Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.102862
Revised: January 29, 2025
Accepted: February 18, 2025
Published online: April 27, 2025
Processing time: 148 Days and 16.7 Hours
Anastomotic leakage (AL) is a serious complication following rectal cancer surgery and is associated with increased recurrence, mortality, extended hospital stays, and delayed chemotherapy. The Onodera prognostic nutritional index (OPNI) and inflammation-related biomarkers, such as the neutrophil-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have been studied in the context of cancer prognosis, but their combined efficacy in predicting AL remains unclear.
To investigate the relationships between AL and these markers and developed a predictive model for AL.
A retrospective cohort study analyzed the outcomes of 434 patients who had undergone surgery for rectal cancer at a tertiary cancer center from 2016 to 2023. The patients were divided into two groups on the basis of the occurrence of AL: One group consisted of patients who experienced AL (n = 49), and the other group did not (n = 385). The investigation applied logistic regression to develop a risk prediction model utilizing clinical, pathological, and laboratory data. The efficacy of this model was then evaluated through receiver operating characteristic curve analysis.
In the present study, 11.28% of the participants (49 out of 434 participants) suffered from AL. Multivariate analysis revealed that preoperative levels of the OPNI, NLR, and PLR emerged as independent risk factors for AL, with odds ratios of 0.705 (95%CI: 0.641-0.775, P = 0.012), 1.628 (95%CI: 1.221-2.172, P = 0.024), and 0.994 (95%CI: 0.989-0.999, P = 0.031), respectively. These findings suggest that these biomarkers could effectively predict AL risk. Furthermore, the proposed predictive model has superior discriminative ability, as demonstrated by an area under the curve of 0.910, a sensitivity of 0.898, and a specificity of 0.826, reflecting its high level of accuracy.
The risk of AL in rectal cancer surgery patients can be effectively predicted by assessing the preoperative levels of serum nutritional biomarkers and inflammatory indicators, emphasizing their importance in the preoperative evaluation process.
Core Tip: This study introduces a novel approach to predict anastomotic leakage (AL) risk in colorectal cancer (CRC) patients. It explored the combined application of the Onodera prognostic nutritional index, neutrophil-to-lymphocyte ratio, and platelet-to-lymphocyte ratio. The results demonstrate that this combination significantly enhances the accuracy of AL prediction, providing a more reliable tool for personalized management of patients with CRC.
- Citation: Zhang ZY, Li KJ, Zeng XY, Wang K, Sulayman S, Chen Y, Zhao ZL. Early prediction of anastomotic leakage after rectal cancer surgery: Onodera prognostic nutritional index combined with inflammation-related biomarkers. World J Gastrointest Surg 2025; 17(4): 102862
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/102862.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.102862
Colorectal cancer (CRC) is a significant digestive system malignancy, with an estimated 1.9 million new cases reported worldwide in 2020 and approximately 935000 deaths linked to the disease. CRC is the third most frequently diagnosed cancer and the second primary contributor to cancer-related deaths, and these statistics emphasize the extensive health burden posed by CRC[1].
CRC treatment primarily involves surgical interventions, radiotherapy, chemotherapy, and targeted therapy. With advancements in surgical methods, such as improved resection and anastomosis techniques and the utilization of modern instruments, surgery continues to be the cornerstone of CRC management. While these advancements have enhanced the prospects for successful radical treatment, complications from surgery are still a concern. Among them, anastomotic leakage (AL) is a major postoperative complication that significantly contributes to abdominal infections, sepsis, extended hospitalization, high reoperation rates, and increased short- and long-term morbidity and mortality[2,3]. Consequently, it is crucial for medical professionals to identify the risk factors that contribute to AL following colorectal surgeries.
The precise cause of AL remains elusive; however, current research indicates that it likely arises from a convergence of multiple elements, including advanced age, obesity, smoking, alcohol intake, and surgeon experience. The duration of colorectal surgery and patient-related factors, such as nutritional state, inflammation, and immune function, also significantly contribute to the risk of AL[4]. Researchers have identified several biomarkers, including the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio, which are indicators of the AL risk[5]. Furthermore, new indices have been introduced in recent years, such as the systemic immune-inflammatory index (SII), the Onodera prognostic nutritional index (OPNI), the pan-immune inflammatory value, and the systemic inflammatory response index, all of which have demonstrated associations with survival rates in various malignancies[6-8].
Timely detection and prevention are vital for reducing the likelihood of potential AL. Early identification and assessment of the severity of this complication play a decisive role in lowering postoperative mortality and enhancing patient care quality. Considering the link between AL and factors such as nutrition, inflammation, and immunity, investigating markers that could be integrated into standard blood tests to predict AL in colorectal surgery patients with CRC might aid in predicting and hindering AL development, ultimately contributing to improved treatment outcomes.
The objective of this study was to create an efficient predictive model of AL, focusing on nutritional and inflammation-related biomarkers, with the intent of precisely evaluating the risk of AL in patients after rectal cancer surgery. This model offers a straightforward and accurate assessment tool for clinicians, ultimately improving patient management strategies and enhancing overall survival.
Employing the Cochran formula, we estimated the required sample size by considering a 10% event rate, a 5% margin of error, and a 95% confidence level. This computation led to a required sample size of 139 patients. Over the course of the study, from January 2016 to December 2023, a total of 434 patients with rectal cancer were recruited from the Cancer Hospital of Xinjiang Medical University, significantly surpassing the initially calculated sample size. The eligibility criteria for participation were as follows: (1) Had a confirmed diagnosis of rectal cancer; (2) Were 18 years of age or older; (3) Had undergone radical rectal cancer surgery; and (4) Had complete preoperative data and examination records available. Individuals were excluded from the study if they: (1) Had distant metastasis at the time of enrollment; (2) Had received radiotherapy or chemotherapy before surgical intervention; (3) Had significant preexisting complications; (4) Had undergone Miles or Hartman surgery without anastomosis; or (5) Were diagnosed with nonprimary rectal cancer. The participating patients provided written informed consent after having been fully apprised of the study details. This research was conducted ethically after receiving clearance from the Ethics Committee of Xinjiang Medical University Cancer Hospital (Approval No: K-2024170).
The patient data utilized in this study were obtained solely from our electronic information system, which provides comprehensive information on participants, including their age, gender, body mass index, medical history of diabetes mellitus and hypertension as comorbidities, smoking and alcohol intake, and preoperative factors such as intestinal obstruction and tumor specificity (T stage, N stage, TNM stage, tumor differentiation, neural infiltration, and choroidal invasion). We also recorded various hematological parameters, including white blood cell (WBC), hemoglobin (Hb), platelet (Plt), lymphocyte (LYM), neutrophil, and monocyte (MONO) counts. To facilitate deeper analysis, we derived additional markers: OPNI = albumin (ALB) + 5 × MONO count (109/L); NLR = neutrophil count (109/L)/LYM count
Patients were classified into two categories based on the development of AL within two weeks postsurgery: The AL group, which included 49 patients, and the non-AL group, which consisted of 385 patients. AL was diagnosed according to the following criteria: (1) The presence of unexplained abdominal pain, bloating, or an uncontrolled postoperative fever, either with or without signs of peritoneal irritation; (2) Alterations in drainage patterns, characterized by a substantial rise in pelvic drainage volume, often containing gas, pus, or feces; (3) Digital rectal exam revealing a palpable anastomosis leak; (4) Imaging studies that revealed contrast medium extravasation, bowel discontinuity, or the presence of air pockets adjacent to the anastomosis; and (5) Significant laboratory results showing elevated WBC count, an increased neutrophil count, and a higher than normal neutrophil percentage[9].
Continuous variable data are presented as median values with their respective quartiles (Q1 to Q3). Statistical analysis was conducted using IBM SPSS software, version 26.0, and group differences were assessed via nonparametric tests, specifically the Wilcoxon rank-sum test. For categorical variables, data are shown as frequencies (n) and corresponding percentages (%). To analyze categorical data across different groups, we employed Fisher's exact test when the expected frequency was less than 5; otherwise, the χ2 test was used. To examine the risk factors associated with postoperative AL, we applied both univariate and multivariate logistic regression models. The diagnostic efficacy of serum nutritional indices in predicting postoperative AL was assessed through receiver operating characteristic (ROC) curve analysis. In the R platform, a predictive model was developed with the "rms" package to construct nomograms[10,11]. The performance of various serum nutritional and inflammatory markers, as well as the predictive models, was evaluated by contrasting their sensitivity, specificity, and area under the curve (AUC) values. The predictive model's calibration was assessed and depicted graphically through a calibration analysis utilizing the "rms" package to ensure visual representation of the model's performance. A statistically significant difference was considered to be a P value less than 0.05.
In a study involving 434 patients who had undergone rectal cancer surgery, 49 participants (accounting for 11.28% of the group) experienced postoperative AL. A comparison of the clinical and pathological features between patients with AL (n = 49) and those without AL (n = 385) is shown in Table 1. The baseline characteristics of the entire patient group (n = 434) were assessed across diverse variables. The prevalence of AL was significantly greater among males than females (P = 0.056), with a male-to-female ratio tending toward significance in the AL group. Notably, a history of diabetes mellitus was significantly associated with the development of AL (P = 0.046), suggesting that individuals with diabetes are more likely to develop AL. This finding implies that those with diabetes mellitus preoperatively might necessitate a more conservative surgical approach. There were no statistically significant differences in smoking habits, alcohol use, or history of hypertension between the two groups. Importantly, diabetes, along with other factors, shaped the demographic and clinical profiles of the study participants. They offer valuable insights into the variables that might influence the study's outcome measures, even though smoking, alcohol consumption, and hypertension did not show any notable disparities between the groups. Nevertheless, diabetes has emerged as a key parameter with implications for the results under examination.
| Characteristic | Anastomotic leakage group (n = 49) | No anastomotic leakage group (n = 385) | Statistic | P value |
| Age | 0.96 | 0.32821 | ||
| < 60 | 173 (44.9) | 18 (37.5) | ||
| ≥ 60 | 212 (55.1) | 30 (62.5) | ||
| Gender | 3.66 | 0.05621 | ||
| Male | 220 (57.1) | 35 (71.4) | ||
| Female | 165 (42.9) | 14 (28.6) | ||
| BMI | 0.26232 | |||
| < 18.5 | 11 (2.9) | 4 (8.2) | ||
| 18.5-24 | 169 (43.9) | 22 (44.9) | ||
| 24-28 | 159 (41.3) | 19 (38.8) | ||
| ≥ 28 | 46 (11.9) | 4 (8.2) | ||
| Smoking | 0.87 | 0.35221 | ||
| No | 268 (69.8) | 31 (63.3) | ||
| Yes | 116 (30.2) | 18 (36.7) | ||
| Drinking | 1.61 | 0.20421 | ||
| No | 308 (80.2) | 43 (87.8) | ||
| Yes | 76 (19.8) | 6 (12.2) | ||
| History of hypertension | 2.03 | 0.15521 | ||
| No | 295 (76.6) | 33 (67.3) | ||
| Yes | 90 (23.4) | 16 (32.7) | ||
| Cardiac insufficiency | 0.13332 | |||
| No | 371 (96.4) | 45 (91.8) | ||
| Yes | 14 (3.6) | 4 (8.2) | ||
| History of diabetes | 3.97 | 0.04621 | ||
| No | 330 (85.7) | 47 (95.9) | ||
| Yes | 55 (14.3) | 2 (4.1) | ||
| History of abdominal surgery | 0.48 | 0.48721 | ||
| No | 289 (75.1) | 39 (79.6) | ||
| Yes | 96 (24.9) | 10 (20.4) | ||
| ASA | 0.46232 | |||
| 1-2 | 369 (95.8) | 46 (93.9) | ||
| 3-4 | 16 (4.2) | 3 (6.1) | ||
| Bowel obstruction | 0.31332 | |||
| No | 366 (95.1) | 45 (91.8) | ||
| Yes | 19 (4.9) | 4 (8.2) | ||
| Black stool | 0.04 | 0.83921 | ||
| No | 163 (42.3) | 20 (40.8) | ||
| Yes | 222 (57.7) | 29 (59.2) | ||
| Differentiated degree | > 0.99932 | |||
| Poorly | 33 (8.6) | 4 (8.2) | ||
| Moderately | 317 (82.3) | 41 (83.7) | ||
| Well | 35 (9.1) | 4 (8.2) | ||
| Nerve invasion | 1.01 | 0.31421 | ||
| Negative | 320 (83.3) | 38 (77.6) | ||
| Positive | 64 (16.7) | 11 (22.4) | ||
| Intravascular tumor emboli | 0.00 | 0.95521 | ||
| Negative | 313 (81.3) | 40 (81.6) | ||
| Positive | 72 (18.7) | 9 (18.4) | ||
| T stage | 0.46432 | |||
| T1 | 20 (5.2) | 2 (4.2) | ||
| T2 | 59 (15.3) | 4 (8.3) | ||
| T3 | 281 (73.0) | 37 (77.1) | ||
| T4 | 25 (6.5) | 5 (10.4) | ||
| N stage | 0.58532 | |||
| N0 | 224 (58.2) | 31 (63.3) | ||
| N1 | 101 (26.2) | 9 (18.4) | ||
| N2 | 56 (14.5) | 9 (18.4) | ||
| N3 | 4 (1.0) | 0 (0.0) | ||
| Tumor stage | 0.19 | 0.65921 | ||
| 1-2 | 223 (57.9) | 30 (61.2) | ||
| 3-4 | 162 (42.1) | 19 (38.8) |
Table 2 outlines the baseline characteristics of the 434 participants, highlighting the disparities in laboratory indices between the two groups. There were no significant differences in WBC, Plt, or neutrophil counts, or serum carcinoembryonic antigen or carbohydrate antigen 199 levels between the groups with and without AL. However, substantial variations were noted in the Hb concentration, MONO, LYM count, NLR and PLR. All these parameters exhibited statistically notable disparities, with P values less than 0.001, indicating significant differences between the AL group and the non-AL group. These findings imply that nutritional status and inflammation-related biomarkers, as represented by the OPNI, are distinct between the groups, indicating that AL status is likely to be correlated with various hematological and serological markers. These findings suggest a potential influence of systemic nutritional status and the immune response on the occurrence of AL.
| Indicators | Anastomotic leakage group (n = 49)1 | No anastomotic leakage group (n = 385)1 | Statistic | P value |
| WBC | 6.00 (5.18, 7.29) | 6.39 (4.63, 7.87) | 9333.00 | 0.9052 |
| Hb | 135 (120, 148) | 120 (103, 138) | 12738.00 | < 0.0012 |
| Plt | 244 (199, 292) | 225 (195, 302) | 9945.00 | 0.5362 |
| LYM | 1.83 (1.49, 2.23) | 1.34 (1.13, 1.64) | 14667.50 | < 0.0012 |
| NE | 3.59 (2.88, 4.35) | 4.05 (2.84, 5.76) | 7957.50 | 0.0752 |
| MONO | 0.44 (0.34, 0.55) | 0.51 (0.39, 0.64) | 7374.00 | 0.0132 |
| OPNI | 50.5 (47.4, 53.2) | 42.5 (39.3, 45.6) | 16750.50 | < 0.0012 |
| NLR | 1.94 (1.46, 2.59) | 3.20 (2.03, 4.22) | 4799.00 | < 0.0012 |
| PLR | 131 (101, 173) | 179 (123, 251) | 5934.50 | < 0.0012 |
| CEA (%) | 2.123 | 0.1453 | ||
| Normal | 238 (61.8) | 25 (51.0) | ||
| High | 147 (38.2) | 24 (49.0) | ||
| CA199 (%) | 0.002 | 0.9653 | ||
| Normal | 284 (73.8) | 36 (73.5) | ||
| High | 101 (26.2) | 13 (26.5) |
Rectal cancer patients' risk factors for AL were assessed via univariate and multivariate logistic regression analyses. The preliminary univariate analysis, summarized in Table 3, identified seven noteworthy factors with ORs: Hb at 0.980 (95%CI: 0.969-0.991, P < 0.001), LYM count at 0.105 (95%CI: 0.048-0.231, P < 0.001), MONO at 6.567 (95%CI: 1.659-25.995, P = 0.007), neutrophil count with an OR of 1.312 (95%CI: 1.129-1.523, P < 0.001), NLR at 0.702 (95%CI: 0.642-0.768, P < 0.001), PLR at 1.838 (95%CI: 1.47-2.298, P < 0.001), and OPNI with an OR of 1.008 (95%CI: 1.004-1.012, P < 0.001). However, subsequent multivariate logistic regression revealed that the OPNI, NLR, and PLR were the sole independent predictors of AL risk, with adjusted ORs of 0.705 (95%CI: 0.641-0.775, P = 0.012), 1.628 (95%CI: 1.221-2.172, P = 0.024), and 0.994 (95%CI: 0.989-0.999, P = 0.031), respectively. Notably, the statistical relevance of Hb, LYM, MONO, and neutrophils disappeared in the multivariate assessment, suggesting possible moderation by other variables. Diabetes history was not significantly linked to AL risk in univariate assessments and was not significantly associated with AL risk in multivariate evaluations.
| Variables | Univariate | Multivariate | ||||||||
| β | SE | Z | P value | OR (95%CI) | β | SE | Z | P value | OR (95%CI) | |
| Hb | -0.021 | 0.006 | -3.634 | < 0.001 | 0.980 (0.969-0.991) | 0.001 | 0.008 | 0.127 | 0.899 | 1.001 (0.986-1.016) |
| LYM | -2.251 | 0.400 | -5.625 | < 0.001 | 0.105 (0.048-0.231) | 0.455 | 0.743 | 0.613 | 0.540 | 0.634 (0.148-2.719) |
| MONO | 1.882 | 0.702 | 2.681 | 0.007 | 6.567 (1.659-25.995) | 1.109 | 1.119 | 0.991 | 0.322 | 3.031 (0.338-27.160) |
| NE | 0.271 | 0.076 | 3.556 | < 0.001 | 1.312 (1.129-1.523) | 0.150 | 0.212 | 0.710 | 0.478 | 0.860 (0.568-1.303) |
| OPNI | -0.354 | 0.045 | -7.781 | < 0.001 | 0.702 (0.642-0.768) | -0.350 | 0.048 | -7.227 | < 0.001 | 0.705 (0.641-0.775) |
| NLR | 0.609 | 0.114 | 5.337 | < 0.001 | 1.838 (1.470-2.298) | 0.488 | 0.147 | 3.317 | < 0.001 | 1.628 (1.221-2.172) |
| PLR | 0.008 | 0.002 | 4.348 | < 0.001 | 1.008 (1.004-1.012) | -0.006 | 0.003 | -2.026 | 0.043 | 0.994 (0.989-0.999) |
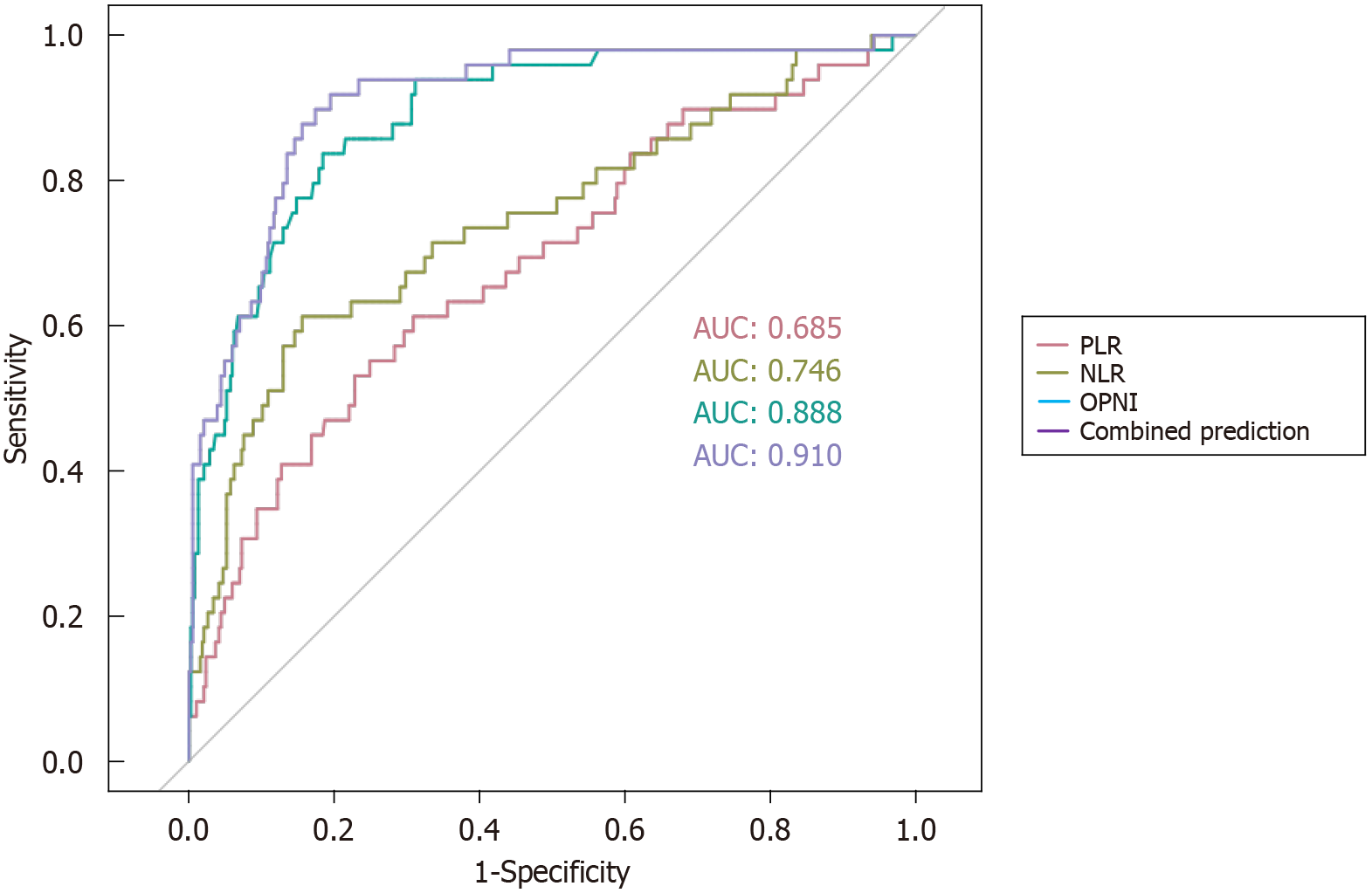
This research focused on assessing the predictive accuracy of three preoperative serum biomarkers-the OPNI, NLR, and PLR-in determining the probability of AL after laparoscopic surgical procedures. ROC curve analysis revealed that a preoperative OPNI level less than 46.60 had an AUC of 0.888 (95%CI: 0.845-0.950), corresponding to a sensitivity of 83.7% and a specificity of 81.6%. An NLR threshold of 2.98 exhibited an AUC of 0.746 (95%CI: 0.660-0.817), indicating 61.2% sensitivity and 84.4% specificity. The optimal cutoff for the PLR was 158.15, which was associated with an AUC of 0.686 (95%CI: 0.601-0.763), along with a sensitivity and specificity of 61.2% and 69.1%, respectively. Notably, when all three biomarkers (OPNI, NLR, and PLR) were combined for AL prediction, the composite AUC increased to 0.910 (95%CI: 0.900-0.929), suggesting a sensitivity of 89.8% and a specificity of 82.6%. These results suggest that assessing all three biomarkers concurrently is more advantageous than evaluating them independently, as visualized in Figure 1.
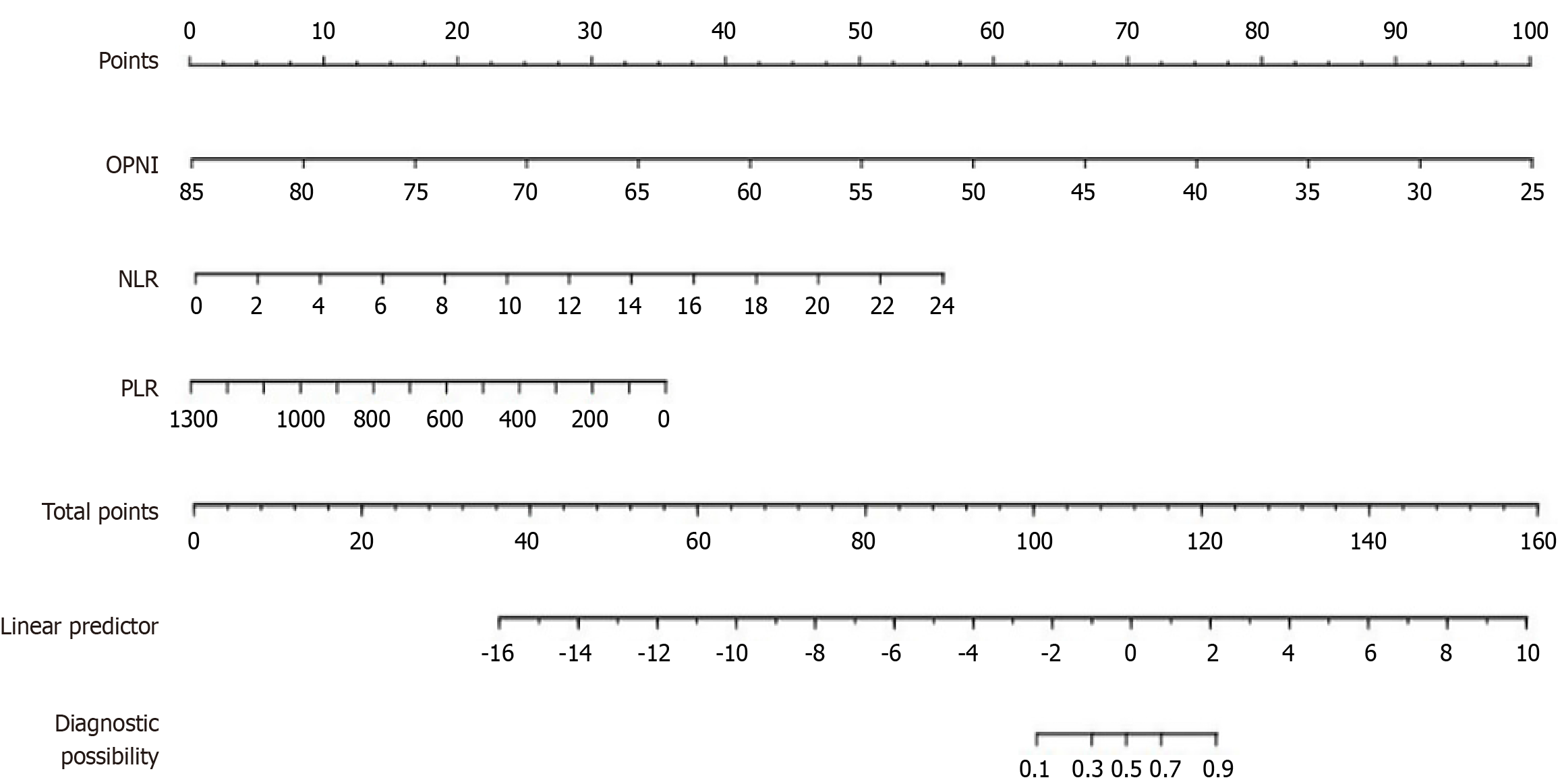
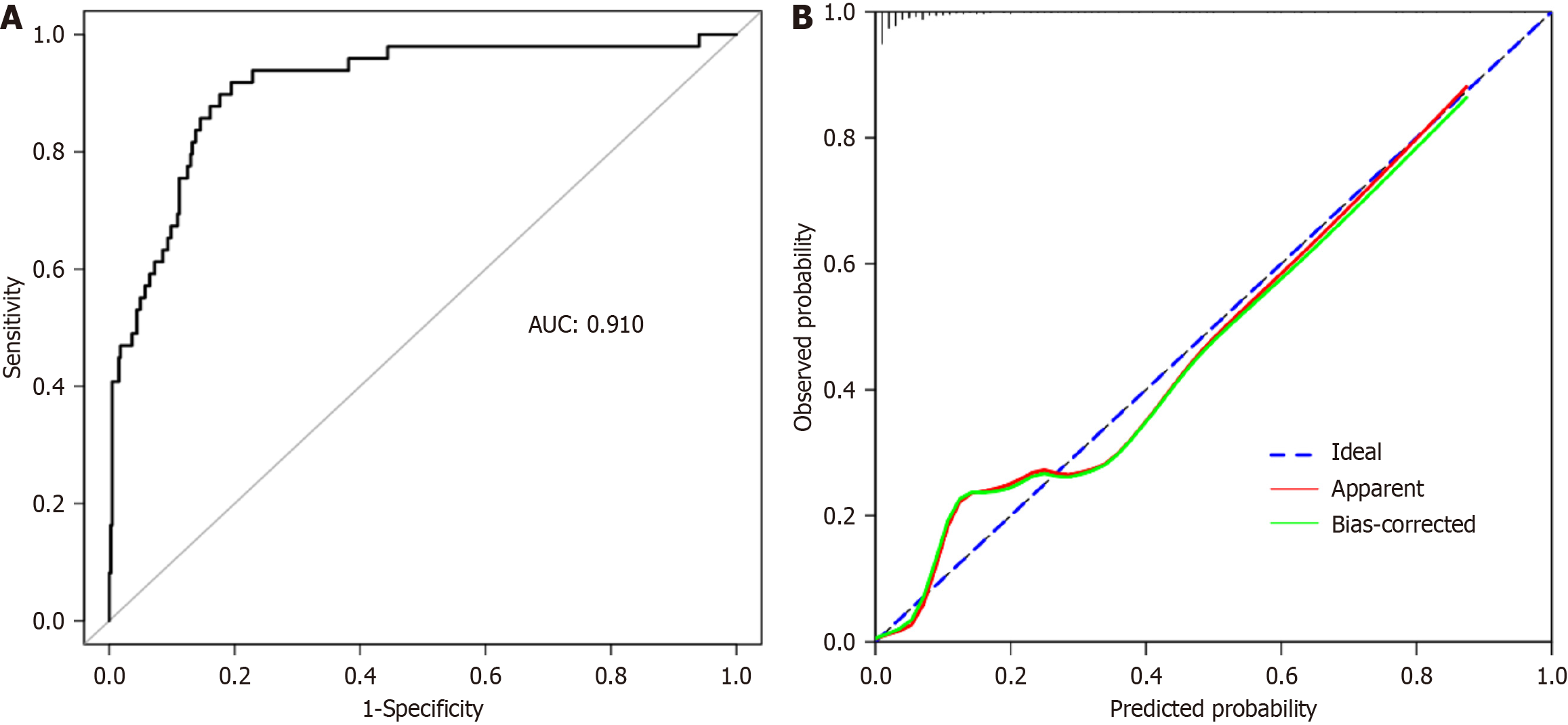
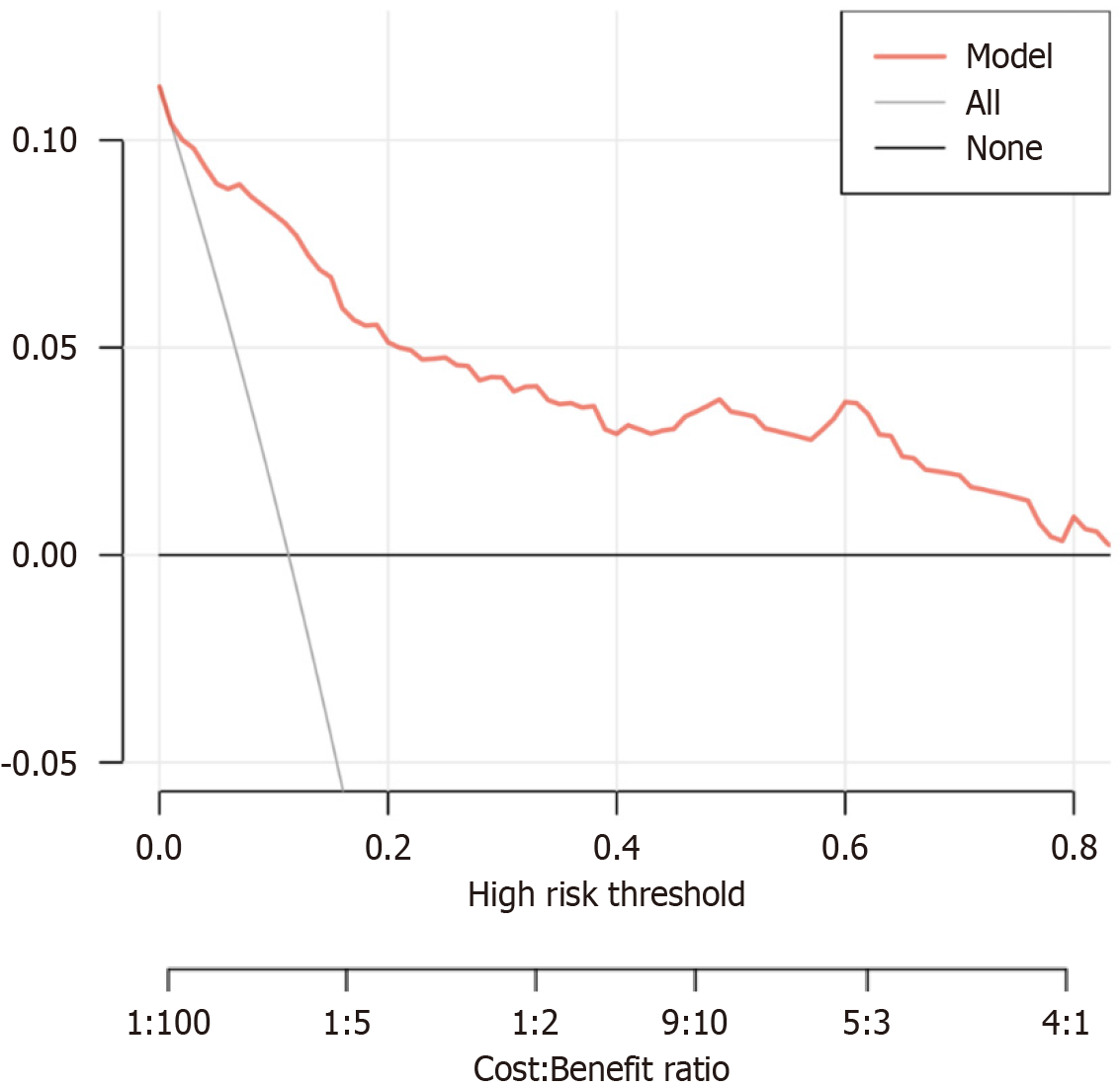
Using multivariate logistic regression analysis, we developed a predictive model for the risk of AL in patients after rectal cancer surgery. This model was based on three independent risk factors: The OPNI, NLR, and PLR. As depicted in Figure 2, this model exhibits robust predictive accuracy, as demonstrated by ROC curve analysis. The model boasts an impressive AUC of 0.910 (95%CI: 0.864-0.956), as depicted in Figure 3A, indicating superior discriminative ability. The predictive model's calibration was evaluated through a calibration plot (Figure 3B), which displayed an almost perfect alignment with the 45-degree reference line, reflected by a fit index of 0.820. This alignment underscores the high level of consistency between the predicted and observed AL rates. To ascertain the clinical relevance of the model, we employed decision curve analysis (DCA), as illustrated in Figure 4. DCA is an intuitive and effective tool that helps clinicians judge when to use model-generated risk by comparing the net benefit of the model at different decision threshold scores.
Rectal cancer ranks among the most prevalent cancers globally, with surgery serving as the primary therapeutic approach for this disease. The goal of this procedure is the complete excision of the tumor, along with the associated lymphatic bed and any adjacent compromised organs. Over time, surgical techniques have significantly advanced; however, the risk of AL persists. AL is a particularly severe postoperative complication that significantly affects patients, their families, and society at large. It can lead to early reoperations to address complications such as infections and leaks, resulting in increased hospitalization periods, diminished quality of life for patients, and considerable economic strain on both individuals and health care systems. Despite the maturity of surgical methods, the challenge of preventing AL remains.
AL is another factor that increases the rate of recurrence and mortality, and it hinders or delays the administration of adjuvant chemotherapy. A meta-analysis reviewed 14 studies evaluating the impact of AL on a total of 11353 oncology patients and revealed that AL leads to worse overall survival and reduced cancer-specific survival rates[12]. AL usually develops within 5-7 days after surgery. Timely diagnosis and intervention for AL pose significant challenges because patients often exhibit mild symptoms when they first present. Consequently, detecting AL at an early stage is highly important for improving the prognosis of rectal cancer patients, thereby decreasing hospitalization duration and minimizing health care expenses.
Rectal cancer patients with malnutrition are at increased risk of experiencing postoperative AL due to delayed wound healing, weakened immunity, and increased susceptibility to infection. Nutrition significantly impacts the speed of recovery after surgery, the long-term prognosis, and the probability of postoperative complications. Among the markers of malnutrition, low ALB levels are particularly common, with depleted levels often indicating malnutrition. A robust body of research clearly demonstrated a correlation between preoperative ALB levels and the incidence of postoperative complications, particularly AL[13,14]. The immune system's health also plays a pivotal role in shaping cancer patients' clinical outcomes[15]. To encompass both nutritional status and immune function, the OPNI is used, which is calculated on the basis of serum ALB levels and LYM counts[16]. The OPNI has been shown to be effective in predicting post
Hypoalbuminemia is a common manifestation of malnutrition, and ALB plays a key role in maintaining immune function and the anti-inflammatory response. Low ALB levels may lead to decreased immunity and an excessive inflammatory response in the postoperative period, thereby increasing the risk of AL. Therefore, preoperative assessment of a patient's nutritional status and immune function via the OPNI can help identify high-risk patients and provide a basis for postoperative rehabilitation and the prevention of complications.
The NLR, a concept first introduced by Zahorec[20], has gained recognition as a crucial indicator of systemic inflammation in the field of critical care medicine. The NLR is acknowledged as a crucial parameter for predicting disease outcomes, as it surpasses the individual predictive strength of WBC, neutrophil, or LYM counts in evaluating inflammatory conditions[21,22]. Comprehensive studies have established that an increased NLR is consistently associated with increased frequencies of postoperative complications and reduced survival odds in patients with CRC[5,23-25]. Notably, it is associated with an increased likelihood of AL following rectal cancer surgery. Importantly, Miyakita et al[26] reported that rectal cancer patients with a preoperative NLR of 2.21 or more had strikingly greater odds (4.51 times) of experiencing postoperative AL (P = 0.0329).
While the OPNI, NLR, and PLR have demonstrated promising predictive value for AL, it is essential to compare these markers with commonly used predictors such as C-reactive protein (CRP) and ALB. CRP has been well established as a reliable predictor of AL in patients who have undergone colorectal surgery. A systematic review and meta-analysis revealed that the CRP level had an AUC ranging from 0.706 to 0.910 in the prediction of AL, with improved predictive accuracy when measured perioperatively[27]. Similarly, Messias et al[28] highlighted the utility of the CRP level in excluding AL after colorectal surgery, with an AUC within this range. Additionally, Choi et al[29] reported that CRP levels could predict AL in patients with enhanced recovery after surgery. On the other hand, the level of ALB, an important marker of nutritional status, has also been linked to postoperative complications. In their study, Shimura and colleagues reported that serum ALB levels measured during the perioperative period could effectively predict the occurrence of AL in patients who underwent curative surgery for CRC. Their findings revealed an AUC ranging from 0.675 to 0.832[30]. Hu et al[31] further confirmed that relative decreases in the serum ALB concentration are predictive of AL in female patients after sphincter-preserving rectal surgery. In our study, the combined use of the OPNI, NLR, and PLR resulted in a superior AUC of 0.910, which outperformed CRP and ALB as predictive tools for AL. Our model achieved a high sensitivity (89.8%) and specificity (82.6%), emphasizing the comprehensive advantage of integrating nutritional and inflammatory markers for more accurate preoperative risk stratification in rectal cancer patients.
Paliogiannis et al[5] demonstrated a positive connection between an increased NLR and the incidence of postoperative AL in patients who underwent elective colorectal surgery. Our study bolsters these findings, as we also noted significantly higher NLRs among patients who experienced AL after undergoing radical rectal cancer surgery. Further analysis revealed that an increased NLR was an independent risk factor for AL in our multifactorial assessments. These results suggest that the NLR could serve as a significant predictor of impending AL in the context of rectal cancer surgery, possibly enabling clinicians to introduce prophylactic strategies in advance. Neutrophils play a dominant role in the inflammatory response, whereas LYM are involved in regulating immune homeostasis. Preoperatively, an elevated NLR usually reflects a more severe chronic inflammatory state in a patient, and an excessive inflammatory response may lead to impaired local blood flow and immunosuppression, which in turn affects the healing ability of the anastomosis. Therefore, an elevated preoperative NLR may suggest that patients are more likely to develop AL postoperatively.
Notably, the PLR, another inflammatory biomarker, is calculated as the division of the Plt count by the LYM count. Plts contribute to inflammation by secreting cytokines and chemokines that aggravate the inflammatory environment of tumors[32]. The PLR has garnered recognition as a crucial marker of inflammation. Numerous studies have linked an elevated PLR to a poor prognosis in different types of cancers, including nasopharyngeal carcinoma, prostate cancer, lung cancer, and CRC[33]. Our research revealed a statistically robust association through multifactorial analysis, demon
Recent research has underscored the importance of preoperative nutritional and immune status in determining the outcomes of surgical procedures for malignant tumors[34,35]. In addition, recent studies have shown that the inflammatory response influences tumor growth by creating a favorable environment for tumor progression[36]. In this context, various serum-based parameters, such as the OPNI, NLR, MLR, PLR, and SII, have been explored as potential prognostic indicators for estimating the likelihood of perioperative AL in rectal cancer patients. After thorough data analysis, we opted for the OPNI, NLR, and PLR as key indicators due to their comprehensive reflection of the patient's nutritional state, immune status, and inflammatory response, which are crucial factors in assessing the patient's overall condition. Furthermore, the NLR, PLR, and OPNI are valued for their simplicity, speed, affordability, and minimal invasiveness, requiring only a peripheral blood sample for calculation. Numerous studies have investigated the significance of these indices in relation to postoperative complications and survival prognosis, although their value for the prediction of postoperative AL in rectal cancer patients remains underexplored.
In this study, a robust prediction model has been developed and meticulously evaluated for predicting the risk of AL after radical surgery in patients with rectal cancer. This model centers on the influential role of preoperative serum nutritional and inflammatory biomarkers in determining the likelihood of AL. Exhibiting exceptional accuracy, this model boasts an AUC of 0.910 on the ROC curve, indicating a remarkable level of sensitivity and specificity in predicting AL risk. We opted for ROC analysis, a clinically relevant and efficacious method, to evaluate our model's discriminatory potential, particularly given its proficiency in determining the performance of our predictive tool.
Although this study has several strengths, it also has several limitations, one of which is the relatively small sample size, which raises concerns about the generalizability of the findings to larger populations and potentially compromises the robustness of the statistical inferences. While the initial sample size calculation suggested a sample of 154 patients, we included 434 patients, which provides a more robust foundation for our conclusions. However, the limitation still stands as it might affect external validity when applied to broader demographics and could raise concerns about the reliability of statistical significance in different populations. As this study relied mainly on preoperative data, it failed to fully consider the impact of relevant intraoperative factors (e.g., surgical approach, duration of surgery, etc.) on the risk of AL. Therefore, the role of these intraoperative factors has not been evaluated in this study. Future studies may include intraoperative factors in the analysis to further optimize and improve the prediction model of AL and enhance its clinical application value and accuracy. In addition, as a retrospective analysis, selection bias and information bias may exist, particularly because the data were collected from a single institution. As this study had a single-center design, the findings may not be fully applicable to other settings with different patient populations or health care environments. This could restrict the external applicability of the results. Furthermore, since the study design was retrospective in nature, we acknowledge the inherent limitations in controlling for all potential confounding factors, which could have affected the outcomes. To increase the precision and validity of the findings, future research should focus on larger-scale, multicenter, and prospective study designs, which will allow broader patient representation and more reliable conclusions. The incorporation of external validation through independent datasets and prospective cohort studies will be crucial for verifying the generalizability of our results across different institutions and geographic regions. Undoubtedly, implementing these approaches would strengthen the reliability and robustness of the findings, enabling a more extensive assessment of the suitability of the predictive model for clinical application.
We conducted a retrospective study analyzing clinical data from 434 rectal cancer patients who had undergone surgery, with a specific emphasis on preoperative serum nutritional and inflammation biomarkers, such as the OPNI, NLR, and PLR. By constructing a predictive model based on these biomarkers, we identified their potential in predicting the risk of AL. The developed model demonstrated an impressive AUC of 0.910. This study highlights the pivotal role of nutritional and inflammatory status in determining the risk of adverse surgical outcomes, contributing to a more comprehensive understanding of the factors that influence these outcomes. This finding stresses the necessity of incorporating nutritional and inflammatory evaluations into preoperative planning to optimize patient care. Such comprehensive evaluations might minimize postoperative complications, facilitate faster patient recovery, and potentially improve the long-term prognosis of rectal cancer patients.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69663] [Article Influence: 13932.6] [Reference Citation Analysis (45)] |
| 2. | Hoek VT, Buettner S, Sparreboom CL, Detering R, Menon AG, Kleinrensink GJ, Wouters MWJM, Lange JF, Wiggers JK; Dutch ColoRectal Audit group. A preoperative prediction model for anastomotic leakage after rectal cancer resection based on 13.175 patients. Eur J Surg Oncol. 2022;48:2495-2501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 3. | Chiarello MM, Fransvea P, Cariati M, Adams NJ, Bianchi V, Brisinda G. Anastomotic leakage in colorectal cancer surgery. Surg Oncol. 2022;40:101708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 132] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 4. | Vasiliu EC, Zarnescu NO, Costea R, Neagu S. Review of Risk Factors for Anastomotic Leakage in Colorectal Surgery. Chirurgia (Bucur). 2015;110:319-326. [PubMed] |
| 5. | Paliogiannis P, Deidda S, Maslyankov S, Paycheva T, Farag A, Mashhour A, Misiakos E, Papakonstantinou D, Mik M, Losinska J, Scognamillo F, Sanna F, Feo CF, Cherchi G, Xidas A, Zinellu A, Restivo A, Zorcolo L. Blood cell count indexes as predictors of anastomotic leakage in elective colorectal surgery: a multicenter study on 1432 patients. World J Surg Oncol. 2020;18:89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 6. | Lin F, Zhang LP, Xie SY, Huang HY, Chen XY, Jiang TC, Guo L, Lin HX. Pan-Immune-Inflammation Value: A New Prognostic Index in Operative Breast Cancer. Front Oncol. 2022;12:830138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 75] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 7. | Feng Y, Zhang N, Wang S, Zou W, He Y, Ma JA, Liu P, Liu X, Hu C, Hou T. Systemic Inflammation Response Index Is a Predictor of Poor Survival in Locally Advanced Nasopharyngeal Carcinoma: A Propensity Score Matching Study. Front Oncol. 2020;10:575417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 8. | Yilmaz H, Yersal Ö. Prognostic significance of novel inflammatory markers in extensive-stage small-cell lung cancer. J Cancer Res Ther. 2022;18:691-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 9. | Rahbari NN, Weitz J, Hohenberger W, Heald RJ, Moran B, Ulrich A, Holm T, Wong WD, Tiret E, Moriya Y, Laurberg S, den Dulk M, van de Velde C, Büchler MW. Definition and grading of anastomotic leakage following anterior resection of the rectum: a proposal by the International Study Group of Rectal Cancer. Surgery. 2010;147:339-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 732] [Cited by in RCA: 1119] [Article Influence: 69.9] [Reference Citation Analysis (5)] |
| 10. | Degiuli M, Elmore U, De Luca R, De Nardi P, Tomatis M, Biondi A, Persiani R, Solaini L, Rizzo G, Soriero D, Cianflocca D, Milone M, Turri G, Rega D, Delrio P, Pedrazzani C, De Palma GD, Borghi F, Scabini S, Coco C, Cavaliere D, Simone M, Rosati R, Reddavid R; collaborators from the Italian Society of Surgical Oncology Colorectal Cancer Network Collaborative Group. Risk factors for anastomotic leakage after anterior resection for rectal cancer (RALAR study): A nationwide retrospective study of the Italian Society of Surgical Oncology Colorectal Cancer Network Collaborative Group. Colorectal Dis. 2022;24:264-276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 104] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 11. | Qin Q, Huang B, Wu A, Gao J, Liu X, Cao W, Ma T, Kuang Y, Guo J, Wu Q, Shao B, Guan Q, Yao H, Zhang X, Wang H; Chinese Radiation Intestinal Injury Research Group. Development and Validation of a Post-Radiotherapy Prediction Model for Bowel Dysfunction After Rectal Cancer Resection. Gastroenterology. 2023;165:1430-1442.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 12. | Wang S, Liu J, Wang S, Zhao H, Ge S, Wang W. Adverse Effects of Anastomotic Leakage on Local Recurrence and Survival After Curative Anterior Resection for Rectal Cancer: A Systematic Review and Meta-analysis. World J Surg. 2017;41:277-284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 131] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 13. | Telem DA, Chin EH, Nguyen SQ, Divino CM. Risk factors for anastomotic leak following colorectal surgery: a case-control study. Arch Surg. 2010;145:371-6; discussion 376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 171] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 14. | Tsalikidis C, Mitsala A, Mentonis VI, Romanidis K, Pappas-Gogos G, Tsaroucha AK, Pitiakoudis M. Predictive Factors for Anastomotic Leakage Following Colorectal Cancer Surgery: Where Are We and Where Are We Going? Curr Oncol. 2023;30:3111-3137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 73] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 15. | Bruni D, Angell HK, Galon J. The immune contexture and Immunoscore in cancer prognosis and therapeutic efficacy. Nat Rev Cancer. 2020;20:662-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1378] [Cited by in RCA: 1191] [Article Influence: 198.5] [Reference Citation Analysis (7)] |
| 16. | Onodera T, Goseki N, Kosaki G. [Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients]. Nihon Geka Gakkai Zasshi. 1984;85:1001-1005. [PubMed] |
| 17. | Kang J, Yang G, Wang D, Lin Y, Wang Q, Luo H. The Clinical Application Value of the Prognostic Nutritional Index for the Overall Survival Prognosis of Patients with Esophageal Cancer: A Robust Real-World Observational Study in China. Comput Math Methods Med. 2022;2022:3889588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 18. | Xie H, Wei L, Yuan G, Liu M, Tang S, Gan J. Prognostic Value of Prognostic Nutritional Index in Patients With Colorectal Cancer Undergoing Surgical Treatment. Front Nutr. 2022;9:794489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 19. | Tamai M, Kiuchi J, Kuriu Y, Arita T, Shimizu H, Ohashi T, Konishi H, Yamamoto Y, Morimura R, Shiozaki A, Ikoma H, Kubota T, Fujiwara H, Okamoto K, Otsuji E. Clinical impact of postoperative prognostic nutritional index in colorectal cancer patients undergoing adjuvant chemotherapy. Am J Cancer Res. 2021;11:4947-4955. [PubMed] |
| 20. | Zahorec R. Ratio of neutrophil to lymphocyte counts--rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy. 2001;102:5-14. [PubMed] |
| 21. | Wang CY, Li XL, Ma XL, Yang XF, Liu YY, Yu YJ. Preoperative neutrophil-to-lymphocyte ratio predicts symptomatic anastomotic leakage in elderly colon cancer patients: Multicenter propensity score-matched analysis. World J Gastrointest Surg. 2024;16:438-450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 22. | Kumarasamy C, Sabarimurugan S, Madurantakam RM, Lakhotiya K, Samiappan S, Baxi S, Nachimuthu R, Gothandam KM, Jayaraj R. Prognostic significance of blood inflammatory biomarkers NLR, PLR, and LMR in cancer-A protocol for systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e14834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 120] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 23. | Tan F, Xu K, Qi X, Gao P, Liu M, Yao Z, Zhang N, Yang H, Zhang C, Xing J, Cui M, Su X. Neutrophil-to-Lymphocyte Ratio as an Early Predictor of Symptomatic Anastomotic Leakage in Patients after Rectal Cancer Surgery: A Propensity Score-Matched Analysis. J Pers Med. 2022;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Mazaki J, Katsumata K, Kasahara K, Tago T, Wada T, Kuwabara H, Enomoto M, Ishizaki T, Nagakawa Y, Tsuchida A. Neutrophil-to-lymphocyte ratio is a prognostic factor for colon cancer: a propensity score analysis. BMC Cancer. 2020;20:922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (4)] |
| 25. | Zhao X, Zhou Y, Liu B, Shen Y, Qian J, Zhang X, Zhao H. Preoperative Neutrophil-Lymphocyte Ratio (NLR)-Binding Fibrinogen-Albumin Ratio (FAR) Is Superior to Platelet-Lymphocyte Ratio (PLR)-Binding Fibrinogen-Albumin Ratio (FAR) and Lymphocyte-Monocyte (LMR)-Binding Fibrinogen-Albumin Ratio (FAR) as Predictors of Survival in Surgical Patients with Colorectal Adenocarcinoma. Med Sci Monit. 2023;29:e939442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 26. | Miyakita H, Sadahiro S, Saito G, Okada K, Tanaka A, Suzuki T. Risk scores as useful predictors of perioperative complications in patients with rectal cancer who received radical surgery. Int J Clin Oncol. 2017;22:324-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 27. | Yeung DE, Peterknecht E, Hajibandeh S, Hajibandeh S, Torrance AW. C-reactive protein can predict anastomotic leak in colorectal surgery: a systematic review and meta-analysis. Int J Colorectal Dis. 2021;36:1147-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 28. | Messias BA, Botelho RV, Saad SS, Mocchetti ER, Turke KC, Waisberg J. Serum C-reactive protein is a useful marker to exclude anastomotic leakage after colorectal surgery. Sci Rep. 2020;10:1687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 29. | Choi JDW, Kwik C, Shanmugalingam A, Allan L, Khoury TE, Pathmanathan N, Toh JWT. C-Reactive Protein as a Predictive Marker for Anastomotic Leak Following Restorative Colorectal Surgery in an Enhanced Recovery After Surgery Program. J Gastrointest Surg. 2023;27:2604-2607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 30. | Shimura T, Toiyama Y, Hiro J, Imaoka H, Fujikawa H, Kobayashi M, Ohi M, Inoue Y, Mohri Y, Kusunoki M. Monitoring perioperative serum albumin can identify anastomotic leakage in colorectal cancer patients with curative intent. Asian J Surg. 2018;41:30-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 31. | Hu K, Tan K, Shang Q, Li C, Zhang Z, Huang B, Zhao S, Li F, Zhang A, Li C, Liu B, Tong W. Relative decline in serum albumin help to predict anastomotic leakage for female patients following sphincter-preserving rectal surgery. BMC Surg. 2023;23:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 32. | Shi C, Pamer EG. Monocyte recruitment during infection and inflammation. Nat Rev Immunol. 2011;11:762-774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2564] [Cited by in RCA: 2367] [Article Influence: 157.8] [Reference Citation Analysis (0)] |
| 33. | Templeton AJ, McNamara MG, Šeruga B, Vera-Badillo FE, Aneja P, Ocaña A, Leibowitz-Amit R, Sonpavde G, Knox JJ, Tran B, Tannock IF, Amir E. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst. 2014;106:dju124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2534] [Cited by in RCA: 2409] [Article Influence: 200.8] [Reference Citation Analysis (3)] |
| 34. | Schwegler I, von Holzen A, Gutzwiller JP, Schlumpf R, Mühlebach S, Stanga Z. Nutritional risk is a clinical predictor of postoperative mortality and morbidity in surgery for colorectal cancer. Br J Surg. 2010;97:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 279] [Article Influence: 17.4] [Reference Citation Analysis (3)] |
| 35. | Alifano M, Mansuet-Lupo A, Lococo F, Roche N, Bobbio A, Canny E, Schussler O, Dermine H, Régnard JF, Burroni B, Goc J, Biton J, Ouakrim H, Cremer I, Dieu-Nosjean MC, Damotte D. Systemic inflammation, nutritional status and tumor immune microenvironment determine outcome of resected non-small cell lung cancer. PLoS One. 2014;9:e106914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 135] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 36. | Diakos CI, Charles KA, McMillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493-e503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1819] [Cited by in RCA: 1748] [Article Influence: 145.7] [Reference Citation Analysis (4)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/