Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.104726
Revised: January 28, 2025
Accepted: February 27, 2025
Published online: April 27, 2025
Processing time: 89 Days and 13.7 Hours
Rectal cancer has increased in incidence, and surgery remains the cornerstone of multimodal treatment. Pelvic anatomy, particularly a narrow pelvis, poses cha
To investigate the relationship between radiologically assessed pelvic anatomy and surgical outcomes as well as the impact on local recurrence following rectal cancer surgery.
We retrospectively analyzed 107 patients with rectal adenocarcinoma treated with elective rectal surgery between January 1, 2017, and September 1, 2022. Pel
When patients were categorized based on CT measurements into narrow and normal/wide pelvis groups, a significant association was observed with male sex, and a lower BMI was more common in the narrow pelvis group (P = 0.002 for both). A significant association was found between a narrow pelvic structure, indicated by low PCI, and increased surgical morbidity (P = 0.049). Advanced age (P = 0.003) and male sex (P = 0.020) were significantly correlated with higher surgical morbidity. Logistic regression analysis identified four parameters that were significantly correlated with local recurrence: older age, early perioperative readmission, longer operation time, and a lower number of dissected lymph nodes (P < 0.05). However, there were no significant differences between the narrow and normal/wide pelvis groups in terms of the operation time, estimated blood loss, or overall local recurrence rate (P > 0.05).
MRI-based pelvimetry may be valuable in predicting surgical difficulty and morbidity in rectal cancer surgery, as indicated by the PCI. The observed correlation between low PCI and increased surgical morbidity suggests the potential importance of a preoperative MRI-based pelvimetric evaluation. In contrast, CT-based pelvimetry did not show significant differences in predicting surgical outcomes or cancer recurrence, indicating that the utility of pelvimetry alone may be limited in these respects.
Core Tip: This study investigates the role of pelvimetry in predicting surgical and oncological outcomes in rectal cancer patients. Using computed tomography (CT)-based pelvic inlet area measurements and magnetic resonance imaging-based pelvic cavity index (PCI), the study identifies lower PCI as a significant predictor of surgical morbidity. However, CT-based pelvimetry showed no significant correlation with operation time or local recurrence. Multivariate analysis highlights age, sex, and PCI as independent risk factors for morbidity. While pelvimetry is useful for assessing surgical complexity, its value in predicting oncological outcomes remains limited, warranting further validation through larger multicenter studies.
- Citation: Ay OF, Firat D, Özçetin B, Ocakoglu G, Ozcan SGG, Bakır Ş, Ocak B, Taşkin AK. Role of pelvimetry in predicting surgical outcomes and morbidity in rectal cancer surgery: A retrospective analysis. World J Gastrointest Surg 2025; 17(4): 104726
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/104726.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.104726
The increasing incidence of rectal cancer highlights its importance as a major health concern, with surgery being the cornerstone of multimodal treatment[1,2]. Total mesorectal excision (TME) plays a critical role in rectal cancer surgery by significantly improving survival rates and reducing the risk of local recurrence[3]. However, ensuring optimal surgical and oncological outcomes can be challenging, particularly in patients with a narrow pelvis, as it is a recognized risk factor for increased postoperative complications. This is primarily because of the technical difficulties associated with performing TME in a limited pelvic cavity[4].
Though it is not widely adopted in routine clinical practice due to time constraints, pelvimetry provides valuable insights into bony pelvic anatomy and its relationship with surgical and oncological outcomes[5,6]. Pelvimetry can be performed using either magnetic resonance (MR) imaging (MRI) or computed tomography (CT) scans, which offer both 2D and 3D imaging capabilities. Although CT pelvimetry provides the convenience of simple assessment and quick and thorough scanning, MR pelvimetry provides a superior evaluation of adjacent structures and the mesorectum because of its heightened sensitivity to soft tissues[7].
The structure of the pelvic bone in two dimensions can be determined by measuring various parameters such as the interspinous distance, transverse inlet diameter, and midsagittal diameter[8]. These measures can be approximated in 3D using certain calculations and software[9]. There is growing interest in this field, as more studies have highlighted its potential benefits in surgical planning and risk assessment[10].
In this study, we employed two different imaging techniques by incorporating both CT-based- and MRI-based pelvimetric measurements. The CT-based method provides 2D analysis of the pelvic inlet area[11], allowing for a basic and effective assessment of the pelvic structure. MRI-based pelvimetric assessments, specifically the pelvic cavity index (PCI), were used to provide a detailed 3D evaluation of the pelvic structure. PCI was chosen because of its ability to integrate multiple anatomical measurements, offering a more comprehensive understanding of pelvic anatomy. This method has been validated in previous studies for its relevance in assessing surgical complexity, particularly in confined pelvic spaces, and for its predictive value for surgical and oncological outcomes[12]. By employing PCI, we aimed to enhance the precision of pelvic assessment and address the limitations of two-dimensional measurements.
By integrating both imaging modalities, our study sought to address gaps in the literature, as most reports relied on either CT or MRI alone. We aimed to provide a more robust understanding of how pelvic anatomy influences surgical difficulty, postoperative morbidity, and oncological outcomes of rectal cancer surgery. Ultimately, our findings could inform clinical practice, help optimize surgical planning, and improve patient outcomes.
The primary objective of our study was to investigate the relationship between radiologically assessed pelvic anatomy and surgical outcomes (perioperative morbidity and mortality, operation time, estimated blood loss, length of hospital stay, pelvic infection, local recurrence, anastomotic leak, readmission, estimated blood loss) in patients with rectal cancer. The secondary objective was to evaluate the relationship between the pelvic structure and local recurrence following rectal cancer surgery.
We hypothesized that rectal cancer patients with a narrow pelvic structure, as assessed by pelvimetric analysis, would have poorer surgical outcomes and lower oncological surgical quality. The oncological surgical quality in our study was evaluated based on the total number of dissected lymph nodes reported in routine pathology reports.
Our study retrospectively analyzed cases of rectal surgery (low anterior resection, abdominoperineal resection) performed electively as open or laparoscopic procedures in patients diagnosed with "rectal adenocarcinoma" at the General Surgery Clinic of SBÜ Bursa Yüksek İhtisas Training and Research Hospital between January 01, 2017 and September 01, 2022. A total of 107 patients were included in this study. The patients included in the study were over 18 years of age and had clinical, pathological, and surgical data accessible through the hospital's data system and archives. For radiological parameters, patients with available CT or pelvic MRI data were included.
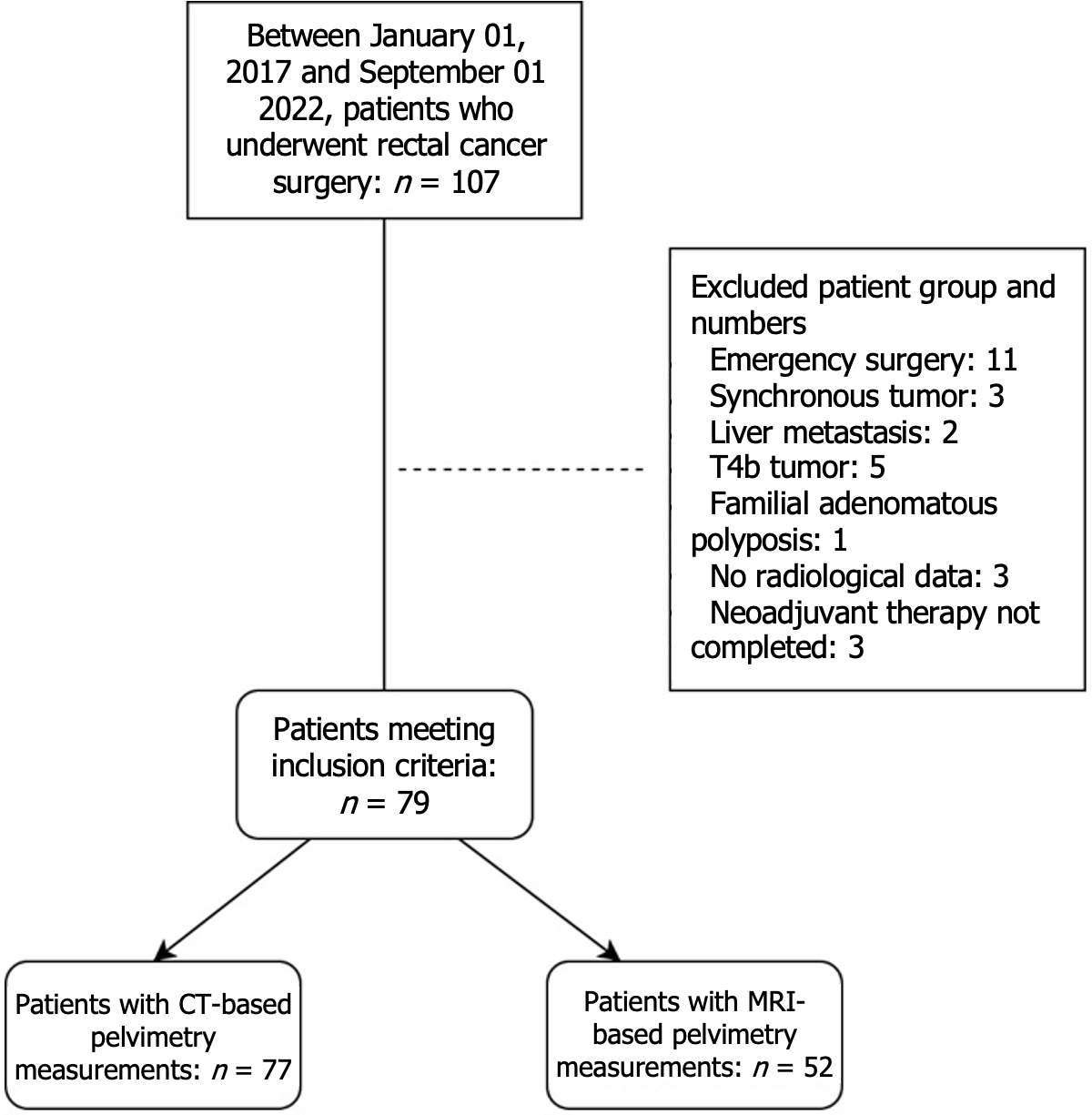
The exclusion criteria were designed to eliminate pathologies that could interfere with the establishment of a direct relationship between pelvic structure and surgical or oncological outcomes. This approach was adopted to ensure the homogeneity of the study population and was consistent with the methodologies described in the literature. The exclusion criteria included patients who underwent emergency Hartmann's procedure (n = 11), patients who underwent surgery for synchronous tumors (n = 3), patients who did not complete neoadjuvant therapy (n = 3), patients with adjacent organ invasion (T4b tumor, n = 5), patients with distant metastasis (n = 2), patients who underwent surgery for familial adenomatous polyposis (n = 1), and patients without accessible radiological data (n = 3; Figure 1). Preoperative, operative, and postoperative data were obtained from the hospital data system and the patient files.
All patients diagnosed with rectal adenocarcinoma in our clinic were evaluated for treatment planning by a multidisciplinary tumor board consisting of the Departments of General Surgery, Oncology, Radiology, Pathology, and Nuclear Medicine. Patients were evaluated using complete colonoscopy, routine blood tests (including complete blood count, biochemistry, and tumor markers such as CEA, CA 19-9, CA 15-3, and radiological examinations (thoracic and abdominal CT and pelvic MRI). In our study, the rectal tumor localization was classified as upper (10-15 cm), middle (5-10 cm), or lower (0-5 cm). Patients were staged and treated according to the National Comprehensive Cancer Network colorectal cancer TNM guidelines. Neoadjuvant chemoradiotherapy, managed by the oncology and radiation oncology departments, consisted of two weeks of radiotherapy and two cycles of 5-fluorouracil infusion. The proposed treatment plan was implemented with patient consent; however, the tumor board did not convene for 9 mo due to quarantine measures during the pandemic, and patient management during this period was determined through consultations.
Surgical planning was based on the individual experience of the primary surgeon, tumor localization, preoperative evaluation, and preference for laparoscopic or open procedures. The rectal cancer surgeries evaluated in this study included open low anterior resection, laparoscopic low anterior resection, and abdominoperineal resection. TME was performed in all surgical cases using a standardized laparoscopic technique for each surgeon using the double-stapling technique (dividing the rectum with a linear stapler and creating anastomosis with a circular stapler).
Patient demographics and characteristic parameters [age, sex, American Society of Anesthesiologists Score (ASA), body mass index (BMI)], neoadjuvant status, operative data [open/laparoscopic low anterior resection, miles operation, ileostomy status, tumor localization (upper, middle, lower)], pathological results (tumor size, mesorectum status, T-N stage, pathological stage, lymph node count, lymphovascular invasion), surgical outcomes (operative time, estimated blood loss, hospital stay, pelvic infection, local recurrence, anastomotic leak, readmission, morbidity, and mortality), radiological pelvic assessment data (pelvic inlet area measured by CT and PCI by MRI), and postoperative follow-up data (follow-up duration, adjuvant therapy, disease-free survival, overall survival, and recurrence) were collected.
Surgical difficulty was assessed by evaluating operation time, estimated blood loss, length of hospital stay, and perioperative morbidity. The assessment of specimen quality was determined by lymph node count, as the status of the mesorectum was not consistently included in the pathology reports. Due to the low mortality rate, it was not possible to perform statistical analysis on overall survival and disease-free survival. Oncological outcomes were assessed based on local recurrence.
Pelvic infection was defined as the presence of a collection in the pelvic region and elevated inflammatory parameters without anastomotic leakage. Local recurrence was defined as any recurrent tumor growth in the pelvic or perineal region as confirmed by clinical, radiological, or pathological assessments without time restriction. Pathological variables were obtained from routine pathology reports; however, mesolectal status" was not consistently reported during the study period and was thus not included as a variable in the statistical analysis.
Operative morbidity, as defined in the literature, refers to temporary or permanent disorders that occur between the day of surgery and beginning of data collection. In our study, the postoperative morbidity within 30 days was used as a variable. Surgical mortality, as defined in the literature, refers to deaths occurring within the first 30 days postoperatively regardless of the cause (in-hospital or out-of-hospital). Early recurrence was defined as recurrence at the surgical site or distant organ metastasis within the first year after the surgery.
Two different methods were used for pelvimetric analysis in the included patients: CT-based- and MRI-based. The definition of "narrow pelvis" was based on the following threshold values for the CT-based method and quantitative analysis for the MRI-based method.
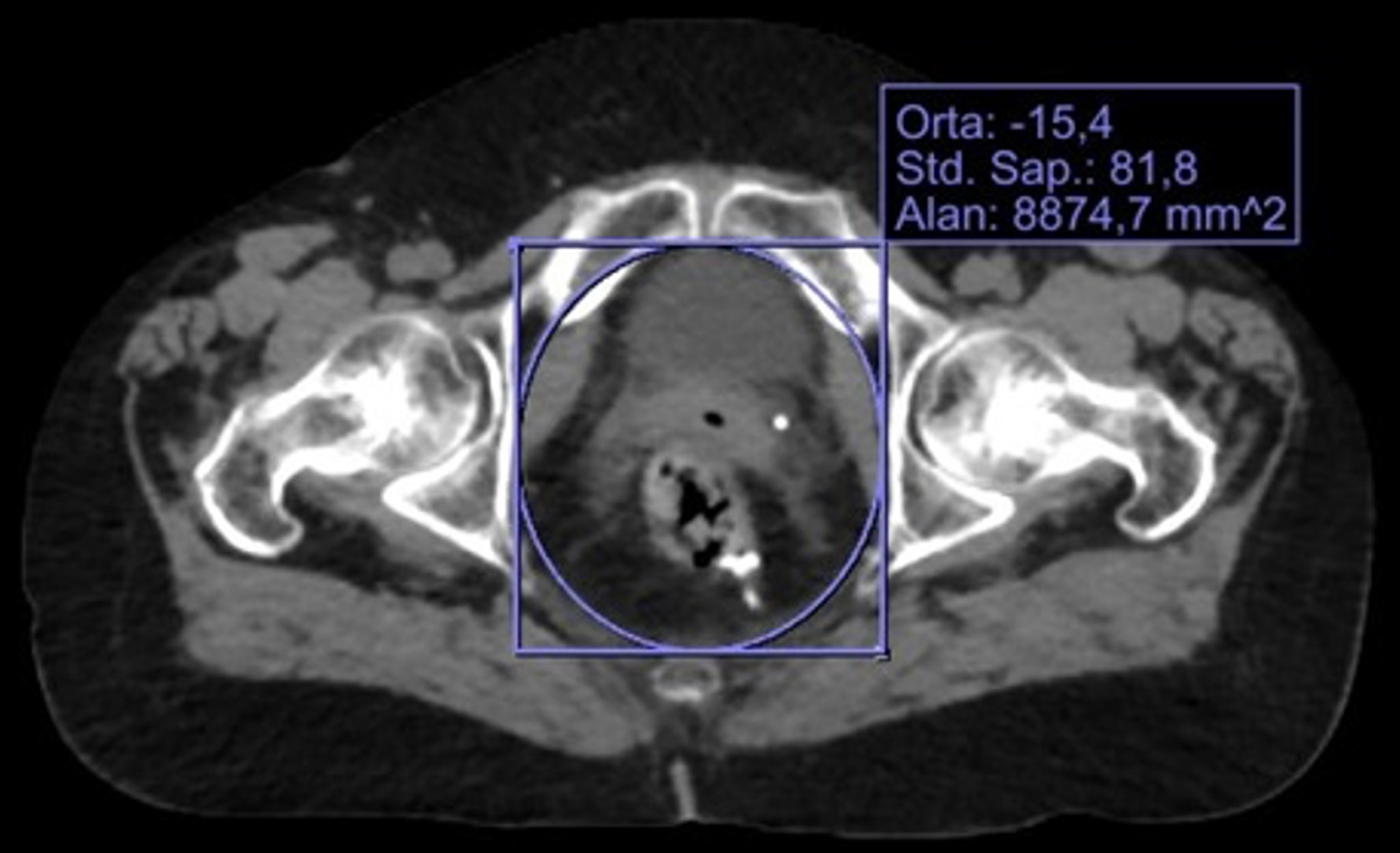
CT-based pelvimetry: Radiological assessment of the pelvic structure using CT (CT pelvimetry) was performed using a multidetector CT scanner (Revolution Maxima, GE Health Care, United States), and CT datasets were analyzed using the Sekans Ankara Picture Archiving and Communication System (Figure 2). Pelvic inlet area measurement and classification of pelvic structure as narrow or wide were conducted using the automatic measurement method described by Noh et al[6], with the threshold value (8800 mm²) they determined. To determine the area, the anteroposterior (AP) pelvic diameter (distance from the posterior border of the symphysis pubis to the tip of the coccyx) and the transverse pelvic diameter (distance between the inner borders of the lateral bony pelvis perpendicular to the AP diameter) were considered. Patients with a pelvic area < 8800 mm² were classified as having a narrow pelvis, whereas those with an area ≥ 8800 mm² were classified as having a normal/wide pelvis (Figure 2). CT Pelvimetry reflects the characteristics of the pelvic structure based on a 2D approach.
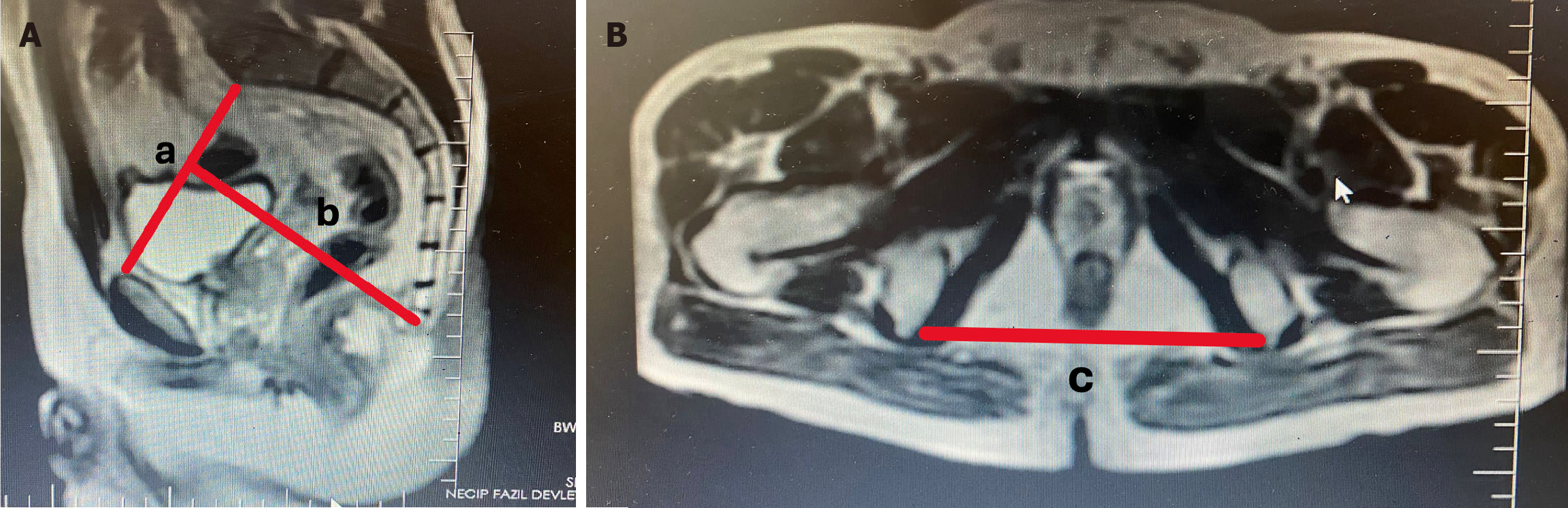
MRI-based pelvimetry and PCI: Pelvic MRI examinations were performed using Philips Gyroscan NT15 Intera 1.5. MRI-based pelvimetric measurements were performed in sagittal and axial sections of the T2 phase with the support of a radiology clinic. The PCI, which reflects the 3D character of the pelvic structure, was calculated using the formula (pelvic inlet × interspinous distance/pelvic depth; Figure 3)[9]. PCI was first described by Hong et al[13] in 2009 to assess the impact of the pelvic structure on robot-assisted laparoscopic prostatectomy. According to this calculation, a wider and shallower pelvic structure will have a higher PCI (cm) value, whereas a narrow and deep pelvis will have a lower PCI value.
Methods for data analysis: The Shapiro-Wilk test was used to determine whether continuous variables followed a normal distribution. Mean and standard deviation values were provided for variables with a normal distribution, whereas median, minimum, and maximum values were provided for variables that did not follow a normal distribution. Based on the results of the normality test, comparisons between two groups were made using the Mann-Whitney U test and independent t-test. If the number of groups exceeded two, the Kruskal-Wallis test was used. If overall significance was found after the Kruskal-Wallis test, post- hoc subgroup analyses were conducted using the Dunn-Bonferroni test. Categorical variables were compared between the groups using Pearson's χ2 test, Fisher's exact χ2 test, and Fisher-Freeman-Halton test. The relationship between continuous variables was determined by correlation analysis, and Pearson and Spearman correlation coefficients were calculated. The factors affecting local recurrence and surgical morbidity were investigated using logistic regression analysis. Receiver operating characteristic analysis was performed to evaluate the effectiveness of BT mm² measurement in identifying the groups associated with recurrence and surgical outcomes. Statistical analyses were performed using the SPSS (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 28.0. Armonk, NY: IBM Corp.) and MedCalc (MedCalc Software bv, Ostend, Belgium; https://www.medcalc.org; 2022) software, and a P value < 0.05 was considered statistically significant.
Bias minimization strategies: Efforts were made to minimize potential sources of bias in this study. Strict inclusion and exclusion criteria were applied to ensure a homogeneous study population, which included only patients with complete clinical, radiological, and pathological data. Retrospective data collection was performed using reliable sources, such as electronic medical records, imaging studies, and surgical notes, to ensure accuracy and consistency. Additionally, to reduce observer bias, all pelvic measurements were conducted by experienced radiologists who were blinded to the clinical outcomes. Statistical adjustments were made using multivariate analyses to control for confounding variables such as age, sex, and BMI. These measures collectively aimed to enhance the validity and reliability of the study’s findings.
Management of incomplete data: For each parameter with missing values, cases specific to that parameter were excluded from the analysis. This approach ensures that only complete and reliable data contributed to the evaluation of each variable. Patients with missing key parameter values were excluded from the study to maintain consistency and validity of the analysis. This method minimized the impact of incomplete data on the study outcomes and preserved the robustness of the findings. The proportion of cases excluded due to missing data was reported to ensure transparency.
Rationale for sensitivity analysis exclusion: No formal sensitivity analysis was conducted in this study. However, the robustness of the findings was supported by strict adherence to the inclusion and exclusion criteria, which ensured a homogeneous study population. For parameters with missing data, a complete case analysis approach was employed to minimize bias and maintain the validity of the results. Given the limited sample size, sensitivity analyses were deemed unnecessary because the dataset was comprehensive for the selected variables and outcomes. Further studies with larger cohorts are needed to explore the potential impact of alternative analytical methods.
Table 1 summarizes the demographic, clinical, radiological, surgical, and pathological characteristics of patients included in the study. The average number of dissected lymph nodes in the surgeries was found to be 14.75, and the PCI value was 10.40. It was observed that 48.10% of the patients received neoadjuvant therapy, and 67.60% of the patients were directed to adjuvant therapy. The mean follow-up duration was 32 mo (Table 1).
| Items | Value |
| Age (years; n = 79) | 66.32 ± 11.87 |
| Gender (n = 79) | |
| Male | 54 (68.35) |
| Female | 25 (31.65) |
| BMI (kg/m2; n = 73) | 25.90 (19:44) |
| Tumor localization (n = 79) | |
| Upper rectum | 30 (37.97) |
| Middle rectum | 16 (20.25) |
| Lower rectum | 33 (41.77) |
| ASA score (n = 78) | |
| I | 1 (1.28) |
| II | 30 (38.46) |
| III | 46 (58.97) |
| IV | 1 (1.28) |
| Operation type (n = 79) | |
| Open low anterior | 36 (45.57) |
| Laparoscopic low anterior | 37 (46.84) |
| Miles | 6 (7.59) |
| Operation time (min; n = 78) | 237.12 ± 59.82 |
| Blood loss cc (n = 78) | 125 (50:200) |
| Tumor stage (n = 78) | |
| 0 | 5 (6.41) |
| 1 | 21 (26.92) |
| 2A | 17 (21.79) |
| 2B | 3 (3.85) |
| 3A | 14 (17.95) |
| 3B | 9 (11.54) |
| 3C | 9 (11.54) |
| Tumor size (cm; n = 77) | 3.50 (0:17) |
| Pelvic inlet area-CT mm2 (n = 77) | 8428.65 ± 1232.66 |
| Fecal diversion (n = 73) | |
| Exist | 48 (65.75) |
| None | 20 (27.40) |
| Lymphovascular invasion (n = 74) | 23 (29.90) |
| Average number of lymph nodes dissected (n = 77) | 14.75 ± 7.99 |
| PCI (n = 52) | 10.40 ± 2 |
| Neoadjuvant therapy (n = 79) | 38 (48.10) |
| Adjuvant therapy (n = 74) | 50 (67.60) |
| Follow-up period (months; n = 75) | 32 (0:70) |
Data on BMI for six patients, ASA score for one patient, and surgical parameters for one patient were unavailable. Because fecal diversion was excluded except for patients who underwent Miles operations, the sample size was 73. Pelvimetric analysis was performed on 77 patients with CT images from 52 patients and MRI images from 79 patients. Due to the absence of data in the pathology report, tumor stage in one patient, tumor size in two patients, lymph node count in two patients, and lymphovascular status in five patients could not be assessed.
Table 2 shows the comparison of demographic, pathological, and clinical variables among the patient groups based on CT measurements. Male sex and BMI were significantly associated with a narrow pelvis (both P = 0.002). The proportion of male patients was higher in the narrow pelvis group, and their mean BMI (25.42 kg/m²) was lower than that in the normal pelvis group (28.93 kg/m²). No significant differences were observed in other variables (P > 0.05). The mean PCI value, reflecting the three-dimensional structure of the pelvis, was lower in the group classified as having a narrow pelvis based on two-dimensional CT, with an average of 10.13 compared to 10.8, but this difference was not statistically significant (Table 2). For this table, data on BMI for six patients, operation time for one patient, pathological tumor stage for one patient, tumor size for two patients, and lymph node count for one patient were unavailable. There was a total of 52 patients with MRI images.
| n | Narrow | n | Normal | P value | |
| Age (years) | 48 | 67.13 ± 11.84 | 29 | 64.86 ± 11.96 | 0.421a |
| Gender (n = 79) | |||||
| Male | 48 | 39 (81.25) | 29 | 14 (48.28) | 0.002b |
| Female | 9 (18.75) | 15 (51.72) | |||
| BMI (kg/m2) | 46 | 25.42 ± 3.96 | 2 | 28.93 ± 5.15 | 0.002a |
| Tumor localization | |||||
| Upper rectum | 48 | 20 (41.67) | 29 | 10 (34.48) | 0.532b |
| Middle rectum | 11 (22.92) | 5 (17.24) | |||
| Lower rectum | 17 (35.42) | 14 (48.27) | |||
| ASA score | |||||
| I | 48 | 1 (2.08) | 28 | 0 | 0.318c |
| II | 15 (31.25) | 14 (50) | |||
| III | 31 (64.58) | 14 (50) | |||
| IV | 1 (2.08) | 0 | |||
| Operation type | |||||
| Open low anterior | 48 | 24 (50) | 29 | 11 (37.93) | 0.281c |
| Laparoscopic low anterior | 22 (45.83) | 14 (48.28) | |||
| Miles | 2 (4.17) | 4 (13.79) | |||
| Tumor stage | |||||
| 0 | 47 | 1 (2.13) | 29 | 4 (13.79) | 0.604c |
| 1 | 13 (27.66) | 8 (27.59) | |||
| 2A | 11 (23.40) | 6 (20.69) | |||
| 2B | 2 (4.26) | 1 (3.45) | |||
| 3A | 7 (14.89) | 5 (17.24) | |||
| 3B | 6 (12.77) | 3 (10.34) | |||
| 3C | 7 (14.89) | 2 (6.90) | |||
| Tumor size (cm) | 46 | 4 (0:17) | 29 | 3 (0:7) | 0.052d |
| PCI | 31 | 10.13 ± 1.99 | 21 | 10.80 ± 2 | 0.236a |
Table 3 focuses on the surgical outcomes of the patient groups based on CT measurements. In the comparison based on CT-assisted pelvimetry, no significant differences were found in the surgical outcomes between the study groups (P > 0.05). For this table, data on the operation time for one patient, estimated blood loss for one patient, and lymph node count for two patients were unavailable. Because we had two cases of perioperative surgical mortality, the recurrence parameter was evaluated with n = 75. Additionally, as two of our patients were lost due to coronavirus disease 2019 (COVID-19) infection (not considering surgical mortality within 30 days postoperatively), the readmission parameter was evaluated based on 72 patients. For the anastomotic leak variable, after excluding patients who underwent Miles operation, the number of patients for whom CT-assisted pelvimetry analysis could be performed was 70 (Table 3).
| n | Narrow | n | Normal | P value | |
| Operation time (min) | 47 | 237.66 ± 57.43 | 29 | 233.97 ± 65.33 | 0.797a |
| Blood loss (cc) | 48 | 137.50 (50:200) | 28 | 125 (50:200) | 0.723d |
| Length of stay (day) | 48 | 8 (4:32) | 29 | 7 (4:21) | 0.462d |
| Readmission | |||||
| Exist | 44 | 8 (53.33) | 28 | 7 (46.67) | 0.487b |
| None | 36 (63.16) | 21 (36.84) | |||
| Dissected lymph nodes | 46 | 15 (1:42) | 29 | 13 (2:30) | 0.229d |
| Pelvic infection | |||||
| Exist | 48 | 6 (60) | 29 | 4 (40) | > 0.99c |
| None | 42 (62.69) | 25 (37.31) | |||
| Recurrence | 45 | 9 (20) | 30 | 6 (20) | 0.943b |
| Anastomosis leakage | 44 | 5 (62.50) | 26 | 3 (37.50) | > 0.99c |
| Perioperative morbidity | 48 | 19 (70.37) | 29 | 8 (29.63) | 0.285b |
| Surgical mortality | 48 | 1 (50) | 29 | 1 (50) | 0.645c |
Table 4 addresses the comparison of patient groups with and without perioperative morbidity. Significant differences were observed in terms of age (P = 0.003), sex distribution (P = 0.020), and the PCI (P = 0.049). Patients in the morbidity group had a higher average age (71.70 years) than those without morbidity (63.52 years). The proportion of male patients was significantly higher in the morbidity group (85.19%) than in the nonmorbidity group (59.62%). Additionally, the mean PCI value was lower in the morbidity group (9.62) than in the non-morbidity group (10.78). No significant differences were found between the groups for any other variables (P > 0.05; Table 4).
| n | Exist | n | None | P value | |
| Age (year) | 27 | 71.70 ± 9.16 | 52 | 63.52 ± 12.22 | 0.003a |
| Gender | |||||
| Male | 27 | 23 (85.19) | 52 | 31 (59.62) | 0.020b |
| Female | 4 (14.81) | 21 (40.38) | |||
| BMI (kg/m2) | 23 | 27.60 (19:44) | 50 | 24.90 (19.50:37.70) | 0.316d |
| Tumor localization | |||||
| Upper rectum | 27 | 12 (44.44) | 52 | 18 (34.62) | 0.588b |
| Middle rectum | 4 (14.81) | 12 (23.08) | |||
| Lower rectum | 11 (40.74) | 22 (42.31) | |||
| ASA score | |||||
| I | 27 | 0 | 51 | 1 (1.96) | 0.583c |
| II | 11 (40.74) | 19 (37.25) | |||
| III | 15 (55.56) | 31 (60.78) | |||
| IV | 1 (3.70) | 0 | |||
| Operation time (min) | 27 | 240 (130:360) | 51 | 240 (150:360) | 0.782d |
| Blood loss (cc) | 26 | 137.50 (50:200) | 52 | 125 (50:200) | 0.355d |
| Tumor size | 25 | 3.50 (0.03:17) | 52 | 3.75 (0:8.40) | 0.836d |
| Pelvic inlet area-CT (mm2) | 27 | 8449.29 ± 1043.45 | 50 | 8417.50 ± 1333.64 | 0.915a |
| Number of dissected lymph nodes | 25 | 13 (3:32) | 52 | 15 (1:42) | 0.372d |
| PCI | 17 | 9.62 ± 1.74 | 35 | 10.78 ± 2.03 | 0.049a |
In the subgroup analysis (Table 5), the operative time was significantly longer in the open low-anterior group than in the laparoscopic low-anterior group (P = 0.040). Similarly, the length of hospital stay was significantly longer in the open low anterior group than in the laparoscopic low anterior group (P < 0.001). No statistically significant differences were found among the groups in any of the other parameters (P > 0.05).
| | n | Open low anterior | n | Laparoscopic low anterior | n | Miles | P value |
| Operation time (min) | 36 | 240 (165:360) | 36 | 210 (130:360) | 6 | 270 (180:360) | 0.023c |
| Length of hospital stay (days) | 36 | 9.50 (5:32) | 37 | 7 (4:32) | 6 | 9 (6:19) | < 0.001c |
| Readmission | |||||||
| Present | 33 | 7 (21.21) | 35 | 6 (17.14) | 6 | 2 (33.33) | 0.684b |
| Absent | 26 (78.79) | 29 (82.86) | 4 (66.67) | ||||
| Dissected lymph nodes | 34 | 15 (1:29) | 34 | 12 (2:42) | 6 | 20 (2:30) | 0.389c |
| Pelvic infection | |||||||
| Present | 36 | 7 (19.44) | 37 | 3 (8.11) | 6 | 0 | 0.308b |
| Absent | 29 (80.56) | 34 (91.89) | 6 (100) | ||||
| Local recurrence | 34 | 6 (17.60) | 35 | 9 (25.70) | 6 | 0 | 0.378a |
| Anastomotic leakage | 33 | 5 (15.15) | 36 | 3 (8.33) | 0 | 0 | 0.527a |
| Surgical morbidity | 36 | 15 (41.67) | 37 | 11 (29.73) | 1 | 1 (16.67) | |
| Surgical mortality | 36 | 3 (8.33) | 37 | 2 (5.41) | 6 | 0 | 0.783a |
No significant differences were found between the groups with or without recurrence based on patients' demographic, pathological, and pelvimetric characteristics (P > 0.05; Table 6).
| n | Exist | n | None | P value | |
| Age | 15 | 70.13 ± 10.02 | 60 | 64.90 ± 12.29 | 0.132a |
| Gender | |||||
| Male | 15 | 9 (60) | 60 | 41 (68.33) | 0.540b |
| Female | 6 (40) | 19 (31.67) | |||
| BMI (kg/m2) | 14 | 25.40 (19:37.70) | 56 | 25.60 (19.50:44) | 0.959d |
| Tumor localization | |||||
| Upper rectum | 15 | 5 (33.33) | 60 | 23 (38.33) | 0.768b |
| Middle rectum | 4 (26.67) | 11 (18.33) | |||
| Lower rectum | 6 (40) | 26 (43.33) | |||
| ASA score | |||||
| I | 15 | 0 | 59 | 1 (1.69) | 0.339c |
| II | 6 (40) | 22 (37.29) | |||
| III | 8 (53.33) | 36 (61.02) | |||
| IV | 1 (6.67) | 0 | |||
| Tumor stage | |||||
| 0 | 15 | 2 (13.33) | 59 | 3 (5.08) | 0.720c |
| 1 | 4 (26.67) | 16 (27.12) | |||
| 2A | 3 (20) | 13 (22.03) | |||
| 2B | 0 | 3 (5.08) | |||
| 3A | 2 (13.33) | 11 (18.64) | |||
| 3B | 3 (20) | 5 (8.47) | |||
| 3C | 1 (6.67) | 8 (13.56) | |||
| Tumor size (cm) | 15 | 3 (0.04:8.40) | 58 | 4 (0:17) | 0.612d |
| Pelvic inlet area-CT (mm2) | 15 | 8476.85 ± 1134.93 | 59 | 8431.43 ± 1292.23 | 0.901a |
| Number of dissected lymph nodes (n) | 15 | 12.33 ± 4.97 | 58 | 15.10 ± 8.67 | 0.240a |
| PCI | 11 | 10.50 ± 2.54 | 40 | 10.41 ± 1.87 | 0.915a |
To identify risk factors for surgical morbidity, variables were first analyzed using univariate logistic regression, and those with P < 0.25 were included in a multivariate logistic regression model (Table 7). The final model identified age, sex, and PCI as significant factors that influence morbidity. The model demonstrated a good fit with the data (Hosmer-Lemeshow test, P = 0.167) and was statistically significant (P < 0.001).
| Univariate logistic regression model | Multivariate logistic regression model | |||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age (years) | 1.07 (1.02-1.12) | 0.005 | ||
| Gender (male) | 3.90 (1.18-12.90) | 0.026 | ||
| BMI | 1.05 (0.94-1.16) | 0.402 | ||
| Tumor localization (upper rectum) | 0.592 | |||
| Middle rectum | 0.50 (0.13-1.92) | 0.313 | ||
| Lower rectum | 0.75 (0.27-2.10) | 0.583 | ||
| ASA score | 1.15 (0.48-2.76) | 0.747 | ||
| Operation type (open low anterior) | 0.375 | |||
| Laparoscopic low anterior | 0.59 (0.23-1.56) | 0.289 | ||
| Miles | 0.28 (0.03-2.65) | 0.267 | ||
| Operation duration | 1 (0.99-1.01) | 0.637 | ||
| Blood loss (cc) | 1 (0.99-1.02) | 0.325 | ||
| Tumor stage (0) | 0.993 | |||
| 1 | 1.33 (0.14-12.82) | 0.803 | ||
| 2A | 1.23 (0.24-6.36) | 0.804 | ||
| 2B | 1.09 (0.20-6.01) | 0.920 | ||
| 3A | 0 | 0.999 | ||
| 3B | 1.11 (0.19-6.49) | 0.907 | ||
| 3C | 0.57 (0.07-4.64) | 0.601 | ||
| Tumor size | 1.10 (0.91-1.33) | 0.311 | ||
| Pelvic inlet area-CT (mm2) | 1 (1-1) | 0.913 | ||
| Amount of dissected lymph node | 0.970 (0.91-1.03) | 0.348 | ||
| PCI | 0.71 (0.52-1.01) | 0.056 | ||
The variables were initially examined using univariate logistic regression to determine risk factors contributing to local recurrence. Only variables with P < 0.25 were included in the multivariate logistic regression model. Variables that met the requirements and were included in the multivariate model were age, surgery time, total number of lymph nodes, and occurrence of readmission. A forward selection method was used for variable selection. The findings are displayed in Table 8 and demonstrate that the model is an appropriate fit for the data based on the Hosmer-Lemeshow test (P = 0.694) and is statistically significant (P = 0.009). Each additional unit of operation time was linked to a 1% reduction in the likelihood of experiencing local recurrence. In addition, individuals who experienced readmission had a risk of recurrence that was 6.07 times higher than patients who never underwent readmission (Table 8).
| Univariate logistic regression model | Multivariate logistic regression model | |||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age (years) | 1.04 (0.99-1.09) | 0.134 | ||
| Gender (male) | 1.44 (0.45-4.62) | 0.542 | ||
| BMI | 0.99 (0.87-1.12) | 0.854 | ||
| Tumor localization (upper rectum) | 0.770 | |||
| Middle rectum | 1.67 (0.37-7.48) | 0.501 | ||
| Lower rectum | 1.06 (0.29-3.95) | 0.929 | ||
| ASA score | 1.29 (0.44-3.78) | 0.639 | ||
| Operation type (open low anterior) | 0.721 | |||
| Laparoscopic low anterior | 1.62 (0.51-55.17) | 0.419 | ||
| Miles | 0 | 0.999 | ||
| Operation duration | 0.99 (0.98-1) | 0.067 | 0.99 (0.97-0.99) | 0.039 |
| Blood loss (cc) | 1 (0.99-1.02) | 0.548 | ||
| Tumor stage (0) | 0.807 | |||
| 1 | 5.33 (0.34-82.83) | 0.232 | ||
| 2A | 2 (0.19-20.97) | 0.563 | ||
| 2B | 1.85 (0.16-20.94) | 0.621 | ||
| 3A | 0 | 0.999 | ||
| 3B | 1.46 (0.11-18.96) | 0.775 | ||
| 3C | 4.80 (0.39-59.90) | 0.223 | ||
| Tumor size | 0.94 (0.74-1.20) | 0.635 | ||
| Pelvic inlet area-CT (mm2) | 1 (1-1) | 0.900 | ||
| Amount of dissected lymph node | 0.95 (0.88-1.03) | 0.239 | ||
| PCI | 1.02 (0.732-1.43) | 0.894 | ||
| Length of stay | 1.03 (0.94-1.12) | 0.503 | ||
| Re-admission (exist) | 3.63 (1.04-12.72) | 0.044 | 6.07 (1.42-25.97) | 0.015 |
| Pelvic infection (exist) | 2.25 (0.49-10.29) | 0.296 | ||
This study explored the associations of MRI-based PCI measurements (3D) and CT-based pelvic inlet area assessments (2D) with surgical outcomes, perioperative morbidity, and local recurrence. Although a narrow pelvis was associated with longer operative times and increased estimated blood loss, these differences were not statistically significant. Additionally, low PCI values demonstrated a significant correlation with increased perioperative morbidity (P = 0.049); however, no notable differences in other surgical and oncological outcomes were observed between the narrow and normal/wide pelvis groups.
In statistical analyses, we evaluated the morbidity parameter as a categorical variable, and we did not utilize the numerical Clavien-Dindo classification. The morbidities observed included atelectasis, pneumonia, urinary retention, postoperative ileus, wound infection, anastomotic leakage, and pelvic collection. Two patient deaths were caused by sepsis and multi-organ failure that occurred following anastomotic leakage, while the other death was a result of delayed bleeding from the epigastric artery during the laparoscopic procedure. Statistical analysis of surgical morbidity cases revealed a strong association between advanced age (P = 0.003) and male sex (P = 0.020).
Several studies have specifically investigated the correlation between morbidity and pelvimetry in rectal cancer surgery, with an emphasis on anastomotic leakage. Tsuruta et al[14] found that using MRI to identify a narrow pelvis with a PCI value of 13 was useful for predicting anastomotic leaks. According to Atasoy et al[9], pelvic depth is linked to anastomotic leaks on MRI. Conversely, the research conducted by Zur Hausen et al[15] failed to find any correlation between pelvimetric readings and anastomotic leakage. Consistent with Zur Hausen et al[15], our study did not identify a statistically significant relationship between pelvimetric parameters and anastomotic leakage (P > 0.05).
Several studies have examined the correlation between operative time and pelvimetry, which can be considered as an indicator of surgical complexity in rectal cancer surgery. Yan et al[16] conducted CT-based pelvimetric analysis and found that the interspinous distance, length of the pelvic inlet, and distance from the sacral promontory to the coccyx tip were associated with longer operating times. Zhou et al[17] found that the AP diameter of the pelvic inlet, AP diameter of the pelvic outlet, height of the pubic symphysis, sacrococcygeal distance, sacrococcygeal-pubic angle, and diameter of the upper pubis to the coccyx affect the duration of surgery when using CT pelvimetry. A study by Noh et al[6] found that a narrow pelvis did not have an impact on the duration of the surgical procedure, as determined by CT scans. Consistent with Noh et al[6], we did not identify any correlation between pelvimetric parameters and operative time or estimated blood loss (P > 0.05).
In the literature, the relationship between surgical quality and pelvimetry in rectal cancers has been analyzed using specimen evaluation and circumferential resection margin (CRM) status[7]. According to the literature, a short interspinous distance has a negative effect on the TME quality[2,15]. The usefulness of pelvimetry in predicting involvement of CRM is debated in the literature. Yamaoka et al[18] were unable to establish a statistical relationship, but Chau et al[19] found statistical significance. In our study, surgical quality was evaluated using the lymph node count reported in pathology assessments. CT-based pelvimetry revealed no significant differences in lymph node counts between patients with narrow and normal pelvises (P > 0.05).
Upon comparing the demographic and patient characteristic data of the cases in our study with existing studies on pelvimetry in rectal cancer surgery, we believe that our study included a relatively higher proportion of elderly patients with comorbidities. The patient profile in this study was predominantly male, which aligns with the previous findings. Additionally, the average BMI in this study was higher than that in Eastern studies[6,9,15,20,21]. This highlights the importance of considering patient characteristics in the context of pelvimetry-related outcomes and their generalizability to broader populations.
When comparing our surgical outcomes with the literature, we observed that our average operative time was longer (237.12 min), and our estimated blood loss was higher (125 cc). We attributed these differences to the quality of documentation and the fact that the surgeries were performed by 11 surgeons in a general surgery clinic. Other statistics included the average number of removed lymph nodes (14.75), laparoscopic surgery rate (46%), conversion to open surgery rate (13%), surgical morbidity rate (34%), mortality rate (2.5%), average hospital stay (7.4 d), and anastomotic leakage rate (11%). We consider the shorter average hospital stay in comparison with the existing literature to be a positive aspect. Additionally, our morbidity rate and surgical outcomes are partially aligned with those reported in the literature[6,20,21].
Various techniques have been used to assess the relationship between pelvic anatomy and rectal cancer surgery. Analysis of CT-based research in the literature showed that Noh et al[6] observed a higher incidence of pelvic surgical site infection and local recurrence in patients with a narrow pelvic inlet area. However, they did not establish a correlation between surgical outcomes and pelvimetry. According to Bolshinsky et al[11], patients with a wide pelvic inlet showed a statistically significant improvement in quality. The research conducted by Gojayev et al[22] did not yield any statistically significant results in their study on the association between pelvic linear diameters and CRM involvement. Narrow pelvis categorization was correlated with male sex in three studies. Yasar et al[10] did not find any statistically significant relationship between pelvimetric measurements using 8 pelvic lengths, 2 angles, and operation time. Analysis of our data showed a statistically significant association between male sex and BMI with CT-based narrow pelvis classification (P = 0.002). At this point, it is important to remember that research in the literature indicates that the male sex is a risk factor for anastomotic leakage, thereby contributing to surgical morbidity[23,24].
Evaluation of MRI-based pelvimetric studies has revealed that a narrow pelvic inlet is linked to CRM involvement[25], incomplete TME[26], and intraoperative blood loss[9,27]. Multiple studies have demonstrated that shortened interspinous and intertubercular distances correlate with poor surgical outcomes. de'Angelis et al[28] showed that a small intertuberous distance increases the probability of conversion from laparoscopic to open surgery. Escal et al[29] found that a small intertuberous distance was linked to a higher surgical complexity in their scoring system.
A comparison between CT and MR pelvimetry revealed that while there is agreement in the literature on several length measurements, MR pelvimetry is more accurate for measuring the posterior sagittal diameter of the pelvic inlet and AP pelvic outlet diameter[30]. At this juncture, we believe that it is crucial to assess various methodologies and verify diverse approaches. Therefore, our objective was to enhance surgical prediction by incorporating parameters from different techniques. Based on our dataset, we observed a correlation between the reduced pelvic inlet area determined by CT and low PCI values determined by MR; however, this correlation was not statistically significant.
Based on our study, these data suggest that preoperative pelvic measurements are not significantly correlated with local recurrence. However, we found a correlation between local recurrence and readmission, longer operative time, age, and fewer lymph node dissections (P = 0.009). The local recurrence rate in our study was 18%, whereas a case series by Tersteeg et al[31] reported a rate of 4.4%. This study aimed to evaluate the effects of neoadjuvant therapy on local recurrence. The results showed that the clinical stage and lymph node count were significantly correlated with local recurrence. The study by Giesen et al[32] involved 1548 patients. They reported a local recurrence rate of 5% after a follow-up period of 36 months. This study compared local recurrence and survival rates based on the lymph node stage. In rectal cancer, the N stage has a substantial impact on local recurrence but no influence on survival. Based on two extensive case series in the literature, it is evident that the current local recurrence rate is higher for rectal cancers. This highlights the need to improve the effectiveness of multimodal treatment for rectal cancer and enhance the quality of surgical procedures.
This study has limitations. In nature, a dataset may contain both losses and biases. The small sample size of the patients might have failed to meet the criteria for scientific validation and generalization. A potential source of heterogeneity may have arisen from the inclusion of patients with upper, middle, and lower rectal cancers in the patient group. The evaluation of pre- and post-treatment responses in patients undergoing neoadjuvant therapy should be effective. Additionally, the absence of standardized reporting for TME quality restricts the comprehensive evaluation of surgical quality, which is crucial for understanding oncological outcomes. The variability in surgical outcomes may also have been influenced by the wide range of surgeons involved, highlighting the operator-dependent nature of rectal cancer surgery. The time interval hindered the collection of data on 5-year local recurrence, overall survival, and disease-free survival. The use of pre-established cut-off values for pelvimetric measurements from external studies may not fully reflect population-specific anatomical variations, further challenging the generalizability of the results. Finally, disruptions caused by the COVID-19 pandemic, including interruptions in multidisciplinary tumor board meetings, may have affected treatment planning and outcomes for a subset of patients. Given the limited scope of this study to a single institution, it is imperative to corroborate the results of additional research. Further investigation is necessary within a broader scope with scrupulous planning.
This retrospective analysis explored the impact of pelvimetric assessments using both CT and MRI on surgical morbidity and local recurrence after rectal cancer surgery. Our findings demonstrate that a narrow pelvic structure, as indicated by a low PCI, is significantly positively correlated with surgical morbidity. However, contrary to our expectations, having a narrow or wide pelvic structure did not show a statistically significant impact on surgical outcomes such as operation time, estimated blood loss, or local recurrence within the first year postoperatively. These results suggest that while pelvimetry may be valuable in predicting surgical difficulty and morbidity, it may not be sufficient to predict local recurrence. In our study, we uniquely utilized both CT and MRI techniques for pelvic anatomy evaluation, providing a combination of two-dimensional and three-dimensional analyses. This approach offers a more comprehensive assessment of the pelvic anatomy than most existing studies, which typically rely on either CT or MRI alone. By employing both modalities, we provided a broader perspective in the context of surgical planning. In conclusion, pelvimetry could be a helpful tool for surgical risk stratification and for improving surgeon-patient communication, particularly in cases involving a complex pelvic anatomy. However, its limited role in predicting cancer recurrence underscores the importance of integrating other patient- and tumor-related factors. Large-scale, multicenter, and prospective studies are necessary to explore its clinical value and establish its broader applicability. Moreover, exploring the potential of combining pelvimetric data with other predictive markers may enhance surgical planning and improve both short- and long-term outcomes in rectal cancer patients.
As the corresponding author, I would like to express my sincere gratitude to Prof. Dr. İsmayil Yılmaz Opr. Dr. Uğur Duman, Opr. Dr. Mehmet Emrah Bayam and Opr. Dr. Mehmet Fatih Erol for their valuable guidance and directives during the preparation of this study, which provided constructive contributions.
| 1. | Yuan W, Wang X, Wang Y, Wang H, Yan C, Song G, Liu C, Li A, Yang H, Gao C, Chen J. Development and validation of a nomogram for predicting operating time in laparoscopic anterior resection of rectal cancer. J Cancer Res Ther. 2023;19:964-971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Chen J, Sun Y, Chi P, Sun B. MRI pelvimetry-based evaluation of surgical difficulty in laparoscopic total mesorectal excision after neoadjuvant chemoradiation for male rectal cancer. Surg Today. 2021;51:1144-1151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 3. | Yamamoto T, Kawada K, Kiyasu Y, Itatani Y, Mizuno R, Hida K, Sakai Y. Prediction of surgical difficulty in minimally invasive surgery for rectal cancer by use of MRI pelvimetry. BJS Open. 2020;4:666-677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 4. | Tschann P, Brock T, Weigl MP, Clemens P, Brunner W, Attenberger C, Königsrainer I, Emmanuel K, Konschake M, Jäger T. Tumour, narrow pelvis and surgery specific factors for total mesorectal excision quality and morbidity following rectal cancer resection. BJS Open. 2023;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 5. | Lorenzon L, Bini F, Landolfi F, Quinzi S, Balducci G, Marinozzi F, Biondi A, Persiani R, D'Ugo D, Tirelli F, Iannicelli E. 3D pelvimetry and biometric measurements: a surgical perspective for colorectal resections. Int J Colorectal Dis. 2021;36:977-986. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 6. | Noh GT, Chung SS, Kim KH, Lee RA. Computed tomography based cross-sectional anatomy of the pelvis predicts surgical outcome after rectal cancer surgery. Ann Surg Treat Res. 2020;99:90-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Zhang Q, Wei J, Chen H. Advances in pelvic imaging parameters predicting surgical difficulty in rectal cancer. World J Surg Oncol. 2023;21:64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 8. | Ma Q, Cheng J, Bao Y, Gao Z, Jiang K, Wang S, Ye Y, Wang Y, Shen Z. Magnetic resonance imaging pelvimetry predicts the technical difficulty of rectal surgery. Asian J Surg. 2022;45:2626-2632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 9. | Atasoy G, Arslan NC, Elibol FD, Sagol O, Obuz F, Sokmen S. Magnetic resonance-based pelvimetry and tumor volumetry can predict surgical difficulty and oncologic outcome in locally advanced mid-low rectal cancer. Surg Today. 2018;48:1040-1051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Yasar NF, Gundogdu E, Yilmaz AS, Badak B, Bayav FD, Ozen A, Oner S. Can 3D radiological calculations predict operational difficulties for rectal cancer?: A single center retrospective analysis. Medicine (Baltimore). 2024;103:e36961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Bolshinsky V, Sweet DE, Vitello DJ, Jia X, Holubar SD, Church J, Herts BR, Steele SR. Using CT-Based Pelvimetry and Visceral Obesity Measurements to Predict Total Mesorectal Excision Quality for Patients Undergoing Rectal Cancer Surgery. Dis Colon Rectum. 2024;67:929-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Kimura N, Yamada Y, Takeshima Y, Otsuka M, Akamatsu N, Hakozaki Y, Miyakawa J, Sato Y, Akiyama Y, Yamada D, Fujimura T, Kume H. The 'prostate-muscle index': a simple pelvic cavity measurement predicting estimated blood loss and console time in robot-assisted radical prostatectomy. Sci Rep. 2022;12:11945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 13. | Hong SK, Lee ST, Kim SS, Min KE, Hwang IS, Kim M, Jeong SJ, Byun SS, Hwang SI, Lee SE. Effect of bony pelvic dimensions measured by preoperative magnetic resonance imaging on performing robot-assisted laparoscopic prostatectomy. BJU Int. 2009;104:664-668. [PubMed] [DOI] [Full Text] |
| 14. | Tsuruta A, Tashiro J, Ishii T, Oka Y, Suzuki A, Kondo H, Yamaguchi S. Prediction of Anastomotic Leakage After Laparoscopic Low Anterior Resection in Male Rectal Cancer by Pelvic Measurement in Magnetic Resonance Imaging. Surg Laparosc Endosc Percutan Tech. 2017;27:54-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 15. | Zur Hausen G, Gröne J, Kaufmann D, Niehues SM, Aschenbrenner K, Stroux A, Hamm B, Kreis ME, Lauscher JC. Influence of pelvic volume on surgical outcome after low anterior resection for rectal cancer. Int J Colorectal Dis. 2017;32:1125-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Yan HH, Lou Z, Sheng J, Zhang W, Fu CG, Meng RG. [Computed tomography pelvimetry as a predictor of technical difficulty in total mesorectal excision]. Zhonghua Wei Chang Wai Ke Za Zhi. 2011;14:846-850. [PubMed] [DOI] [Full Text] |
| 17. | Zhou XC, Su M, Hu KQ, Su YF, Ye YH, Huang CQ, Yu ZL, Li XY, Zhou H, Ni YZ, Jiang YI, Lou Z. CT pelvimetry and clinicopathological parameters in evaluation of the technical difficulties in performing open rectal surgery for mid-low rectal cancer. Oncol Lett. 2016;11:31-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 18. | Yamaoka Y, Yamaguchi T, Kinugasa Y, Shiomi A, Kagawa H, Yamakawa Y, Furutani A, Manabe S, Torii K, Koido K, Mori K. Mesorectal fat area as a useful predictor of the difficulty of robotic-assisted laparoscopic total mesorectal excision for rectal cancer. Surg Endosc. 2019;33:557-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 19. | Chau J, Solomon J, Liberman AS, Charlebois P, Stein B, Lee L. Pelvic dimensions on preoperative imaging can identify poor-quality resections after laparoscopic low anterior resection for mid- and low rectal cancer. Surg Endosc. 2020;34:4609-4615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 20. | Akiyoshi T, Kuroyanagi H, Oya M, Konishi T, Fukuda M, Fujimoto Y, Ueno M, Miyata S, Yamaguchi T. Factors affecting the difficulty of laparoscopic total mesorectal excision with double stapling technique anastomosis for low rectal cancer. Surgery. 2009;146:483-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 154] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 21. | Targarona EM, Balague C, Pernas JC, Martinez C, Berindoague R, Gich I, Trias M. Can we predict immediate outcome after laparoscopic rectal surgery? Multivariate analysis of clinical, anatomic, and pathologic features after 3-dimensional reconstruction of the pelvic anatomy. Ann Surg. 2008;247:642-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 154] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 22. | Gojayev A, Yuksel C, Akbulut S, Erşen O, Bakırarar B, Gülpınar B, Gürsoy Çoruh A, Unal AE, Demirci S. Can CT Pelvimetry Be Used to Predict Circumferential Resection Margin Positivity in Laparoscopic Resection of Middle and Lower Rectum Cancer? Cureus. 2022;14:e31745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 23. | Park JS, Choi GS, Kim SH, Kim HR, Kim NK, Lee KY, Kang SB, Kim JY, Lee KY, Kim BC, Bae BN, Son GM, Lee SI, Kang H. Multicenter analysis of risk factors for anastomotic leakage after laparoscopic rectal cancer excision: the Korean laparoscopic colorectal surgery study group. Ann Surg. 2013;257:665-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 399] [Cited by in RCA: 353] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 24. | Kang J, Lee HB, Cha JH, Hur H, Min BS, Baik SH, Kim NK, Sohn SK, Lee KY. Feasibility and impact on surgical outcomes of modified double-stapling technique for patients undergoing laparoscopic anterior resection. J Gastrointest Surg. 2013;17:771-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Boyle KM, Petty D, Chalmers AG, Quirke P, Cairns A, Finan PJ, Sagar PM, Burke D. MRI assessment of the bony pelvis may help predict resectability of rectal cancer. Colorectal Dis. 2005;7:232-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 79] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 26. | Baik SH, Kim NK, Lee KY, Sohn SK, Cho CH, Kim MJ, Kim H, Shinn RK. Factors influencing pathologic results after total mesorectal excision for rectal cancer: analysis of consecutive 100 cases. Ann Surg Oncol. 2008;15:721-728. [PubMed] [DOI] [Full Text] |
| 27. | Hong JS, Brown KGM, Waller J, Young CJ, Solomon MJ. The role of MRI pelvimetry in predicting technical difficulty and outcomes of open and minimally invasive total mesorectal excision: a systematic review. Tech Coloproctol. 2020;24:991-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 28. | de'Angelis N, Pigneur F, Martínez-Pérez A, Vitali GC, Landi F, Torres-Sánchez T, Rodrigues V, Memeo R, Bianchi G, Brunetti F, Espin E, Ris F, Luciani A; EuMaRCS Study Group. Predictors of surgical outcomes and survival in rectal cancer patients undergoing laparoscopic total mesorectal excision after neoadjuvant chemoradiation therapy: the interest of pelvimetry and restaging magnetic resonance imaging studies. Oncotarget. 2018;9:25315-25331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 29. | Escal L, Nougaret S, Guiu B, Bertrand MM, de Forges H, Tetreau R, Thézenas S, Rouanet P. MRI-based score to predict surgical difficulty in patients with rectal cancer. Br J Surg. 2018;105:140-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 30. | Shen P, Fu J, Zhang W, Chen C, Liu P. A comparative study of two pelvimetry methods: 3D models based on CT and MRI. Eur J Obstet Gynecol Reprod Biol. 2024;296:286-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Tersteeg JJC, van Esch LM, Gobardhan PD, Kint PAM, Rozema T, Crolla RMPH, Schreinemakers JMJ. Early local recurrence and one-year mortality of rectal cancer after restricting the neoadjuvant therapy regime. Eur J Surg Oncol. 2019;45:597-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Giesen LJX, Borstlap WAA, Bemelman WA, Tanis PJ, Verhoef C, Olthof PB; Dutch Snapshot Research Group. Effect of understaging on local recurrence of rectal cancer. J Surg Oncol. 2020;122:1179-1186. [PubMed] [DOI] [Full Text] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/