Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.103494
Revised: January 21, 2025
Accepted: February 11, 2025
Published online: April 27, 2025
Processing time: 128 Days and 16.6 Hours
Hemorrhoids, a prevalent chronic condition globally, significantly impact patients' quality of life. While various surgical interventions, such as external stripping and internal ligation, procedure for prolapse and hemorrhoids, and tissue selecting technique, are employed for treatment, they are often associated with postoperative complications, including unsatisfactory defecation, bleeding, and anal stenosis. In contrast, Xiaozhiling injection, a traditional Chinese me
To investigate the molecular mechanism underlying the therapeutic effect of Xiaozhiling injection in the treatment of internal hemorrhoids.
An internal hemorrhoid model was established in rats, and the rats were randomly divided into a modeling group [control group (CK group)] and a treatment group. One week after injection, Stereo-seq and electron microscopy were used to study the changes in gene expression and subcellular structures in fibroblasts.
Single-cell sequencing revealed differences in the expression and transcript levels of the genes collagen 3 alpha 1, decorin, and actin alpha 2 in fibroblasts between the CK group and the treatment group. Spatial transcriptome analysis revealed that genes of the sphingosine kinase 1 (Sphk1)/sphingosine-1-phosphate (S1P) pathway spatially overlapped with key genes of the transforming growth factor beta 1 pathway, namely, Sphk1, S1P receptor, and transforming growth factor beta 1, in the treatment group. The proportion of fibroblasts was lower in the treatment group than in the CK group, and Xiaozhiling treatment had a significant effect on the proportion of fibroblasts in hemorrhoidal tissue. Immunohistochemistry revealed a significant increase in the expression of a fibroblast marker. Electron microscopy showed that the endoplasmic reticulum of fibroblasts contained a large amount of glycogen, indicating cell activation. Fibroblast activation and the expression of key genes of the Sphk1-S1P pathway could be observed at the injection site, suggesting that after Xiaozhiling intervention, the Sphk1-S1P pathway could be activated to promote fibrosis.
Xiaozhiling injection exerts its therapeutic effects on internal hemorrhoids by promoting collagen synthesis and secretion in fibroblasts. After Xiaozhiling intervention, the Sphk1-S1P pathway can be activated to promote fibrosis.
Core Tip: Single-cell sequencing revealed differences in the expression and transcription levels of genes collagen 3 alpha 1, decorin, and actin alpha 2 in fibroblasts between the control groups and treatment groups. The activation of fibroblasts and the expression of key genes in the sphingosine kinase 1-sphingosine-1-phosphate pathway were observed at the injection site, suggesting that the sphingosine kinase 1-sphingosine-1-phosphate pathway could be activated to promote fibrosis after the intervention of Xiaozhling.
- Citation: Ke MH, Huang SY, Lin WG, Xu ZG, Zheng XX, Liu XB, Cheng YM, Li ZF. Single-nucleus RNA sequencing and spatial transcriptomics reveal the mechanism by which Xiaozhiling injection treats internal hemorrhoids. World J Gastrointest Surg 2025; 17(4): 103494
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/103494.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.103494
Hemorrhoids are a common chronic condition worldwide that affects people’s lives. There are many surgical methods for the treatment of hemorrhoids, including external stripping and internal ligation, the procedure for prolapse and hemorrhoids, and the tissue selecting technique, but these treatments are associated with postoperative complications of different severities, such as unsatisfactory defecation, bleeding, and anal stenosis[1,2]. Due to the anatomy of the rectum and the pathogenesis of hemorrhoids, Xiaozhiling injection is the main treatment for internal hemorrhoids in Chinese medicine. Xiaozhiling injection has clear advantages in the treatment of hemorrhoids as well as a curative effect. It is less invasive and requires less dietary restrictions than other treatments, and it is widely applicable, does not induce a strong systemic inflammatory response, and causes acute inflammation for only a short period of time. It can eliminate the hemorrhoid nucleus, prevent local tissue necrosis, protect and fix the anal cushion, and reduce complications and sequelae such as postoperative bleeding and prolapse[3].
Since its discovery more than 40 years ago, Xiaozhiling injection has been widely used in China and more than 50 other countries and regions, such as Japan and South Korea[4]. Aluminum potassium sulfate and tannic acid (ALTA) injection, which is an improved version of Xiaozhiling, is widely used in Japan. Xiaozhiling injection[5] has good clinical efficacy and high safety in the treatment of grades II-III internal hemorrhoids and can significantly alleviate the symptoms of hematostoecium and prolapse in patients with internal hemorrhoids; moreover, the amount of time and hospital stay required for treatment are short, and it is associated with few and mild postoperative complications. To date, studies on the mechanism underlying the effect of Xiaozhiling have focused on its impact on aseptic inflammation and tissue fibrosis[4], which mainly involves functional changes in fibroblasts. Fibroblasts are normally located in the tissue interstitium and play a key role in maintaining tissue structural integrity, healing, and regeneration[6]. In normal colorectal tissues, fibroblasts are evenly distributed in the lamina propria adjacent to the colorectal mucosal epithelium and in tissues and organs rich in epithelial cells, and secreted collagen and lamin form elastic and viscous tissue structures[7]. Owing to the wide distribution of fibroblasts and their diverse functions, fibroblast activation is considered the key to tissue fibrosis[8,9]. Fibroblasts can secrete cytokines, chemokines, etc. to modify the intensity and duration of the inflammatory response. They can also release interleukin-1β and transforming growth factor-β (TGF-β) to promote the differentiation of fibroblasts into myofibroblasts (Myofs) under the influence of mechanical tension[10]. Through immunohistochemical analysis, Sferra et al[11] found that the expression of TGF-β1 and sphingosine kinase-1 (Sphk1) was elevated in the intestinal mucosa and submucosa of mice with colorectal fibrosis, suggesting that the TGF-β1-mediated Sphk1-sphingosine 1-phosphate (S1P) signaling pathway could facilitate Myof activation and result in fibrosis.
In a previous study, our research group found that the thickness of the submucosal tissue in the hemorrhoidal area changed and fibroblasts proliferated after Xiaozhiling treatment, suggesting that the drug induced some changes in the cell population; however, changes in various cells and the spatial pattern of Sphk1-S1P signaling pathway activation at the single-cell level were not assessed. This study was designed to explore the complexity and diversity of patho
Xiaozhiling injection (Beijing China Resources High-Tech Natural Medicine Co., Ltd.); inhaled isoflurane (Baxter Healthcare Corporation, China); 4% neutral formaldehyde fixative solution; 6% croton oil (Beijing Huawei Ruike Chemical Co., Ltd.); pyridine (Beijing Lambolide Trading Co., Ltd.); ether (Xilong Science Co., Ltd.); horseradish peroxidase-conjugated polyclonal anti-rabbit/mouse IgG (Boster); an anti-TGF-β1 rabbit polyclonal antibody (Proteintech); an anti-Sphk1 rabbit polyclonal antibody (Proteintech); an anti-sphingosine-1-phosphate receptor 1 (S1PR1) rabbit polyclonal antibody (Proteintech); and an alpha smooth muscle actin specific monoclonal antibody (Proteintech) were used in this study.
A total of 20 healthy specific pathogen-free 8-week-old male SD rats weighing 180-220 g were purchased from SCXK Laboratory Animal Co., Ltd. (Shanghai; 2018–0004). The animals were raised in the Laboratory Animal Center of Fujian University of Chinese Medicine [approval No. SYXK (Fujian) 2019-0007]. Standard food was provided by the Laboratory Animal Center of Fujian University of Chinese Medicine. The animals were housed on a 12-hour light/dark cycle at a constant humidity and controlled temperature of 22-25 °C. The experiments were carried out after 1 week of adaptation. All operations were performed in accordance with the ethics requirements of the Fujian Academy of Traditional Chinese Medicine (approval No. FJATCM-IAEC2018015). The rats were treated in accordance with the Guiding Opinions on the Kindness of Laboratory Animals[13]. Twenty rats were randomly divided into a control group (CK group) and a treatment group (10 rats in each group) after the internal hemorrhoid model was successfully established as previously described[14].
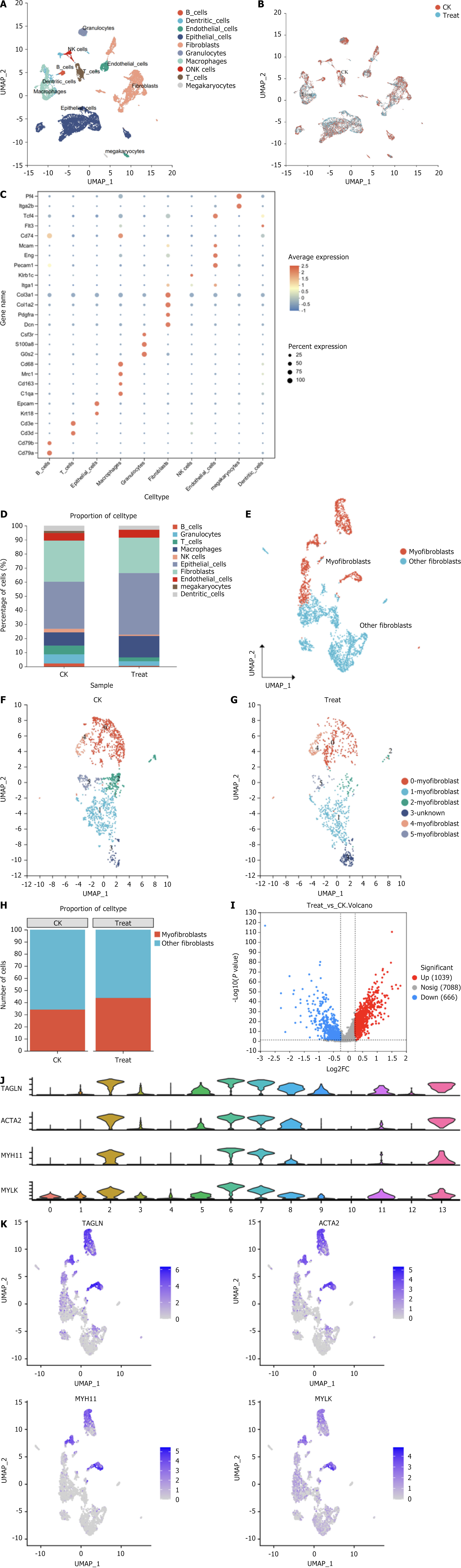
To investigate the differences in the expression and transcript levels of classic fibroblast marker genes in the hemorrhoidal region, single-cell transcriptome sequencing of hemorrhoidal tissue was performed 1 week after model establishment. According to the results of single-cell transcriptome sequencing, a total of 12589 high-quality cells were obtained; there were 7538 cells from the modeling group (CK group), with a median gene number of 1738, and 5051 cells from the Xiaozhiling (treatment group), with a median gene number of 2174. A total of 29 cell clusters were identified using Seurat software for reduction and visualization, and these clusters were found to consist of the following 10 cell types on the basis of the expression of classic markers: B cells, dendritic cells, endothelial cells, epithelial cells, fibroblasts, granulocytes, macrophages, neurons, T cells, and megakaryocytes (Figure 1A). The cell types present in the CK group and treatment group were essentially the same (Figure 1B). Bubble maps of marker genes of each cell type are shown in Figure 1C. There were significant differences in the proportions of the different cell types between the two groups (Figure 1D-G), and the proportion of Myofs cells in the Xiaozhiling group significantly increased (Figure 1H and I). Gene display of the large groups of differentially expressed genes in fibroblasts and smaller cell clusters revealed that the expression of classic marker genes of Myofs (actin alpha 2, myosin heavy chain 11, myosin light chain kinase, and transgelin) changed (Figure 1J and K). These findings suggested that Myof proliferation was stimulated after drug intervention.
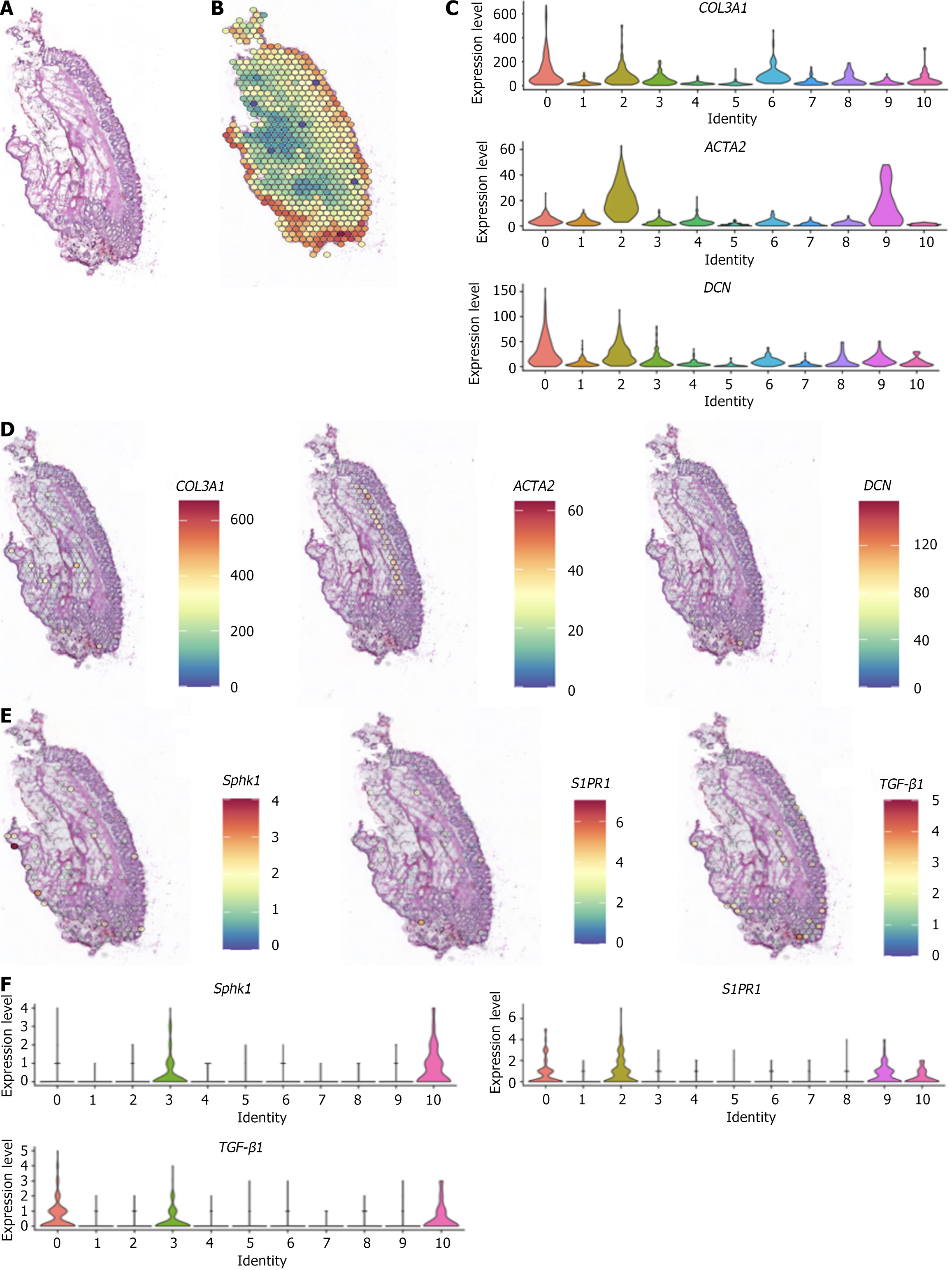
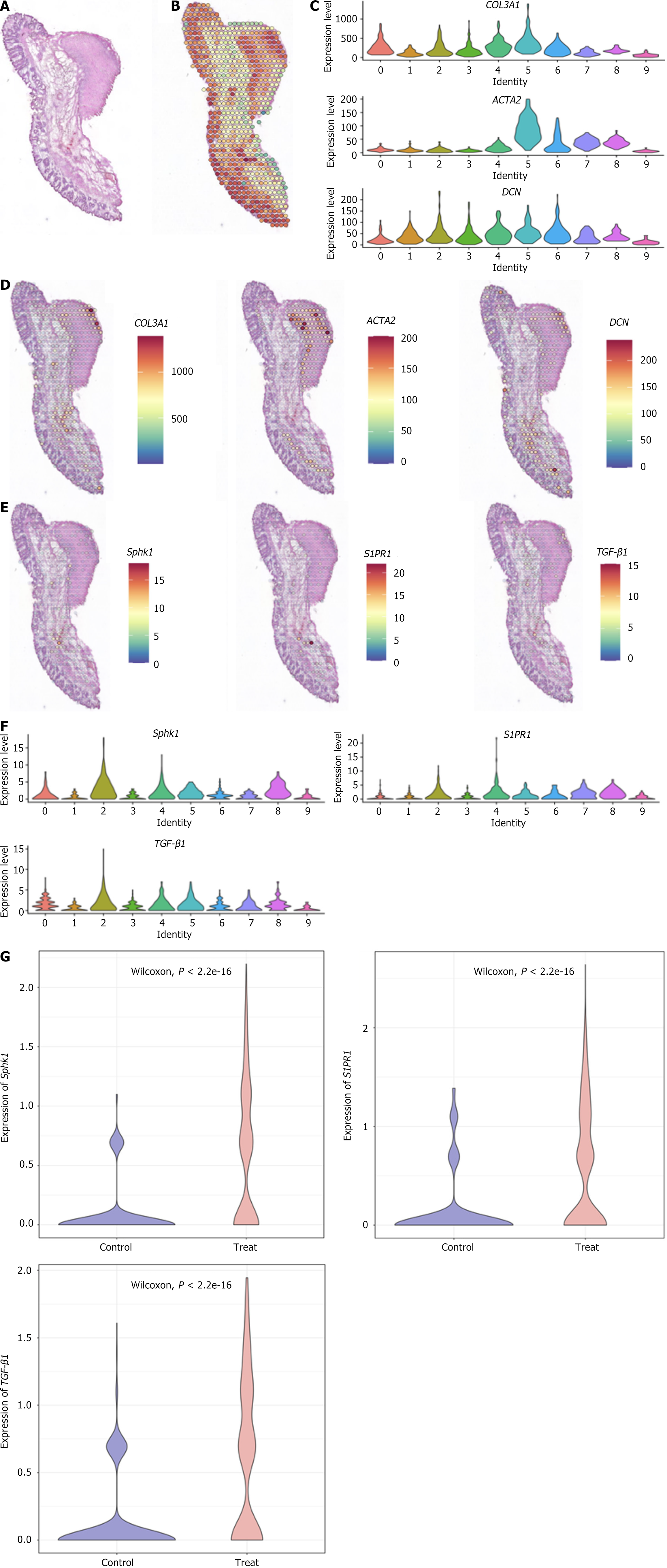
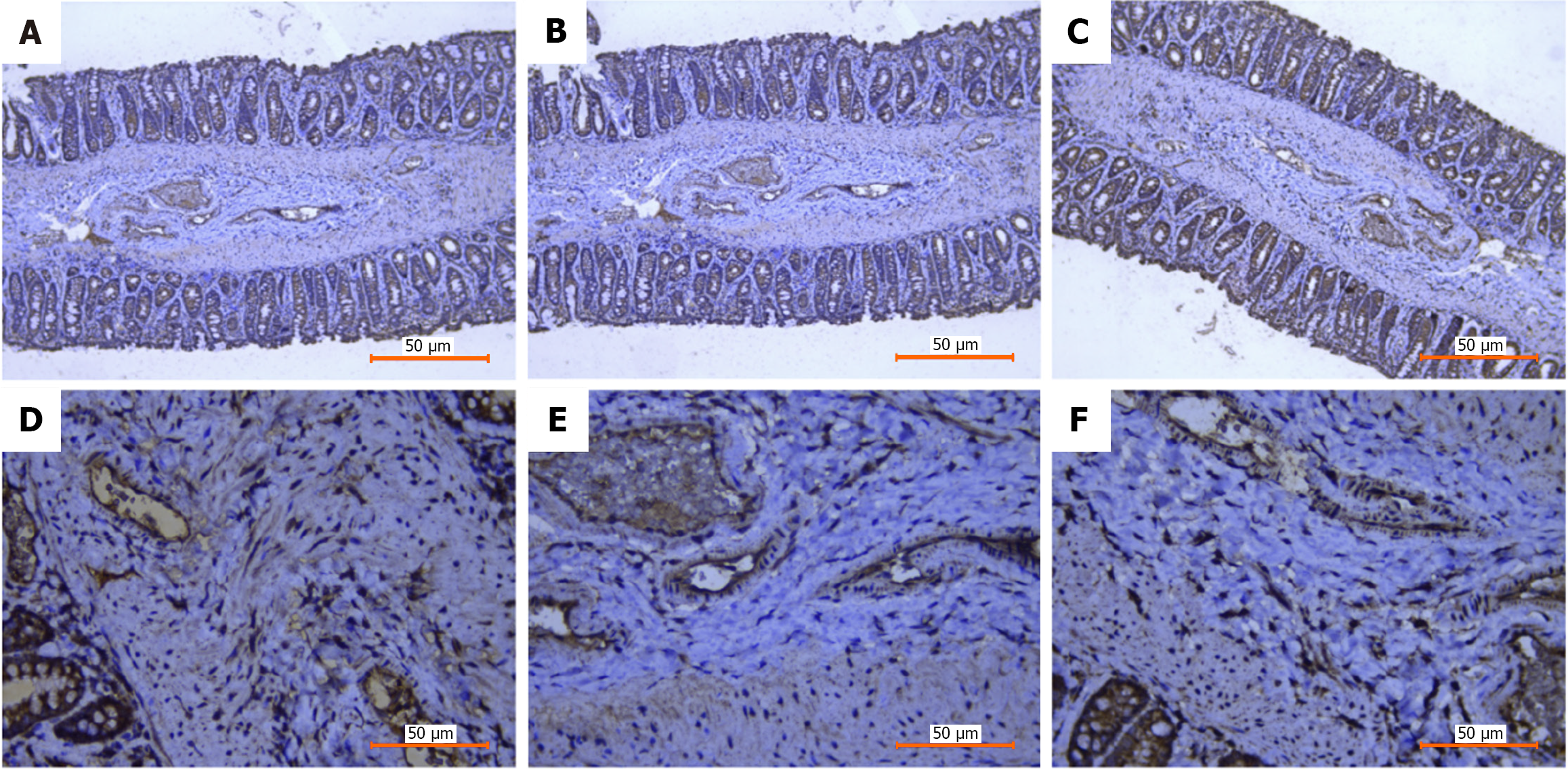
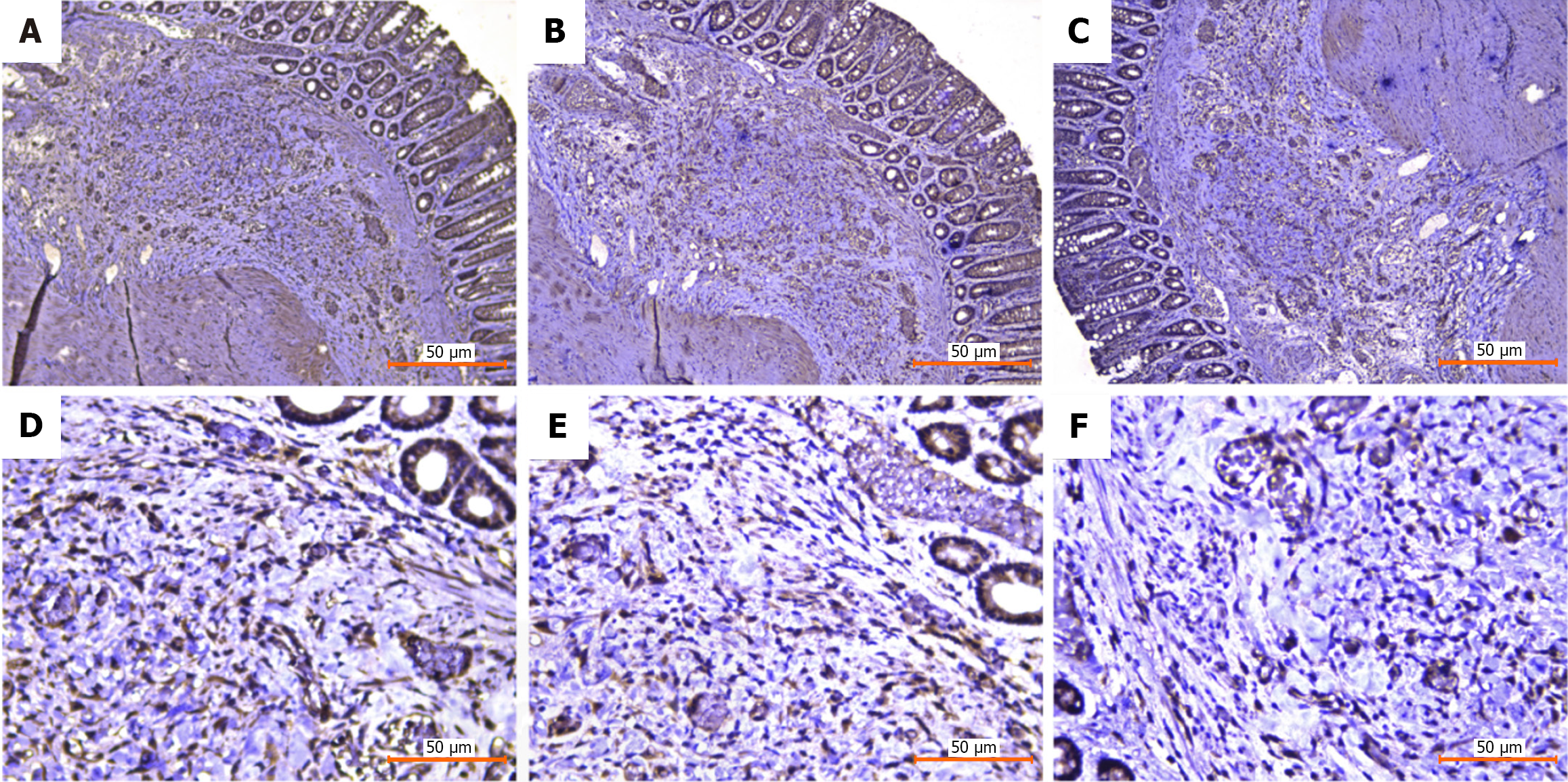
The spatial expression patterns of Sphk1, S1PR1, and TGF-β1, key genes of the Sphk1-S1P and TGF-β1 pathways, after Xiaozhiling injection were analyzed. Since single-cell transcriptome sequencing revealed differences in the transcriptome of fibroblasts before and after intervention, spatial transcriptome sequencing was performed to further explore changes in the spatial location of fibroblasts and to determine whether there is a relationship between the TGF-β1 pathway and the spatial location of fibroblasts. The spatial localization of cell types in internal hemorrhoid tissue was analyzed via spatial transcriptome sequencing. The total number of genes expressed in internal hemorrhoid tissue (Figure 2A and B) was 17590, with the median number of genes being 4943; the total number of genes expressed in hemorrhoid tissue one week after Xiaozhiling injection was 17792 (Figure 3A and B), with the median number of genes being 5980. The differences in the expression of classic fibroblast marker genes (collagen 3 alpha 1, decorin, and actin alpha 2) in tissues before and after drug intervention were subsequently explored (Figure 2C and Figure 3C), as were the spatial localization of these genes (Figure 2D and Figure 3D). The expression levels of fibroblast marker genes after drug intervention were significantly greater than those before intervention. The spatial localization of key genes of the Sphk1-S1P and TGF-β1 pathways, i.e., Sphk1, S1PR1, and TGF-β1, was assessed (Figure 2E and Figure 3E). The expression levels of Sphk1-S1P and TGF-β1 pathway-related genes (Figure 2F, Figure 3F and G) were significantly increased in areas where fibroblasts were concentrated after Xiaozhiling injection. These findings suggest that the Sphk1-S1P pathway is activated after the injection of Xiaozhiling.
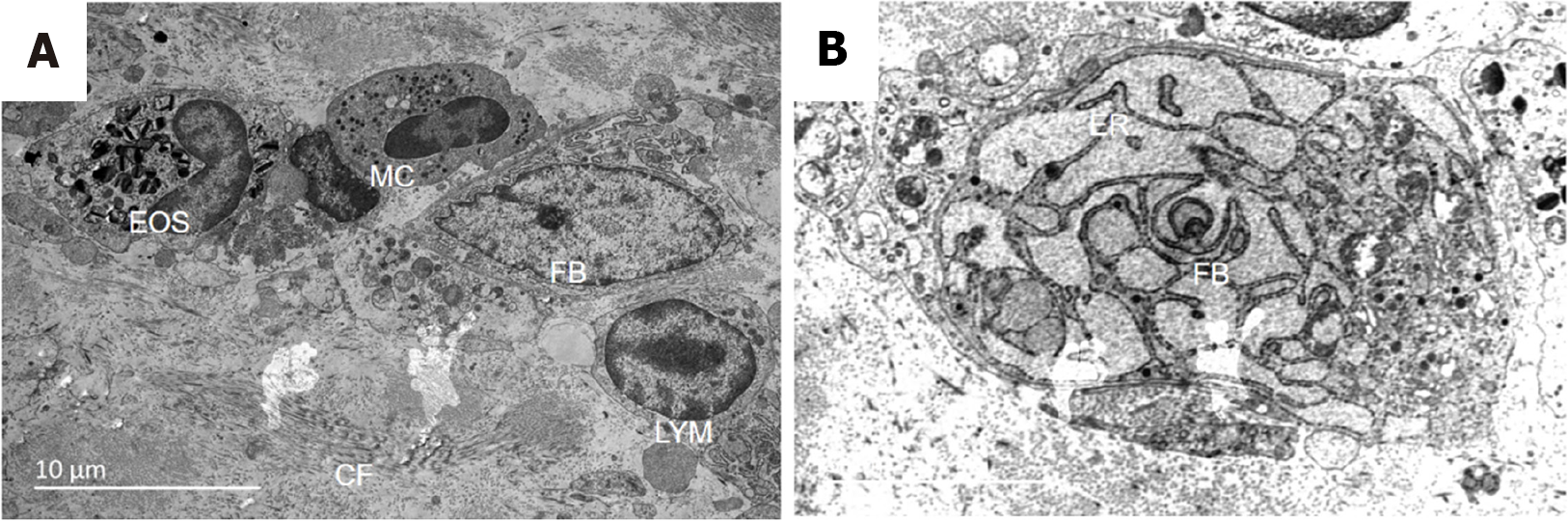
According to spatiotemporal omics localization and transmission electron microscopy, the endoplasmic reticulum of fibroblasts was obviously active, and the endoplasmic reticulum was rich in glycogen. These findings suggest that the fibroblasts were activated after Xiaozhiling injection, which is consistent with the spatiotemporal omics results (Figure 4).
Changes in the expression of the Sphk1-S1P signaling pathway-related proteins Sphk1, S1PR1, and TGF-β1 before and after injection were detected via immunohistochemistry. Tissues were collected from the hemorrhoidal area before and after drug intervention, and the changes in the expression of the Sphk1-S1P signaling pathway-related proteins Sphk1, S1PR1, and TGF-β1 before and after injection were detected via immunohistochemistry. According to the immunohistochemical staining results, the submucosal thickness increased significantly, and the expression of Sphk1, S1PR1, and TGF-β1, and other related indicators increased after Xiaozhiling injection, indicating that the Sphk1-S1P signaling pathway was activated. These results are consistent with the spatial transcriptome data (Figures 5 and 6).
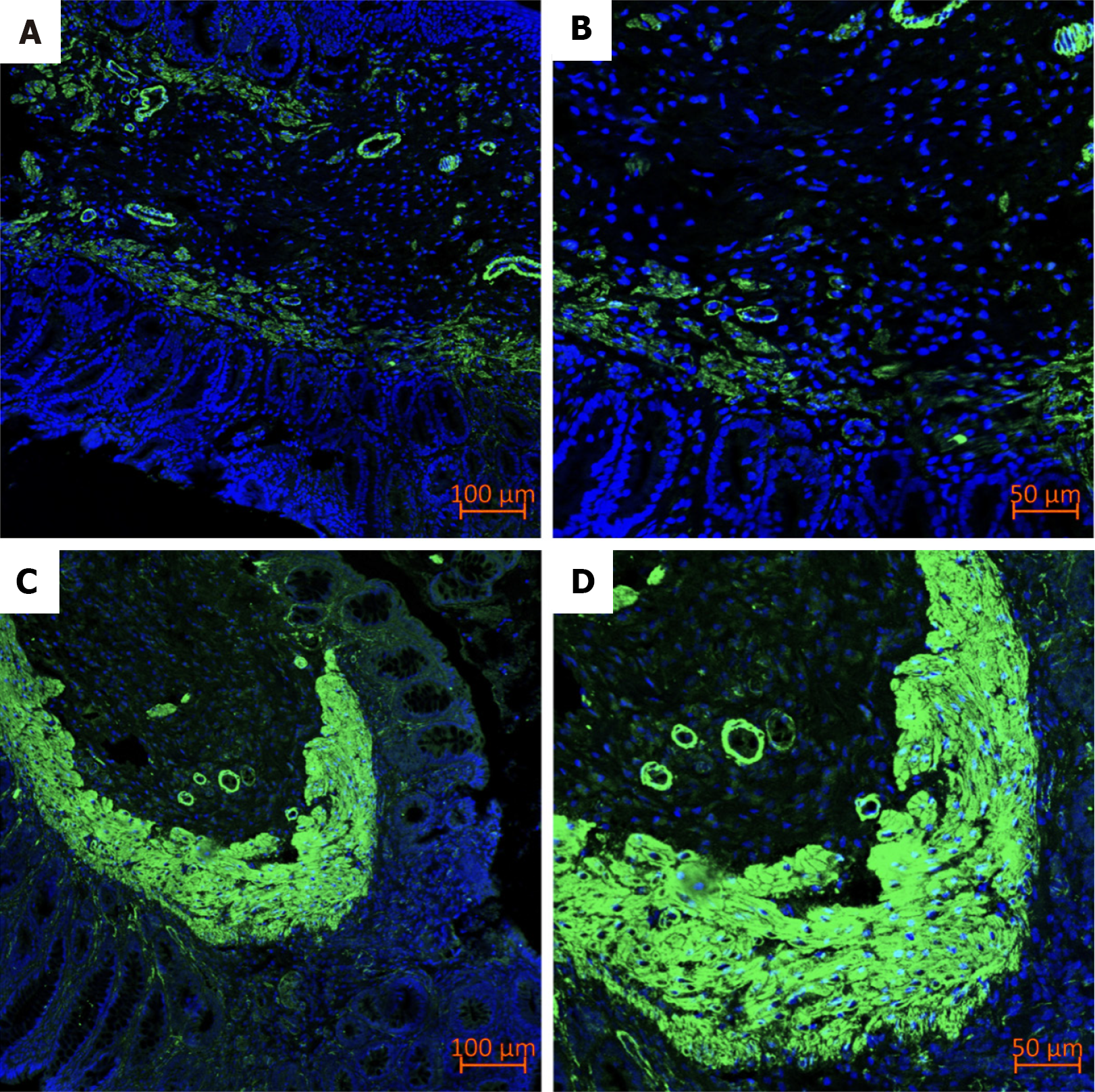
Immunofluorescence staining suggested that alpha-smooth muscle actin (α-SMA) became highly expressed after Xiaozhiling injection (Figure 7).
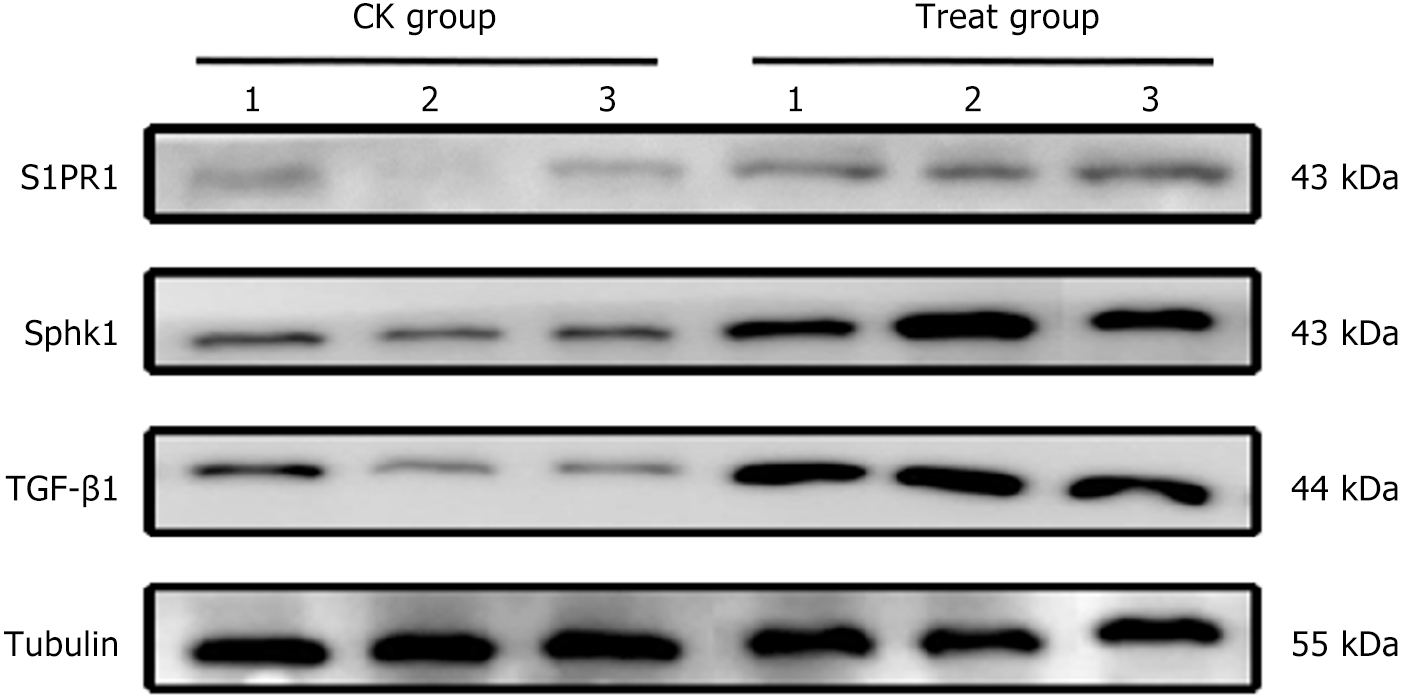
Compared with the CK group, the protein expression of Sphk1, TGF-β1, and S1PR1 in the rectal tissue of the treatment group was significantly elevated (P < 0.05) (Figure 8).
Renowned experts in anorectal diseases applied the traditional Chinese medicine principle that “acid can be convergent, and astringency can be fixed”, and selected gallnut and alum, which possess both sour and astringent properties, to formulate Xiaozhiling. ALTA injection, widely used in Japan, is a modified version of Xiaozhiling. Japanese researchers extracted tannic acid and aluminum potassium sulfate from traditional Chinese medicine to serve as the primary components in the preparation of this medication.
Spatiotemporal transcriptomics analysis has been widely used in the medical field. Qin et al[15] used single-cell RNA sequencing and spatial transcriptome sequencing technologies to characterize the heterogeneity and spatial structure of rectal cancer tissue. The study of cell dynamics in 29 colorectal cancer patients via this technique suggested that adjuvant chemotherapy can modulate the tumor microenvironment by reshaping cancer-associated fibroblast populations. Chen et al[16] used this technology to study the mechanism by which Buzhong Yiqi decoction alleviates rectal prolapse and reported that the expression of mouse platelet-derived growth factor A and mouse platelet-derived growth factor B in smooth muscle cells, pericytes, and fibroblasts in the treatment group was higher than that of the control group. Using this technique, researchers can identify temporal and spatial changes in cell populations, track the precise location of each cell, and determine how cells contribute to the development of disease and the effects of drug interventions over time.
To explore the effects of drug intervention on spatial gene expression patterns, spatial transcriptomics was used to analyze the spatial distribution of genes related to the Sphk1-S1P signaling pathway before Xiaozhiling injection and 1 week after intervention. 10 × Visium spatial transcriptome sequencing and corresponding single-cell RNA sequencing were used to characterize heterogeneity and differences in spatial structure before and after tissue intervention and to explore the mechanism of spatial-temporal changes in detail. Specifically, through single-cell spatial omics technology, we used 10 × Visium spatial transcriptome sequencing and 10 × single-cell RNA sequencing to reveal the heterogeneity of the cell populations and spatial gene expression patterns in internal hemorrhoid tissues before and after Xiaozhiling injection and explored the potential mechanism by which Xiaozhiling injection inhibits the progression of internal hemorrhoids.
There were changes in various parameters at different time points after Xiaozhiling injection; specifically, the thickness of the submucosa was the highest one week after drug intervention and then gradually decreased, suggesting that the transition period of inflammation and fibrosis may be one of the key turning points. In addition, the endoplasmic reticulum of fibroblasts was active and rich in glycogen, as observed via transmission electron microscopy. Therefore, one week after Xiaozhiling injection, single-cell and spatial omics experiments were performed. Notably, the proportion of fibroblasts in the Xiaozhiling group was lower than that of the model group, but the proportion of inflammatory cells was greater in the Xiaozhiling group than in the model group. Considering that inflammation occurred within one week after drug intervention, fibroblasts did not rapidly proliferate temporarily in this period. In the model group, there were 1507 Myofs, while in the Xiaozhiling group, there were 837 Myofs. These results indicated that Xiaozhiling had a significant effect on the proportion of fibroblasts in hemorrhoidal tissue but that the rapid proliferation of fibroblasts did not occur one week after drug intervention. On the basis of the above studies, it is currently speculated that TGF-β1 can significantly upregulate the expression of the Sphk1 gene and protein in fibroblasts and that the mechanism by which Xiaozhiling induces fibrosis is related to the Sphk1-S1P signaling pathway.
TGF-β1 is a multifunctional cytokine that is involved in multiple processes, such as inflammatory infiltration, cell proliferation, cell differentiation, and extracellular matrix deposition in tissue fibrosis, and can be activated by multiple mechanisms[17]. During fibrosis, it is derived mainly from macrophages, and its main effects are as follows: (1) TGF-β1 induces the activation of fibroblasts into Myofs and increases the expression of the Myof marker protein α-SMA[18]; and (2) It promotes the deposition of extracellular matrix and increases the synthesis of matrix components (Col I and Col III). S1P is an important lipid metabolite produced by the phosphorylation of sphingosine, which can act on intracellular targets or be exported outside the cell via related protein transporters and bind to a group of S1P receptors (S1PR, S1P1-5). This leads to the activation of different signaling pathways and subsequent cellular responses. Sphk1 is one of the key enzymes that catalyzes the phosphorylation of S1P. After activation, Sphk1 migrates from the cytoplasm to the cell membrane and catalyzes the phosphorylation of S1P in the catalytic component of the membrane[19]. TGF-β1 can significantly upregulate the gene and protein expression of Sphk1 in fibroblasts and promote fibrosis through the Sphk1/S1P pathway. Studies have confirmed that TGF-β1 affects the Sphk1-S1P signaling pathway to promote Myof activation in the intestine, liver, heart, lung, and other tissues. It also promotes α-SMA and collagen expression, resulting in fibrosis (Figure 2)[20,21]. For example, TGF-β1, a proven profibrotic factor in renal interstitial fibrosis, can inhibit the degradation of the extracellular matrix and promote the production of collagen[22]. S1P is a bioactive sphingolipid metabolite that is produced by the phosphorylation of sphingosine by the highly conserved protein Sphk1, and the Sphk1/S1P signaling pathway can participate in the development of fibrosis and inflammation[23].
The spatial localization of key genes of the Sphk1-S1P and TGF-β1 pathways, i.e., Sphk1, S1PR1, and TGF-β1, was compared (Figure 2E and Figure 3E). The expression levels of Sphk1-S1P and TGF-β1 pathway-related genes (Figure 2F and Figure 3F) were significantly increased in the area where fibroblasts were concentrated after Xiaozhiling injection. The above results reveal that fibroblasts and key genes of the Sphk1-S1P pathway are activated at the injection site. Xiaozhiling was injected into the submucosa of the hemorrhoidal area, and through the upregulation of TGF-β1, a classic marker gene of the Sphk1-S1P signaling pathway, was specifically activated at the injection site, where it regulated MyoF activation. These findings suggest that, after Xiaozhiling injection, the Sphk1-S1P pathway can be activated to promote fibrosis.
| 1. | Li CY, Yuan P. [Minimally Invasive Techniques in Hemorrhoidal Surgery and Prospects]. Shaanxi Yixue Zazhi. 2020;49:915-918. |
| 2. | Wei K, Wei ZZ, Zhao Y. [Clinical effect analysis of moderate to severe hemorrhoids treated with anastomotic suprahemorrhoidal mucosal cricothyrotomy combined with sclerotherapy injection]. Linchuang Yiyao Wenxian Dianzi Zazhi. 2020;7:55-57. [DOI] [Full Text] |
| 3. | Yu TJ, Jiang L, Li WS, Tang MH, Jia YT, Yang XD. [Clinical Efficacy of Xiaozhiling Injection in Treatment of 60 Cases of Internal Hemorrhoids Under Endoscope]. Zhongguo Gangchang Zazhi. 43:23-25. |
| 4. | Chen Q, Li HS. [A review of the development of Anti-Hemorrhoidal Spirit at home and abroad for more than 40 years since its introduction]. Jiangsu Zhongyiyao. 2020;52:76-80. [DOI] [Full Text] |
| 5. | Anorectal Branch of Chinese Medical Doctor Association; Colorectal Surgery Group; Surgical Society of Chinese Medical Association. [Chinese expert consensus on the treatment of hemorrhoids by injection (2023 edition)]. Zhonghua Weichang Waike Zazhi. 2023;26:1103-1111. |
| 6. | Buechler MB, Pradhan RN, Krishnamurty AT, Cox C, Calviello AK, Wang AW, Yang YA, Tam L, Caothien R, Roose-Girma M, Modrusan Z, Arron JR, Bourgon R, Müller S, Turley SJ. Cross-tissue organization of the fibroblast lineage. Nature. 2021;593:575-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 802] [Article Influence: 160.4] [Reference Citation Analysis (3)] |
| 7. | Griffin MF, desJardins-Park HE, Mascharak S, Borrelli MR, Longaker MT. Understanding the impact of fibroblast heterogeneity on skin fibrosis. Dis Model Mech. 2020;13:dmm044164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 145] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 8. | Talbott HE, Mascharak S, Griffin M, Wan DC, Longaker MT. Wound healing, fibroblast heterogeneity, and fibrosis. Cell Stem Cell. 2022;29:1161-1180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 662] [Cited by in RCA: 552] [Article Influence: 138.0] [Reference Citation Analysis (4)] |
| 9. | Shi S, Li MF, Wang L, Gu L, Tian YX, Shu H. [Effects of lipopolysaccharide on expression of fibrosis-related factors in dermal fibroblast and hypertrophic scar formation]. Jiefangjun Yixue Zazhi. 2020;45:604-610. |
| 10. | Younesi FS, Son DO, Firmino J, Hinz B. Myofibroblast Markers and Microscopy Detection Methods in Cell Culture and Histology. Methods Mol Biol. 2021;2299:17-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (3)] |
| 11. | Sferra R, Pompili S, Ventura L, Dubuquoy C, Speca S, Gaudio E, Latella G, Vetuschi A. Interaction between sphingosine kinase/sphingosine 1 phosphate and transforming growth factor-β/Smads pathways in experimental intestinal fibrosis. An in vivo immunohistochemical study. Eur J Histochem. 2018;62:2956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Jiang YJ, Jing ZH, Feng J, Xu X. [Recent Advances in Spatiotemporal Omics Technology]. Zhongguo Shengwu Gongcheng Zazhi. 2024;44:19-31. [DOI] [Full Text] |
| 13. | Shi XP, Zong AN, Tao J, Wang LZ. [Study of instructive notions with respect to caring for laboratory animals]. Zhongguo Yike Daxue Xuebao. 2007;04:493. |
| 14. | Ke M, Huang S, Lin H, Xu Z, Li X, Li Z, Chen F, Wu H. Establishment and study of a rat internal haemorrhoid model. Sci Rep. 2023;13:21385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (3)] |
| 15. | Qin P, Chen H, Wang Y, Huang L, Huang K, Xiao G, Han C, Hu J, Lin D, Wan X, Zheng Y, Liu Y, Li G, Yang H, Ye S, Luo M, Fu Y, Xu H, Wen L, Guo Z, Shen X, Li Z, Wang C, Chen X, Wang L, Sun L, Ren D, Wu L, Wang J, Liu S, Lin H. Cancer-associated fibroblasts undergoing neoadjuvant chemotherapy suppress rectal cancer revealed by single-cell and spatial transcriptomics. Cell Rep Med. 2023;4:101231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 16. | Chen HX, Xiao GZ, Liu F, Lin HC. [Study on the mechanism of Buzhong Yiqi Decoction in the treatment of rectal prolapse: related analysis of single-cell transcriptome sequencing based on surgical excision specimens]. Jiezhichang Gangmen Waike. 2023;29:38-46. [DOI] [Full Text] |
| 17. | Tzavlaki K, Moustakas A. TGF-β Signaling. Biomolecules. 2020;10:487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 702] [Cited by in RCA: 627] [Article Influence: 104.5] [Reference Citation Analysis (4)] |
| 18. | Brenmoehl J, Miller SN, Hofmann C, Vogl D, Falk W, Schölmerich J, Rogler G. Transforming growth factor-beta 1 induces intestinal myofibroblast differentiation and modulates their migration. World J Gastroenterol. 2009;15:1431-1442. [PubMed] [DOI] [Full Text] |
| 19. | Adams DR, Pyne S, Pyne NJ. Sphingosine Kinases: Emerging Structure-Function Insights. Trends Biochem Sci. 2016;41:395-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 20. | Xiu L, Chang N, Yang L, Liu X, Yang L, Ge J, Li L. Intracellular sphingosine 1-phosphate contributes to collagen expression of hepatic myofibroblasts in human liver fibrosis independent of its receptors. Am J Pathol. 2015;185:387-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 21. | Zou X, Yu B, Xu LH, Ma JW, Liu XJ. [Effect of Evodiamine on Renal Fibrosis in Rats with Chronic Renal Failure by Regulating Sphk1/S1P Signaling Pathway]. Guangzhou Zhongyiyao Daxue Xuebao. 2024;41:1560-1566. [DOI] [Full Text] |
| 22. | Nie LH, Su AS, Zhu SP. [Effect of Manshenkangning prescription on kidneys of renal interstitial fibrosis rats caused by adenine]. Zhongguo Linchuang Yaolixue Zazhi. 2019;35:1255-1259. [DOI] [Full Text] |
| 23. | Xiao JL, Zou X, Dan JP. [Effects of alfentanil on myocardial fibrosis in rats with acute myocardial infarction by regulating the Sphk1/S1P signaling pathway]. Zhongguo Yaofang. 2024;35:955-960. |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/