Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.105096
Revised: February 4, 2025
Accepted: March 3, 2025
Published online: April 27, 2025
Processing time: 76 Days and 4.2 Hours
Intraductal oncocytic papillary neoplasm (IOPN) of the pancreas is an extremely rare pancreatic tumor, with only sporadic cases reported in the literature. IOPN is difficult to diagnose and highly prone to misdiagnosis. IOPN carries a certain risk of progressing to invasive cancer. Surgical resection is the primary treatment for IOPN. According to the existing literature reports, the vast majority of patients with IOPN of the pancreas undergo open surgery, while only one case of laparoscopic surgery have been reported.
This report presents two cases of IOPN in elderly female patients, aged 60 and 61. Both patients were asymptomatic, and their pancreatic masses were discovered incidentally. Preoperative diagnosis of IOPN is challenging and prone to misdiagnosis. In the first case, the patient underwent a laparoscopic distal pancreatectomy and splenectomy. The surgical procedure spanned 342 minutes, with an estimated intraoperative blood loss of around 100 mL. The patient experienced an uneventful postoperative recovery and was discharged on the 8th postoperative day. For the second case, a laparoscopic pancreaticoduodenectomy was per
These cases demonstrate that laparoscopic surgery can be considered as one of the treatment options for IOPN of the pancreas.
Core Tip: Intraductal oncocytic papillary neoplasm (IOPN) of the pancreas is extremely rare. The mechanism of the disease is not fully understood. Here, we reported 2 cases of pancreatic IOPN that were treated with laparoscopic surgery, covering its clinical manifestations, imaging findings, pathological morphological and immunophenotype, clinical treatment and prognosis, aiming to provide references for clinicians and improve the level of diagnosis and treatment of similar cases. Since a certain proportion of cases may be associated with invasive cancer, surgery is the preferred treatment. Laparoscopic surgery can be considered as one of the treatment options for IOPN of the pancreas.
- Citation: Wu GZ, Lu LN, Lin HP, Wang XY, Yu SA, Yu M. Laparoscopic management of intraductal oncocytic papillary neoplasm of the pancreas: Two case reports and review of literature. World J Gastrointest Surg 2025; 17(4): 105096
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/105096.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.105096
Intraductal oncocytic papillary neoplasm (IOPN) of the pancreas is a rare pancreatic tumor and a precancerous lesion that may develop into pancreatic cancer. Pancreatic IOPN was first reported by Adsay et al[1] in 1996. Previously, pancreatic IOPN was believed to be a subtype of pancreatic intraductal papillary mucinous neoplasm (IPMN). However, due to its unique morphology, mucin content, and molecular characteristics, IOPN is now considered a high-grade intraepithelial neoplasm. Thus, in the new 2019 World Health Organization classification of digestive system tumors, IOPN is classified separately from IPMN[2,3]. Following the recent molecular and clinicopathological data revealing the rather substantial differences between IOPNs and IPMNs[4]. Specifically, IOPN does not exhibit the typical mutations observed in IPMN, such as KRAS, GNAS, and RNF43[5]. More recently, recurrent gene fusions have been described in genes encoding protein kinase A (PRKACA and PRKACB) in these tumors, which are not found in any of their clinicopathological differential diagnoses, further cementing their distinction as a separate entity. Furthermore, the clinical behavior is also distinct, demonstrating a significantly better prognosis than in IPMN, even in the context of metastasis and recurrence[6,7].
Currently, few articles have reported IOPN of the pancreas, and its pathogenesis and clinical features remain largely unclear. In addition, due to the lack of distinctive laboratory and imaging manifestations, preoperative diagnosis is challenging, and misdiagnosis is common. IOPN carries a certain risk of progressing to invasive cancer. Surgical resection is the primary treatment for pancreatic IOPN patients. According to the existing literature reports, the vast majority of pancreatic IOPN patients undergo open surgery, whereas laparoscopic surgery has hardly been reported. Compared with traditional open surgery, laparoscopic surgery boasts advantages like reduced trauma, smaller incisions, less blood loss, and quicker recovery[8]. Minimally invasive surgery represents the trend in surgical development. Owing to the complexity and safety considerations of pancreatic surgery, the adoption of minimally invasive techniques in pancreatic surgery has progressed more slowly than in other surgical fields. Recent research indicates that, once the learning curve is overcome, laparoscopic minimally invasive pancreatic surgery is both safe and viable, and can yield treatment outcomes comparable to those of open surgery[9,10]. Nonetheless, this report presents two cases of pancreatic IOPN that were treated with laparoscopic surgery.
Case 1: The patient was a 60-year-old female who was admitted to the hospital on May 1, 2024 due to a pancreatic mass discovered half a month prior.
Case 2: A 61-year-old female was admitted to the hospital on March 11, 2024 due to a pancreatic mass found 5 days prior.
Case 1: Two weeks before admission, the patient underwent surgical treatment for a lower limb fracture at another hospital. During that period, a chest computed tomography (CT) scan revealed an occupying lesion in the body and tail of the pancreas. A subsequent abdominal enhanced CT scan suggested an occupying lesion in the body of the pancreas, possibly a neuroendocrine tumor. The patient attended our hospital for further evaluation and treatment. No symptoms such as abdominal pain, bloating, nausea, vomiting, or dizziness was found.
Case 2: Five days before admission, the patient was being treated at another hospital for a urinary tract infection. B-ultrasound indicated a hypoechoic lesion in front of the right side of the pancreas. The patient showed no discomfort such as abdominal pain or bloating.
Case 1: The patient had no history of abdominal surgery, smoking, or alcohol consumption.
Case 2: She had a history of cholecystectomy and hypertension.
All other personal and family medical history was noncontributory.
Case 1: No jaundice of the skin or sclera. The abdomen was soft, with no tenderness or rebound tenderness in the upper abdomen. No abdominal mass was palpable. Murphy’s sign was negative and shifting dullness was negative. Bilateral renal punch was negative. The left lower limb was fixed with a brace.
Case 2: No jaundice of the skin or sclera. The abdomen was soft, without tenderness or rebound tenderness in the upper abdomen. No abdominal mass was palpable. Murphy’s sign was negative. Shifting dullness was negative. Bilateral renal punch was negative.
Case 1: Blood routine, biochemistry, tumor markers, etc., were all normal.
Case 2: Blood routine and biochemistry results were normal. Carbohydrate antigen (CA) 19-9 was 204.91 U/mL.
Case 1: Abdominal enhanced CT indicated a mass in the tail of the pancreas. The mass had a clear boundary and was approximately 30 mm in size. A neuroendocrine tumor was highly suspected. Abdominal enhanced magnetic resonance (MR) indicated a cystic-solid mass in the pancreatic tail, with an uneven T2 signal. The diffusion weighted imaging signal was high and the apparent diffusion coefficient showed partially low-level signal. The boundary was clear, with a maximum section size of about 35 mm × 32 mm. After contrast enhancement, the solid portion of the mass was enhanced. The lesion appeared to be connected to the pancreatic duct (Figure 1A-D). MR imaging suggested a high possibility of a low-grade malignant tumor.
Case 2: Abdominal enhanced CT suggested an occupying lesion in the pancreaticoduodenal space, possibly indicating a gastrointestinal stromal tumor. Abdominal enhanced MR revealed an occupying lesion in the head of the pancreas, raising suspicion of a solid pseudopapillary tumor or neuroendocrine tumor (Figure 1E-H). Moreover, preoperative endoscopic ultrasonography also suggested an occupying lesion in the head of the pancreas. Puncture pathology revealed mucinous substances, inflammatory cells, histiocytes, and a small amount of glandular epithelium.
Postoperative routine pathology indicated an IOPN in the body and tail of the pancreas with high-grade intraepithelial neoplasia. The immunohistochemistry results revealed positive MUC6, positive MUC5AC, negative MUC2, and positive hepatocyte paraffin 1 (HepPar1) and cluster of differentiation (CD) 117 (Figure 2A-F).
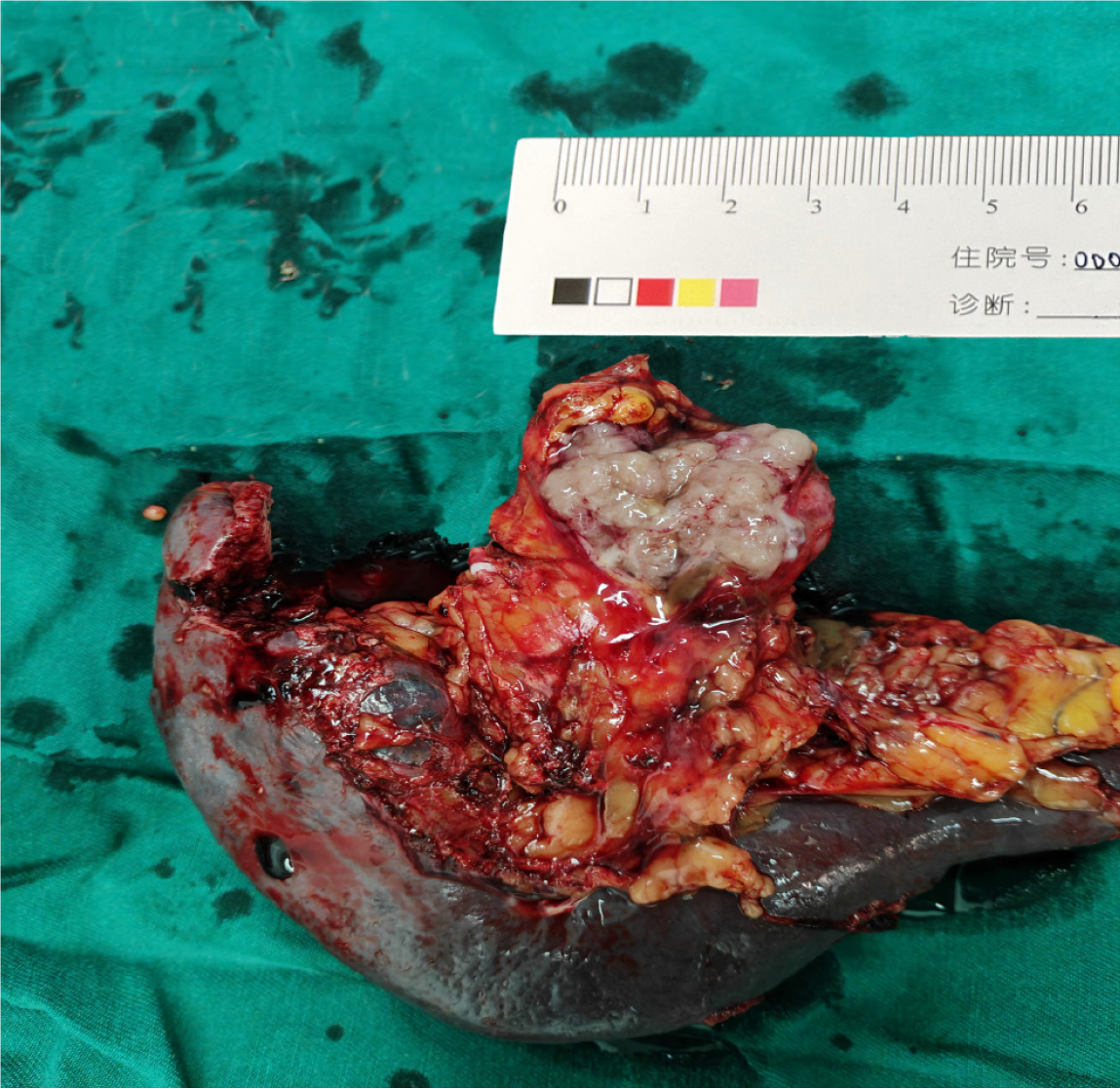
After completing preoperative preparations, the patient underwent laparoscopic distal pancreatectomy and splenectomy. Intraoperative rapid pathology indicated pancreatic mucinous adenocarcinoma. Consequently, a residual pancreatic resection was performed on the left side of the portal vein, accompanied by retroperitoneal lymph node dissection. Upon dissecting the specimen, the tumor was found to be located in the tail of the pancreas, showing a grayish-white appearance with a maximum diameter of 3.5 cm (Figure 3). The operation lasted for 342 minutes and the intraoperative blood loss was approximately 100 mL.
After meticulously completing preoperative preparations, laparoscopic pancreaticoduodenectomy was performed on March 20, 2024. The tumor in the head of the pancreas was measured at 4.5 cm × 3.6 cm × 1.8 cm during postoperative pathology. The operation lasted for 431 minutes with an intraoperative blood loss of approximately 50 mL.
The patient was discharged on postoperative day 8 with an excellent postoperative course. No recurrence was observed during the 8-month postoperative follow-up. The patient is entirely competent to engage in normal life and work activities, and expresses profound contentment with the therapeutic efficacy. Considering the potential for recurrence or malignant transformation of pancreatic IOPN, we recommend that patients undergo an abdominal enhanced CT examination every six months after surgery. Additionally, we will conduct continuous tracking.
The patient exhibited an excellent postoperative course and was discharged on postoperative day 24. Two months after the surgery, the tumor marker CA19-9 dropped to the normal level. No recurrence was observed during the 10-month postoperative follow-up. The patient is entirely competent to engage in normal life and work activities, and expresses profound contentment with the therapeutic efficacy. Considering the potential for recurrence or malignant transformation of pancreatic IOPN, we recommend that patients undergo an abdominal enhanced CT examination every six months after surgery. Additionally, we will conduct continuous tracking.
IOPN shows similar clinical manifestations to those of IPMN, including abdominal pain, bloating, and jaundice. Approximately 30% of patients are asymptomatic, and the tumor is detected incidentally during imaging examination[11,12]. In terms of laboratory tests, no specific markers indicate the presence of this disease, although a few patients may show elevated CEA and CA19-9 Levels[13-15].
IOPN is more common in middle-aged and elderly individuals and can occur in any part of the pancreas, with a higher prevalence in the pancreatic head. IOPN demonstrates varying imaging characteristics and may present as cystic, cystic-solid, or solid lesions, which is similar to IPMN. Currently, only a few reports have explored the imaging differentiation between IOPN and other pancreatic tumors. Therefore, preoperative diagnosis remains extremely challenging. Wang et al[16] summarized 66 cases of IOPN reported in the literature. Among these, only 8 cases were correctly diagnosed as IOPN preoperatively, 14 cases were diagnosed as IPMN, 13 as pancreatic ductal adenocarcinoma (PDAC), 10 as mucinous cystic neoplasm, and 1 as a neuroendocrine tumor. Most patients with IOPN undergo surgical treatment as they have been diagnosed with conditions like IPMN and PDAC. Surgical methods include pancreaticoduodenectomy, duodenum-preserving pancreatic head resection, distal pancreatectomy and splenectomy, total pancreatectomy, etc[17]. In this report, for case 1, the preoperative diagnosis was considered neuroendocrine tumor? For case 2, the preoperative diagnoses were considered to be stromal tumor? Solid pseudopapillary tumor? Neuroendocrine tumor? This is similar to the previously reported results. Interestingly, all the suspected diseases under consideration required surgical resection, which made the surgical indications definitely clear. Notably, positron emission tomography (PET)-CT may facilitate the preoperative diagnosis of IOPN. Literature reports that the standard uptake value of IOPN on PET-CT is much higher than that of general pancreatic cancer. Therefore, PET-CT is a potential diagnostic method for preoperative differentiation[18], which may be attributed to eosinophilic cells being highly active in metabolism, and non-pancreatic oncocytomas also showing strong positive manifestations on PET-CT[19,20]. However, it’s a pity that neither of the two patients in this report underwent a complete PET-CT examination before surgery. In the future, for similar pancreatic tumor patients with unclear preoperative diagnoses, a PET-CT examination can be completed. In the future, we will devote more efforts to studying the role and value of PET-CT in the diagnosis of IOPN. If the preoperative diagnosis can be made more definite, both doctors and patients will be able to face the situation more calmly.
Currently, the differentiation between IOPN and IPMN mainly relies on pathological examinations. Adsay et al[1] reported that IOPN has an average diameter of 6.0 cm (1.6 cm-15.0 cm). Tumors usually grow in or are connected to the dilated pancreatic duct, presenting as a cystic or cystic-solid lesion. Grayish-white or grayish-brown, soft, and fragile papillae can be seen in the cyst, or may also appear as grayish-white nodules attached to the wall. However, if the tumor cells grow in a solid form, the dilated pancreatic duct structure may not be obvious, hindering the identification of intraductal lesions through imaging and macroscopic observation. In addition, IOPN generally produces less mucus than other types of IPMN[21]. Under low magnification, a significant difference between IOPN and IPMN is that most tumor cells contain abundant eosinophilic granules in their cytoplasm. However, the mucus differentiation of IOPN is often not obvious, and goblet cells containing mucus or epithelial luminal spaces containing mucus are commonly observed between tumor cells, forming a sieve-like structure. The nuclei of IOPN tumors are round or oval in shape, displaying obvious atypia, such as mitotic figures, nuclear pleomorphism, coarse chromatin, and prominent nucleoli, etc. Based on its complex structure (complex branching papillae) and nuclear atypia, all IOPN are accompanied by high-grade dysplasia, which is not found in IPMN[22]. Approximately 30% of IOPN may be accompanied by associated invasive carcinomas. The invasive carcinomas have the characteristics of eosinophilic cells similar to those of the intraductal tumors[23]. Wang et al[16] summarized the pathological characteristics of 66 patients with pancreatic IOPN from 24 published literatures. The results revealed that all 66 patients had severe dysplasia or invasive carcinoma; specifically, 59% showed severe dysplasia and 41% showed invasive carcinoma. In this report, case 2 demonstrated invasive carcinoma components. IOPN generally exhibits an excellent prognosis. Notably, even in cases where it is complicated by invasive carcinoma, the 5-year survival rate remains 100%, and the 10-year survival rate stands at 94%. When performing surgical resection, it is necessary to ensure a negative surgical margin to reduce the risk of recurrence. Intriguingly, recurrence can sometimes occur as late as 10 years following the mass resection. However, even if such a recurrence does transpire, the prognosis following re-operation remains highly promising[7]. The two patients in this report were followed up for only 8 months and 10 months respectively. The follow-up period is still too short. In the future, we will continue to follow up these two patients. It is recommended to conduct an enhanced abdominal CT examination every six months.
Similar to IPMN, IOPN typically shows positive results for cytokeratin 7, cytokeratin 8, cytokeratin 18 and cytokeratin 19. However, positive MUC6 and MUC5AC, and negative for MUC2 are also observed, which can be used to differentiate between IPMN and IOPN. Meanwhile, HepPar1 and CD117 may also show positive expressions in IOPN[24,25]. In these two cases, immunohistochemical staining revealed positive expressions of MUC6 and MUC5AC, while the expression of MUC2 was negative, which helped to rule out the possibility of other tumors and further confirmed the diagnosis of IOPN. In case 1, both HepPar1 and CD117 exhibited positive expressions. In case 2, immunohistochemistry results showed positive results for Syn and CK-P, which were immunohistochemical markers representing neuroendocrine differentiation and microinvasion respectively.
The PubMed database was searched, revealing a case diagnosed with IOPN of the pancreas with prominent invasion that was treated with laparoscopic pancreatosplenectomy, proximal gastrectomy, and transverse colectomy. This was the first and only case reported by Nomaru et al[26] in 2023, and all the other reported cases underwent opensurgeries. Previous reports have focused on the pathological features and diagnosis of IOPN. Compared with open surgery, laparoscopic pancreatic surgery has advantages such as less intraoperative bleeding and faster postoperative recovery[27,28]. Moreover, it will not augment the incidence of complications, including postoperative infection and intra-abdominal bleeding[29]. However, whether these advantages are applicable to pancreatic IOPN remains unknown, and this requires research with a larger sample size.
The pancreas is located deep in the body, surrounded by numerous large blood vessels, and often involves reconstruction after resection, which makes the surgery highly challenging. Therefore, the application of laparoscopic surgery in pancreatic operations lagged behind that of organs such as the gastrointestinal tract and liver. Previously, we had an adequate reserve of laparoscopic techniques and were proficient in performing laparoscopic radical gastrectomy and liver cancer resection. Without sufficient technical reserves, one should refrain from carrying out laparoscopic pancreatic surgery. At the same time, laparoscopy offers an excellent field of view and magnification effect, enabling more precise operations. Compared with open surgery, laparoscopic surgery may achieve more thorough lymph node dissection and a higher R0 resection rate[30,31].
The current report presents two cases of patients with pancreatic IOPN treated by laparoscopic surgery, including one case of laparoscopic pancreaticoduodenectomy and one case of laparoscopic distal pancreatectomy and splenectomy. Both of our patients were discharged smoothly after surgery without complications such as bleeding, pancreatic fistula, or reoperation. The patients and their family members were very satisfied with the treatment outcomes. The two patients had a good prognosis and developed no recurrence during the follow-up period. Therefore, laparoscopic surgery for pancreatic IOPN is a safe and effective method. Nevertheless, further research is required to confirm these findings. Therefore, we recommend that hospitals with rich experience in laparoscopic surgeries can attempt laparoscopic surgery for pancreatic IOPN.
However, this study still has some limitations. The main limitation is that only two patients were included, resulting in a small sample size, which prevents relevant statistical analysis. We hope to include more IOPN patients in future studies to conduct further research on the differentiation of IOPN from other pancreatic tumors, as well as studies on the laparoscopic and open surgical treatments for IOPN.
IOPN of the pancreas is extremely rare. The clinical manifestations, laboratory examinations, and imaging findings are all atypical, making preoperative diagnosis difficult. Thorough histopathological and molecular analysis is essential for an accurate diagnosis. Surgery is the preferred treatment, and while a certain proportion of cases may be associated with invasive cancer, the surgical prognosis remains favorable. Neither of the two patients had perioperative complications or recurrence. Laparoscopic surgery can be considered as one of the treatment options for IOPN of the pancreas. Certainly, this requires verification with more cases and long-term follow-up.
We’d like to thank Dr. Shi HQ and Dr. Wang MY for processing the pathological images.
| 1. | Adsay NV, Adair CF, Heffess CS, Klimstra DS. Intraductal oncocytic papillary neoplasms of the pancreas. Am J Surg Pathol. 1996;20:980-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 168] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 2. | Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, Washington KM, Carneiro F, Cree IA; WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3203] [Cited by in RCA: 2873] [Article Influence: 478.8] [Reference Citation Analysis (5)] |
| 3. | Esposito I, Häberle L, Yavas A, Kraft A, Gaida MM. [Intraductal neoplasms of the pancreas]. Pathologe. 2021;42:472-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Innocenti L, Rotondo MI, Donati F, Boggi U, Campani D. Intraductal oncocytic papillary neoplasm (IOPN): two case reports and review of the literature. Transl Cancer Res. 2023;12:663-672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 5. | Basturk O, Tan M, Bhanot U, Allen P, Adsay V, Scott SN, Shah R, Berger MF, Askan G, Dikoglu E, Jobanputra V, Wrzeszczynski KO, Sigel C, Iacobuzio-Donahue C, Klimstra DS. The oncocytic subtype is genetically distinct from other pancreatic intraductal papillary mucinous neoplasm subtypes. Mod Pathol. 2016;29:1058-1069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 6. | Marchegiani G, Mino-Kenudson M, Ferrone CR, Warshaw AL, Lillemoe KD, Fernández-del Castillo C. Oncocytic-type intraductal papillary mucinous neoplasms: a unique malignant pancreatic tumor with good long-term prognosis. J Am Coll Surg. 2015;220:839-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 7. | Wang T, Askan G, Adsay V, Allen P, Jarnagin WR, Memis B, Sigel C, Seven IE, Klimstra DS, Basturk O. Intraductal Oncocytic Papillary Neoplasms: Clinical-Pathologic Characterization of 24 Cases, With An Emphasis on Associated Invasive Carcinomas. Am J Surg Pathol. 2019;43:656-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 8. | Pfister M, Probst P, Müller PC, Antony P, Klotz R, Kalkum E, Merz D, Renzulli P, Hauswirth F, Muller MK. Minimally invasive versus open pancreatic surgery: meta-analysis of randomized clinical trials. BJS Open. 2023;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 9. | Abu Hilal M, van Ramshorst TME, Boggi U, Dokmak S, Edwin B, Keck T, Khatkov I, Ahmad J, Al Saati H, Alseidi A, Azagra JS, Björnsson B, Can FM, D'Hondt M, Efanov M, Espin Alvarez F, Esposito A, Ferrari G, Groot Koerkamp B, Gumbs AA, Hogg ME, Huscher CGS, Ielpo B, Ivanecz A, Jang JY, Liu R, Luyer MDP, Menon K, Nakamura M, Piardi T, Saint-Marc O, White S, Yoon YS, Zerbi A, Bassi C, Berrevoet F, Chan C, Coimbra FJ, Conlon KCP, Cook A, Dervenis C, Falconi M, Ferrari C, Frigerio I, Fusai GK, De Oliveira ML, Pinna AD, Primrose JN, Sauvanet A, Serrablo A, Smadi S, Badran A, Baychorov M, Bannone E, van Bodegraven EA, Emmen AMLH, Giani A, de Graaf N, van Hilst J, Jones LR, Levi Sandri GB, Pulvirenti A, Ramera M, Rashidian N, Sahakyan MA, Uijterwijk BA, Zampedri P, Zwart MJW, Alfieri S, Berti S, Butturini G, Di Benedetto F, Ettorre GM, Giuliante F, Jovine E, Memeo R, Portolani N, Ruzzenente A, Salvia R, Siriwardena AK, Besselink MG, Asbun HJ; Collaborators. The Brescia Internationally Validated European Guidelines on Minimally Invasive Pancreatic Surgery (EGUMIPS). Ann Surg. 2024;279:45-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 74] [Article Influence: 37.0] [Reference Citation Analysis (1)] |
| 10. | Fung G, Sha M, Kunduzi B, Froghi F, Rehman S, Froghi S. Learning curves in minimally invasive pancreatic surgery: a systematic review. Langenbecks Arch Surg. 2022;407:2217-2232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 11. | Jyotheeswaran S, Zotalis G, Penmetsa P, Levea CM, Schoeniger LO, Shah AN. A newly recognized entity: intraductal "oncocytic" papillary neoplasm of the pancreas. Am J Gastroenterol. 1998;93:2539-2543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Munley J, Chang MD, Thomas RM. Intraductal Oncocytic Papillary Neoplasm of the Pancreas. J Gastrointest Surg. 2021;25:319-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Nakaya M, Nakai Y, Takahashi M, Fukukura Y, Sato K, Kameda A, Tashiro Y, Kageyama S, Sofue K, Nakano T, Yoshimitsu K, Marugami N, Takeyama N, Tanaka M, Hasegawa K, Watadani T. Intraductal oncocytic papillary neoplasm of the pancreas: clinical and radiological features compared to those of intraductal papillary mucinous neoplasm. Abdom Radiol (NY). 2023;48:2483-2493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 14. | Assarzadegan N, Babaniamansour S, Shi J. Updates in the Diagnosis of Intraductal Neoplasms of the Pancreas. Front Physiol. 2022;13:856803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (5)] |
| 15. | Paolino G, Basturk O, Esposito I, Hong SM, Brosens LA, Tarcan Z, Wood LD, Gkountakos A, Omori Y, Mattiolo P, Ciulla C, Marchegiani G, Pea A, Bevere M, De Robertis R, D'Onofrio M, Salvia R, Cheng L, Furukawa T, Scarpa A, Adsay V, Luchini C. Comprehensive Characterization of Intraductal Oncocytic Papillary Neoplasm of the Pancreas: A Systematic and Critical Review. Mod Pathol. 2024;37:100554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 16. | Wang YZ, Lu J, Jiang BL, Guo JC. Intraductal oncocytic papillary neoplasm of the pancreas: A systematic review. Pancreatology. 2019;19:858-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Jabłońska B, Braszczok Ł, Szczęsny-Karczewska W, Dubiel-Braszczok B, Lampe P. Surgical treatment of pancreatic cystic tumors. Pol Przegl Chir. 2017;89:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (3)] |
| 18. | Ghaneh P, Hanson R, Titman A, Lancaster G, Plumpton C, Lloyd-Williams H, Yeo ST, Edwards RT, Johnson C, Abu Hilal M, Higginson AP, Armstrong T, Smith A, Scarsbrook A, McKay C, Carter R, Sutcliffe RP, Bramhall S, Kocher HM, Cunningham D, Pereira SP, Davidson B, Chang D, Khan S, Zealley I, Sarker D, Al Sarireh B, Charnley R, Lobo D, Nicolson M, Halloran C, Raraty M, Sutton R, Vinjamuri S, Evans J, Campbell F, Deeks J, Sanghera B, Wong WL, Neoptolemos JP. PET-PANC: multicentre prospective diagnostic accuracy and health economic analysis study of the impact of combined modality 18fluorine-2-fluoro-2-deoxy-d-glucose positron emission tomography with computed tomography scanning in the diagnosis and management of pancreatic cancer. Health Technol Assess. 2018;22:1-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 86] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 19. | Blake MA, McKernan M, Setty B, Fischman AJ, Mueller PR. Renal oncocytoma displaying intense activity on 18F-FDG PET. AJR Am J Roentgenol. 2006;186:269-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 20. | Kim DJ, Chung JJ, Ryu YH, Hong SW, Yu JS, Kim JH. Adrenocortical oncocytoma displaying intense activity on 18F-FDG-PET: a case report and a literature review. Ann Nucl Med. 2008;22:821-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 21. | Klöppel G, Basturk O, Schlitter AM, Konukiewitz B, Esposito I. Intraductal neoplasms of the pancreas. Semin Diagn Pathol. 2014;31:452-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 22. | Kallen ME, Naini BV. Intraductal Oncocytic Papillary Neoplasms of the Pancreas. Arch Pathol Lab Med. 2016;140:992-996. [PubMed] [DOI] [Full Text] |
| 23. | Kang MJ, Lee KB, Jang JY, Han IW, Kim SW. Evaluation of clinical meaning of histological subtypes of intraductal papillary mucinous neoplasm of the pancreas. Pancreas. 2013;42:959-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Basturk O, Chung SM, Hruban RH, Adsay NV, Askan G, Iacobuzio-Donahue C, Balci S, Zee SY, Memis B, Shia J, Klimstra DS. Distinct pathways of pathogenesis of intraductal oncocytic papillary neoplasms and intraductal papillary mucinous neoplasms of the pancreas. Virchows Arch. 2016;469:523-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 25. | Mattiolo P, Hong SM, Paolino G, Rusev BC, Marchegiani G, Salvia R, Andrianello S, Capelli P, Piccoli P, Parolini C, Scarpa A, Lawlor RT, Luchini C. CD117 Is a Specific Marker of Intraductal Papillary Mucinous Neoplasms (IPMN) of the Pancreas, Oncocytic Subtype. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 26. | Nomaru R, Ishida Y, Kitaguchi T, Koga T, Tsuchiya N, Nakashima R, Sato K, Hamada Y, Hasegawa S, Hirai F. Preoperatively diagnosed intraductal oncocytic papillary neoplasm of the pancreas with prominent invasion: a case report. Clin J Gastroenterol. 2023;16:772-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Reis PCA, Bittar V, Almirón G, Schramm AJ, Oliveira JP, Cagnacci R, Camandaroba MPG. Laparoscopic Versus Open Pancreatoduodenectomy for Periampullary Tumors: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Gastrointest Cancer. 2024;55:1058-1068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 28. | Xia N, Li J, Wang Q, Huang X, Wang Z, Wang L, Tian B, Xiong J. Safety and effectiveness of minimally invasive central pancreatectomy versus open central pancreatectomy: a systematic review and meta-analysis. Surg Endosc. 2024;38:3531-3546. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 29. | Hong C, Liu W. Effect of laparoscopic and open distal pancreatectomy on postoperative wound complications in patients with pancreatic cancer: A meta-analysis. Int Wound J. 2024;21:e14708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 30. | Hariri HM, Perez SB, Turner KM, Wilson GC. Minimally Invasive Pancreas Surgery: Is There a Benefit? Surg Clin North Am. 2024;104:1083-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 31. | Riviere D, van den Boezem PB, Besselink MG, van Laarhoven CJ, Kooby DA, Vollmer CM, Davidson BR, Gurusamy KS. Minimally invasive versus open pancreatoduodenectomy in benign, premalignant, and malignant disease. Cochrane Database Syst Rev. 2024;7:CD014017. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/