Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.102980
Revised: December 26, 2024
Accepted: March 3, 2025
Published online: April 27, 2025
Processing time: 120 Days and 23.5 Hours
Congenital intestinal atresia (CIA) is a common intestinal malformation in the neonatal period, and surgery is currently the main treatment method. The choice of postoperative feeding is crucial for the recovery of gastrointestinal function in children.
To compare and analyze the effects of different postoperative feeding methods on gastrointestinal function reconstruction in newborns with CIA.
Twenty-six children diagnosed with neonatal CIA, treated with minimally invasive surgery at Shijiazhuang Maternal and Child Health Hospital between January 2021 and May 2024, were selected for this single-center prospective randomized controlled study. They were divided into two groups using envelope randomization: Enteral nutrition (EN) group (n = 13) and parenteral nutrition (PN) group (n = 13). Baseline and clinical characteristics were collected, and recovery time of bowel sounds and time to first defecation were used as eva
The time to bowel sound recovery (51.54 vs 65.85, P = 0.013) and first defecation (58.15 vs 76.62, P < 0.001) was shorter in the EN group compared to the PN group. Clinical improvements in the EN group, including discharge weight (P = 0.044), hospital stay (P = 0.027), white blood cell count (P = 0.023), albumin content (P = 0.013), and direct bilirubin content (P = 0.018), were also better than those in the PN group. No substantial differences in postoperative complications were found between the groups. Correlation analysis indicated that abdominal infection and operation time may relate to time to first defecation. Linear regression analysis demonstrated a considerable association between EN feeding and shorter time to first defecation. Abdominal infection and an operation time > 2 hours may be risk factors for prolonged time to first defecation.
EN substantially promotes the recovery of gastrointestinal function after CIA in neonates and can improve clinical outcomes in children. Future research should explore optimal EN practices to enhance clinical application and child health.
Core Tip: This study demonstrated that enteral nutrition (EN) significantly accelerates gastrointestinal function recovery in neonates with congenital intestinal atresia after surgery compared to parenteral nutrition. EN reduces bowel sound recovery time and time to first defecation, while improving nutritional status and clinical outcomes. These findings highlight the importance of prioritizing EN in postoperative feeding strategies for neonatal congenital intestinal atresia to enhance recovery and overall prognosis.
- Citation: Kang HL, Zhao YZ. Association between postoperative feeding patterns and gastrointestinal function reconstruction after congenital intestinal atresia in neonates. World J Gastrointest Surg 2025; 17(4): 102980
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/102980.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.102980
Congenital intestinal atresia (CIA) is one of the most common intestinal malformations observed in the neonatal period, affecting approximately 1 in 5000 to 1 in 10000 live births[1]. Although the incidence of CIA is relatively low, it is a severe condition that often leads to substantial feeding difficulties and intestinal obstruction symptoms after birth. This condition markedly affects the growth and development of newborns, their quality of life, and can even pose life-threatening risks[2]. The pathogenesis of CIA is not fully understood; however, it is believed to be associated with small intestinal insufficiency during embryonic development, vascular accident, inflammatory infections, and genetic variations[3,4]. Surgery is currently the preferred treatment for CIA, because it can restore intestinal patency by removing the atretic segment of the intestine[5]. With advancements in medical technology, particularly the increased use of minimally invasive surgical techniques, the outcomes of surgical treatment for CIA in newborns have markedly improved, leading to enhanced postoperative survival rates[6]. Postoperative recovery in these children is influenced not only by the quality of the intestinal surgery but also by the reconstruction of intestinal function. Studies have indicated that faster recovery of intestinal function post-surgery correlates with a lower risk of surgery-related complications[7]. Therefore, the reconstruction of intestinal function serves as a key indicator for evaluating the efficacy of surgical treatment for CIA in children.
The choice of postoperative feeding mode has long been a key research focus in neonatal surgery. Currently, enteral nutrition (EN) and parenteral nutrition (PN) are two common nutritional support methods for newborns after surgery. EN is digested and absorbed via the gastrointestinal tract, stimulating the normal secretion of gastric acid, digestive fluids, and other digestive secretions; facilitate gallbladder contraction and gastrointestinal peristalsis; and contribute to the restoration of intestinal function[8]. However, EN may lead to feeding intolerance, insufficient nutrient intake, and other issues in children whose gastrointestinal function does not recover in the early postoperative period. In contrast, PN can bypass the damaged intestine, provide essential nutritional support, reduce intestinal burden, and facilitate intestinal rest and recovery[9]. However, long-term PN may lead to intestinal mucosal atrophy, intestinal flora imbalance, cholestasis, and other complications, which can adversely affect the recovery of intestinal function[10]. Therefore, selecting an appropriate feeding method to promote the reconstruction of gastrointestinal function after neonatal congenital atresia has become a focus of clinicians’ attention.
Some studies have investigated the clinical effects of EN and PN after CIA in newborns; however, these studies have not investigated the relationship between EN and gastrointestinal functional reconstruction[11]. For children with CIA following surgery, the reconstitution of gastrointestinal function is critical for long-term prognosis and quality of life. Identifying the factors influencing intestinal function recovery in children after CIA surgery is essential for formulating timely, targeted intervention strategies to promote postoperative recovery and improve prognosis. This study employed a prospective randomized controlled design to investigate the association between different feeding patterns and neonatal gastrointestinal functional reconstruction after CIA surgery. The findings of this study provide a theoretical basis for selecting postoperative feeding methods for newborns with CIA and will help optimize clinical treatment. Additionally, this study is anticipated to provide useful references for the postoperative feeding management of other types of congenital digestive tract malformations.
A total of 26 pediatric patients with CIA treated at Shijiazhuang Maternal and Child Health Hospital were included in this study, with a case collection period from January 2021 to May 2024. All patients met the following inclusion criteria: (1) Neonates diagnosed with CIA based on a comprehensive clinical evaluation; (2) Patients eligible for surgical intervention for CIA; and (3) Patients with complete clinical data. The exclusion criteria were as follows: (1) Patients with other severe congenital malformations or genetic disorders; (2) Patients with severe malnutrition or complications before surgery; (3) Patients with severe impairment or necrosis of intestinal function before surgery; and (4) Patients who abandoned treatment for various reasons. This study was approved by the Shijiazhuang Maternal and Child Health Hospital Ethics Committee, and all guardians of the children voluntarily provided informed consent.
A single-center prospective randomized controlled design was employed to investigate the effects of postoperative feeding patterns on gastrointestinal function reconstruction after CIA. All children were randomly assigned to either the EN group or the PN group immediately after surgery, with 13 patients in each group. The grouping was conducted using envelope randomization to ensure both randomness and blinding during the allocation process. After surgery, once it was confirmed that the patients met the inclusion criteria, a third party not involved in the follow-up treatment or evaluation assigned them to groups according to the instructions in the envelope, ensuring the fairness of the grouping results.
Basic characteristics of the patients were collected, including age, sex, birth weight, location and type of CIA, duration of surgery, presence of abdominal infection, gestational duration, and length of bowel resection. Post-intervention characteristics of the patients were collected, including discharge weight, length of stay, complications, white blood cell (WBC) count, C-reactive protein, hemoglobin, albumin (ALB), and direct bilirubin (DB). After surgery, the abdomen was auscultated every 2 hours, and the time of the first recovery of bowel sounds was recorded, with clear, continuous bowel sounds lasting at least 3 minutes serving as the recovery standard. The time of the first excretion of fetal stool or formed stool after the surgery was recorded. Bowel sound recovery time and time to first defecation were used as indices to evaluate postoperative gastrointestinal function reconstruction.
In the EN group, once the vital signs of the children stabilized postoperatively, an appropriate amount of enteral nutrient solution was administered through a nasogastric or fistula tube as soon as feasible. In the early phase, infant formula or hydrolyzed protein formula, which is easily digestible and absorbable, was utilized as the EN solution. A small number of meals were provided, with the initial volume of each feeding kept moderate to avoid excessive intestinal burden. Generally, feeding started at 5-10 mL each time, gradually increasing to 20-30 mL or more, depending on the tolerance of the child. Feedings typically occurred 8-12 times per day to ensure the child received sufficient nutritional support. Simultaneously, the child’s swallowing function and oral motor capacity were regularly evaluated in preparation for subsequent oral feeding. In the PN group, central or peripheral intravenous parenteral nutritional support was provided after surgery, including glucose, amino acids, fat milk, electrolytes, vitamins, and trace elements. The infusion volume and rate were adjusted based on the weight and nutritional requirements of the children, with a gradual transition to EN after the restoration of gastrointestinal function.
SPSS 26.0 statistical software was used for data analysis. Continuous variables were described as mean ± SD and analyzed using independent sample t-tests. Categorical variables were described in terms of frequency and percentage, and comparisons between groups were conducted using the χ2 test or Fisher’s exact test. Spearman’s correlation tests were employed to analyze the association between the time of the first bowel movement after surgery and other variables. In addition, with the time to first defecation as the dependent variable, a linear regression model was used to analyze factors influencing postoperative gastrointestinal function recovery. All statistical tests were two-tailed, and statistical significance was set at P < 0.05.
A total of 26 neonatal patients with CIA were included in this study. The mean age of the PN group was 21.85 ± 14.15 hours, while the mean age of the EN group was 25.08 ± 16.12 hours, with no statistically significant difference between the two groups (P = 0.592) (Table 1). In addition, there were no substantial differences between the two groups in terms of sex, gestational duration, birth weight, length of intestinal resection, location and type of CIA, surgical duration, or the presence of abdominal infection (Table 1).
| Variables | Total (n = 26) | PN group (n = 13) | EN group (n = 13) | Statistic | P value |
| Age (hour), mean ± SD | 23.46 ± 14.95 | 21.85 ± 14.15 | 25.08 ± 16.12 | t = -0.54 | 0.592 |
| Sex | - | 1.000 | |||
| Female | 14 (53.85) | 7 (53.85) | 7 (53.85) | ||
| Male | 12 (46.15) | 6 (46.15) | 6 (46.15) | ||
| Gestation days (day), mean ± SD | 256.00 ± 14.93 | 253.54 ± 14.76 | 258.46 ± 15.28 | t = -0.84 | 0.412 |
| Birth weight (kg), mean ± SD | 2.87 ± 0.58 | 2.84 ± 0.58 | 2.91 ± 0.61 | t = -0.31 | 0.761 |
| Intestinal resection length (cm), mean ± SD | 23.14 ± 7.58 | 23.90 ± 7.62 | 22.37 ± 7.76 | t = 0.51 | 0.615 |
| CIA location | - | 1.000 | |||
| Ileum | 13 (50.00) | 7 (53.85) | 6 (46.15) | ||
| Duodenum | 9 (34.62) | 4 (30.77) | 5 (38.46) | ||
| Colon | 4 (15.38) | 2 (15.38) | 2 (15.38) | ||
| CIA type | - | 1.000 | |||
| I and II type | 11 (42.31) | 6 (46.15) | 5 (38.46) | ||
| III and IV type | 15 (57.69) | 7 (53.85) | 8 (61.54) | ||
| Surgical duration | - | 0.434 | |||
| < 2 hours | 13 (50.00) | 5 (38.46) | 8 (61.54) | ||
| ≥ 2 hours | 13 (50.00) | 8 (61.54) | 5 (38.46) | ||
| Abdominal infection | - | 1.000 | |||
| No | 17 (65.38) | 8 (61.54) | 9 (69.23) | ||
| Yes | 9 (34.62) | 5 (38.46) | 4 (30.77) |
The mean discharge weight of patients in the PN group was 3.09 kg, which was significantly higher than that of 3.56 kg in the EN group (P = 0.044) (Table 2). The mean length of hospital stay was significantly longer in the PN group than in the EN group (48.23 vs 35.62, P = 0.027) (Table 2). Regarding serum markers, there were significant differences in WBC (8.96 vs 7.98, P = 0.023), ALB (31.67 vs 34.29, P = 0.013), and DB (19.47 vs 12.31, P = 0.018) levels between the PN and EN groups (Table 2). No significant differences were observed in C-reactive protein (1.07 vs 1.15, P = 0.390) or hemoglobin (106.00 vs 111.92, P = 0.401) levels between the two groups (Table 2).
| Variables | Total (n = 26) | PN group (n = 13) | EN group (n = 13) | Statistic | P value |
| Discharge weight (kg), mean ± SD | 3.33 ± 0.60 | 3.09 ± 0.56 | 3.56 ± 0.57 | t = -2.13 | 0.044a |
| Hospital stays (day), mean ± SD | 41.92 ± 14.88 | 48.23 ± 15.01 | 35.62 ± 12.24 | t = 2.35 | 0.027a |
| WBC (109/L), mean ± SD | 8.47 ± 1.13 | 8.96 ± 1.04 | 7.98 ± 1.02 | t = 2.43 | 0.023a |
| CRP (μg/mL), mean ± SD | 1.11 ± 0.22 | 1.07 ± 0.21 | 1.15 ± 0.23 | t = -0.88 | 0.390 |
| HGB (g/L), mean ± SD | 108.96 ± 17.57 | 106.00 ± 14.81 | 111.92 ± 20.12 | t = -0.85 | 0.401 |
| ALB (IU/L), mean ± SD | 32.98 ± 2.78 | 31.67 ± 3.12 | 34.29 ± 1.63 | t = -2.69 | 0.013a |
| DB (μmol/L), mean ± SD | 15.89 ± 7.95 | 19.47 ± 8.14 | 12.31 ± 6.14 | t = 2.53 | 0.018a |
Postoperative complications occurred in six patients (46.15%) in the PN group and three patients (23.08%) in the EN group, with no significant difference (P = 0.411) (Table 3). In the PN group, there were three cases of abdominal distension, two cases of emesis, and one case of incision infection after the intervention. In the EN group, there was one case of abdominal distension, two cases of emesis, and no cases of incision infection. No significant differences were found in the subgroup analysis of complications between the two groups (P > 0.05) (Table 3).
| Type | PN group (n = 13) | EN group (n = 13) | P value |
| Abdominal distension | 3 (23.08) | 1 (7.69) | 0.593 |
| Emesis | 2 (15.38) | 2 (15.38) | 1.000 |
| Incision infection | 1 (7.69) | 0 (0.00) | 1.000 |
| Total | 6 (46.15) | 3 (23.08) | 0.411 |
The recovery time of bowel sounds in the PN group was 65.85 ± 13.10 hours, while in the EN group it was 51.54 ± 13.91 hours (Table 4). The recovery time for bowel sounds was significantly shorter in the EN group than in the PN group (P = 0.013) (Table 4). The time to first defecation was 76.62 ± 12.04 hours in the PN group and 58.15 ± 12.95 hours in the EN group, indicating that the defecation recovery speed in the EN group was significantly faster than that in the PN group (P < 0.001) (Table 4). In summary, EN substantially promoted gastrointestinal function reconstruction after CIA surgery.
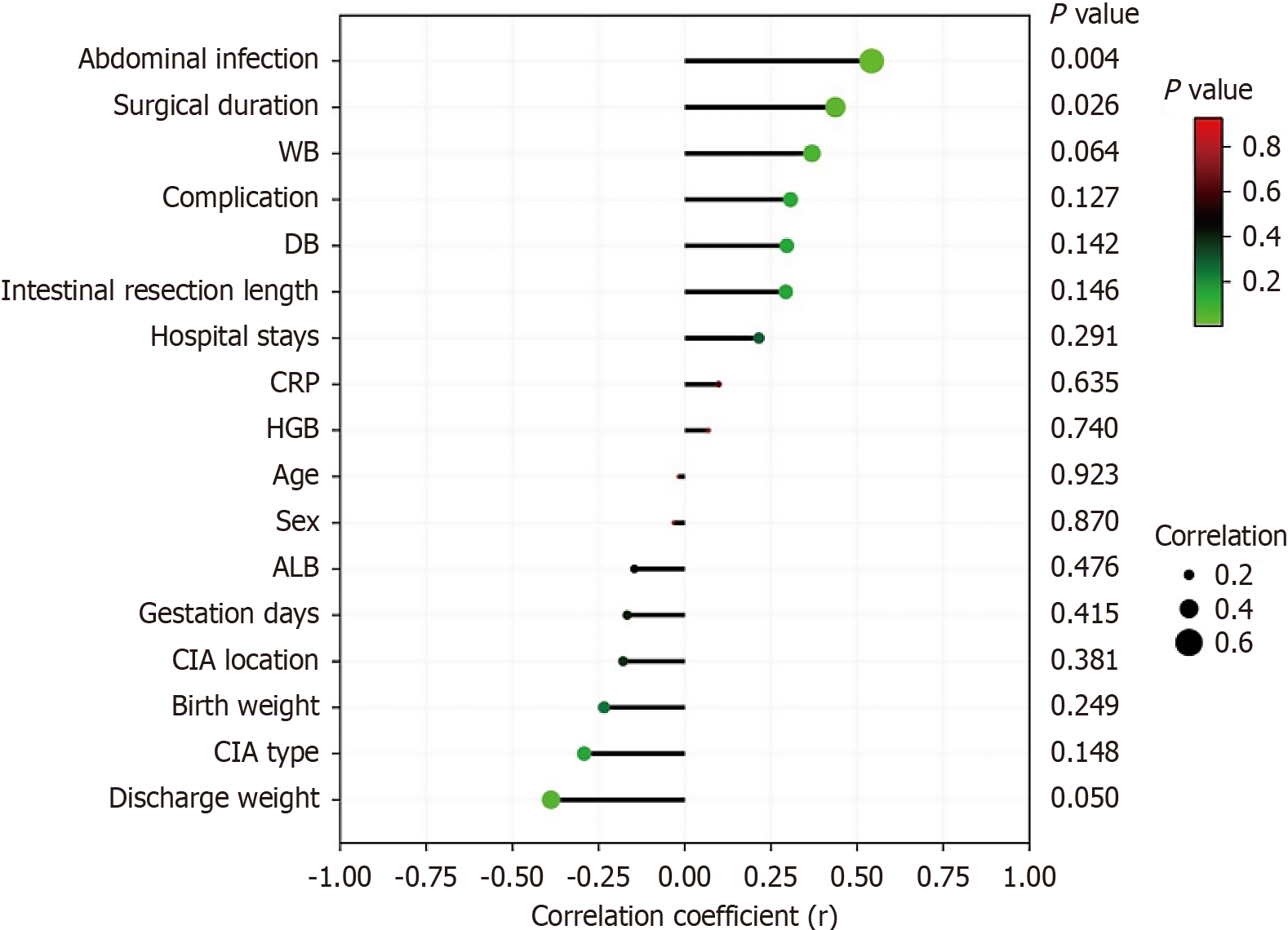
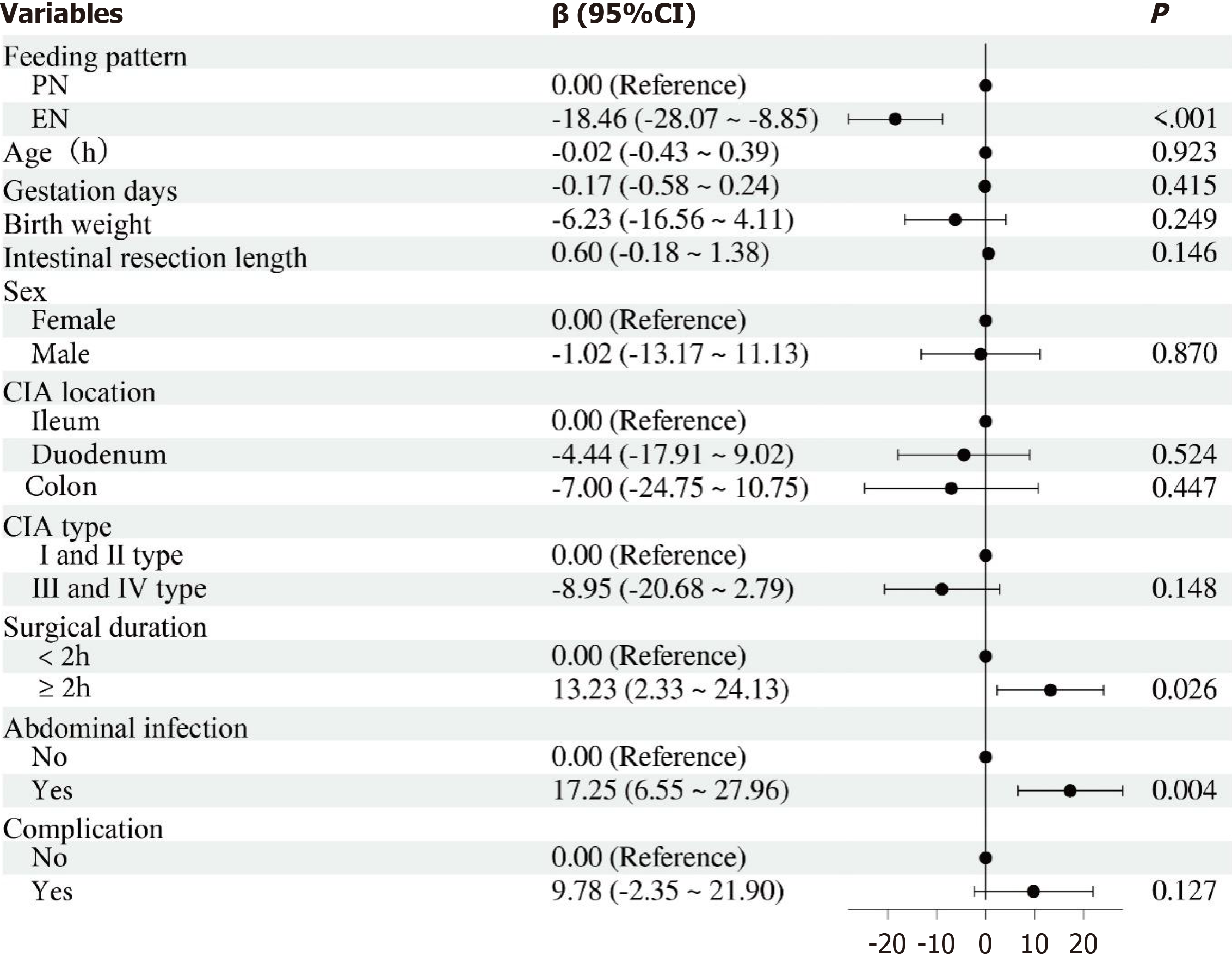
Spearman’s correlation analysis demonstrated a significant association between prolonged time to first defecation and the presence of abdominal infection (P = 0.004) (Figure 1). There was also a significant association between an operation time > 2 hours and delayed first defecation (P = 0.024) (Figure 1). The time to first defecation was used as the index of intestinal function recovery for linear regression analysis. The results indicated that EN feeding was associated with improved postoperative gastrointestinal function recovery (P < 0.001) (Figure 2). In addition, an operation time > 2 hours and the presence of abdominal infection limited the effectiveness of intestinal function recovery after CIA surgery (P < 0.001) (Figure 2).
CIA can lead to neonatal intestinal obstruction, which poses a serious threat to the lives of affected children[12]. Surgery is the only viable option for treating CIA in neonates, and the choice of postoperative feeding mode is of great significance for the recovery of gastrointestinal function and the long-term prognosis of these patients[13]. Although intravenous hyperalimentation is sufficient to correct malnutrition in the short term, long-term use may lead to multiple complications, such as hepatic steatosis, cholestasis, and intestinal flora imbalance[14]. EN can directly provide nutri
One of the core findings was that, compared to PN, EN markedly reduced the time to bowel sound recovery and the time to first bowel movement in children with congenital atresia after surgery. These results directly support the beneficial role of EN in promoting postoperative recovery of gastrointestinal function. The recovery of bowel sounds and the occurrence of the first defecation are important indicators of restored gastrointestinal function[16]. Their early manifestation indicates recovery of intestinal motility and enhancement of intestinal barrier function. By directly stimulating the intestinal mucosa, EN promotes the secretion of intestinal hormones such as motilin and cholecystokinin, which are crucial for regulating intestinal movement and facilitating digestion and absorption[16]. In addition, EN helps maintain the balance of intestinal flora, reduces the reproduction of harmful bacteria, and promotes the recovery of intestinal function[17].
This study also found that EN positively affects clinical outcomes. Patients who received EN were discharged with higher body weight, had a shorter hospital stay, and exhibited lower WBC counts and DB levels, along with higher ALB levels. The comprehensive improvement in these indices reflects the advantages of EN in enhancing the overall nutritional status of children, reducing the inflammatory response, and promoting the recovery of liver function. The increase in discharge weight directly reflects improved nutritional status, while the shorter hospital stay indicates more effective use of medical resources and a reduced burden on families. A decrease in WBC count may indicate a reduction in the postoperative systemic inflammatory response, which is a positive indicator for recovery[18]. As one of the main proteins in the plasma, the increase in ALB content reflects improvements in nutritional status and intestinal function in the EN group[19]. DB is closely associated with liver metabolism and excretion. A decrease in DB typically indicates improved liver function or unobstructed bile excretion, both of which are positive signs of liver function recovery[20]. The study also identified that abdominal infection and surgery duration greater than 2 hours as independent risk factors for prolonged recovery of bowel function in patients. As a common complication of CIA, abdominal infection can exacerbate the intestinal inflammatory response and destroy the intestinal mucosal barrier, thereby increasing surgical risks and prolonging the recovery of intestinal function[21]. Prolonged surgery duration may augment surgical trauma, affect blood circulation, and disrupt nerve regulation of the intestine, subsequently delaying recovery of intestinal function. Therefore, in clinical practice, considerable attention should be devoted to the prevention and treatment of abdominal infections, and efforts should be made to minimize surgical duration to mitigate adverse impacts on intestinal function.
In addition, the results of correlation analyses also showed that EN was significantly associated with shorter time to first bowel movement. The clinical significance of this finding lies in the fact that EN may promote the secretion of intestinal hormones (e.g., gastric motility and cholecystokinin) through direct stimulation of the intestinal mucosa, thereby regulating intestinal motility and promoting digestion and absorption[22]. In addition, abdominal infections were significantly associated with prolonged time to first bowel movement. Abdominal infections may exacerbate the intestinal inflammatory response and disrupt the intestinal mucosal barrier, thereby increasing the risk of surgery and prolonging the recovery of bowel function[23]. This finding highlights the importance of prevention and treatment of abdominal infections in clinical practice, as well as minimising trauma and controlling the duration of surgery to mitigate adverse effects on bowel function. Surgical times longer than 2 hours were associated with delayed recovery of bowel function. This may be due to the fact that longer surgical time may increase surgical trauma, affect blood circulation, and interfere with neuromodulation of the bowel, thus delaying the recovery of bowel function. This finding suggests that we should strive to optimise the surgical process and reduce unnecessary surgical time in our clinical practice to promote rapid recovery of bowel function in children after surgery.
Based on the findings of this study, we propose the following strategies for the clinical application of EN: First, EN should be initiated as soon as possible in children with CIA after surgery to fully utilize its promoting effect on gastrointestinal function recovery. Second, during EN implementation, the intestinal tolerance of children should be closely monitored, and the type, concentration, and infusion rate of the nutrient solution should be adjusted promptly to avoid the occurrence of excessive intestinal burden or insufficient nutrition. Finally, for children with abdominal infections or prolonged surgery durations, more proactive intervention measures should be taken, such as enhancing anti-infection treatment and optimizing the surgical plan, to shorten the recovery time of intestinal function and improve clinical outcomes.
Nevertheless, this study has some limitations. First, being a single-center study with a relatively small sample size may have introduced selection bias. Secondly, the follow-up time of this study is limited, and only the short-term effects of feeding methods on patients can be observed. Longer follow-up is needed to further evaluate the long-term benefits of both feeding regimens. Finally, the choice of feeding mode may be influenced by a variety of factors, such as financial burdens and consumption of medical resources, which are crucial considerations in clinical decision-making. This study indicates that EN has substantial advantages in promoting gastrointestinal function recovery after CIA surgery in newborns and can improve clinical outcomes in pediatric patients. Future research should focus on exploring the best practices for EN to better serve clinical practice and the health of pediatric patients.
Through prospective randomized controlled trials, this study revealed that EN was substantially superior in facilitating the restoration of gastrointestinal function in newborns after CIA surgery. Specifically, the recovery time of bowel sounds and the first defecation in the EN group was markedly shorter than in the PN group, thereby directly substantiating that EN can effectively stimulate intestinal motility and expedite recovery of intestinal function. Secondly, EN positively influences the nutritional status of children, mitigates systemic inflammatory responses, and promotes liver metabolic functions. Additionally, intraperitoneal infection and surgery duration exceeding 2 hours were identified as independent risk factors for prolonged recovery of bowel function in patients. In conclusion, the results of this study support prioritizing EN as a feeding modality after neonatal CIA to promote the rapid recovery of gastrointestinal function and enhance clinical outcomes. Future studies can further refine the implementation plan of EN and explore its application in diverse clinical scenarios to offer more precise and effective nutritional support strategies in clinical practice.
| 1. | Koenig SM, Russell RT, Quevedo OG, Chen MK. Intestinal Atresias: A Ten-Year Evaluation of Outcomes. J Surg Res. 2024;296:130-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 2. | Jiang JH, Tsai YW, Lee SY, Chuang JH. Biliary atresia associated with small-intestine atresia: A condition of high morbidity and mortality. Asian J Surg. 2022;45:1897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Sabbatini S, Ganji N, Chusilp S, Balsamo F, Li B, Pierro A. Intestinal atresia and necrotizing enterocolitis: Embryology and anatomy. Semin Pediatr Surg. 2022;31:151234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Dannheim K, Ouahed J, Field M, Snapper S, Raphael BP, Glover SC, Bishop PR, Bhesania N, Kamin D, Thiagarajah J, Goldsmith JD. Pediatric Gastrointestinal Histopathology in Patients With Tetratricopeptide Repeat Domain 7A (TTC7A) Germline Mutations: A Rare Condition Leading to Multiple Intestinal Atresias, Severe Combined Immunodeficiency, and Congenital Enteropathy. Am J Surg Pathol. 2022;46:846-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Kudo H, Kazama T, Fukuzawa T, Ando R, Okubo R, Sakurai T, Hashimoto M, Endo Y, Nio M, Wada M. Utility of the web excision with pre-membranous incision for congenital intestinal atresia-type I and stenosis. BMC Pediatr. 2024;24:454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | Fung ACH, Lee MK, Lui MPK, Lip LY, Chung PHY, Wong KKY. Primary anastomosis is the preferred surgical approach for proximal intestinal atresia: a retrospective 20-year analysis. Pediatr Surg Int. 2023;39:99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Lin Q, Sun X, She M. Analysis of influencing factors of gastrointestinal function recovery after congenital intestinal atresia in children. Minerva Gastroenterol (Torino). 2023;69:319-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Razzaghy J, Shukla VV, Gunawan E, Reeves A, Nguyen K, Salas AA. Early and exclusive enteral nutrition in infants born very preterm. Arch Dis Child Fetal Neonatal Ed. 2024;109:378-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 9. | Morris EE, Miller NC, Marka NA, Super JL, Nagel EM, Gonzalez JD, Demerath EW, Ramel SE. Randomized Trial of Early Enhanced Parenteral Nutrition and Later Neurodevelopment in Preterm Infants. Nutrients. 2022;14:3890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 10. | Wang YS, Shen W, Yang Q, Lin R, Tang LX, Bai RM, Yang D, Zhang J, Zhang YJ, Yu WT, Song SR, Kong J, Song SY, Mao J, Tong XM, Li ZK, Wu F, Lin XZ. Analysis of risk factors for parenteral nutrition-associated cholestasis in preterm infants: a multicenter observational study. BMC Pediatr. 2023;23:250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 11. | Wang J, Xu H, Wang J, Xiao D. Evaluation of postoperative feeding strategies in children with intestinal atresia: A single-center retrospective study. Front Pediatr. 2022;10:953852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Choi G, Je BK, Kim YJ. Gastrointestinal Emergency in Neonates and Infants: A Pictorial Essay. Korean J Radiol. 2022;23:124-138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 13. | Zheng Z, Jin Z, Gao M, Tang C, Gong Y, Huang L, Liu Y, Wang J. Comparison of Hand-Sewn with Stapled Anastomosis in Neonatal Intestinal Atresia Surgery: A Randomized Controlled Study. J Laparoendosc Adv Surg Tech A. 2022;32:696-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Brossier DW, Tume LN, Briant AR, Jotterand Chaparro C, Moullet C, Rooze S, Verbruggen SCAT, Marino LV, Alsohime F, Beldjilali S, Chiusolo F, Costa L, Didier C, Ilia S, Joram NL, Kneyber MCJ, Kühlwein E, Lopez J, López-Herce J, Mayberry HF, Mehmeti F, Mierzewska-Schmidt M, Miñambres Rodríguez M, Morice C, Pappachan JV, Porcheret F, Reis Boto L, Schlapbach LJ, Tekguc H, Tziouvas K, Parienti JJ, Goyer I, Valla FV; Metabolism Endocrinology and Nutrition section of the European Society of Pediatric and Neonatal Intensive Care (ESPNIC). ESPNIC clinical practice guidelines: intravenous maintenance fluid therapy in acute and critically ill children- a systematic review and meta-analysis. Intensive Care Med. 2022;48:1691-1708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 15. | Shores DR, Alaish SM, Aucott SW, Bullard JE, Haney C, Tymann H, Nonyane BAS, Schwarz KB. Postoperative Enteral Nutrition Guidelines Reduce the Risk of Intestinal Failure-Associated Liver Disease in Surgical Infants. J Pediatr. 2018;195:140-147.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 16. | Ma M, Zheng Z, Zeng Z, Li J, Ye X, Kang W. Perioperative Enteral Immunonutrition Support for the Immune Function and Intestinal Mucosal Barrier in Gastric Cancer Patients Undergoing Gastrectomy: A Prospective Randomized Controlled Study. Nutrients. 2023;15:4566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 17. | Liu Z, Li N, Dang Q, Liu L, Wang L, Li H, Han X. Exploring the roles of intestinal flora in enhanced recovery after surgery. iScience. 2023;26:105959. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 18. | Zhu B, Feng X, Jiang C, Mi S, Yang L, Zhao Z, Zhang Y, Zhang L. Correlation between white blood cell count at admission and mortality in COVID-19 patients: a retrospective study. BMC Infect Dis. 2021;21:574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 19. | Godala M, Gaszyńska E, Walczak K, Małecka-Wojciesko E. Evaluation of Albumin, Transferrin and Transthyretin in Inflammatory Bowel Disease Patients as Disease Activity and Nutritional Status Biomarkers. Nutrients. 2023;15:3479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 20. | Liao FM, Chang KC, Wu JF, Chen HL, Ni YH, Chang MH. Direct Bilirubin and Risk of Biliary Atresia. Pediatrics. 2022;149:e2021053073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Saldana-Morales FB, Kim DV, Tsai MT, Diehl GE. Healthy Intestinal Function Relies on Coordinated Enteric Nervous System, Immune System, and Epithelium Responses. Gut Microbes. 2021;13:1-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 22. | Doley J. Enteral Nutrition Overview. Nutrients. 2022;14:2180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 23. | Huston JM, Barie PS, Dellinger EP, Forrester JD, Duane TM, Tessier JM, Sawyer RG, Cainzos MA, Rasa K, Chipman JG, Kao LS, Pieracci FM, Colling KP, Heffernan DS, Lester J; Therapeutics and Guidelines Committee. The Surgical Infection Society Guidelines on the Management of Intra-Abdominal Infection: 2024 Update. Surg Infect (Larchmt). 2024;25:419-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 55] [Article Influence: 27.5] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/