Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.112405
Revised: October 28, 2025
Accepted: December 12, 2025
Published online: March 27, 2026
Processing time: 196 Days and 4.7 Hours
Post-endoscopic retrograde cholangiopancreatography (ERCP) acute pancreatitis (PEP) is a severe postoperative inflammatory complication following ERCP, characterized by rapid onset and potentially life-threatening systemic manifestations. The integrity of the intestinal mucosal barrier plays a pivotal role in preventing secondary infections and systemic inflammatory response syndrome in the postoperative period. Traditional Chinese medicine has shown promising therapeutic potential in postoperative management and mucosal barrier protec
To investigate the efficacy of Dachengqitang (DCQT) in ameliorating PEP through enhancement of intestinal mucosal barrier function and suppression of M1 macrophage polarization.
Bioinformatics analysis was performed on differential gene expression and functional enrichment using the GEO database entry GSE54774. Subsequently, in vivo and in vitro experiments were conducted, including sham ope
Differential analysis of GSE54774 identified 468 differentially expressed genes, with enrichment observed in blood microparticle, primary lysosome, and azurophil granule pathways. In vivo experiments demonstrated that compared to the PEP model group, DCQT treatment significantly enhanced the expression of intestinal mucosal barrier proteins ZO-1 and Occludin-1, reduced the population of CD86-positive pro-inflammatory macrophages, and promoted IL-10 secretion by M2 macrophages in the postoperative period. In vitro Caco-2 cell cultures showed that compared to the oxygen-glucose deprivation group, DCQT improved ZO-1 and Occludin-1 expression and enhanced mitochondrial membrane potential.
This study demonstrates that DCQT effectively enhances intestinal mucosal barrier function and reduces post
Core Tip: Post-endoscopic retrograde cholangiopancreatography (ERCP) acute pancreatitis is a serious complication involving intestinal mucosal barrier dysfunction and excessive inflammation. This study integrates bioinformatics analysis with in vivo and in vitro models to investigate the protective effects of Dachengqitang (DCQT). The results demonstrate that DCQT enhances intestinal barrier integrity by upregulating ZO-1 and Occludin-1, reduces M1 macrophage activation, and promotes M2 polarization with increased IL-10 expression. DCQT also improves mitochondrial membrane potential in epithelial cells. These findings highlight the therapeutic potential of DCQT in managing postoperative inflammatory responses following ERCP.
- Citation: Zheng L, Wang J, Xu MQ, Jiang W, Qian L, Ge Y, Feng MK, Zhu YM, Wang MJ, Sun SS, Liu CK, Wang XM, Zhang C. Dachengqitang mitigates endoscopic retrograde cholangiopancreatography-induced pancreatitis. World J Gastrointest Surg 2026; 18(3): 112405
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/112405.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.112405
Post-endoscopic retrograde cholangiopancreatography (ERCP) acute pancreatitis (PEP) constitutes one of the most sig
Impairment of intestinal barrier function represents a critical pathogenic mechanism in PEP development. Disruption of this barrier facilitates bacterial translocation and systemic endotoxemia, thereby perpetuating and amplifying the inflammatory response. Understanding these pathophysiological mechanisms is essential for developing targeted therapeutic strategies to mitigate PEP incidence and severity while optimizing perioperative management.
Intestinal barrier integrity serves as a critical defense mechanism against translocation of luminal pathogens and toxins, maintaining intestinal homeostasis and preventing systemic inflammation following ERCP. Given that barrier dys
Macrophages function as key effector cells in the post-procedural immune response, exhibiting phenotypic plasticity in response to environmental stimuli. M1-polarized macrophages, characterized by their proinflammatory profile, play a pivotal role in PEP pathogenesis through secretion of proinflammatory cytokines (IL-6, IL-1β, TNF-α) and chemokines that amplify tissue inflammation. Consequently, inhibiting M1 macrophage polarization represents a rational therapeutic target for PEP management[4,5].
Dachengqitang (DCQT), a classical Chinese herbal formulation documented in the "Shanghan Lun" (Treatise on Cold Damage Disorders), has been prescribed for gastrointestinal dysfunction and abdominal distension for over 1800 years. Contemporary pharmacological investigations have identified multiple mechanisms through which DCQT promotes gastrointestinal health: (1) Enhancement of gastrointestinal motility and suppression of bacterial translocation; (2) Attenuation of intestinal inflammation via modulation of NF-κB signaling; and (3) Preservation of intestinal barrier integrity through regulation of tight junction protein expression in experimental colitis models. These mechanistic insights provide a robust scientific foundation for investigating DCQT's therapeutic potential in protecting intestinal barrier function during PEP. The constituent herbs possess documented anti-inflammatory properties[6,7], and emerging evidence suggests that DCQT exhibits immunomodulatory and antioxidant activities relevant to post-procedural care. However, the precise molecular mechanisms underlying DCQT's protective effects on intestinal barrier function in PEP remain to be fully elucidated.
This study explores the potential of DCQT to mitigate postoperative intestinal mucosal damage in PEP. We propose that DCQT operates through a dual mechanism: Enhancing mitochondrial membrane potential to restore mucosal integrity, while curbing intestinal macrophage activation and associated inflammation, ultimately leading to reduced disease severity and improved recovery.
Performing differential gene analysis on the data sourced from the GEO database GSE54774[8,9].
Using data sourced from the GEO database (GSE54774), we conducted Gene Ontology and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses by applying R language packages. This process involved generating various visualizations, including bubble charts, circular charts, and bar charts[10,11].
With visualization of these networks done using Cytoscape 3.6.1. In order to investigate the biological processes were analyzed further within the STRING database.
The obstructive model of acute pancreatitis was developed by ligating the pancreatic duct where the gastric and duodenal lobes meet, while preserving the bile duct and its associated artery, as previously described and modified[12]. SAP model treated with DCQT (SAP + DCQT). The animals were fasted for 12 hours and deprived of water for 6 hours before surgery. The DCQT treatment group received a single gastric infusion of 10% DCQT solution (9.18 g/kg) 15 minutes before modeling, while the control group and SAP group received an equal amount of 0.9% sodium chloride solution. Anesthesia was induced by intraperitoneal injection of 10% chloral hydrate (300 mg/kg), followed by a midline abdominal incision. The bile duct and pancreatic duct opening at the duodenal papilla were closed with a non-damaging metal clip. A needle from a syringe was inserted into the pancreatic duct towards the duodenal opening, and 3.5% sodium taurocholate (0.1 mL/100 g body weight) was injected at a rate of 0.1 mL/min. The abdomen was closed 10 minutes later. In the control group, after opening the abdomen, the duodenum was turned over and the pancreas was palpated a few times before closing the abdomen. The survival of the rats was observed.
Macrophage depletion was performed according to the following references[13,14]. Four groups of mice were randomly assigned, including Sham + PBS group (Sham; n = 4), SAP group (n = 4), SAP + DCQT group (SAD; n = 4), and SAP + DCQT + Cls group (MMP; n = 4). Intraperitoneal administration of CIs or control liposome (PBS) with a 24-hour interval before the SAP procedure was implemented.
Caco-2 cells were sourced from the Cell Bank of the Chinese Academy of Sciences and randomly divided into four groups: The PBS group (n = 4), the oxygen-glucose deprivation (OGD) group (n = 4), and the OGD + DCQT group (n = 4).
The OGD model was selected based on its pathophysiological relevance to PEP. Severe acute pancreatitis, including PEP, is frequently accompanied by splanchnic hypoperfusion and intestinal ischemia due to systemic inflammatory response and microcirculatory dysfunction. Unlike single-cytokine models (TNF-α or IFN-γ alone), OGD simultaneously simulates the ischemic and metabolic stress conditions that occur in vivo during severe pancreatitis, providing a more comprehensive representation of the clinical scenario. Furthermore, this model serves as a complementary approach to our in vivo PEP model, allowing specific investigation of DCQT's direct protective effects on intestinal epithelial cells under stress conditions, independent of immune cell involvement.
The research employed immunofluorescence on sections from both frozen and paraffin-embedded tissues. Samples, either cells or tissues, were secured onto glass slides or within centrifuge tubes and fixed with a 4% paraformaldehyde solution to preserve their morphology and protein localization. The primary antibodies used were against CD86, ZO-1, and Occludin. After the appropriate incubation with the secondary antibodies, the samples were examined using a fluorescence microscope[15,16].
Mitochondrial membrane potential was assessed according to the protocol provided with the assay kit. The cells were first extensively rinsed and then incubated with the JC-1 reagent for 20 minutes at a temperature of 37 °C. Subsequently, the dual emission fluorescence of JC-1 was recorded using a RuoChuang imaging system[17].
Analysis of the data was performed using GraphPad Prism 9.0. We assessed data distribution normality with the Kolmogorov-Smirnov test. For comparing multiple groups, a one-way ANOVA test followed by Tukey's HSD test was conducted. Data are expressed as the mean ± SD. A threshold for statistical significance was set at P < 0.05.
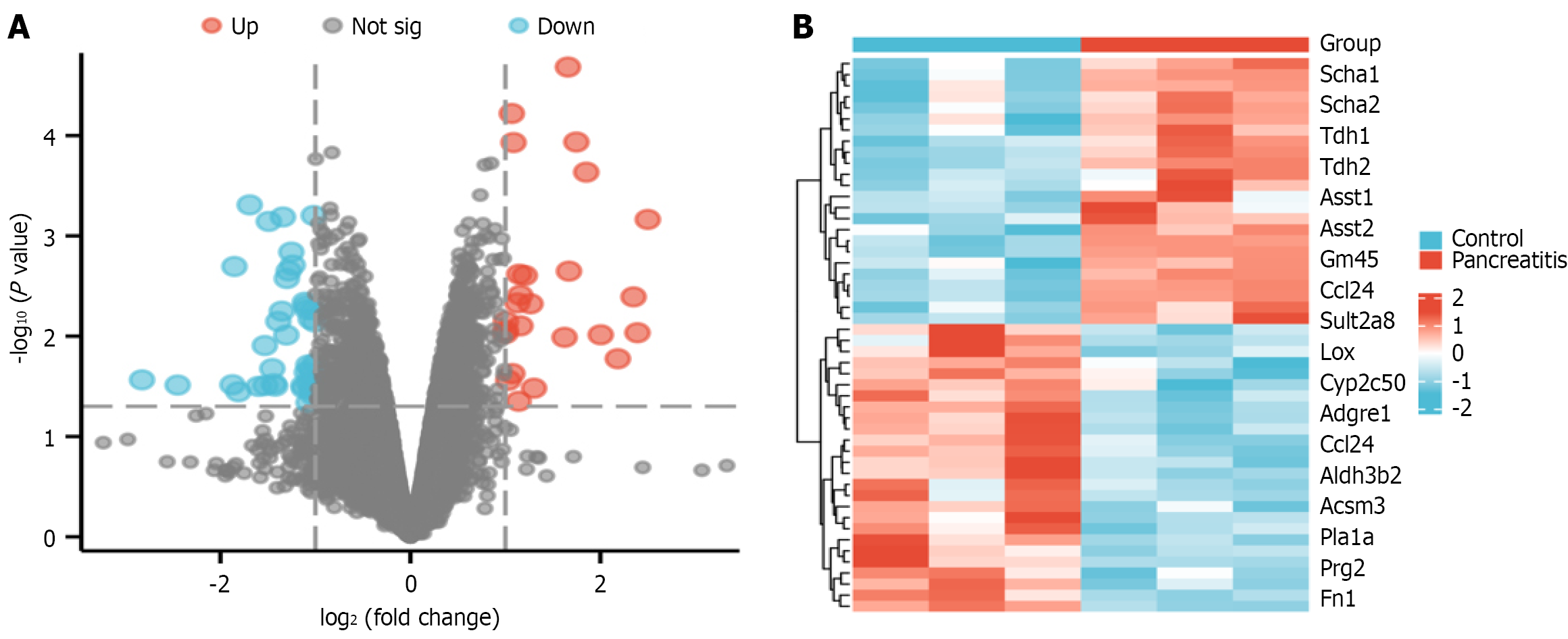
Examine the figures for representations of the heatmap and volcano plot highlighting differential gene expression. A sum of 468 genes were detected using the specified criteria. Notably, BMP4 exhibits increased expression in AP samples as depicted in Figure 1.
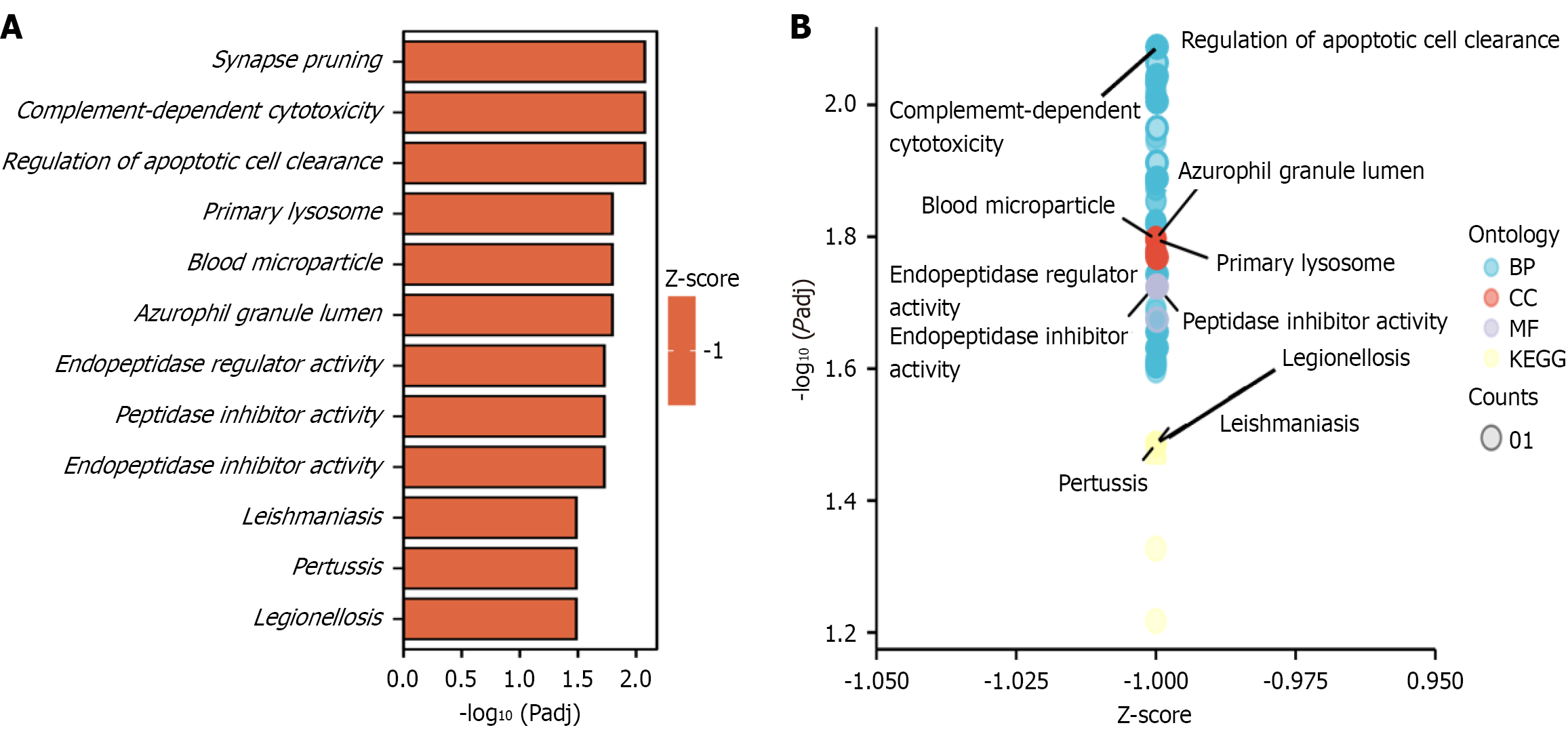
Comparison of inflammatory markers in control and experimental group. most of these factors are associated with blood microparticles (blood mps), primary lysosomes, azurophil granules, and the vacuolar lumen (Figure 2).
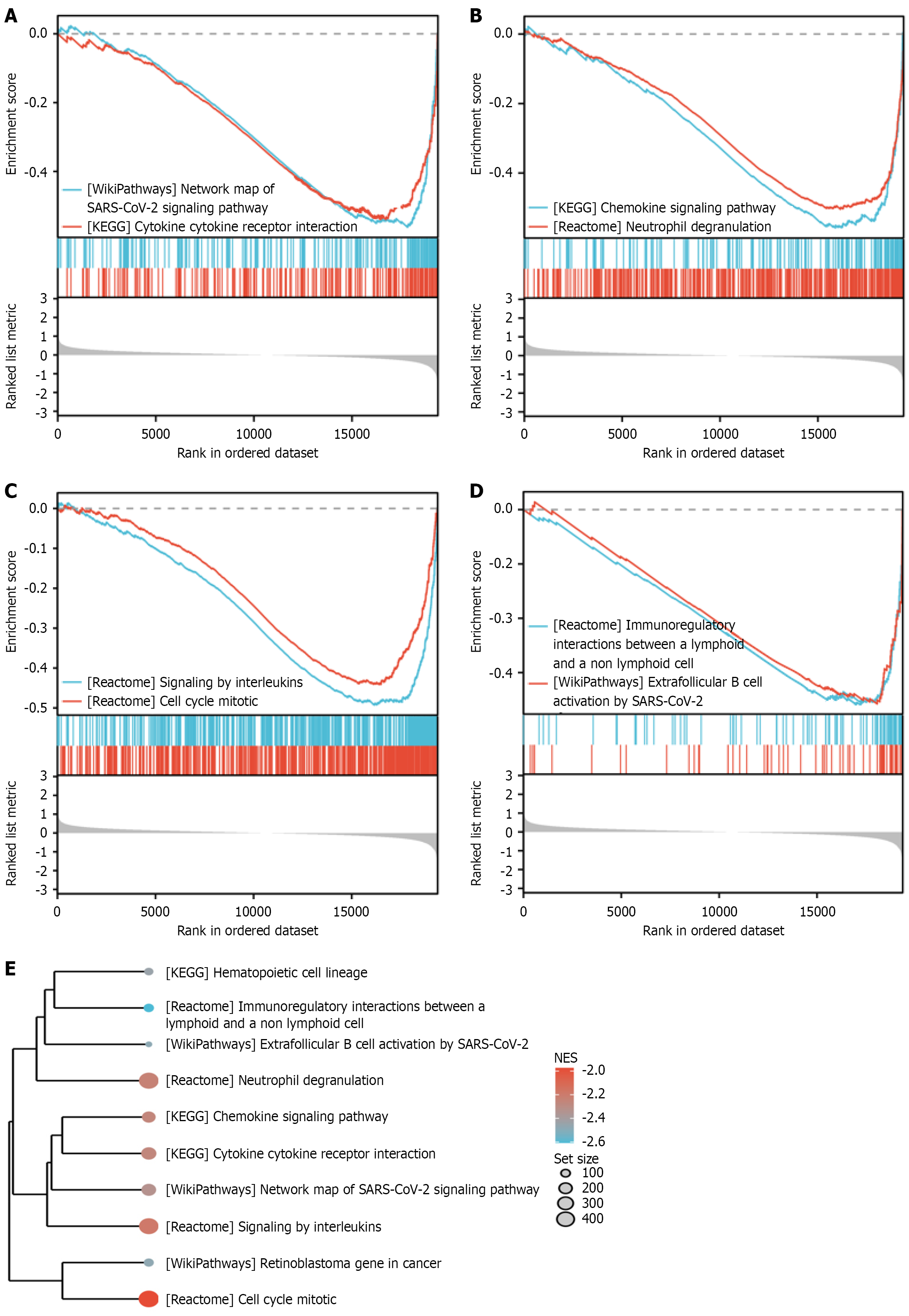
The enriched genes showed significant involvement in various biological processes, including interactions between lymphoid and non-lymphoid cells, reactome neutrophil degranulation, reactome signaling by interleukins, reactome cell cycle mitotic, wp retinoblastoma gene in cancer, KEGG hematopoietic cell lineage (Figure 3).
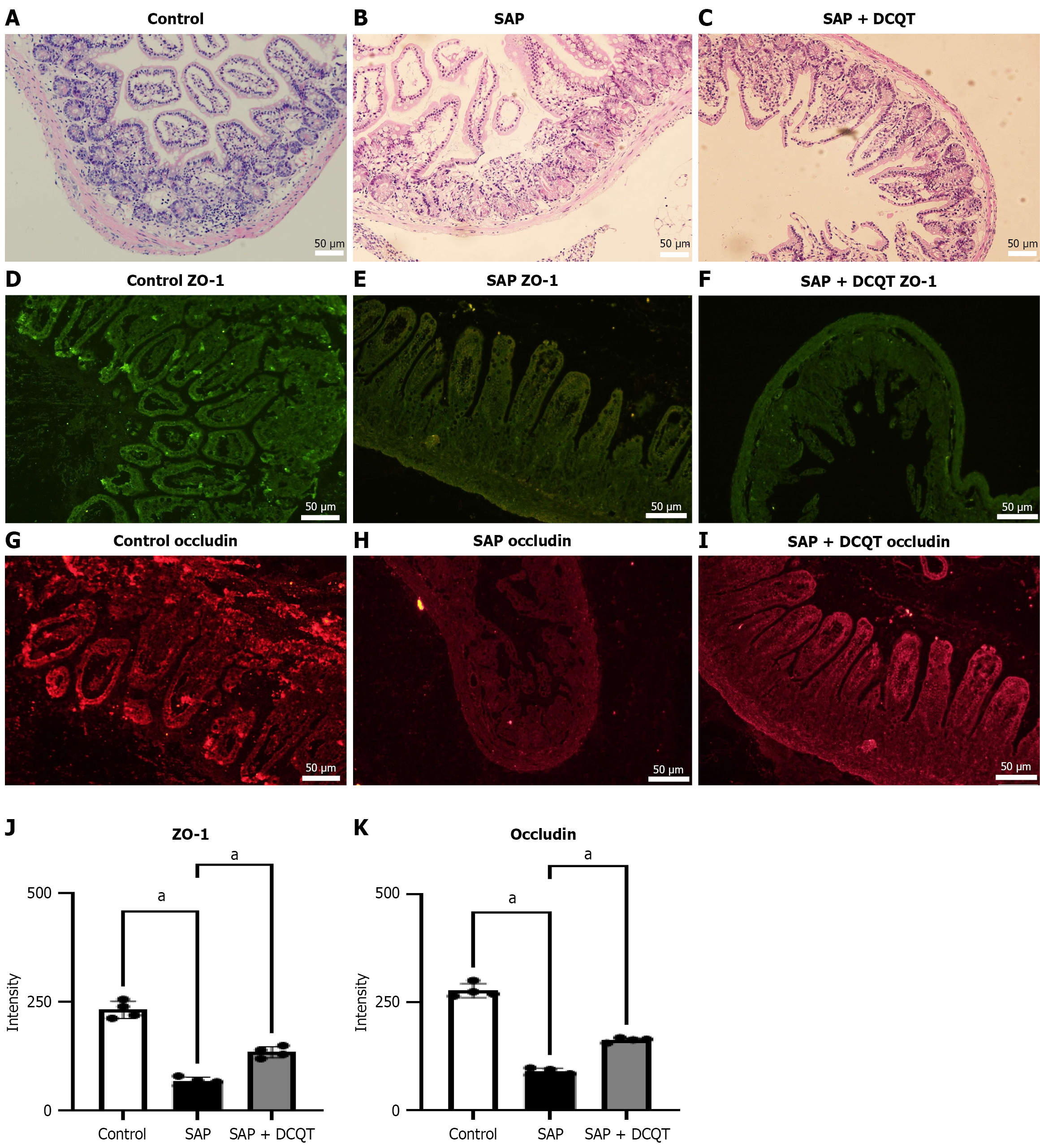
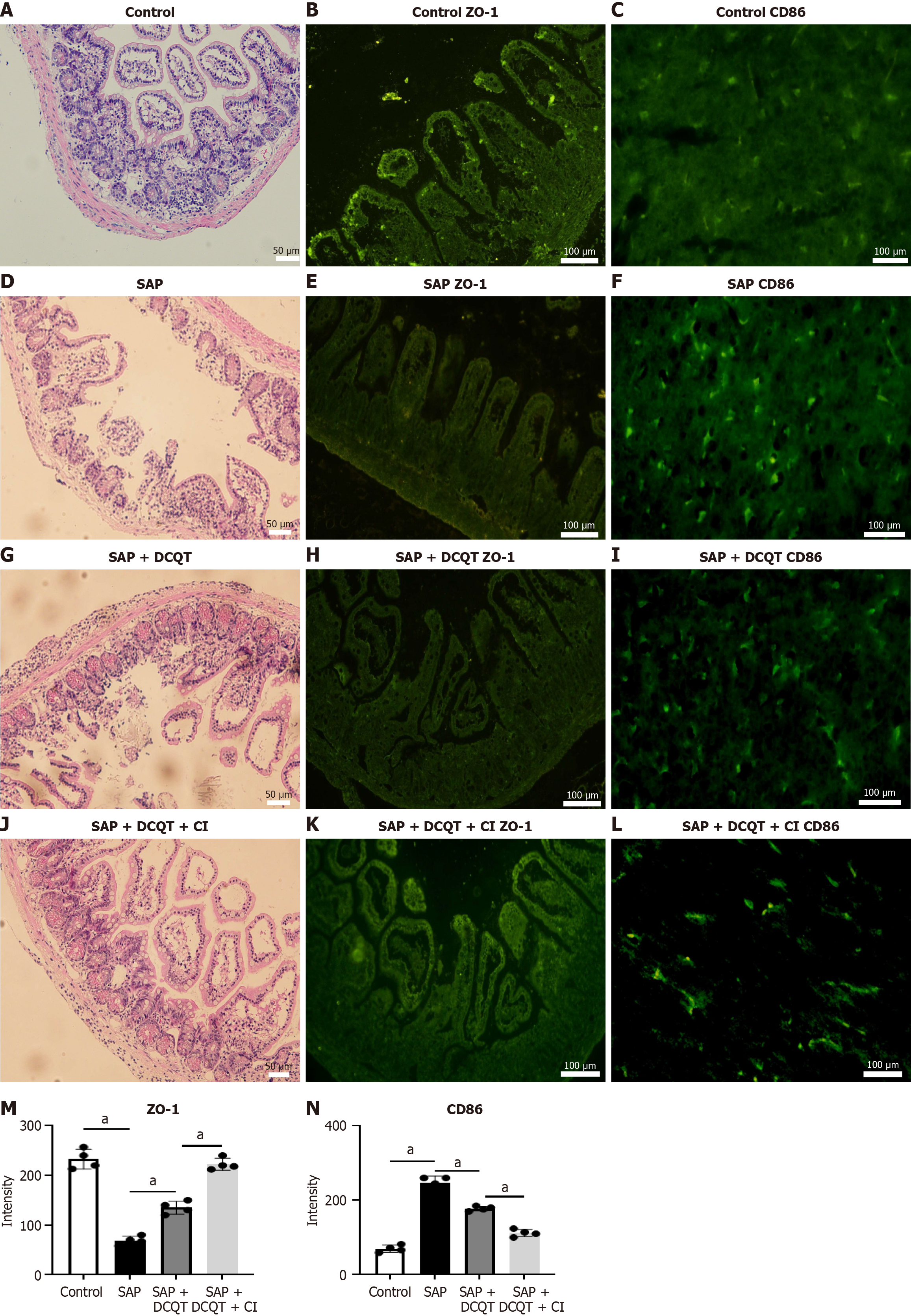
To investigate the impact of DCQT on injury, we delivered DCQT through intraperitoneal injections to mitigate the effects. The severity of injury was observed to be reduced in DCQT-administered mice, as evidenced by hematoxylin and eosin staining (Figure 4A-C). A substantial reduction in the intestinal injury marker was noted 24 hours post AP treatment via immunofluorescence analysis (Figure 4D-I). Additionally, treatment with DCQT led to a significant upregulation in the protein levels of occludin and ZO-1, as illustrated in the corresponding immunofluorescence photographs (Figure 4J and K).
As shown in Figure 5, the intestinal morphology and expression of tight junction protein ZO-1 and macrophage marker CD86 were examined across different experimental groups. H&E staining revealed that SAP induced significant intestinal mucosal damage compared to the control group, while DCQT treatment markedly improved intestinal tissue morpho
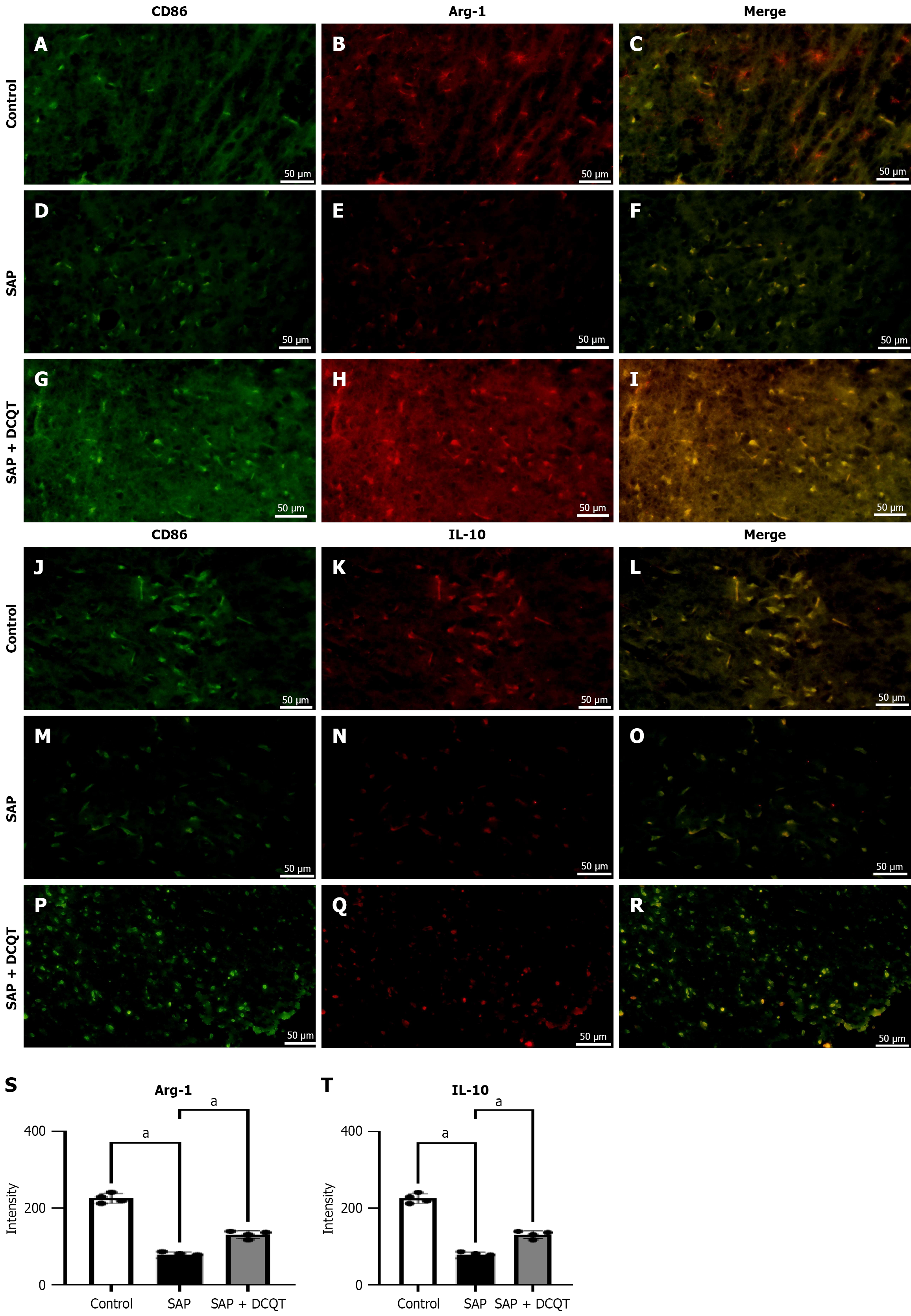
In vivo experiments demonstrated that DCQT stimulates the expression of Arg-1 and IL-10, promoting the M2 macrophage phenotype. Following the induction of diabetes with streptozotocin, there was a marked decrease in the fluorescence intensity of Arg-1 (Figure 6A-I) and IL-10 (Figure 6J-T) as compared to the levels before treatment. The fluorescence intensity further diminished with DCQT administration.
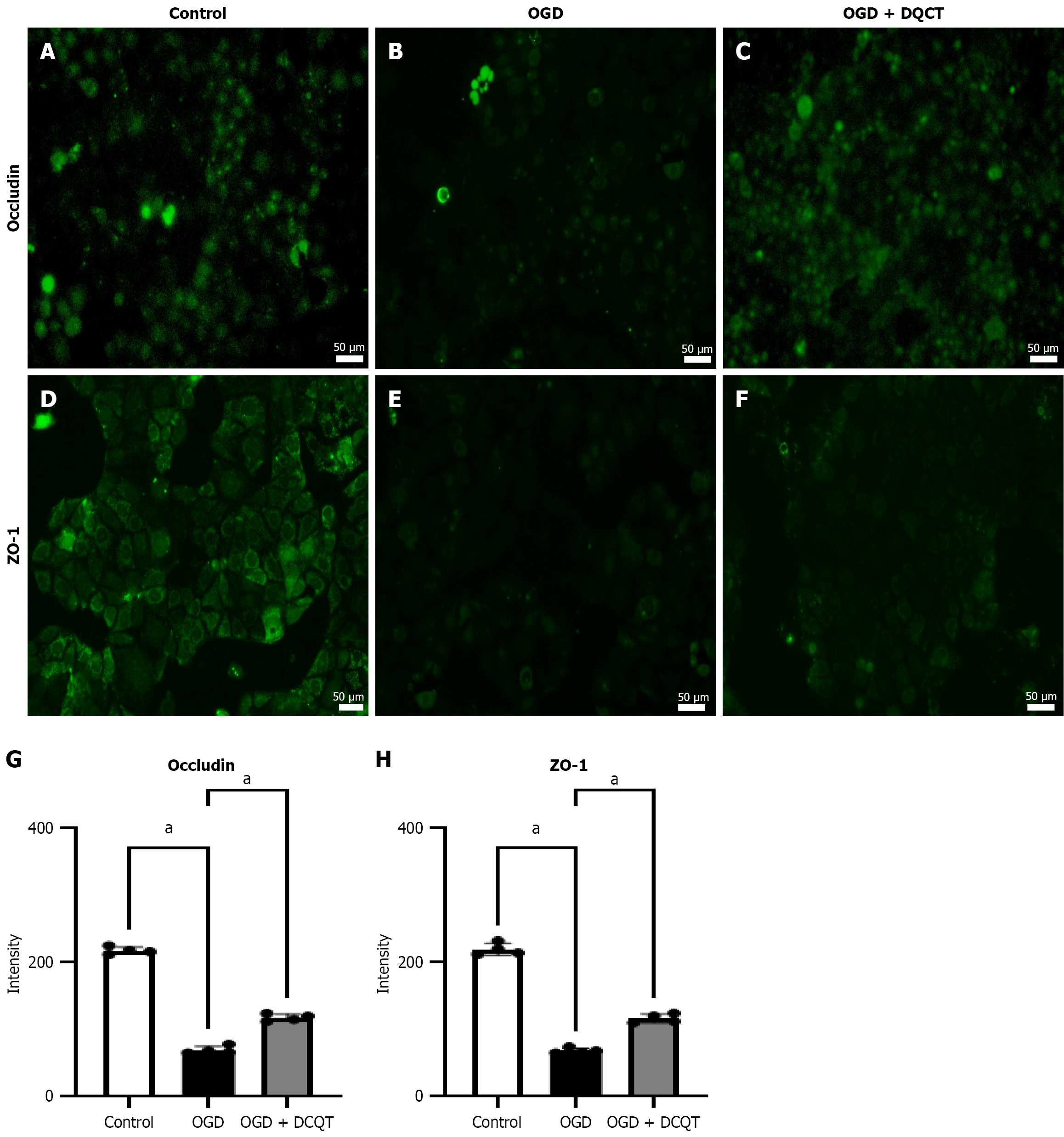
Fluorescence intensities of occludin (Figure 7A-C) and ZO-1 (Figure 7D-F) significantly decreased following OGD treat
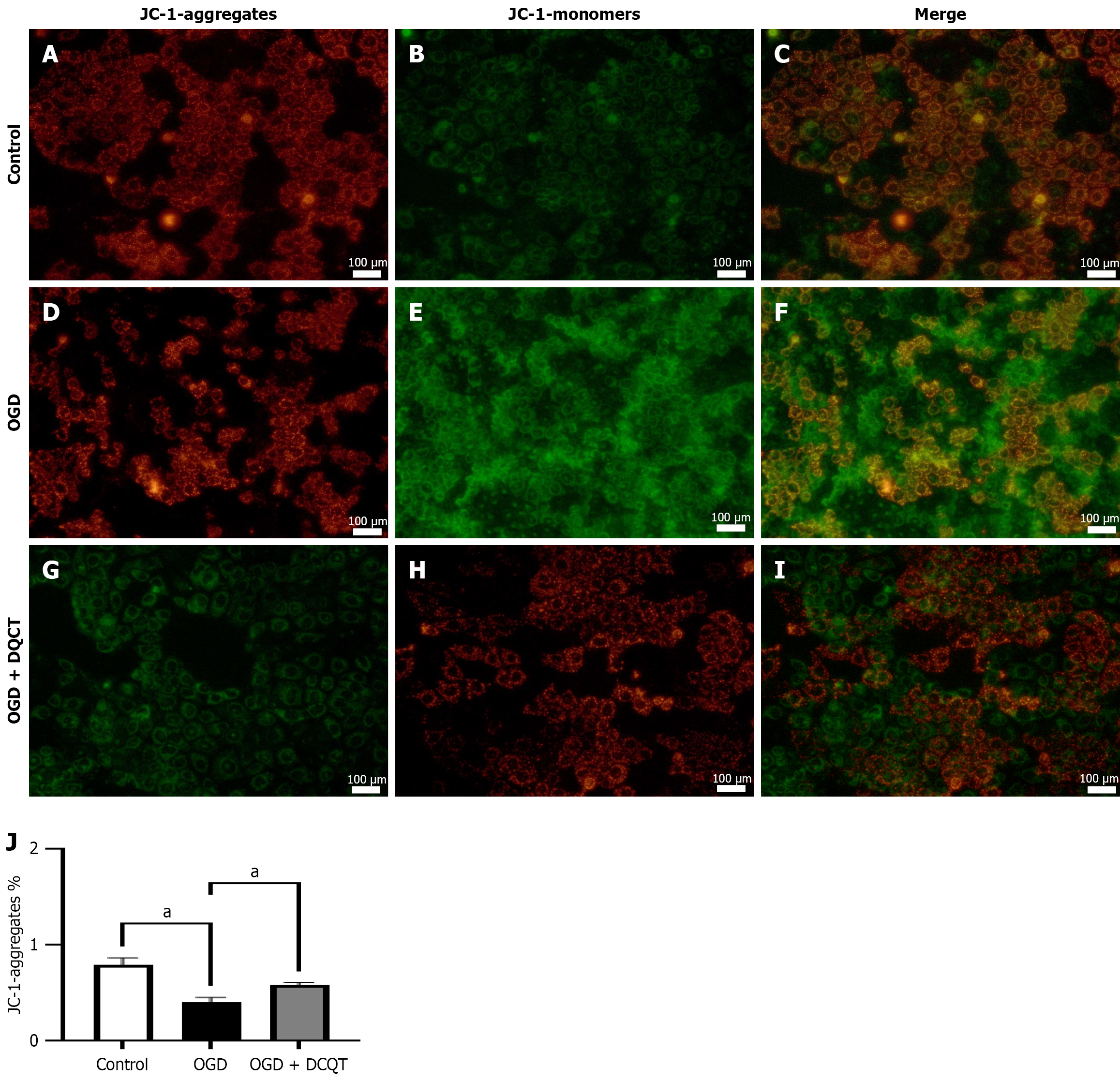
In this study, we assessed the effects of dexamethasone on mitochondrial dysfunction in OGD induced Caco-2 cells. Our results show that, compared to cells exposed only to high glucose conditions, the addition of remifentanil resulted in reduced JC-1 monomer levels and enhanced mitochondrial membrane potential. This indicates that dexamethasone may mitigate the mitochondrial damage induced by high glucose levels (Figure 8).
Post-ERCP pancreatitis represents a potentially life-threatening complication with acute onset and variable severity ranging from mild to severe disease[18-20]. This procedure-related pancreatitis may progress to SIRS and multi-organ dysfunction following endoscopic intervention. The pathophysiological mechanisms underlying PEP are multifactorial, encompassing procedure-induced pancreatic enzyme activation, mechanical trauma from endoscopic instrumentation, and amplification of inflammatory cascades[18-21]. A pivotal contributor to PEP progression is compromised intestinal barrier integrity. Barrier disruption facilitates bacterial translocation and endotoxin passage from the intestinal lumen into systemic circulation during the vulnerable peri-procedural period, thereby amplifying the inflammatory response. Elucidating these mechanistic pathways is essential for developing evidence-based therapeutic interventions to reduce PEP burden and optimize clinical outcomes.
The present study demonstrates the intricate relationship among intestinal barrier function, immune cell phenotype modulation, and inflammatory processes in PEP pathogenesis. The barrier-protective effects of DCQT are particularly significant, as intestinal barrier integrity is increasingly recognized as a critical determinant of systemic inflammatory responses following ERCP.
The intestinal mucosal barrier serves as a primary defense mechanism preventing bacterial and endotoxin translocation into systemic circulation, particularly during the peri-procedural period[22,23]. In PEP, barrier compromise results in increased intestinal permeability "leaky gut", which triggers or exacerbates systemic inflammation. Our findings demonstrate that DCQT enhances barrier function, potentially through upregulation of tight junction proteins and reduction of paracellular permeability. Restoration of barrier integrity may play a pivotal role in attenuating systemic inflammatory responses in PEP and facilitating recovery.
Macrophages exhibit remarkable phenotypic plasticity, polarizing toward M1 (proinflammatory) or M2 (anti-inflammatory) phenotypes in response to local microenvironmental cues[24,25]. The DCQT-induced shift toward M2 polariza
Gene expression profiling and pathway analysis provide molecular insights into DCQT's mechanisms of action. Down
DCQT may activate PI3K/Akt signaling, which upregulates tight junction proteins (ZO-1, Occludin) and enhances barrier function. The observed improvement in mitochondrial membrane potential (JC-1 assay) suggests DCQT confers mito
Our data showing reduced CD86+ M1 macrophages and increased Arg-1+/IL-10+ M2 macrophages suggest that DCQT inhibits NF-κB/STAT1 signaling while promoting STAT6/PPARγ activation-ey regulators of M1/M2 polarization. DCQT may modulate the intestinal cytokine milieu, shifting the balance from proinflammatory mediators (IL-6, TNF-α) toward anti-inflammatory cytokines (IL-10, TGF-β).
We hypothesize that DCQT exerts barrier-protective effects through dual mechanisms: Direct cytoprotection via PI3K/Akt and Nrf2 pathway activation in epithelial cells, and indirect protection through immunomodulation of macrophage phenotypes via NF-κB/STAT signaling. Attenuated M1 macrophage activation may reduce local inflammatory burden, thereby further preserving epithelial barrier integrity.
Future validation studies will include: Western blot analysis of NF-κB, STAT1, STAT6, PI3K/Akt, and Nrf2 pathway components; pharmacological inhibitor studies to validate specific signaling cascade involvement; and transcriptomic profiling (RNA-seq) to comprehensively map DCQT-regulated genes and pathways.
The concordance between in vivo and in vitro findings strengthens the translational relevance of our results. DCQT's protective effects observed in the PEP animal model are recapitulated in intestinal epithelial cell cultures, demonstrating system-level improvements (enhanced ZO-1/Occludin-1 expression, reduced macrophage activation) paralleled by cellular-level benefits (improved intercellular tight junctions, preserved mitochondrial membrane potential).
Our findings have substantial clinical implications for PEP management. If DCQT's protective effects translate to human patients undergoing ERCP, it may offer a novel therapeutic approach, particularly for patients with severe complications or inadequate responses to conventional prophylactic strategies. Rigorously designed clinical trials are warranted to evaluate DCQT's safety and efficacy in PEP prevention and treatment. Furthermore, complete elucidation of the molecular mechanisms underlying DCQT's effects on intestinal barrier function and macrophage polarization may facilitate development of targeted pharmacological agents for pathway-specific interventions in PEP management.
In conclusion, our study conducted in pre-clinical rat models establishes the multifaceted therapeutic effect of DCQT in post-ERCP pancreatitis. DCQT alleviates the condition by enhancing the intestinal mucosal barrier, modulating macrophage polarization, and regulating specific inflammatory pathways, thereby demonstrating significant potential as a postoperative treatment.
| 1. | Padula D, Mauro A, Maggioni P, Kurihara H, Di Sabatino A, Anderloni A. Practical approach to acute pancreatitis: from diagnosis to the management of complications. Intern Emerg Med. 2024;19:2091-2104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 2. | Yang J, Wu B, Sha X, Lu H, Pan LL, Gu Y, Dong X. Intestinal GSTpi deficiency exacerbates the severity of experimental hyperlipidemic acute pancreatitis. Int Immunopharmacol. 2024;137:112363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 3. | Zhang J, Lu X, Ge Y. Nurse-Led Care at Home Visit Versus Standard Care in Patients with Mild Acute Pancreatitis: A Retrospective Analysis. Dig Dis Sci. 2024;69:2775-2783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 4. | Chen Y, Lin S, Wang L, Zhang Y, Chen H, Fu Z, Zhang M, Luo H, Liu J. Reinforcement of the intestinal mucosal barrier via mucus-penetrating PEGylated bacteria. Nat Biomed Eng. 2024;8:823-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 80] [Article Influence: 40.0] [Reference Citation Analysis (1)] |
| 5. | Qiu Q, Geng Z, Wang L, Zuo L, Deng M, Zhang H, Yang Y, Wang Y, Zhao Z, Wen H, Wang Q, Wang Y, He X, Li J, Wang Y, Zhang X, Liu M, Song X. Peiminine ameliorates Crohn's disease-like colitis by enhancing the function of the intestinal epithelial barrier through Nrf2/HO1 signal. Int Immunopharmacol. 2024;136:112380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 6. | Chen C, Li M, Liu X, Fan J, Zhang H, Lin S, Yin L, Fichna J, Li Y. Traditional Chinese Medicine Da-Cheng-Qi-Tang Ameliorates Impaired Gastrointestinal Motility and Intestinal Inflammatory Response in a Mouse Model of Postoperative Ileus. Evid Based Complement Alternat Med. 2020;2020:9074069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 7. | Takashima K, Matsushima M, Hashimoto K, Nose H, Sato M, Hashimoto N, Hasegawa Y, Kawabe T. Protective effects of intratracheally administered quercetin on lipopolysaccharide-induced acute lung injury. Respir Res. 2014;15:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 8. | Clough E, Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, Marshall KA, Phillippy KH, Sherman PM, Lee H, Zhang N, Serova N, Wagner L, Zalunin V, Kochergin A, Soboleva A. NCBI GEO: archive for gene expression and epigenomics data sets: 23-year update. Nucleic Acids Res. 2024;52:D138-D144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 263] [Article Influence: 131.5] [Reference Citation Analysis (1)] |
| 9. | Alameer A, Chicco D. geoCancerPrognosticDatasetsRetriever: a bioinformatics tool to easily identify cancer prognostic datasets on Gene Expression Omnibus (GEO). Bioinformatics. 2022;38:1761-1763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 10. | Dvorak P, Hlavac V, Hanicinec V, Rao BH, Soucek P. Genes divided according to the relative position of the longest intron show increased representation in different KEGG pathways. BMC Genomics. 2024;25:649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 11. | Stojković D, Gašić U, Uba AI, Zengin G, Rajaković M, Stevanović M, Drakulić D. Chemical profiling of Anthriscus cerefolium (L.) Hoffm., biological potential of the herbal extract, molecular modeling and KEGG pathway analysis. Fitoterapia. 2024;177:106115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 12. | Niu M, Zhang X, Song P, Li L, Wen L. Intraductal pressure in experimental models of acute and chronic pancreatitis in mice. Pancreatology. 2022;22:917-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 13. | Terao R, Lee TJ, Colasanti J, Pfeifer CW, Lin JB, Santeford A, Hase K, Yamaguchi S, Du D, Sohn BS, Sasaki Y, Yoshida M, Apte RS. LXR/CD38 activation drives cholesterol-induced macrophage senescence and neurodegeneration via NAD(+) depletion. Cell Rep. 2024;43:114102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 14. | Zhou Q, Cao C, Bao Y, Sun T, Yao Adzraku S, Hao X, Li Y, Yuan S, Huang Y, Xu K, Qiao J, Ju W, Zeng L. Macrophage depletion damages hematopoiesis partially through inhibition of cell homing and expansion after hematopoietic cell transplantation. Int Immunopharmacol. 2024;130:111760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 15. | Galati DF, Asai DJ. Immunofluorescence Microscopy. Curr Protoc. 2023;3: e842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 16. | Tomimatsu K, Fujii T, Bise R, Hosoda K, Taniguchi Y, Ochiai H, Ohishi H, Ando K, Minami R, Tanaka K, Tachibana T, Mori S, Harada A, Maehara K, Nagasaki M, Uchida S, Kimura H, Narita M, Ohkawa Y. Precise immunofluorescence canceling for highly multiplexed imaging to capture specific cell states. Nat Commun. 2024;15:3657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 17. | Małota K, Student S, Świątek P. Low mitochondrial activity within developing earthworm male germ-line cysts revealed by JC-1. Mitochondrion. 2019;44:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 18. | Janssens LP, Yamparala A, Martin J, O'Meara J, Harmsen WS, Sathi T, Lemke E, Abu Dayyeh BK, Bofill-Garcia A, Petersen BT, Storm AC, Topazian M, Vargas EJ, Chandrasekhara V, Law RJ. Incidence of Post-ERCP Pancreatitis in Patients Receiving Rectal Indomethacin vs. Compounded Rectal Diclofenac Prophylaxis. Dig Dis Sci. 2024;69:3970-3978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 19. | Michael FA, Feldmann C, Erasmus HP, Kubesch A, Goerguelue E, Knabe M, Abedin N, Heilani M, Hessz D, Graf C, Walter D, Finkelmeier F, Mihm U, Lingwal N, Zeuzem S, Bojunga J, Friedrich-Rust M, Dultz G. A novel ultrasound-based algorithm for the detection of pancreatic stents placed for prophylaxis of post-ERCP pancreatitis: a prospective trial. Ultraschall Med. 2025;46:177-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 20. | Vaithiyam VS, Sachdeva S, Singh AK, Dalal A. Clinical impact of pancreatic steatosis on the risk of post-ERCP pancreatitis: Fact or fiction! Gastrointest Endosc. 2024;100:579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 21. | Bozkurt S, Güner A, Kadıoğlu H, Keçe C, Reis E, Coşkun H. The effects of different mechanisms on the development of post-ERCP pancreatitis in an ERCP model in rats. Turk J Gastroenterol. 2013;24:469-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (2)] |
| 22. | Li F, Wang Z, Cao Y, Pei B, Luo X, Liu J, Ge P, Luo Y, Ma S, Chen H. Intestinal Mucosal Immune Barrier: A Powerful Firewall Against Severe Acute Pancreatitis-Associated Acute Lung Injury via the Gut-Lung Axis. J Inflamm Res. 2024;17:2173-2193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 23. | Xu H, Wen Q, Hu H, Yang S, Lu L, Hu X, Li H, Huang X, Li N. Electroacupuncture at ST36 modulates the intestinal microecology and may help repair the intestinal barrier in the rat model of severe acute pancreatitis. Microb Biotechnol. 2024;17:e14401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 24. | Dror S, Lucotti S, Asao T, Li J, Wortzel I, Berger LS, Matei I, Boudreau N, Zhang H, Jones D, Bromberg J, Lyden D. Tumour-derived Extracellular Vesicle and Particle Reprogramming of Interstitial Macrophages in the Lung Pre-Metastatic Niche Enhances Vascular Permeability and Metastatic Potential. Res Sq. 2024;rs.3.rs-4462139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 25. | Nafiz TN, Sankar P, Mishra LK, Rousseau RP, Saqib M, Subbian S, Parihar SP, Mishra BB. Differential requirement of Formyl Peptide Receptor 1 in macrophages and neutrophils in the host defense against Mycobacterium tuberculosis Infection. Res Sq. 2024;rs.3.rs-4421561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |