Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.116957
Revised: December 12, 2025
Accepted: December 26, 2025
Published online: February 27, 2026
Processing time: 93 Days and 16.2 Hours
Hepatic haemangiomas are the most common benign liver lesions, often found on incidental imaging. Although often asymptomatic, they can lead to abdominal pain, bleeding and rarely rupture. Surgery is indicated in symptomatic cases or in case of complications. To evaluate current surgical management strategies for hepatic haemangiomas, focusing on recent advances in minimally invasive and robotic surgery, imaging, embolization, and perioperative care. It also explores the emerging role of artificial intelligence (AI) in preoperative planning, intraoperative guidance, and postoperative monitoring, and proposes evidence-based management algorithms reflecting current practice and future directions. This minireview synthesizes the recent literature on the surgical management of hepatic haemangiomas, with emphasis on advances in surgical strategies, perioperative protocols available to date, and postoperative care. The contemporary role of diagnostic imaging and AI in improving preoperative evaluation is also explored. Advances in minimally invasive techniques, improved intraoperative navigation and improved recovery protocols have significantly reduced surgical morbidity. The integration of AI has improved lesion characterization and sur
Core Tip: Hepatic haemangiomas are common benign liver lesions that typically require surgery only when symptomatic or complicated. Recent progress in minimally invasive and robotic techniques, enhanced intraoperative guidance, and optimized recovery pathways has significantly improved surgical safety and outcomes. Meanwhile, artificial intelligence is emerging as a valuable tool for precise lesion assessment and individualized surgical planning. Continued refinement of patient selection and standardized perioperative protocols will be key to maximizing treatment effectiveness.
- Citation: Rozani S, Vougas V. Advancements in hepatic haemangioma surgery: An update-review of protocols, techniques, and postoperative outcomes. World J Gastrointest Surg 2026; 18(2): 116957
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/116957.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.116957
Hepatic haemangiomas are the most common benign liver lesions and are often found incidentally on imaging studies. The lesions are generally asymptomatic and so many patients are unaware of their status[1]. However, hepatic haemangiomas may present with abdominal pain, gastrointestinal bleeding or, in unusual cases, rupture. Treatment strategies for hepatic haemangiomas have evolved considerably in recent years, particularly with regard to surgical techniques, diagnostic methods and post-operative care[1,2]. This study provides a comprehensive review in this era of rapid technological advances, proposing algorithms for current surgical management.
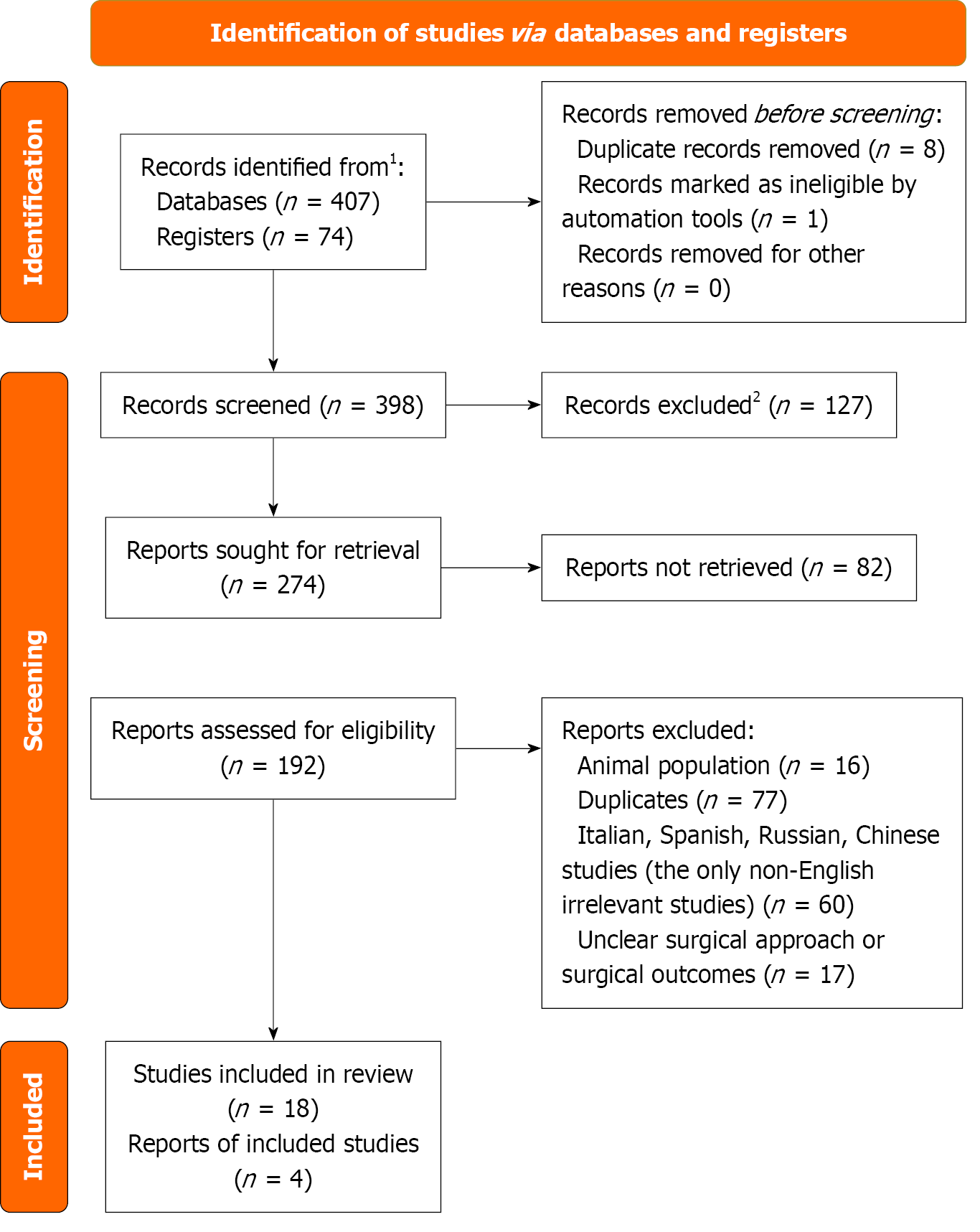
The review was conducted according to the PRISMA 2020 standard guidelines (Figure 1). Both authors contributed equally to each part of this manuscript. Eligibility criteria were developed using the population, intervention, comparator and outcome framework. An electronic search of titles, abstracts and keywords in PubMed, Scopus/MEDLINE and Google Scholar databases was performed based on the research question. Current literature in the Google Scholar, PubMed, and Scopus/MEDLINE databases in a time window of five-years (2020-2025) was reviewed. The first search was conducted in July 2024 and the second search in October 2025. The search terms used were (“hepatic haemagioma”) AND (“liver haemagioma”) AND “hepatic haemagioma surgery”) AND (“liver surgery”), AND (“surgical management”) AND (“giant hepatic haemagioma”). All full-text articles that reported data on robotic liver surgery, hepatic haemangiomas and artificial intelligence (AI) were included in this review duplicates were removed from the search results and then suitable titles and abstracts were searched.
(1) Only studies involving human populations were included; (2) Titles and abstracts were reviewed to identify whether they addressed the research question; (3) The references of the selected articles were also studied for identification of further pertinent studies; (4) Mainly research studies and observational studies were included, while data from previous reviews regarding with the topic were also used; and (5) Mostly studies in English and a non-English study of first-author related to the topic were included.
(1) Abstracts for which there was no full text available were excluded; and (2) Articles in which there was no clear description about surgical approaches and surgical outcomes after robotic liver surgery were excluded to allow comparison and increase reliability of our manuscript. The articles were then qualitatively analysed.
The present review is an update-scoping review as it attempts a thorough study and collection of all thus far investigated surgical outcomes that have a significant impact on the hepatic haemangioma surgery. Furthermore, it emphasizes on suggesting new interdisciplinary guidelines according to the recent studies. Finally, we attempt a comprehensive investigation of the current approaches and the future perspectives in the light of AI.
Hepatic haemangiomas are usually asymptomatic and are usually detected incidentally on imaging tests such as ultra
Recently, impressive strides have been made in the diagnostic evaluation of hepatic hemangiomas, which have a direct impact on surgical decisions. Diagnostic imaging has become more accurate, allowing earlier and more precise detection of lesions and better preoperative planning[2,3]. Contrast-enhanced ΜRI (ΜRI and CT) has become the gold standard for the diagnosis of hepatic hemangiomas since it is able to distinguish hemangiomas from other hepatic lesions with a high degree of sensitivity and specificity[4]. In addition, latest imaging techniques, such as contrast-enhanced dynamic MRI, provide a comprehensive insight of the vascular components, thus aiding in the planning of surgical procedures, especially involving resection or embolization[5].
Regarding to ultrasound, contrast-enhanced ultrasound has also been investigated in the evaluation of liver lesions and has been found to be effective in detecting small lesions and provides real-time dynamic imaging of the haemangioma flow[6]. Vascular mapping and angiography are key aspects in preoperative assessments. Through angiography or CT angiography, surgeons can assess the vascular supply of haemangiomas, especially in larger lesions or central locations. This can significantly influence the resection or embolization planning[7].
Advances in recent surgical procedures and methods have led to better patient outcomes for hepatic hemangiomas, especially when it comes to reducing risks and recovery periods. The use of laparoscopic liver surgery has been under significant attention for its minimally invasive approach, which translates into advantages such as less postoperative pain, shorter hospitalization, and quicker recovery times when compared to traditional open surgery[8]. Moreover, robotic-assisted laparoscopic liver resections have enhanced the precision of such interventions, allowing greater accuracy in hemangioma resections as well as less trauma inflicted upon the surrounding liver tissue[7].
Enucleation, a procedure that involves excision of the haemangioma without disturbing the surrounding liver tissue, has become increasingly popular over the years as the procedure of choice for the treatment of peripheral lesions[3]. Wedge resections, on the other hand, are usually reserved when enucleation is not possible, especially in cases with larger or central lesions. This approach is the removal of a wedge-shaped portion of the liver that includes the hemangioma and is used when a less extensive resection is necessary to preserve liver function[3,7]. In patients with large symptomatic hemangiomas or in whom surgery is not an option, selective hepatic artery embolization (HAE) has emerged as a common option. This procedure minimizes the size of the hemangioma by interrupting its blood supply, which is particularly useful to avoid rupture or bleeding[9].
Liver transplantation can be performed in very exceptional cases where haemangiomas are complicated by advanced cirrhosis or liver failure. However, it should be noted that this is still the last option because of the risks involved in organ transplantation[10]. Besides, intraoperative technologies such as intraoperative ultrasound and near-infrared fluorescence imaging, for example, indocyanine green, have greatly improved the ability of surgeons to accurately identify haemangiomas and determine their proximity to surrounding structures. Therefore, this has improved surgical accuracy and reduced the risk of complications[10,11].
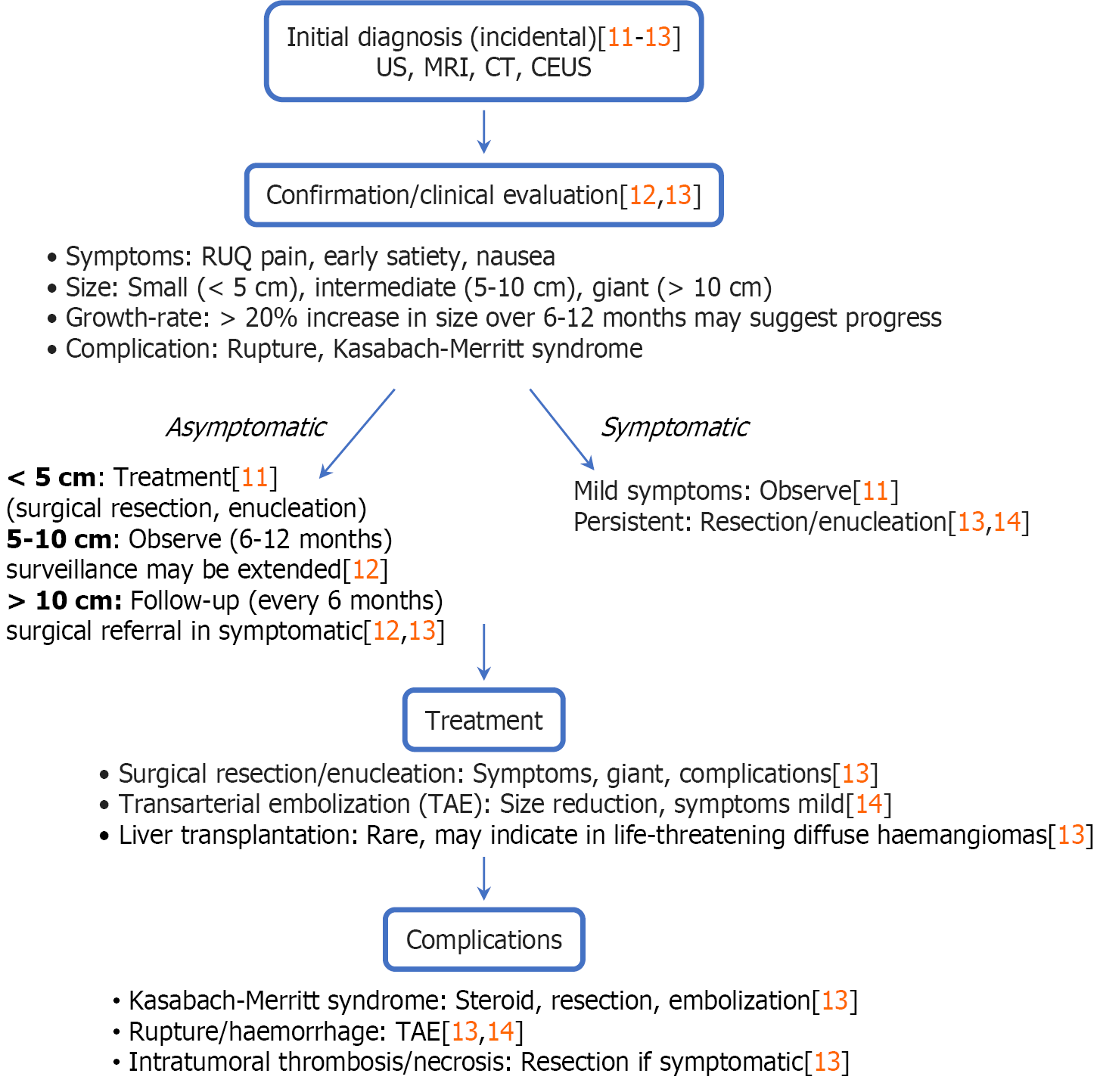
According to the latest studies, the following management algorithm could be proposed for the intraoperative management of hepatic haemangiomas (Figure 2).
The widespread use of AI in Robotic Surgery and Minimally Invasive techniques has made tremendous strides, especially in liver surgery. Early studies on robotic surgeries, guided by AI, offer greater precision and improved surgical outcomes by providing surgeons with improved visualization, stability, and control during the procedure[3,12-14]. It appears that AI-powered robotic systems can aid in precise tissue dissection, reducing damage to surrounding liver tissue and enhancing the safety of enucleation or resection[3]. AI algorithms can adjust robotic instruments in real time based on anatomical variations, ensuring that the hemangioma is removed while minimizing damage to critical structures. In addition, intraoperative AI tools, such as augmented reality (AR) and real-time imaging, help guide surgeons during laparoscopic and robotic liver resections. AI algorithms can integrate preoperative imaging data into the surgical field, providing real-time feedback on the location of the hemangioma and surrounding vascular structures[5,15].
AI has transformed preoperative planning by providing advanced visualization, risk assessment, and decision-making tools. In liver surgery, and especially in complex cases such as giant hemangiomas, AI has the potential to improve spatial perception and supports surgeons with real-time guidance[16]. The integration of AR with 3D liver models created with AI allows virtual reconstructions to be superimposed on the surgical field, improving orientation and facilitating the identification of deep or hidden structures. Intraoperative ultrasound with the help of AI can automatically detect tumor boundaries, vascular pathways, and gallbladder anatomy, increasing accuracy in technically difficult areas[16,17]. New-generation hemostatic devices equipped with AI analyze tissue characteristics to predict the risk of bleeding and optimize energy delivery, while robotic platforms with built-in AI improve accuracy, reduce tremor, and provide alerts near vital structures[17-19]. The integration of machine learning algorithms trained on previous surgical procedures can even suggest optimal resection levels[17,20]. Recent advances in automated liver segmentation using 3D-CNN and U-Net architectures, which achieve high accuracy and reduced processing time, highlight the potential of AI-based methods to support planning for lesions in anatomically challenging areas[17,21]. A study using a 3D CNN Deep Medic model for liver segmentation on abdominal CT reported a dice similarity coefficient of 93.14%, with accuracy, sensitivity, and specificity of 94.36%, 94.57%, and 91.86%, respectively[21]. These results are based on retrospective data without exten
The post-operative management of patients operated on for hepatic haemangiomas has also improved significantly, particularly in terms of reducing complications and maximising recovery. Complications in the postoperative period include a range of issues such as bleeding, infection, bile leakage and, in a few cases, recurrence of the haemangioma. However, advances in surgical techniques, particularly minimally invasive surgery, coupled with enhanced perioperative management, have served to reduce the incidence of these complications[5]. Meticulous observation during the imme
In terms of postoperative monitoring and outcome prediction, AI systems are increasingly being used to monitor patients and predict potential complications, such as intraoperative infection, bleeding, or hemangioma recurrence. AI-based systems can continuously monitor patient vital signs, laboratory results, and other parameters to identify early signs of complications[15]. AI algorithms can analyze patient data in real time to predict complications such as liver dysfunction, bleeding, or infection, allowing for early intervention and improved recovery times[9,15]. This can lead to shorter hospital stays and better long-term outcomes. Postoperatively, AI can also help with long-term imaging surveil
| Ref. | Procedures | Outcomes |
| Li et al[15] | Hemangioma enucleation: Pringle maneuver applied for portal blood occlusion. Blunt dissection along boundary between hemangioma and normal liver parenchyma. Small vessels and bile ducts transected with ultrasonic knife; large vessels clipped with Hem-o-Lok. Left lateral lobectomy: Incision along falciform ligament; Glisson sheath and left hepatic vein transected with Endo-GIA stapler. Hemostasis with bipolar electrocoagulation, argon spray, or suturing. Specimen retrieval: Removed via specimen bag through trocar; fragmented if needed. Two abdominal drains placed. Intraoperative management: Low central venous pressure (0-4 cm H2O). Intermittent portal occlusion (15 minutes block + 5 minutes release) | Patients: 58 in total (special location group SLG: 27; normal location group NLG: 31). Intraoperative findings: Operation time: SLG 258.4 ± 49.2 minutes vs NLG 186.2 ± 51.8 minutes (P < 0.001). Blood loss: SLG 466.7 ± 235.7 mL vs NLG 259.3 ± 92.6 mL (P < 0.001). Blood transfusion: SLG 29.6% vs NLG 6.5% (P = 0.047). Conversion to laparotomy: SLG 14.8%, NLG 0% (not significant, P = 0.089). No perioperative deaths. Postoperative recovery: Exhaust time: SLG 3.8 ± 1.4 days vs NLG 2.9 ± 0.8 days (P = 0.03). Hospital stay: SLG 11.5 ± 3.4 days vs NLG 7.3 ± 2.6 days (P < 0.01). Postoperative complications: Pleural effusion, ascites, biliary leakage, DVT. Conclusions: Laparoscopic enucleation in special segments is technically challenging with higher intraoperative bleeding risk; safe and feasible with proper planning, skilled team, and readiness to convert to laparotomy; preserves liver parenchyma and achieves effective tumor resection |
| Xu et al[10] | Total patients: 357. Total hemangiomas: 378. Age (mean): 49.2 ± 10.5 years. Lesion size: 5.0-21.2 cm. Location: 273 subcapsular (laparoscopic ablation), 105 intraparenchymal (CT-guided percutaneous ablation). Procedures: Laparoscopic thermal ablation for subcapsular hemangiomas. CT-guided percutaneous thermal ablation for intraparenchymal hemangiomas. One ablation session for 369 lesions; two sessions for 9 lesions. Technical success rate: 100% | Complete ablation achieved in 361/378 hemangiomas (95.5%). Incomplete ablation in 17 lesions (4.5%). Major complication rate: 2.0% (7/357 patients). Symptom relief in 224 symptomatic patients: 96.4% complete disappearance, 3.6% partial improvement. Ablated lesion shrinkage progressive; 11.4% of hemangiomas almost completely disappeared over time. Median follow-up: 67 months (range 12-124 months) |
| Zhou et al[4] | Total: 1171 patients, median age 45, 63% women, 53.5% solitary hemangioma, 46.5% multiple. Lesion growth: 38% stable, 60% slow (< 1 cm/year), 2% rapid (≥ 1 cm/year). Score based on symptoms, size, location, growth rate (0-2 each): Score ≥ 4 → surgery recommended, score < 4 → observation; alternatives: TAE or RFA | Score ≥ 4: Surgery → 97% symptom-free, 9% complications, 1.8% needed intervention, observation → 44% symptom-free, 40% required intervention. Score < 4: Surgery → 94% symptom-free, 12% complications, observation → 88% symptom-free, 4% required intervention |
Follow-up after surgery is recommended in all patients and this usually includes regular imaging to diagnose recur
The management of hepatic haemangiomas has evolved significantly with the advancement of minimally invasive surgical techniques, improved perioperative protocols, and enhanced diagnostic imaging. Laparoscopic and robotic-assisted approaches have reduced postoperative morbidity, shortened recovery time, and improved surgical precision, particularly for lesions in challenging anatomical locations. Enucleation remains the preferred parenchymal-sparing technique, while resection and embolization continue to play important roles in selected cases. These developments support a more individualized and safer approach to surgical decision-making.
AI has emerged as a transformative tool in both preoperative planning and intraoperative navigation. AI-enhanced imaging, 3D segmentation, and AR guidance allow for improved lesion characterization and more accurate surgical mapping, which can be particularly valuable in complex or giant haemangiomas. Early postoperative applications of AI, including complication prediction and automated imaging surveillance, further highlight its potential to optimize patient outcomes and streamline postoperative care. As these technologies continue to mature, their integration into hepatic surgery is expected to deepen.
Despite these advances, the current evidence base has limitations that should be acknowledged. Much of the available literature consists of retrospective or observational studies with heterogeneous methodologies, limiting the ability to draw definitive conclusions. Variability in surgical indications, lesion classification, and outcome reporting further restricts comparability across studies. Additionally, while AI-assisted tools show promise, they remain in early stages of clinical validation and are not yet widely accessible. Future prospective studies and standardized clinical guidelines will be essential to refine patient selection, validate emerging technologies, and optimize management strategies for hepatic haemangiomas. The majority of studies included in this review are retrospective or observational, with limited sample sizes and heterogeneous methodologies. Surgical indications, lesion classification, and outcome reporting varied across studies, reducing comparability. Evidence on AI-assisted surgical planning and intraoperative guidance is preliminary, often based on pilot studies or single-center experiences, with limited external validation. Overall, while these studies provide valuable insights into emerging techniques and technologies, the quality of evidence remains moderate to low, highlighting the need for prospective, multicenter trials and standardized reporting frameworks.
Surgical management of hepatic hemangiomas has also undergone remarkable transformation with advances in diagnostic imaging, surgical techniques, and postoperative management that have translated into improved patient outcomes. The application of laparoscopic and robotic assistance, along with preoperative vascular mapping and embolization, has enhanced the safety and effectiveness of surgery even in complex cases. While surgery is still the first-line treatment for symptomatic hemangiomas, the development of non-surgical treatments, including HAE, offers useful options in patients who are not surgical candidates. Continued research and development in this area guarantee even greater advances in both diagnostic and therapeutic techniques for hepatic hemangiomas.
Advancements in the surgical treatment of hepatic hemangiomas has facilitated more sophisticated protocols, less invasive surgical techniques and more effective postoperative care, ultimately improving outcomes for patients. Preoperative imaging and embolization have developed to be part of the protocol, providing greater accuracy of resections with less bleeding. Minimally invasive surgical strategies such as laparoscopic or robotic-assisted surgery have shown potential to improve efficacious recovery time and reduce complications. Even though recurrence rates remain low of hepatic hemangiomas, ongoing long-term follow-up and research into best practices of surgical management is ongoing, so the management of hepatic hemangiomas is continuously getting better.
Future directions may consider the implementation of advanced therapies and technologies, such as improved imaging systems and newer hemostatic devices to further refine surgical management and postoperative care. Additionally, as the population ages and the recognition of the impact of disease increases, it is likely that surgical management will continue to advance in ways that address the beneficial aspects of patient care (both clinical and quality of life) for patients with hepatic hemangioma.
| 1. | Kacała A, Dorochowicz M, Matus I, Puła M, Korbecki A, Sobański M, Jacków-Nowicka J, Patrzałek D, Janczak D, Guziński M. Hepatic Hemangioma: Review of Imaging and Therapeutic Strategies. Medicina (Kaunas). 2024;60:449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 2. | Xie QS, Chen ZX, Zhao YJ, Gu H, Geng XP, Liu FB. Outcomes of surgery for giant hepatic hemangioma. BMC Surg. 2021;21:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 3. | Aziz H, Brown ZJ, Baghdadi A, Kamel IR, Pawlik TM. A Comprehensive Review of Hepatic Hemangioma Management. J Gastrointest Surg. 2022;26:1998-2007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 4. | Zhou CM, Cao J, Chen SK, Tuxun T, Apaer S, Wu J, Zhao JM, Wen H. Retrospective analysis based on a clinical grading system for patients with hepatic hemangioma: A single center experience. World J Gastrointest Surg. 2024;16:2047-2053. [PubMed] [DOI] [Full Text] |
| 5. | Zhang ZH, Jiang C, Li JX. Reconsideration of the clinical management of hepatic hemangioma. World J Gastrointest Surg. 2024;16:3623-3628. [PubMed] [DOI] [Full Text] |
| 6. | Sandulescu LD, Urhut CM, Sandulescu SM, Ciurea AM, Cazacu SM, Iordache S. One stop shop approach for the diagnosis of liver hemangioma. World J Hepatol. 2021;13:1892-1908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 7. | Ketchum WA, Lin-Hurtubise KM, Ochmanek E, Ishihara K, Rice RD. Management of Symptomatic Hepatic "Mega" Hemangioma. Hawaii J Med Public Health. 2019;78:128-131. [PubMed] |
| 8. | Zhang W, Huang ZY, Ke CS, Wu C, Zhang ZW, Zhang BX, Chen YF, Zhang WG, Zhu P, Chen XP. Surgical Treatment of Giant Liver Hemangioma Larger Than 10 cm: A Single Center's Experience With 86 Patients. Medicine (Baltimore). 2015;94:e1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 9. | Boecker J, Oldhafer KJ. CHAPTER 8 - Hepatic hemangioma. In: Treatment and Management of Tropical Liver Disease. Amsterdam: Elsevier, 2025: 60-64. [DOI] [Full Text] |
| 10. | Xu L, Wu S, Kong J, Ke S, Yin T, Guo S, Ning C, Wang X, Li S, Ding J, Li A, Kong X, Wang Q, Xu Y, Gao J, Sun W. Thermal ablation of hepatic hemangioma: A multi-center experience with long-term outcomes. Eur J Radiol. 2023;164:110842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Ying X, Dong S, Zhao Y, Chen Z, Jiang J, Shi H. Research Progress on Contrast-Enhanced Ultrasound (CEUS) Assisted Diagnosis and Treatment in Liver-Related Diseases. Int J Med Sci. 2025;22:1092-1108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 12. | Frenette C, Mendiratta-Lala M, Salgia R, Wong RJ, Sauer BG, Pillai A. ACG Clinical Guideline: Focal Liver Lesions. Am J Gastroenterol. 2024;119:1235-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 13. | Reguram R, Ghonge A, Tse J, Dhanasekaran R. Practical approach to diagnose and manage benign liver masses. Hepatol Commun. 2024;8:e0560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 14. | Torkian P, Li J, Kaufman JA, Jahangiri Y. Effectiveness of Transarterial Embolization in Treatment of Symptomatic Hepatic Hemangiomas: Systematic Review and Meta-analysis. Cardiovasc Intervent Radiol. 2021;44:80-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 15. | Li H, Duan X, Wu Z, Qin Y. Feasibility of laparoscopic enucleation for hemangioma in special hepatic segments. Front Surg. 2022;9:1111307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Panagakis A, Katsaros I, Sotiropoulou M, Mylonakis A, Despotidis M, Sourgiadakis A, Sakarellos P, Kapiris S, Vergadis C, Schizas D, Felekouras E, Vailas M. Artificial Intelligence and 3D Reconstruction in Complex Hepato-Pancreato-Biliary (HPB) Surgery: A Comprehensive Review of the Literature. J Pers Med. 2025;15:610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Rozani S, Weber G, Grützmann R. [Surgical management of giant hemangiomas in the caudate lobe: an overview on the intraoperative management and role of artificial intelligence (AI) in improvement of the surgical results]. Chirurgie (Heidelb). 2025;96:918-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Schulze A, Haselbeck-Köbler M, Brandenburg JM, Daum MTJ, März K, Hornburg S, Maurer H, Myers F, Reichert G, Bodenstedt S, Nickel F, Kriegsmann M, Wielpütz MO, Speidel S, Maier-Hein L, Müller-Stich BP, Mehrabi A, Wagner M. Aliado - A design concept of AI for decision support in oncological liver surgery. Eur J Surg Oncol. 2025;51:108669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Pomohaci MD, Grasu MC, Băicoianu-Nițescu AŞ, Enache RM, Lupescu IG. Systematic Review: AI Applications in Liver Imaging with a Focus on Segmentation and Detection. Life (Basel). 2025;15:258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Zhu D, Tulahong A, Abuduhelili A, Liu C, Aierken A, Lin Y, Jiang T, Lin R, Shao Y, Aji T. Machine Learning Prognostic Model for Post-Radical Resection Hepatocellular Carcinoma in Hepatitis B Patients. J Hepatocell Carcinoma. 2025;12:353-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 21. | Humady K, Al-Saeed Y, Eladawi N, Elgarayhi A, Elmogy M, Sallah M. Efficient liver segmentation with 3D CNN using computed tomography scans. 2022 Preprint. Available from: arXiv:2208.13271. [DOI] [Full Text] |
| 22. | Veerankutty FH, Jayan G, Yadav MK, Manoj KS, Yadav A, Nair SRS, Shabeerali TU, Yeldho V, Sasidharan M, Rather SA. Artificial Intelligence in hepatology, liver surgery and transplantation: Emerging applications and frontiers of research. World J Hepatol. 2021;13:1977-1990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/