Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.113773
Revised: November 23, 2025
Accepted: January 9, 2026
Published online: March 27, 2026
Processing time: 161 Days and 4 Hours
Traumatic liver rupture has high mortality (10%-15%) but current prognostic tools show limited accuracy. This study develops an integrated model combining se
To investigate the predictive value of serum biochemical indicators combined with imaging scores for the prognosis of patients with traumatic liver rupture, establish a comprehensive prediction model, and provide scientific evidence for early clinical identification of high-risk patients.
A single-center retrospective cohort study was conducted, including 186 patients with traumatic liver rupture treated from June 2020 to June 2024. Patient baseline data, serum biochemical indicators within 2 hours of admission (including liver function indicators, coagulation function, inflammatory indicators), and imaging scores were collected. The primary outcome was adverse prognosis within 30 days, including death, need for emergency surgical intervention, severe com
Among 186 patients, 142 cases (76.3%) had good prognosis and 44 cases (23.7%) had poor prognosis. Age, liver function indicators, coagulation function indica
The comprehensive prediction model established by combining serum biochemical indicators with imaging scores can effectively predict the prognosis of patients with traumatic liver rupture, providing important scientific evidence for early clinical identification of high-risk patients.
Core Tip: This study established a comprehensive prognostic prediction model for traumatic liver rupture by integrating serum biochemical indicators with clinical assessment and imaging evaluation. In a cohort of 186 patients, the integrated model demonstrated superior predictive performance compared to single-modality approaches. The model showed excellent discrimination and clinical utility, offering a practical tool for early identification of high-risk patients and guiding individualized treatment strategies in traumatic liver rupture management. All predictive variables are routinely available within 2 hours of hospital admission, ensuring rapid risk assessment without additional costs or specialized testing.
- Citation: Yin H, Liu Y, Chen JT, Li Z, Pan Y, Zhu F. Predictive value of serum biochemical indicators combined with imaging scores for prognosis of traumatic liver rupture. World J Gastrointest Surg 2026; 18(3): 113773
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/113773.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.113773
Traumatic liver rupture is one of the most common and dangerous types of injury in abdominal trauma, accounting for 35%-45% of all blunt abdominal trauma, with a mortality rate of 10%-15%[1]. With the increasing incidence of accidental injuries such as traffic accidents and falls from height, the incidence of traumatic liver rupture is showing an increasing trend year by year, and has become an important clinical challenge faced by trauma surgery[2]. Early accurate assessment of patient prognosis is of great significance for formulating reasonable treatment strategies and improving patient survival rates.
Currently, prognostic assessment of traumatic liver rupture mainly relies on clinical scoring systems, such as the Injury Severity Score (ISS) and Glasgow Coma Scale (GCS)[3]. However, these traditional scoring systems are mainly based on anatomical injury severity and consciousness status, making it difficult to comprehensively reflect the physiological functional status and injury severity of the body, with limited predictive accuracy. In recent years, the value of serum biochemical indicators in trauma prognosis assessment has gradually received attention, as liver function indicators, coagulation function, and inflammatory indicators can sensitively reflect the degree of liver injury and the body's inflammatory response status[4].
Imaging examination, particularly contrast-enhanced computed tomography (CT) scanning, is the gold standard for diagnosing and evaluating traumatic liver rupture[5]. CT imaging-based scoring systems, such as the Liver Trauma CT Severity Score (LTSS), can objectively quantify the anatomical characteristics of liver injury, including injury extent, hematoma size, intraperitoneal bleeding volume, etc., providing important information for prognostic assessment[6]. However, single imaging scores often ignore the patient’s physiological functional status, which may lead to insufficient predictive accuracy.
Serum biochemical indicators can reflect the body’s physiological functional status, while imaging scores objectively assess anatomical injury severity, and both have good complementarity[7]. Combining serum biochemical indicators with imaging scores is expected to establish a more comprehensive and accurate prognostic prediction model, providing more reliable evidence for clinical decision-making[8]. However, current research on the value of their combined application in prognostic prediction of traumatic liver rupture is still relatively limited.
Therefore, this study aims to explore the predictive value of serum biochemical indicators combined with imaging scores for the prognosis of patients with traumatic liver rupture, establish a comprehensive prediction model, and provide scientific evidence for early identification of high-risk patients and formulation of individualized treatment plans in clinical practice.
This study was a single-center retrospective cohort study. The study protocol was reviewed and approved by the Medical Ethics Committee of our hospital and complied with the ethical principles of the Declaration of Helsinki. As this was a retrospective study, patient informed consent was waived.
Study population: Patients with traumatic liver rupture admitted to our hospital from June 2020 to June 2024 were selected as study subjects.
Inclusion criteria: (1) Age ≥ 18 years; (2) Traumatic liver rupture confirmed by CT or surgery; (3) Time from trauma to hospital admission ≤ 24 hours; (4) Complete clinical data, including serum biochemical indicators and imaging examination data at admission; and (5) Complete follow-up data.
Exclusion criteria: (1) Previous history of liver disease (cirrhosis, liver cancer, chronic hepatitis, etc.); (2) Combined severe trauma to other organs (ISS score > 25 points and Hepatic Injury Scale (HIS) > 3 points)[9]; (3) Use of medications affec
Baseline data collection: The following patient data were collected through the HIS: (1) Demographic characteristics: Age, gender, body mass index; (2) Trauma mechanism: Traffic accident, fall from height, heavy object impact, others; (3) Past medical history: Hypertension, diabetes, heart disease, etc.; (4) Vital signs at admission: Systolic blood pressure, diastolic blood pressure, heart rate, respiratory rate, body temperature; (5) Consciousness status at admission (GCS); and (6) ISS.
Serum biochemical indicator testing: All patients had their first venous blood samples collected within 2 hours of admission, and the following indicators were tested using an automated biochemical analyzer (Beckman Coulter AU5800): (1) Liver function indicators: Alanine aminotransferase, aspartate aminotransferase (AST), total bilirubin, direct bilirubin, albumin, alkaline phosphatase, γ-glutamyl transferase; (2) Coagulation function: Prothrombin time, activated partial thromboplastin time, international normalized ratio (INR); (3) Inflammatory indicators: C-reactive protein (CRP), procalcitonin (PCT), white blood cell count (WBC), neutrophil percentage; and (4) Other indicators: Creatinine, blood urea nitrogen, lactate dehydrogenase, creatine kinase.
All patients underwent abdominal contrast-enhanced CT examination after admission, using a multi-slice spiral CT scanner with scanning parameters: Slice thickness 2 mm, reconstruction slice thickness 1mm, contrast agent dose 1.5 mL/kg body weight, injection rate 3.5 mL/second.
Liver injury grading: According to the 2018 World Society of Emergency Surgery liver trauma grading standard[10], liver injuries were classified into grades I-VI: Grade I was subcapsular hematoma < 10 cm2 or capsular tear depth < 1 cm; grade II was subcapsular hematoma 10-50 cm2 or liver parenchymal tear depth 1-3 cm, length < 10 cm; grade III was subcapsular hematoma > 50 cm2 or liver parenchymal tear depth > 3 cm; grade IV was liver parenchymal tear involving 25%-75% of liver lobe; grade V was liver parenchymal tear involving > 75% of liver lobe or > 3 Couinaud liver segments, or hepatic vein injury; grade VI was liver avulsion.
HIS: According to the American Association for the Surgery of Trauma standard, HIS was graded 1-6: Grade 1 was capsular tear or subcutaneous hematoma; grade 2 was capsular tear with active bleeding, liver parenchymal laceration
Imaging scoring system: The LTSS was used, which included liver injury extent (0-3 points: 0 points for no obvious injury, 1 point for injury involving < 25% liver parenchyma, 2 points for injury involving 25%-50% liver parenchyma, 3 points for injury involving > 50% liver parenchyma), hematoma size (0-2 points: 0 points for no hematoma, 1 point for hematoma diameter < 5 cm, 2 points for hematoma diameter ≥ 5 cm), intraperitoneal bleeding volume (0-3 points: 0 points for no intraperitoneal bleeding, 1 point for small amount of bleeding in pelvis only, 2 points for moderate amount of bleeding in pelvis + paracolic gutters, 3 points for large amount of bleeding in entire abdomen), active bleeding signs (0-2 points: 0 points for no active bleeding, 1 point for suspected active bleeding, 2 points for definite active bleeding). Total score 0-10 points, with higher scores indicating more severe injury. All imaging data were independently reviewed and scored by 2 radiologists with more than 10 years of experience in abdominal imaging diagnosis. When opinions differed, consensus was reached through discussion. Inter-rater reliability was assessed using intraclass correlation coefficient (ICC).
Conservative treatment or surgical treatment was adopted according to the severity of the patient's condition. (1) Conservative treatment included bed rest, fasting, fluid infusion, hemostasis, anti-infection, etc.; and (2) Surgical treatment included liver repair, partial hepatectomy, hepatic artery embolization, etc. Follow-up was conducted through outpatient visits, telephone follow-up, etc., with the follow-up endpoint being 6 months after discharge. All enrolled patients were followed up until January 2025 to ensure completeness of 6-month follow-up data.
Primary outcome indicator was poor prognosis, defined as the occurrence of any of the following events within 30 days of admission: Death, need for emergency surgical intervention, occurrence of serious complications (massive intraperitoneal bleeding, septic shock, multiple organ failure), or need for intensive care unit (ICU) treatment. Secondary outcome indicator was 6-month survival rate.
SPSS 25.0 software was used for statistical analysis. Continuous variables were assessed for normality using the Shapiro-Wilk test. Normally distributed data were expressed as mean ± SD, with between-group comparisons using t-test; non-normally distributed data were expressed as median (interquartile range), with between-group comparisons using Mann-Whitney U test. Categorical variables were expressed as numbers and percentages, with between-group comparisons using χ2 test or Fisher’s exact test.
Univariate analysis was used to screen factors related to prognosis, and variables with P < 0.1 were included in multivariate logistic regression analysis to identify independent predictors. A prediction model was established, and receiver operating characteristic curves were used to evaluate the predictive performance of the model, calculating area under the curve (AUC), sensitivity, specificity, positive predictive value, and negative predictive value. Hosmer-Lemeshow goodness-of-fit test was used to evaluate model calibration. Decision curve analysis (DCA) was used to evaluate the clinical net benefit of the model. Bootstrap resampling (1000 times) was used for internal validation.
Inter-rater reliability for imaging scoring was assessed using ICC, with ICC > 0.75 considered good reliability. All statistical tests were two-sided tests, with P < 0.05 considered statistically significant.
During the study period, a total of 186 patients with traumatic liver rupture were included, including 118 males (63.4%) and 68 females (36.6%), aged 18-78 years, with a mean age of (42.3 ± 15.7) years. According to whether poor prognosis occurred within 30 days, patients were divided into a good prognosis group (n = 142, 76.3%) and a poor prognosis group (n = 44, 23.7%). Comparison of baseline characteristics between the two groups (Table 1).
| Variable | Good prognosis group (n = 142) | Poor prognosis group (n = 44) | Statistical value | P value |
| Demographic characteristics | ||||
| Age (years), mean ± SD | 40.2 ± 14.8 | 48.5 ± 17.2 | t = 3.15 | 0.002 |
| Male | 87 (61.3) | 31 (70.5) | χ2 = 1.25 | 0.264 |
| BMI (kg/m2), mean ± SD | 23.4 ± 3.2 | 24.1 ± 3.8 | t = 1.12 | 0.264 |
| Trauma mechanism | ||||
| Traffic accident | 78 (54.9) | 28 (63.6) | χ2 = 4.83 | 0.185 |
| Fall from height | 35 (24.6) | 12 (27.3) | - | - |
| Heavy object impact | 21 (14.8) | 3 (6.8) | - | - |
| Others | 8 (5.6) | 1 (2.3) | - | - |
| Past medical history | ||||
| Hypertension | 23 (16.2) | 12 (27.3) | χ2 = 2.58 | 0.108 |
| Diabetes | 11 (7.7) | 6 (13.6) | χ2 = 1.42 | 0.233 |
| Heart disease | 8 (5.6) | 4 (9.1) | χ2 = 0.72 | 0.397 |
| Other diseases | 15 (10.6) | 8 (18.2) | χ2 = 1.86 | 0.172 |
| Vital signs | ||||
| Systolic BP (mmHg), mean ± SD | 128.4 ± 18.3 | 108.2 ± 22.1 | t = 5.98 | < 0.001 |
| Diastolic BP (mmHg), mean ± SD | 78.5 ± 12.6 | 68.3 ± 15.2 | t = 4.35 | < 0.001 |
| Heart rate (beats/minute), mean ± SD | 88.7 ± 16.2 | 106.3 ± 21.4 | t = 5.72 | < 0.001 |
| Respiratory rate (breaths/minute), mean ± SD | 19.2 ± 3.4 | 22.8 ± 4.7 | t = 5.21 | < 0.001 |
| Body temperature (°C), mean ± SD | 36.8 ± 0.6 | 36.4 ± 0.8 | t = 3.42 | 0.001 |
| Consciousness and injury scores, mean ± SD | ||||
| GCS score | 14.2 ± 1.8 | 11.8 ± 2.7 | t = 6.35 | < 0.001 |
| ISS score | 12.4 ± 4.3 | 18.7 ± 6.2 | t = 7.12 | < 0.001 |
Comparison of serum biochemical indicators between the two groups showed that liver function indicators, coagulation function indicators, and inflammatory indicators were all significantly worse in the poor prognosis group (Table 2).
| Indicator | Good prognosis group (n = 142) | Poor prognosis group (n = 44) | Statistical value | P value |
| Liver function indicators | ||||
| ALT (U/L) | 156.3 ± 89.2 | 312.7 ± 145.6 | t = 7.65 | < 0.001 |
| AST (U/L) | 168.4 ± 102.3 | 387.9 ± 178.4 | t = 8.92 | < 0.001 |
| TBIL (μmol/L) | 18.7 ± 8.4 | 34.2 ± 15.6 | t = 7.23 | < 0.001 |
| DBIL (μmol/L) | 7.2 ± 3.8 | 15.4 ± 8.2 | t = 7.81 | < 0.001 |
| ALB (g/L) | 38.6 ± 5.2 | 31.4 ± 6.8 | t = 6.94 | < 0.001 |
| ALP (U/L) | 89.3 ± 32.1 | 118.7 ± 45.3 | t = 4.65 | < 0.001 |
| GGT (U/L) | 45.8 ± 28.3 | 78.2 ± 41.6 | t = 5.87 | < 0.001 |
| Coagulation function | ||||
| PT (second) | 12.8 ± 1.6 | 16.4 ± 3.2 | t = 8.76 | < 0.001 |
| APTT (second) | 28.4 ± 4.2 | 38.7 ± 8.1 | t = 9.34 | < 0.001 |
| INR | 1.08 ± 0.14 | 1.42 ± 0.28 | t = 9.12 | < 0.001 |
| Inflammatory indicators | ||||
| CRP (mg/L) | 23.6 ± 18.4 | 67.8 ± 32.5 | t = 9.87 | < 0.001 |
| PCT (ng/mL) | 0.34 ± 0.28 | 1.28 ± 0.84 | t = 8.45 | < 0.001 |
| WBC (× 109/L) | 11.2 ± 4.3 | 16.8 ± 6.7 | t = 6.12 | < 0.001 |
| NEU% (%) | 76.8 ± 9.2 | 85.4 ± 8.6 | t = 5.38 | < 0.001 |
| Other indicators | ||||
| Cr (μmol/L) | 78.3 ± 18.2 | 112.6 ± 35.4 | t = 7.23 | < 0.001 |
| BUN (mmol/L) | 5.8 ± 2.1 | 9.4 ± 3.8 | t = 6.89 | < 0.001 |
| LDH (U/L) | 287.4 ± 84.6 | 468.3 ± 156.2 | t = 8.76 | < 0.001 |
| CK (U/L) | 345.2 ± 168.4 | 612.8 ± 284.7 | t = 6.98 | < 0.001 |
All imaging data were independently reviewed and scored by 2 radiologists with more than 10 years of experience in abdominal imaging diagnosis. The inter-rater reliability ICC for imaging scoring was 0.87 (95%confidence interval: 0.82-0.91), indicating good inter-rater reliability. Imaging scoring results showed that all scores in the poor prognosis group were significantly higher than those in the good prognosis group (Table 3).
| Scoring system | Good prognosis group (n = 142) | Poor prognosis group (n = 44) | Statistical value | P value |
| WSES liver injury grading | ||||
| Grade I | 32 (22.5) | 2 (4.5) | χ2 = 28.47 | < 0.001 |
| Grade II | 45 (31.7) | 8 (18.2) | ||
| Grade III | 38 (26.8) | 12 (27.3) | ||
| Grade IV | 21 (14.8) | 15 (34.1) | ||
| Grade V | 6 (4.2) | 6 (13.6) | ||
| Grade VI | 0 (0) | 1 (2.3) | ||
| HIS score, mean ± SD | 2.4 ± 1.1 | 3.8 ± 1.3 | t = 6.98 | < 0.001 |
| LTSS score, mean ± SD | ||||
| Liver injury extent score | 1.2 ± 0.8 | 2.1 ± 0.9 | t = 6.12 | < 0.001 |
| Hematoma size score | 0.8 ± 0.7 | 1.4 ± 0.6 | t = 5.23 | < 0.001 |
| Intraperitoneal bleeding score | 1.1 ± 0.9 | 2.3 ± 0.8 | t = 7.86 | < 0.001 |
| Active bleeding score | 0.6 ± 0.7 | 1.5 ± 0.7 | t = 7.41 | < 0.001 |
| LTSS total score | 3.7 ± 2.1 | 7.3 ± 2.0 | t = 9.87 | < 0.001 |
The distribution of treatment plans between the two groups is shown in Table 4. The good prognosis group was mainly treated conservatively, while the poor prognosis group was mainly treated surgically, with a statistically significant difference (χ2 = 25.34, P < 0.001) (Table 4).
| Treatment plan | Good prognosis group (n = 142) | Poor prognosis group (n = 44) | χ2 | P value |
| Conservative treatment | 98 (69.0) | 8 (18.2) | 25.34 | < 0.001 |
| Surgical treatment | 44 (31.0) | 36 (81.8) | - | - |
| Liver repair | 28 (19.7) | 18 (40.9) | 7.82 | 0.020 |
| Partial hepatectomy | 12 (8.5) | 12 (27.3) | 9.15 | 0.002 |
| Hepatic artery embolization | 4 (2.8) | 6 (13.6) | 6.87 | 0.009 |
Among the 44 patients in the poor prognosis group, adverse events included: Death in 12 cases (27.3%), need for emergency surgical intervention in 23 cases (52.3%), serious complications in 16 cases (36.4%), and need for ICU treat
The 6-month follow-up results showed that the 6-month survival rate in the good prognosis group was 100% (142/142). In the poor prognosis group, 12 cases died within 30 days, and among the remaining 32 patients, 2 cases died during the 6-month follow-up period. Therefore, the 6-month survival rate in the poor prognosis group was 68.2% (30/44), with a statistically significant difference between the two groups (χ2 = 42.86, P < 0.001).
Univariate analysis was used to screen factors related to prognosis, with variables having P < 0.1 as candidate variables, among which indicators with P < 0.05 showing statistical significance (Table 5). A total of 28 variables met the inclusion criteria for multivariate analysis with P < 0.1.
| Variable | β | SE | Wald χ2 | P value | OR value | 95%CI |
| Baseline characteristics | ||||||
| Age | 0.031 | 0.010 | 9.63 | 0.002 | 1.032 | 1.012-1.053 |
| Vital signs | ||||||
| Systolic BP | -0.034 | 0.007 | 23.47 | < 0.001 | 0.967 | 0.953-0.981 |
| Diastolic BP | -0.047 | 0.014 | 11.29 | 0.001 | 0.954 | 0.928-0.981 |
| Heart rate | 0.056 | 0.009 | 38.45 | < 0.001 | 1.058 | 1.038-1.078 |
| Respiratory rate | 0.211 | 0.044 | 23.12 | < 0.001 | 1.236 | 1.134-1.347 |
| Body temperature | -0.793 | 0.243 | 10.64 | 0.001 | 0.453 | 0.281-0.730 |
| Scoring systems | ||||||
| GCS score | -0.361 | 0.082 | 19.41 | < 0.001 | 0.698 | 0.593-0.821 |
| ISS score | 0.171 | 0.031 | 30.45 | < 0.001 | 1.187 | 1.118-1.260 |
| Serum biochemical indicators | ||||||
| ALT | 0.008 | 0.002 | 16.84 | < 0.001 | 1.008 | 1.005-1.011 |
| AST | 0.009 | 0.002 | 20.25 | < 0.001 | 1.009 | 1.006-1.012 |
| TBIL | 0.093 | 0.017 | 29.78 | < 0.001 | 1.098 | 1.061-1.136 |
| DBIL | 0.136 | 0.029 | 22.04 | < 0.001 | 1.146 | 1.084-1.212 |
| ALB | -0.181 | 0.037 | 24.01 | < 0.001 | 0.834 | 0.777-0.896 |
| ALP | 0.018 | 0.005 | 12.96 | < 0.001 | 1.018 | 1.008-1.028 |
| GGT | 0.025 | 0.005 | 25.00 | < 0.001 | 1.025 | 1.014-1.036 |
| PT | 0.563 | 0.095 | 35.12 | < 0.001 | 1.756 | 1.458-2.115 |
| APTT | 0.181 | 0.029 | 39.07 | < 0.001 | 1.198 | 1.132-1.269 |
| INR | 2.847 | 0.624 | 20.84 | < 0.001 | 17.238 | 5.081-58.484 |
| CRP | 0.051 | 0.008 | 40.64 | < 0.001 | 1.052 | 1.036-1.068 |
| PCT | 2.109 | 0.493 | 18.31 | < 0.001 | 8.245 | 3.142-21.654 |
| WBC | 0.145 | 0.031 | 21.84 | < 0.001 | 1.156 | 1.087-1.229 |
| NEU% | 0.093 | 0.020 | 21.56 | < 0.001 | 1.098 | 1.055-1.143 |
| Cr | 0.030 | 0.005 | 36.00 | < 0.001 | 1.031 | 1.020-1.042 |
| BUN | 0.261 | 0.053 | 24.24 | < 0.001 | 1.298 | 1.168-1.441 |
| LDH | 0.008 | 0.001 | 64.00 | < 0.001 | 1.008 | 1.006-1.010 |
| CK | 0.003 | 0.0005 | 36.00 | < 0.001 | 1.003 | 1.002-1.004 |
| Imaging scores | ||||||
| HIS score | 0.758 | 0.158 | 23.04 | < 0.001 | 2.134 | 1.562-2.915 |
| LTSS total score | 0.629 | 0.084 | 56.07 | < 0.001 | 1.876 | 1.587-2.215 |
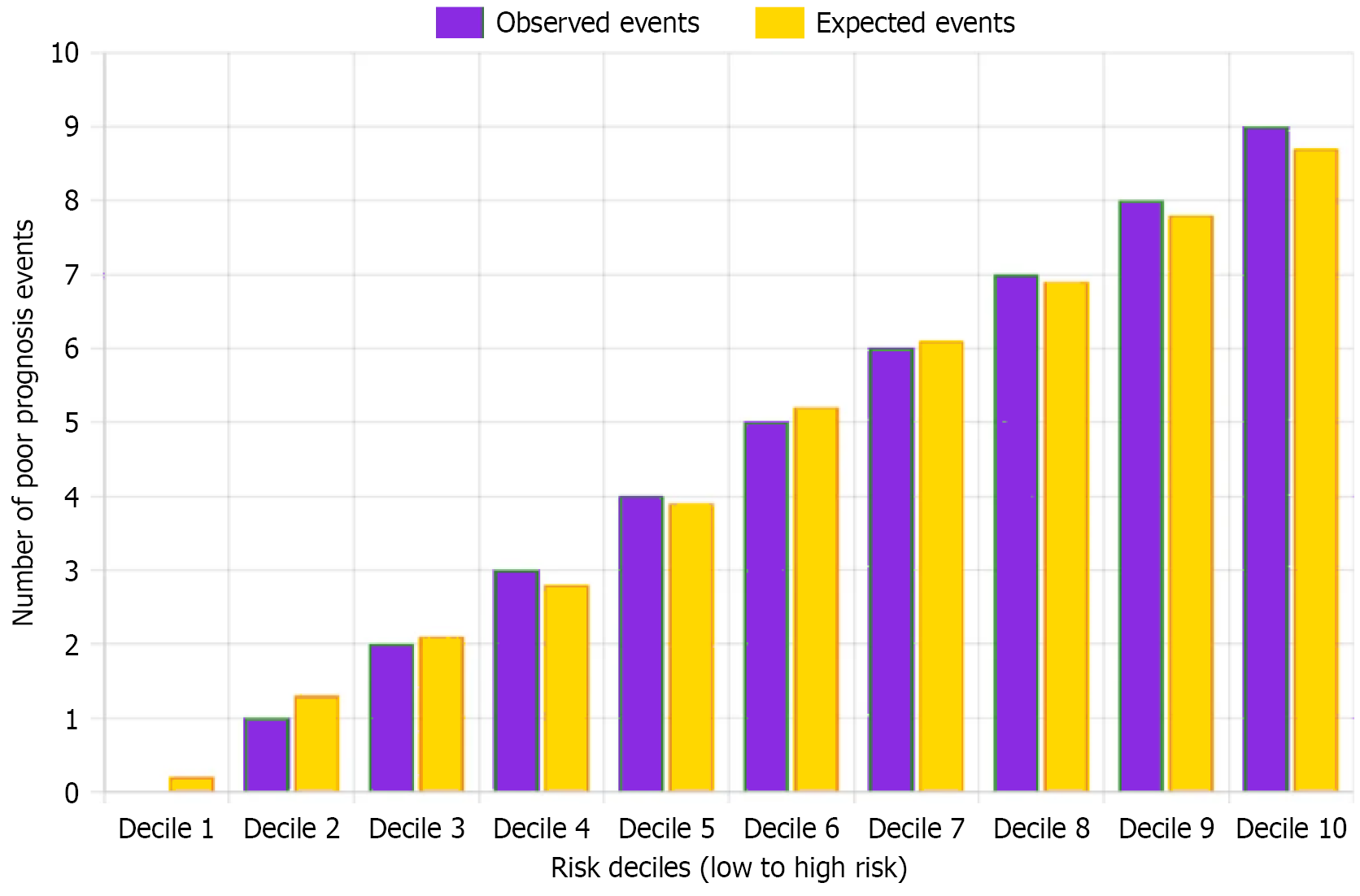
Variables with P < 0.1 in univariate analysis were included in multivariate logistic regression analysis using forward stepwise method. Results showed that AST, INR, CRP, GCS score, and ICC total score were independent predictors of traumatic liver rupture prognosis (Table 6). The Hosmer-Lemeshow goodness-of-fit test for the model was χ2 = 6.42 (P = 0.601), suggesting good model fit (Figure 1).
| Variable | β | SE | Wald χ2 | P value | OR value | 95%CI |
| AST (U/L) | 0.008 | 0.002 | 12.34 | < 0.001 | 1.008 | 1.003-1.013 |
| INR | 2.534 | 0.687 | 13.62 | < 0.001 | 12.596 | 3.281-48.347 |
| CRP (mg/L) | 0.023 | 0.006 | 14.78 | < 0.001 | 1.023 | 1.011-1.036 |
| GCS score | -0.394 | 0.128 | 9.48 | 0.002 | 0.675 | 0.525-0.867 |
| LTSS total score | 0.542 | 0.134 | 16.37 | < 0.001 | 1.720 | 1.322-2.238 |
| Constant | -2.463 | 1.234 | 3.98 | 0.046 | 0.085 | - |
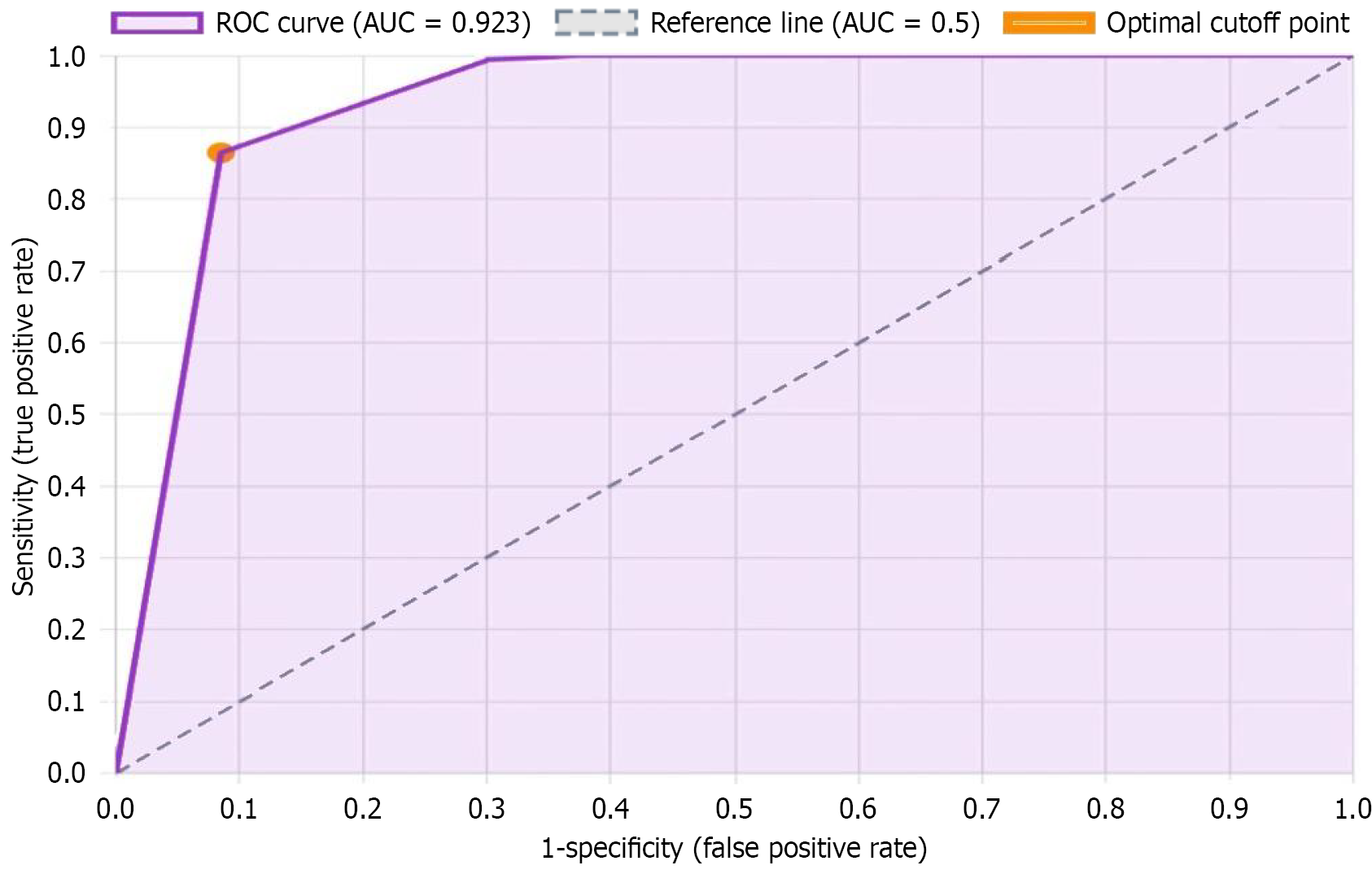
Based on multivariate analysis results, a prediction model was established: Logit(P) = -2.463 + 0.008 × AST + 2.534 × INR + 0.023 × CRP - 0.394 × GCS + 0.542 × LTSS receiver operating characteristic curve analysis showed that the prediction model had an AUC of 0.923 (95% confidence interval: 0.881-0.965), sensitivity of 86.4%, specificity of 91.5%, positive predictive value of 76.0%, negative predictive value of 95.6%, and Youden index of 0.779 (Figure 2).
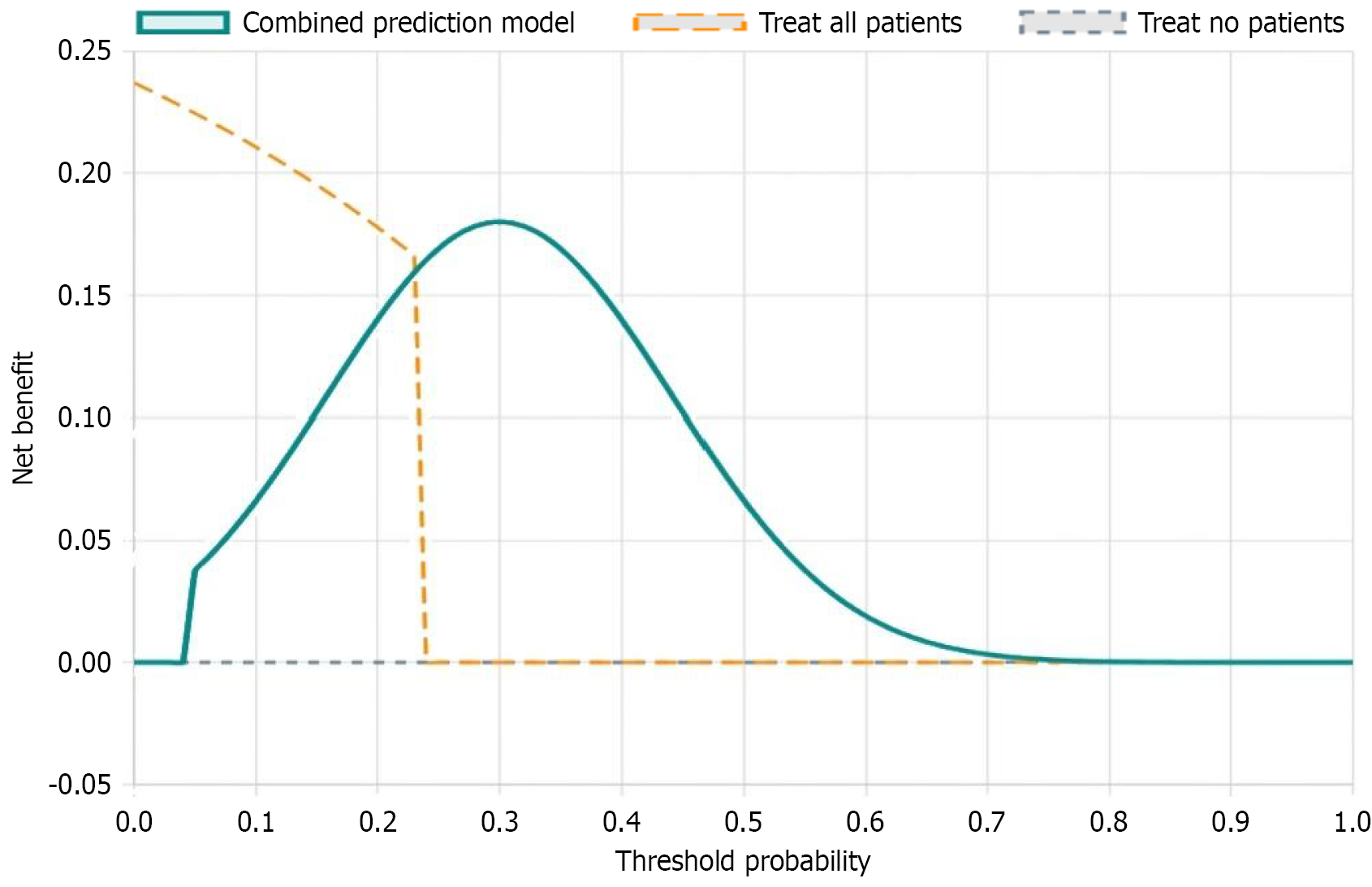
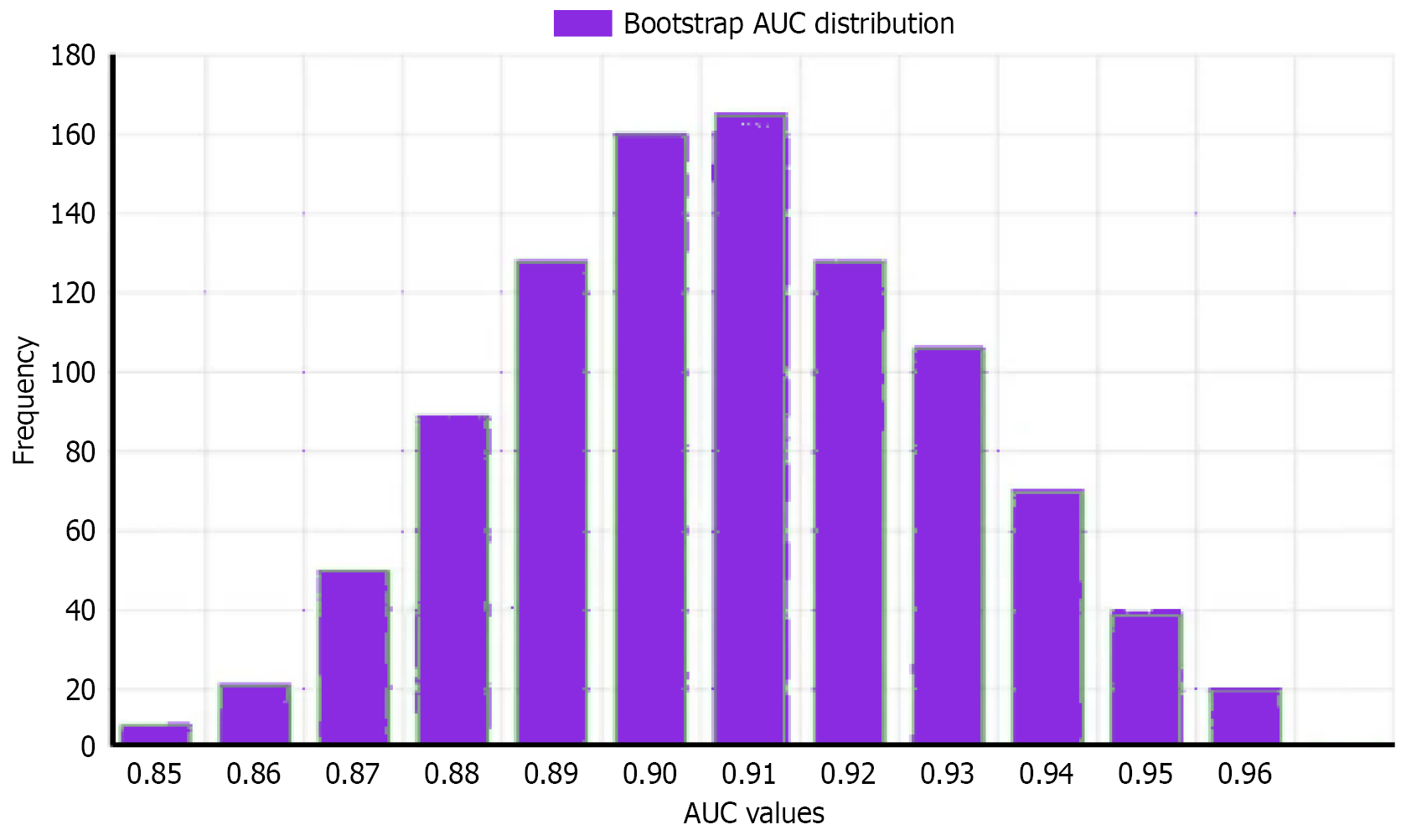
DCA showed that when the threshold probability was between 0.1-0.8, this prediction model had higher clinical net benefit compared to traditional scoring methods (Figure 3). Bootstrap resampling 1000 times for internal validation showed that the corrected AUC was 0.912, indicating that the model had good internal consistency (Figure 4).
Comparison results of predictive performance using serum biochemical indicators alone, imaging scores alone, and combined application (Table 7).
| Predictive indicator/model | AUC | 95%CI | Sensitivity (%) | Specificity (%) | Youden index |
| Single indicators | |||||
| AST | 0.798 | 0.728-0.868 | 75.0 | 78.2 | 0.532 |
| INR | 0.823 | 0.758-0.888 | 79.5 | 80.3 | 0.598 |
| CRP | 0.836 | 0.774-0.898 | 81.8 | 78.9 | 0.607 |
| GCS score | 0.742 | 0.665-0.819 | 72.7 | 71.8 | 0.445 |
| LTSS total score | 0.845 | 0.786-0.904 | 84.1 | 76.8 | 0.609 |
| Combined models | |||||
| Serum biochemical combined | 0.887 | 0.836-0.938 | 84.1 | 85.2 | 0.693 |
| Serum biochemical + imaging combined | 0.923 | 0.881-0.965 | 86.4 | 91.5 | 0.779 |
DeLong test was used to compare the predictive performance of each model. The combined model of serum bio
This study analyzed clinical data from 186 patients with traumatic liver rupture and established a comprehensive prediction model combining serum biochemical indicators with imaging scores. The study found that this model has good predictive performance for predicting 30-day poor prognosis in patients, providing important scientific evidence for early identification of high-risk patients in clinical practice.
This study found that AST, INR, CRP, GCS score, and LTSS total score are independent predictors of prognosis in patients with traumatic liver rupture. The combined prediction model based on these five indicators achieved an AUC of 0.923, significantly superior to the predictive performance of any single indicator alone. This finding has important clinical significance. First, this model covers multiple dimensions reflecting liver injury severity, coagulation function, inflammatory response, consciousness status, and anatomical injury, enabling comprehensive assessment of the patient's pathophysiological status[11]. Second, all indicators in the model are routine clinical examination items that are easily obtainable and cost-effective, with good clinical operability[12].
AST, as a sensitive indicator of hepatocyte injury, rapidly increases after liver trauma, and its level is closely related to the degree of hepatocyte necrosis[13]. Previous studies have shown that the degree of AST elevation is significantly related to the severity and prognosis of traumatic liver rupture, which is consistent with the results of this study[14]. INR reflects coagulation status. Patients with traumatic liver rupture often experience coagulation dysfunction due to factors such as blood loss and liver function impairment. Elevated INR indicates deteriorating coagulation function and is an important warning signal for poor prognosis[15].
This study found that CRP is an independent predictor, suggesting that inflammatory response plays an important role in the prognosis of traumatic liver rupture. After trauma, the body initiates systemic inflammatory response syndrome, and CRP, as an acute-phase reactant protein, reflects the severity of the body’s inflammatory response[16]. Studies have shown that excessive inflammatory response can lead to microcirculatory dysfunction, tissue hypoxia, and multiple organ dysfunction, which are important causes of death in trauma patients[17]. Therefore, early monitoring of CRP levels helps identify patients with high inflammatory response and timely implementation of anti-inflammatory treatment measures.
Some studies have reported the value of PCT in trauma prognosis prediction, but in the multivariate analysis of this study, PCT did not enter the final model. This may be related to PCT mainly reflecting bacterial infection, while early trauma is primarily sterile inflammatory response[18]. In comparison, CRP can better reflect the inflammatory status in early trauma and therefore has stronger predictive value.
GCS score, as a classic tool for assessing consciousness status, showed good predictive value in this study. This finding is consistent with multiple study results, suggesting that the degree of nervous system injury is closely related to the overall prognosis of trauma patients[19]. Decreased GCS score not only reflects the severity of craniocerebral injury but may also indicate deterioration of overall condition, such as shock and hypoxia[20]. Additionally, changes in conscious
LTSS, as an imaging scoring system specifically for evaluating liver trauma, showed good predictive performance in this study. This scoring system comprehensively considers multiple dimensions such as liver injury extent, hematoma size, intraperitoneal bleeding volume, and active bleeding, enabling objective quantification of anatomical liver injury severity[21]. Compared with traditional World Society of Emergency Surgery grading and HIS scoring, LTSS scoring is more detailed and quantified, thus having stronger predictive value[22].
It is worth noting that the inter-rater reliability ICC for imaging scoring in this study was 0.87, indicating good reproducibility of scoring. This has important significance for clinical promotion and application of the scoring system, suggesting that imaging physicians with appropriate training can obtain consistent scoring results[23].
The most important finding of this study is that the comprehensive model combining serum biochemical indicators with imaging scores is significantly superior to using any single indicator alone. This result aligns with the concept of modern precision medicine, which improves disease prediction accuracy by integrating multi-dimensional information[24]. Serum biochemical indicators can reflect the body’s physiological functional status and metabolic changes, while imaging scores objectively assess anatomical injury severity, and both have good complementarity[25].
DCA showed that when the threshold probability was between 0.1-0.8, the combined model had higher clinical net benefit, indicating that this model has practical value in clinical decision-making. When the prediction probability exceeds a certain threshold, clinicians can consider adopting more aggressive treatment measures, such as close monitoring and preventive intervention[26].
Previous research on prognostic prediction of traumatic liver rupture has mainly focused on single indicators or traditional scoring systems. Some studies have reported the relationship between liver enzyme levels and prognosis, but with limited predictive performance[27]. Other studies have explored the predictive value of coagulation function indicators but lacked combined analysis with imaging indicators[28]. This study is the first to systematically integrate serum biochemical indicators and imaging scores, establishing a more comprehensive prediction model.
Compared with similar international studies, this study has a relatively large sample size, more comprehensive included indicators, and improved predictive performance. A similar study reported an AUC of 0.856, which is lower than the 0.923 in this study[29]. This may be related to differences in study population characteristics, included indicators, and modeling methods.
The prediction model established in this study has good clinical application prospects. First, all indicators in the model are routine clinical examination items that do not require additional special testing, with good accessibility[30]. Second, the model has high sensitivity (86.4%) and specificity (91.5%), effectively identifying high-risk patients and avoiding missed diagnosis and misdiagnosis. Third, DCA shows that the model has good clinical net benefit and can provide quantitative support for clinical decision-making.
In practical application, clinicians can calculate prediction probability based on the patient's AST, INR, CRP, GCS score, and LTSS total score. When the probability exceeds a certain threshold, more aggressive treatment measures should be considered, such as ICU monitoring and preventive surgical intervention. This helps improve the prognosis of high-risk patients and reduce mortality[31].
This study has some limitations that need to be recognized. First, as a single-center retrospective study, there is a possibility of selection bias, and the extrapolability of study results may be limited. Second, although Bootstrap internal validation was performed, external cohort validation is lacking, and the universality of the model needs further verification[32]. Third, the study only included short-term prognosis within 30 days and did not evaluate long-term prognostic indicators. While the 30-day endpoint captures the critical early period when most major adverse events occur and clinical decision-making has the most profound impact, future long-term follow-up studies are needed to assess functional recovery, quality of life, and late-onset complications. Fourth, some potential predictive factors such as genetic polymorphisms and nutritional status were not included in the analysis. Additionally, the exclusion of patients with pre-existing liver disease, while methodologically necessary to isolate the independent effect of traumatic injury and avoid confounding from chronic hepatic dysfunction, limits the generalizability of our model to patients with underlying liver conditions. Future studies should develop separate or modified prediction models for this important patient subgroup, as their baseline hepatic reserve and recovery trajectories may differ substantially.
Future research should address several important directions. First, multi-center prospective validation studies are essential to confirm the external validity of our model. Second, dynamic biomarker monitoring through serial mea
In conclusion, this study successfully established a prognostic prediction model for traumatic liver rupture combining serum biochemical indicators with imaging scores. This model has good predictive performance and clinical practicality. AST, INR, CRP, GCS score, and LTSS total score are independent predictors of prognosis, and their combined application is significantly superior to single indicators. This model provides scientific evidence for early identification of high-risk patients and formulation of individualized treatment plans in clinical practice, and is expected to improve the prognosis of patients with traumatic liver rupture. However, multi-center prospective studies still need to be conducted for external validation to ensure the universality and reliability of the model.
| 1. | Kinnunen J, Satopää J, Niemelä M, Putaala J. Coagulopathy and its effect on treatment and mortality in patients with traumatic intracranial hemorrhage. Acta Neurochir (Wien). 2021;163:1391-1401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 2. | Alanezi T, Altoijry A, Alanazi A, Aljofan Z, Altuwaijri T, Iqbal K, AlSheikh S, Molla N, Altuwaijri M, Aloraini A, Altuwaijri F, Aldossary MY. Management and Outcomes of Traumatic Liver Injury: A Retrospective Analysis from a Tertiary Care Center Experience. Healthcare (Basel). 2024;12:131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 3. | Chun M, Zhang Y, Becnel C, Brown T, Hussein M, Toraih E, Taghavi S, Guidry C, Duchesne J, Schroll R, McGrew P. New Injury Severity Score and Trauma Injury Severity Score are superior in predicting trauma mortality. J Trauma Acute Care Surg. 2022;92:528-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 4. | Thakur S, Kumar V, Das R, Sharma V, Mehta DK. Biomarkers of Hepatic Toxicity: An Overview. Curr Ther Res Clin Exp. 2024;100:100737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 86] [Article Influence: 43.0] [Reference Citation Analysis (5)] |
| 5. | Dixe de Oliveira Santo I, Sailer A, Solomon N, Borse R, Cavallo J, Teitelbaum J, Chong S, Roberge EA, Revzin MV. Grading Abdominal Trauma: Changes in and Implications of the Revised 2018 AAST-OIS for the Spleen, Liver, and Kidney. Radiographics. 2023;43:e230040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 6. | Zhao K, Mabud TS, Patel N, Bernstein MP, McDermott M, Bryk H, Taslakian B. Predictors of need for endovascular intervention in hepatic trauma. Abdom Radiol (NY). 2023;48:1131-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 7. | Dana J, Venkatasamy A, Saviano A, Lupberger J, Hoshida Y, Vilgrain V, Nahon P, Reinhold C, Gallix B, Baumert TF. Conventional and artificial intelligence-based imaging for biomarker discovery in chronic liver disease. Hepatol Int. 2022;16:509-522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 8. | Shin H, Hur MH, Song BG, Park SY, Kim GA, Choi G, Nam JY, Kim MA, Park Y, Ko Y, Park J, Lee HA, Chung SW, Choi NR, Park MK, Lee YB, Sinn DH, Kim SU, Kim HY, Kim JM, Park SJ, Lee HC, Lee DH, Chung JW, Kim YJ, Yoon JH, Lee JH. AI model using CT-based imaging biomarkers to predict hepatocellular carcinoma in patients with chronic hepatitis B. J Hepatol. 2025;82:1080-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 9. | Garcia MF, Gomes RT, Pugliesi EC, Santos JPVD, Martino FD, Gomes KHV, Pasquareli DRG, Lenza RDM. Comparação entre o Injury Severity Score (ISS) e o New Injury Severity Score (NISS) na predição da mortalidade de traumas torácicos atendidos em um hospital terciário. Rev Col Bras Cir. 2024;51:e20243652. [DOI] [Full Text] |
| 10. | Coccolini F, Coimbra R, Ordonez C, Kluger Y, Vega F, Moore EE, Biffl W, Peitzman A, Horer T, Abu-Zidan FM, Sartelli M, Fraga GP, Cicuttin E, Ansaloni L, Parra MW, Millán M, DeAngelis N, Inaba K, Velmahos G, Maier R, Khokha V, Sakakushev B, Augustin G, di Saverio S, Pikoulis E, Chirica M, Reva V, Leppaniemi A, Manchev V, Chiarugi M, Damaskos D, Weber D, Parry N, Demetrashvili Z, Civil I, Napolitano L, Corbella D, Catena F; WSES expert panel. Liver trauma: WSES 2020 guidelines. World J Emerg Surg. 2020;15:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 252] [Cited by in RCA: 184] [Article Influence: 30.7] [Reference Citation Analysis (1)] |
| 11. | Liu Y, Sun R, Jiang H, Liang G, Huang Z, Qi L, Lu J. Development and validation of a predictive model for in-hospital mortality in patients with sepsis-associated liver injury. Ann Transl Med. 2022;10:997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 12. | Farzaneh N, Stein EB, Soroushmehr R, Gryak J, Najarian K. A deep learning framework for automated detection and quantitative assessment of liver trauma. BMC Med Imaging. 2022;22:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 13. | Field X, Crichton J, Kong V, Ko J, Laing G, Bruce J, Clarke D. AAST grade of liver injury is not the single most important consideration in decision making for liver trauma. Injury. 2024;55:111526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 14. | Beltzer C, Imach S, Wafaisade A, Lefering R, Kölbel B; TraumaRegister DGU. Use of angioembolization, treatment modalities and mortality in association with blunt liver trauma in Germany - a data analysis of the TraumaRegister DGU®. Langenbecks Arch Surg. 2023;409:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Kumagai K, Mawatari S, Moriuchi A, Oda K, Takikawa Y, Kato N, Oda S, Inoue K, Terai S, Genda T, Shimizu M, Sakaida I, Mochida S, Ido A. Early-phase prothrombin time-international normalized ratio in acute liver injury indicates the timing of therapeutic intervention and predicts prognostic improvement. Hepatol Res. 2023;53:160-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 16. | Wu W, Zhang D, Jin T, Lu T, Zhou F. Progress in the study of biomarkers for early prediction of systemic inflammatory response syndrome after percutaneous nephrolithotomy. Front Immunol. 2023;14:1142346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 17. | Li R, Ye JJ, Gan L, Zhang M, Sun D, Li Y, Wang T, Chang P. Traumatic inflammatory response: pathophysiological role and clinical value of cytokines. Eur J Trauma Emerg Surg. 2024;50:1313-1330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 18. | Bakhtiar A, Haider Kazmi SJ, Asghar MS, Khurshaidi MN, Mazhar S, Khan NA, Ahmed N, Yasmin F, Yaseen R, Hassan M. Accuracy of Procalcitonin Levels for Diagnosis of Culture-Positive Sepsis in Critically Ill Trauma Patients: A Retrospective Analysis. Cureus. 2021;13:e12988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 19. | Schucht JE, Rakhit S, Smith MC, Han JH, Brown JB, Grigorian A, Gondek SP, Smith JW, Patel MB, Maiga AW. Beyond Glasgow Coma Scale: Prehospital prediction of traumatic brain injury. Surgery. 2025;179:108893. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Anestis DM, Marinos K, Tsitsopoulos PP. Comparison of the prognostic validity of three simplified consciousness assessment scales with the Glasgow Coma Scale. Eur J Trauma Emerg Surg. 2023;49:2193-2202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 21. | Huang S, Zhou Z, Qian X, Li D, Guo W, Dai Y. Automated quantitative assessment of pediatric blunt hepatic trauma by deep learning-based CT volumetry. Eur J Med Res. 2022;27:305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 22. | AlShahwan N, Aldeligan SH, Althunayan ST, Alkodari A, Bin Manee M, Albassam FA, Aloraini A, Alburakan A, Mashbari H, AlKanhal A, Nouh T. Outcomes of Traumatic Liver Injuries at a Level-One Tertiary Trauma Center in Saudi Arabia: A 10-Year Experience. Life (Basel). 2025;15:1138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 23. | Akinci D'Antonoli T, Cavallo AU, Vernuccio F, Stanzione A, Klontzas ME, Cannella R, Ugga L, Baran A, Fanni SC, Petrash E, Ambrosini I, Cappellini LA, van Ooijen P, Kotter E, Pinto Dos Santos D, Cuocolo R; EuSoMII Radiomics Auditing Group. Reproducibility of radiomics quality score: an intra- and inter-rater reliability study. Eur Radiol. 2024;34:2791-2804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 24. | Bicciato G, Arnold M, Gebhardt A, Katan M. Precision medicine in secondary prevention of ischemic stroke: how may blood-based biomarkers help in clinical routine? An expert opinion. Curr Opin Neurol. 2022;35:45-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 25. | Kiebish MA, Tekumalla P, Ravipaty S, Dobi A, Srivastava S, Wu W, Patil S, Friss T, Klotz A, Srinivasan A, Cullen J, Rosner IL, Ali A, Laszlo S, Petrovic M, Fleshner N, Garren J, Miller G, Mahaveer Chand N, Rodrigues LO, Granger E, Kellogg MD, Luan S, Diamandis E, Akmaev VR, Sarangarajan R, Bountra C, Freedland SJ, McLeod DG, Narain NR. Clinical utility of a serum biomarker panel in distinguishing prostate cancer from benign prostate hyperplasia. Sci Rep. 2021;11:15052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 26. | Vickers AJ, Van Claster B, Wynants L, Steyerberg EW. Decision curve analysis: confidence intervals and hypothesis testing for net benefit. Diagn Progn Res. 2023;7:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 27. | Shrestha A, Neupane HC, Tamrakar KK, Bhattarai A, Katwal G. Role of liver enzymes in patients with blunt abdominal trauma to diagnose liver injury. Int J Emerg Med. 2021;14:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 28. | Wolf EB, Plante M, Chirila RM. If a patient has cirrhosis, should I correct coagulation abnormalities before a minor invasive procedure? Cleve Clin J Med. 2023;90:541-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 29. | Park JO. Update of systemic chemotherapy & immunotherapy for bile duct cancers. Ann Hepatobiliary Pancreat Surg. 2022;26:S35-S35. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 30. | Efthimiou O, Seo M, Chalkou K, Debray T, Egger M, Salanti G. Developing clinical prediction models: a step-by-step guide. BMJ. 2024;386:e078276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 265] [Cited by in RCA: 231] [Article Influence: 115.5] [Reference Citation Analysis (0)] |
| 31. | Sutton RT, Pincock D, Baumgart DC, Sadowski DC, Fedorak RN, Kroeker KI. An overview of clinical decision support systems: benefits, risks, and strategies for success. NPJ Digit Med. 2020;3:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2192] [Cited by in RCA: 1444] [Article Influence: 240.7] [Reference Citation Analysis (0)] |
| 32. | Collins GS, Dhiman P, Ma J, Schlussel MM, Archer L, Van Calster B, Harrell FE Jr, Martin GP, Moons KGM, van Smeden M, Sperrin M, Bullock GS, Riley RD. Evaluation of clinical prediction models (part 1): from development to external validation. BMJ. 2024;384:e074819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 435] [Cited by in RCA: 357] [Article Influence: 178.5] [Reference Citation Analysis (5)] |