Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.115200
Revised: December 31, 2025
Accepted: February 3, 2026
Published online: April 27, 2026
Processing time: 154 Days and 1.4 Hours
Gastrointestinal disorders (GIDs) frequently develop after abdominal surgery and impede postoperative recovery and quality-of-life improvement.
To determine whether acupuncture and moxibustion (Acu-Mox) can improve clinical outcomes in patients with postabdominal surgery GID.
We selected 105 patients with GID following abdominal procedures between December 2022 and December 2024. Comparative analyses were conducted on 50 patients in the control group receiving conventional Western medical treatment and 55 cases in the research group who additionally received Acu-Mox, with endpoints including overall effectiveness, the Intake, Feeling Nauseated, Emesis, Physical Exam, and Duration of Symptoms (I-FEED) scale score and its normalization time, gastrointestinal function recovery (bowel sound recovery, exhaust/defecation time, first liquid diet intake time), serum inflammatory indicators (C-reactive protein, interleukin-6, tumor necrosis factor-α), and complications (ileus, con
The research group showed a markedly higher total effective treatment rate than the controls. This group also exhibited a notable reduction in posttreatment I-FEED scores, C-reactive protein, interleukin-6, and tumor necrosis factor-α that were statistically lower vs the control group, together with shorter time to I-FEED score normalization, bowel sound recovery, exhaust/defecation, and the first postoperative liquid diet, as well as an obviously lower overall complication rate.
Acu-Mox for post-abdominal surgery GID is more effective than traditional Western medicine, deserving pro
Core Tip: In this study, 105 postabdominal surgery patients with gastrointestinal disorders were selected strictly according to the inclusion and exclusion criteria. Comparative analysis assessed the clinical effects of routine western medicine treatment vs combined western medicine treatment and acupuncture–moxibustion. Multidimensional evaluation results showed higher curative effects of Western medicine + acupuncture–moxibustion therapy on gastrointestinal disorder patients after abdominal surgery. Such therapy alleviates clinical symptoms, promotes postoperative gastrointestinal function recovery, inhibits serum inflammation, and ensures higher clinical safety. The results can provide a better treatment option for these patients.
- Citation: Zhao YC, Che JN, Zhang YQ, Luo NN, Wang DX, Wang ZJ, Zhang WZ, Ma J, Cao XP, Wen X. Acupuncture and moxibustion for postoperative gastrointestinal disorders: An efficacy analysis. World J Gastrointest Surg 2026; 18(4): 115200
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/115200.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.115200
Continuous iterative upgrades in surgical technology have transformed abdominal procedures into minimally invasive laparoscopic approaches; nevertheless, patients still face a high risk of postoperative complications, especially post
With the approval of the Affiliated Hospital of Gansu University of Chinese Medicine Ethics Committee, 105 patients with post-abdominal surgery GID were enrolled (December 2022 to December 2024) for retrospective analysis. The control group (n = 50) received routine care, while the research group (n = 55) received additional Acu-Mox treatment. Baseline characteristics between groups were comparable (P > 0.05), which ensured their comparability.
Patient eligibility: A GID diagnosis postabdominal surgery[11]; age: 18-80 years; GID symptoms (abdominal distension, nausea, vomiting, weakened or disappeared bowel sounds) of varying degrees after standard postoperative fasting (3-5 days); complete medical records.
Ineligibility: Severe postoperative infections or complications; preoperative ileus; chronic constipation/diarrhea; significant cardio-/cerebrovascular diseases or cardiac/hepatic/renal insufficiency; benign/malignant tumors; psy
The control group received conventional Western medical treatment: (1) Patients were kept fasting and water-deprived and placed in a semirecumbent position for continuous GI decompression. Vital signs (e.g., blood pressure and heart rate) and abdominal findings were closely monitored; (2) Nutritional support included intravenous fluid therapy and antibiotic administration to correct water-electrolyte imbalances and maintain acid-base equilibrium; and (3) Patients were assisted with clockwise abdominal massage and encouraged to ambulate early.
In addition to the abovementioned measures, the research group received Acu-Mox treatment. Patients lay in the supine position and underwent routine disinfection, with attention paid to thermal insulation. Based on the Stomach meridian of Foot-Yangming, and in accordance with the principles of local and proximal acupoint selection, meridian-based distal acupoint selection, and avoidance of surgical incision sites, the following acupoints were selected: Zusanli, Yinlingquan, Sanyinjiao, Xiajuxu, Shangjuxu, Taichong, Neiguan, and Tianshu (all selected bilaterally), as well as Hegu and Zhongwan. Qihai, Guanyuan, and Xuehai were additionally selected for patients with qi-blood deficiency. Acu
Overall treatment effectiveness: Markedly effective - normalization of stool frequency and color, passage of flatus within 24 hours, essential disappearance of abdominal pain, distension, nausea, and vomiting, and the return of normal bowel sounds[12]. Effective - the presence of bowel sounds and anal flatus within a 24-hour to 72-hour window, plus marked alleviation of clinical symptoms. Ineffective - a failure to pass flatus beyond 72 hours, persistently weak or absent bowel sounds, and unimproved/worsened initial symptoms. The total effective rate is the percentage of the sum of markedly effective and effective cases out of the total number of cases.
Intake, Feeling Nauseated, Emesis, Physical Exam, and Duration of Symptoms score: This tool evaluates intake, nausea, vomiting, abdominal examination, and symptom duration domains on a scale of 0-3. Patients’ postoperative GI function was judged as normal (0-2 points), postoperative GI intolerance (3-5 points), or POGD (≥ 6 points) based on the total score. In addition, Intake, Feeling Nauseated, Emesis, Physical Exam, and Duration of Symptoms (I-FEED) score recovery time, i.e., the time required to recover to a “normal” state from the beginning of the intervention, was recorded[13].
GI function recovery: Bowel sound recovery, as well as the first flatus/defecation/Liquid diet postoperation, was monitored.
Serum inflammatory biomarkers: Before and 1 week after treatment, each patient provided fasting blood samples (5 mL) for serum separation via centrifugation. Enzyme-linked immunosorbent assays were then conducted for C-reactive protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) quantification.
Complications: Cases suffering from ileus, constipation, bloating, and abdominal pain were counted, and the overall incidence rate in each group was computed.
Data analyses used SPSS 20.0, employing a P < 0.05 significance threshold. Continuous data that followed a normal distribution were statistically described using mean ± SD, with inter-group and intra-group (pre-treatment vs post-treatment) comparisons made with the t-test and the paired t-test, respectively; for the data that did not follow a normal distribution, they were expressed as median (interquartile range) [M (P25, P75)]. Qualitative data, expressed as n (%), were compared by χ2 tests.
Baseline comparability between study cohorts was confirmed, with no notable differences in gender distribution, age, disease course, anesthetic technique, or surgery type (P > 0.05; Table 1).
| Indicators | Control group (n = 50) | Research group (n = 55) | χ2/t | P value |
| Sex | 0.100 | 0.752 | ||
| Male | 27 (54.00) | 28 (50.91) | ||
| Female | 23 (46.00) | 27 (49.09) | ||
| Age (years) | 53.64 ± 7.87 | 56.53 ± 9.79 | 1.657 | 0.101 |
| Disease duration (weeks) | 11.48 ± 3.75 | 12.16 ± 2.95 | 1.037 | 0.302 |
| Anesthetic technique | 0.405 | 0.525 | ||
| General anaesthesia | 26 (52.00) | 32 (58.18) | ||
| Epidural anesthesia | 24 (48.00) | 23 (41.82) | ||
| Type of surgery | 1.181 | 0.758 | ||
| Appendicectomy | 24 (48.00) | 28 (50.91) | ||
| Hepatobiliary surgery | 16 (32.00) | 15 (27.27) | ||
| Gynecological surgery | 7 (14.00) | 6 (10.91) | ||
| Splenectomy | 3 (6.00) | 6 (10.91) |
The research group had higher overall effectiveness than the control group (90.91% vs 74.00%, P < 0.05; Table 2).
| Indicators | Control group (n = 50) | Research group (n = 55) | χ2 | P value |
| Markedly effective | 20 (40.00) | 27 (49.09) | ||
| Effective | 17 (34.00) | 23 (41.82) | ||
| Ineffective | 13 (26.00) | 5 (9.09) | ||
| Overall effectiveness | 37 (74.00) | 50 (90.91) | 5.272 | 0.022 |
Prior to intervention, I-FEED scores were comparable between the groups (P > 0.05). Both groups’ scores decreased posttreatment (P < 0.05), particularly in the research group (P < 0.05). The research group showed a significantly shorter recovery time than the control cohort (P < 0.001; Table 3).
Assessment of GI functional recovery, based on the return of bowel sounds, first flatus, first defecation, and first liquid diet, showed shorter times in the research group, indicating faster recovery than in the controls (P < 0.001; Table 4).
| Indicators | Control group (n = 50) | Research group (n = 55) | t | P value |
| Return of bowel sounds | 32.32 ± 6.02 | 25.91 ± 6.65 | 5.159 | < 0.001 |
| Time to first flatus | 40.94 ± 8.47 | 33.55 ± 5.56 | 5.331 | < 0.001 |
| Time to first defecation | 45.80 ± 8.33 | 39.60 ± 7.56 | 3.998 | < 0.001 |
| Time of first liquid diet | 71.84 ± 21.92 | 52.18 ± 21.00 | 4.692 | < 0.001 |
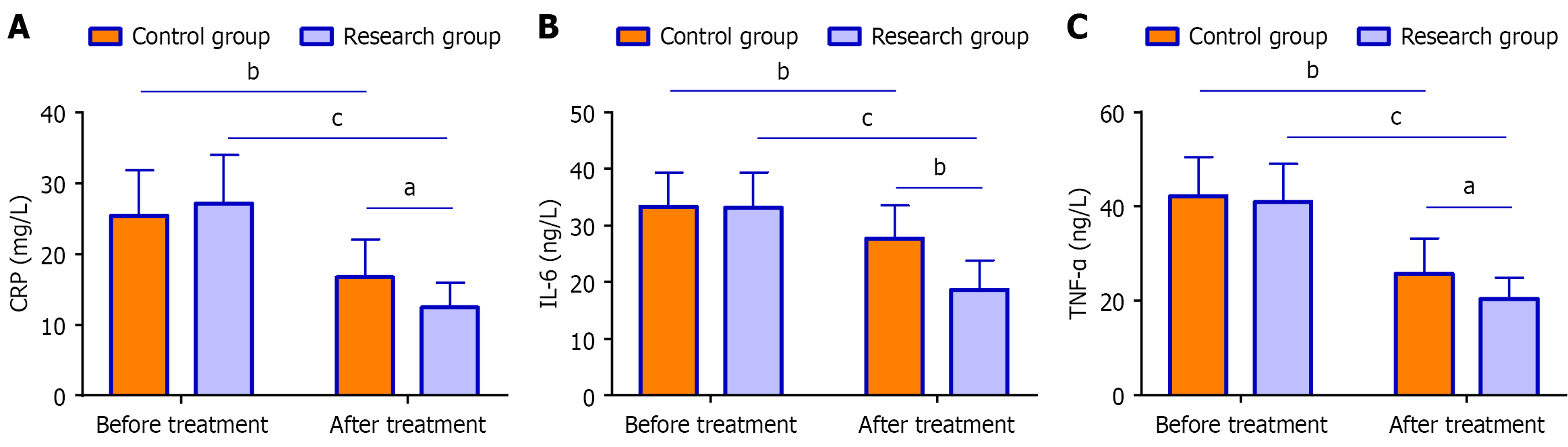
Enzyme-linked immunosorbent assay-based CRP, IL-6, and TNF-α measurements were equivalent between groups prior to treatment (P > 0.05). While both treatments led to significant reductions in all markers postintervention, the research group achieved consistently lower final levels than the controls (P < 0.05; Figure 1).
Safety assessment (Table 5), based on the incidence of complications including ileus, constipation, bloating, and abdominal pain, revealed a statistically significant reduction in the aggregate complication rate in the research group (10.91%) vs the control group (34.00%) (P < 0.01). All of the above complications were mild to moderate in severity, and no severe cases requiring surgical intervention occurred. The patients with relatively obvious symptoms were effectively controlled through standard symptomatic treatments (e.g., laxation, pain relief, GI decompression) without affecting the overall treatment plan.
| Indicators | Control group (n = 50) | Research group (n = 55) | χ2 | P value |
| Ileus | 4 (8.00) | 0 (0.00) | ||
| Constipation | 3 (6.00) | 1 (1.82) | ||
| Bloating | 4 (8.00) | 2 (3.64) | ||
| Abdominal pain | 6 (12.00) | 3 (5.45) | ||
| Total | 17 (34.00) | 6 (10.91) | 8.163 | 0.004 |
In this analysis of 105 patients with postabdominal-surgery GID, those receiving adjunctive Acu-Mox (research group) demonstrated a markedly higher overall efficacy rate than those receiving conventional care (90.91% vs 74.00%). TCM theory conceptualizes postoperative GID as a consequence of vital energy deficiency (qi and blood), organ dysfunction (spleen-stomach), circulatory obstruction (qi stagnation, blood stasis, and meridian obstruction), and systemic imbalance (Yin-Yang)[14]. Therapeutic strategies are therefore directed toward strengthening the body’s resistance, replenishing vital energy, promoting circulation, resolving stagnation, and restoring functional balance. In this study, stimulation of Zusanli by Acu-Mox was considered to replenish qi, regulate vital energy, dispel spleen dryness and dampness, promote the descent of intestinal qi, and alleviate weakness. Yinlingquan is believed to invigorate the spleen and eliminate dampness; Shangjuxu to dredge fu-qi and facilitate the transfer and resolution of accumulated stagnation; Zhongwan to strengthen the middle energizer and regulate qi; Qihai to strengthen the lower energizer, replenish vitality, and circulate qi to dissipate stagnation; and Guanyuan to tonify the kidney, nourish essence, and warm yang to relieve depletion. Xuehai, in combination with Zusanli, Sanyinjiao, and Tianshu, is thought to help warm yang, dispel cold, and regulate the spleen and stomach. The application of moxibustion-related thermal stimulation to these acupoints provides additional benefits, including improved local blood and lymphatic circulation, alleviation and resolution of smooth muscle spasm, and subsequent anti-inflammatory and analgesic effects[15].
Furthermore, Acu-Mox outperformed conventional Western medicine by achieving a more pronounced decrease in the I-FEED score and a faster return to normal scores. Patients receiving Acu-Mox also exhibited quicker recovery of bowel sounds, earlier flatus and defecation, and an accelerated time to their first liquid meal. Li et al[16] similarly reported Acu-Mox’s ability to markedly accelerate the first defecation/exhaust/ambulation and prevent GID in postmajor abdominal surgery patients. Zhou et al[17] applied Acu-Mox to POGD patients after colorectal cancer surgery and observed sig
Serum inflammatory indicator assessment showed significant CRP, IL-6, and TNF-α inhibition by Acu-Mox in postabdominal surgery GID patients, consistent with He et al[19], who reported marked CRP and IL-6 reductions on postoperative day 3 after radical gastrectomy for gastric cancer. Acu-Mox may achieve this serum inflammation inhibition effect in our cohort by stimulating Zusanli, which exerts an enteritis preventive effect through intestinal neurons that mediate vagal nerve signals, or by regulating the inflammatory microenvironment balance through vagal-adrenal pathway activation[20,21]. Acu-Mox can inhibit the activity of nuclear factor κB by regulating the Janus kinase 2/signal transducer and activator of transcription 3 signaling cascade mediated by the α7 nicotinic acetylcholine receptor in macrophages, thereby reducing the release of inflammatory factors in the body[22]. According to our data, postab
Acu-Mox has a definite effect on postabdominal surgery GID patients. It can effectively relieve clinical symptoms, promote postoperative GI function recovery, inhibit serum inflammation, and provide higher clinical safety. These findings may provide better treatment options for such patients.
| 1. | Gojayev A, Erkent M, Aydin HO, Karakaya E, Yildirim S, Moray G. Is laparoscopic surgery safe and feasible in acute adhesive ileus? Medicine (Baltimore). 2023;102:e34894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Alsharqawi N, Alhashemi M, Kaneva P, Baldini G, Fiore JF Jr, Feldman LS, Lee L. Validity of the I-FEED score for postoperative gastrointestinal function in patients undergoing colorectal surgery. Surg Endosc. 2020;34:2219-2226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 3. | Kong Q, Chen LM, Liu CY, Li W, Yin PH. The effect of acupuncture on gastrointestinal recovery after abdominal surgery: a narrative review from clinical trials. Int J Surg. 2024;110:5713-5721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 4. | Wang B, Hu L, Hu X, Han D, Wu J. Exploring perioperative risk factors for poor recovery of postoperative gastrointestinal function following gynecological surgery: A retrospective cohort study. Heliyon. 2024;10:e23706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 5. | Wang Y, Wang L, Ni X, Jiang M, Zhao L. Effect of acupuncture therapy for postoperative gastrointestinal dysfunction in gastric and colorectal cancers: an umbrella review. Front Oncol. 2024;14:1291524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (9)] |
| 6. | Liu L, Yuan X, Yang L, Zhang J, Luo J, Huang G, Huo J. Effect of acupuncture on hormone level in patients with gastrointestinal dysfunction after general anesthesia: A study protocol for a randomized controlled trial. Medicine (Baltimore). 2020;99:e19610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 7. | Ye Z, Wei X, Feng S, Gu Q, Li J, Kuai L, Luo Y, Xi Z, Wang K, Zhou J. Effectiveness and safety of acupuncture for postoperative ileus following gastrointestinal surgery: A systematic review and meta-analysis. PLoS One. 2022;17:e0271580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (3)] |
| 8. | Guo Y, Kong X, Cao Q, Li Y, Zhang Y, Huang J, Li K, Guan Y. Efficacy and safety of acupuncture in postoperative ileus after gynecological surgery: A protocol for system review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2021;100:e24342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Fang Y, Zhu J, Wang Z, Li K, Ding X, Wang N, Chu H. [Mechanism research progress on acupuncture-moxibustion therapy for functional gastrointestinal disorders: review and prospects]. Zhongguo Zhen Jiu. 2025;45:551-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Wang XY, Wang H, Guan YY, Cai RL, Shen GM. Acupuncture for functional gastrointestinal disorders: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2021;36:3015-3026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 11. | Xue J, Xu Z, Wang Q, Hou H, Wei L, Zhang J, Zhao X, Chen L, Ding F, Ma L, Zhao Y, Wang Y, Ma D, Wang T, Liu R, Gan TJ, Robinson N, Frank Y, Su F, Chi Y, Yang D, Liu S, Cui S, Wei Y, Chen Z, Qin Y, Cao L, Chen G, Shu K, Xiao Z, Zhang H, Yu J, Hu Z, Cheng H, Ma W, Liu G, Wang X, Cao X, Gao J, Kong G, Tao Q, Wang B, Wang J, Li H, Lyu C, Zhang Z, Li T, Yang K. Clinical practice guidelines for prevention and treatment of postoperative gastrointestinal disorder with Integrated Traditional Chinese and Western Medicine (2023). J Evid Based Med. 2024;17:207-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 12. | Liu Q, Shi Z, Zhang T, Jiang T, Luo X, Su X, Yang Y, Wei W. Efficacy and Safety of Chinese Herbal Medicine Xiao Yao San in Functional Gastrointestinal Disorders: A meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Front Pharmacol. 2021;12:821802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Wu CY, Lai CJ, Xiao FR, Yang JT, Yang SH, Lai DM, Tsuang FY. Validity of the IFEED classification in assessing postoperative gastrointestinal impairment in patients undergoing elective lumbar spinal surgery with general anesthesia: a prospective observational study. Perioper Med (Lond). 2024;13:50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 14. | Teschke R, Wolff A, Frenzel C, Eickhoff A, Schulze J. Herbal traditional Chinese medicine and its evidence base in gastrointestinal disorders. World J Gastroenterol. 2015;21:4466-4490. [PubMed] [DOI] [Full Text] |
| 15. | Huang KY, Liang S, Hu GY, Zou YY, Lu L, Zhang JB. [From Biological Effects of Local Cutaneous Thermal Stimulation to Moxibustion Therapy]. Zhen Ci Yan Jiu. 2015;40:504-509. [PubMed] |
| 16. | Li WJ, Gao C, An LX, Ji YW, Xue FS, Du Y. Perioperative transcutaneous electrical acupoint stimulation for improving postoperative gastrointestinal function: A randomized controlled trial. J Integr Med. 2021;19:211-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 17. | Zhou T, Wang S, Fan BJ, Zhang LX, Hu SH, Hou W. Clinical efficacy of acupuncture in treating postoperative gastrointestinal dysfunction of colorectal cancer, a systematic review and Meta analysis. Zhen Ci Yan Jiu. 2024;49:208-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 18. | Jang JH, Lee DJ, Bae CH, Ha KT, Kwon S, Park HJ, Hahm DH, Lee H, Kim S. Changes in small intestinal motility and related hormones by acupuncture stimulation at Zusanli (ST 36) in mice. Chin J Integr Med. 2017;23:215-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (4)] |
| 19. | He D, Wang FZ, Zhang Z, Huang F, Chen JJ, Li B. [Effect of low-frequency electrical acupoint stimulation on gastrointestinal motility function following radical gastrectomy in patients with gastric cancer]. Zhen Ci Yan Jiu. 2020;45:51-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Liu S, Fu W, Fu J, Chen G, He Y, Zheng T, Ma T. Electroacupuncture alleviates intestinal inflammation via a distinct neuro-immune signal pathway in the treatment of postoperative ileus. Biomed Pharmacother. 2024;173:116387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 21. | Liu S, Wang Z, Su Y, Qi L, Yang W, Fu M, Jing X, Wang Y, Ma Q. A neuroanatomical basis for electroacupuncture to drive the vagal-adrenal axis. Nature. 2021;598:641-645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 680] [Cited by in RCA: 542] [Article Influence: 108.4] [Reference Citation Analysis (4)] |
| 22. | Yang NN, Yang JW, Ye Y, Huang J, Wang L, Wang Y, Su XT, Lin Y, Yu FT, Ma SM, Qi LY, Lin LL, Wang LQ, Shi GX, Li HP, Liu CZ. Electroacupuncture ameliorates intestinal inflammation by activating α7nAChR-mediated JAK2/STAT3 signaling pathway in postoperative ileus. Theranostics. 2021;11:4078-4089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (4)] |
| 23. | Ying HZ. Impact of laser acupuncture on gastrointestinal function recovery in patients after acute appendicitis surgery: A retrospective clinical study. World J Gastrointest Surg. 2025;17:105897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (2)] |