelinating glia-specific neurofascin (Nfasc NF155) in mice reveals gradual loss of paranodal axoglial junctions and concomitant disorganization of axonal domains. J Neurosci Res. 2009;87:1773-1793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 160] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 71. | Sabater L, Gaig C, Gelpi E, Bataller L, Lewerenz J, Torres-Vega E, Contreras A, Giometto B, Compta Y, Embid C, Vilaseca I, Iranzo A, Santamaría J, Dalmau J, Graus F. A novel non-rapid-eye movement and rapid-eye-movement parasomnia with sleep breathing disorder associated with antibodies to IgLON5: a case series, characterisation of the antigen, and post-mortem study. Lancet Neurol. 2014;13:575-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 503] [Cited by in RCA: 403] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 72. | Pi Y, Zhang LL, Li JC. Anti-IgLON5 disease with distinctive brain MRI findings responding to immunotherapy: A case report. Medicine (Baltimore). 2021;100:e24384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 73. | Gelpi E, Höftberger R, Graus F, Ling H, Holton JL, Dawson T, Popovic M, Pretnar-Oblak J, Högl B, Schmutzhard E, Poewe W, Ricken G, Santamaria J, Dalmau J, Budka H, Revesz T, Kovacs GG. Neuropathological criteria of anti-IgLON5-related tauopathy. Acta Neuropathol. 2016;132:531-543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 186] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 74. | Luong K, Lozier BK, Novis CL, Smith TL, Zuromski LM, Peterson LK. Comparison of three methods for the detection of antibodies against muscle-specific kinase. J Immunol Methods. 2024;526:113627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 75. | Kwon YN, Woodhall M, Sung JJ, Kim KK, Lim YM, Kim H, Kim JE, Baek SH, Kim BJ, Park JS, Seok HY, Kim DS, Kwon O, Park KH, Sohn E, Bae JS, Yoon BN, Kim NH, Ahn SW, Choi K, Oh J, Park HJ, Shin KJ, Lee S, Park J, Kim SH, Seok JI, Bae DW, An JY, Joo IS, Choi SJ, Nam TS, Kim S, Park KJ, Kwon KH, Waters P, Hong YH. Clinical pitfalls and serological diagnostics of MuSK myasthenia gravis. J Neurol. 2023;270:1478-1486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 76. | Spagni G, Gastaldi M, Businaro P, Chemkhi Z, Carrozza C, Mascagna G, Falso S, Scaranzin S, Franciotta D, Evoli A, Damato V. Comparison of Fixed and Live Cell-Based Assay for the Detection of AChR and MuSK Antibodies in Myasthenia Gravis. Neurol Neuroimmunol Neuroinflamm. 2023;10:e200038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 77. | Spagni G, Vincent A, Sun B, Falso S, Jacobson LW, Devenish S, Evoli A, Damato V. Serological Markers of Clinical Improvement in MuSK Myasthenia Gravis. Neurol Neuroimmunol Neuroinflamm. 2024;11:e200313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 78. | Huda S, Waters P, Woodhall M, Leite MI, Jacobson L, De Rosa A, Maestri M, Ricciardi R, Heckmann JM, Maniaol A, Evoli A, Cossins J, Hilton-Jones D, Vincent A. IgG-specific cell-based assay detects potentially pathogenic MuSK-Abs in seronegative MG. Neurol Neuroimmunol Neuroinflamm. 2017;4:e357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 79. | Ruiz-García R, Muñoz-Sánchez G, Naranjo L, Guasp M, Sabater L, Saiz A, Dalmau J, Graus F, Martinez-Hernandez E. Limitations of a Commercial Assay as Diagnostic Test of Autoimmune Encephalitis. Front Immunol. 2021;12:691536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 80. | Sabater L, Planagumà J, Dalmau J, Graus F. Cellular investigations with human antibodies associated with the anti-IgLON5 syndrome. J Neuroinflammation. 2016;13:226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 81. | Binks SNM, Klein CJ, Waters P, Pittock SJ, Irani SR. LGI1, CASPR2 and related antibodies: a molecular evolution of the phenotypes. J Neurol Neurosurg Psychiatry. 2018;89:526-534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 152] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 82. | Muñoz-Sánchez G, Planagumà J, Naranjo L, Couso R, Sabater L, Guasp M, Martínez-Hernández E, Graus F, Dalmau J, Ruiz-García R. The diagnosis of anti-LGI1 encephalitis varies with the type of immunodetection assay and sample examined. Front Immunol. 2022;13:1069368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 83. | Yeo T, Chen Z, Chai JYH, Tan K. Detection of LGI1 and CASPR2 antibodies with a commercial cell-based assay in patients with very high VGKC-complex antibody levels. J Neurol Sci. 2017;378:85-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 84. | Goncalves D, Benaiteau M, Rogemond V, Closs S, Pinto AL, Dhairi M, Villard M, Picard G, Fabien N, Honnorat J. Comparison of commercial and in-house tissue-based and cell-based assays for the detection of autoantibodies targeting neuronal surface proteins: a prospective cohort study. Front Immunol. 2025;16:1563877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 85. | Irani SR, Pettingill P, Kleopa KA, Schiza N, Waters P, Mazia C, Zuliani L, Watanabe O, Lang B, Buckley C, Vincent A. Morvan syndrome: clinical and serological observations in 29 cases. Ann Neurol. 2012;72:241-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 367] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 86. | Kerstens J, Schreurs MWJ, de Vries JM, Neuteboom RF, Brenner J, Crijnen YS, van Steenhoven RW, de Bruijn MAAM, van Sonderen A, van Coevorden-Hameete MH, Bastiaansen AEM, Vermeiren MR, Damoiseaux JGMC, Otten HG, Frijns CJM, Meek B, Platteel ACM, van de Mortel A, Delnooz CCS, Broeren MAC, Verbeek MM, Hoff EI, Boukhrissi S, Franken SC, Nagtzaam MMP, Paunovic M, Veenbergen S, Sillevis Smitt PAE, Titulaer MJ. Autoimmune Encephalitis and Paraneoplastic Neurologic Syndromes: A Nationwide Study on Epidemiology and Antibody Testing Performance. Neurol Neuroimmunol Neuroinflamm. 2024;11:e200318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 87. | Nagata N, Kanazawa N, Mitsuhata T, Iizuka M, Nagashima M, Nakamura M, Kaneko J, Kitamura E, Nishiyama K, Iizuka T. Neuronal surface antigen-specific immunostaining pattern on a rat brain immunohistochemistry in autoimmune encephalitis. Front Immunol. 2022;13:1066830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 88. | Querol L, Siles AM, Alba-Rovira R, Jáuregui A, Devaux J, Faivre-Sarrailh C, Araque J, Rojas-Garcia R, Diaz-Manera J, Cortés-Vicente E, Nogales-Gadea G, Navas-Madroñal M, Gallardo E, Illa I. Antibodies against peripheral nerve antigens in chronic inflammatory demyelinating polyradiculoneuropathy. Sci Rep. 2017;7:14411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 89. | Lyou HJ, Chung YH, Kim MJ, Kim M, Jeon MY, Kim SW, Shin HY, Kim BJ. Clinical Features of Autoimmune Nodopathy With Anti-Neurofascin-155 Antibodies in South Koreans. J Clin Neurol. 2024;20:186-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 90. | Cheng L, Jia B, Wang C, Fu Q, Zhou L. Immunotherapy for autoimmune encephalitis. Cell Death Discov. 2025;11:207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 91. | Vakrakou AG, Karachaliou E, Chroni E, Zouvelou V, Tzanetakos D, Salakou S, Papadopoulou M, Tzartos S, Voumvourakis K, Kilidireas C, Giannopoulos S, Tsivgoulis G, Tzartos J. Immunotherapies in MuSK-positive Myasthenia Gravis; an IgG4 antibody-mediated disease. Front Immunol. 2023;14:1212757. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 92. | Rittel JC, Hudasch D, Doppler K, Bergh FT, Lesser M, Aktas O, Nagel M, Huttner HB, Rostasy K, Tauber S, Friese MA, Malter M, Madlener M, Kraft A, Hoffmann F, Lewerenz J, Senel M, Wickel J, Geis C, Moser A, Wandinger KP, Bartsch T, Leypoldt F, Thaler F, Kümpfel T, Meuth S, Melzer N, Finke C, Prüss H, Stangel M, Sühs KW; German Network for Research on Autoimmune Encephalitis (GENERATE). Intravenous immunoglobulin as first-line acute treatment in adults with autoimmune encephalitis caused by antibodies to NMDAR, LGI1 and CASPR2. J Neurol. 2025;272:287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 93. | Borges LS, Richman DP. Muscle-Specific Kinase Myasthenia Gravis. Front Immunol. 2020;11:707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 94. | Li EC, Zhang TY, Cai MT, Su SY, Shen CH, Lai QL, Zhang YX. Clinical and Paraclinical Characterizations, Management, and Prognosis in DPPX Antibody-Associated Encephalitis: A Systematic Review. Neurol Neuroimmunol Neuroinflamm. 2025;12:e200350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 95. | Shin YW, Lee ST, Park KI, Jung KH, Jung KY, Lee SK, Chu K. Treatment strategies for autoimmune encephalitis. Ther Adv Neurol Disord. 2018;11:1756285617722347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 189] [Cited by in RCA: 159] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 96. | Dalakas MC. IgG4-Mediated Neurologic Autoimmunities: Understanding the Pathogenicity of IgG4, Ineffectiveness of IVIg, and Long-Lasting Benefits of Anti-B Cell Therapies. Neurol Neuroimmunol Neuroinflamm. 2022;9:e1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 97. | Dalakas MC. Update on Intravenous Immunoglobulin in Neurology: Modulating Neuro-autoimmunity, Evolving Factors on Efficacy and Dosing and Challenges on Stopping Chronic IVIg Therapy. Neurotherapeutics. 2021;18:2397-2418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 98. | Heine J, Ly LT, Lieker I, Slowinski T, Finke C, Prüss H, Harms L. Immunoadsorption or plasma exchange in the treatment of autoimmune encephalitis: a pilot study. J Neurol. 2016;263:2395-2402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 99. | Zografou C, Vakrakou AG, Stathopoulos P. Short- and Long-Lived Autoantibody-Secreting Cells in Autoimmune Neurological Disorders. Front Immunol. 2021;12:686466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 100. | Marino M, Basile U, Spagni G, Napodano C, Iorio R, Gulli F, Todi L, Provenzano C, Bartoccioni E, Evoli A. Long-Lasting Rituximab-Induced Reduction of Specific-But Not Total-IgG4 in MuSK-Positive Myasthenia Gravis. Front Immunol. 2020;11:613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 72] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 101. | Jiao L, Xiang Y, Li S, Zhang F, Ruan X, Guo S. Efficacy of low dose rituximab in treatment-resistant CIDP with antibodies against NF-155. J Neuroimmunol. 2020;345:577280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 102. | Sanders DB, Wolfe GI, Benatar M, Evoli A, Gilhus NE, Illa I, Kuntz N, Massey JM, Melms A, Murai H, Nicolle M, Palace J, Richman DP, Verschuuren J, Narayanaswami P. International consensus guidance for management of myasthenia gravis: Executive summary. Neurology. 2016;87:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 986] [Cited by in RCA: 817] [Article Influence: 81.7] [Reference Citation Analysis (1)] |
| 103. | Nosadini M, Mohammad SS, Ramanathan S, Brilot F, Dale RC. Immune therapy in autoimmune encephalitis: a systematic review. Expert Rev Neurother. 2015;15:1391-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 170] [Article Influence: 15.5] [Reference Citation Analysis (1)] |
| 104. | Dinoto A, Ferrari S, Mariotto S. Treatment Options in Refractory Autoimmune Encephalitis. CNS Drugs. 2022;36:919-931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 105. | El-Salem K, Yassin A, Al-Hayk K, Yahya S, Al-Shorafat D, Dahbour SS. Treatment of MuSK-Associated Myasthenia Gravis. Curr Treat Options Neurol. 2014;16:283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 106. | Godil J, Barrett MJ, Ensrud E, Chahin N, Karam C. Refractory CIDP: Clinical characteristics, antibodies and response to alternative treatment. J Neurol Sci. 2020;418:117098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 107. | Scheibe F, Ostendorf L, Reincke SM, Prüss H, von Brünneck AC, Köhnlein M, Alexander T, Meisel C, Meisel A. Daratumumab treatment for therapy-refractory anti-CASPR2 encephalitis. J Neurol. 2020;267:317-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 108. | Jang S, Kim SY, Kim WJ, Chae JH, Kim KJ, Lim BC. A case of pediatric anti-leucine-rich glioma inactivated 1 encephalitis with faciobrachial dystonic seizure. Brain Dev. 2023;45:348-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 109. | Krogias C, Hoepner R, Müller A, Schneider-Gold C, Schröder A, Gold R. Successful treatment of anti-Caspr2 syndrome by interleukin 6 receptor blockade through tocilizumab. JAMA Neurol. 2013;70:1056-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/