Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.102730
Revised: December 16, 2024
Accepted: February 11, 2025
Published online: April 27, 2025
Processing time: 152 Days and 19.5 Hours
Heterotopic gastric mucosa (HGM) is the most common type of epithelial hetero
We reported a middle-aged man admitted to the hospital due to abdominal pain, whose gastroduodenoscopy revealed a polypoid uplift in the duodenum. The attending physician directly removed the lesion using a snare, histopathological analysis demonstrated multidirectional cellular differentiation on the basis of duodenal HGM. The hematoxylin and eosin stains of the duodenal lesion demon
We reported the first case of gastric adenocarcinoma of fundic gland mucosa arising in the duodenum on the basis of HGM. Although HGM is mostly benign, there is also a risk of carcinogenesis.
Core Tip: This case represents the first reported of gastric adenocarcinoma of fundic gland mucosa in the duodenum arising from heterotopic gastric mucosa. The article highlights a rare occurrence, which could be of great interest to medical professionals encountering similar cases. The collaboration between the gastroenterology and pathology departments in different hospitals shows the importance of interdisciplinary teamwork in diagnosing and treating complex medical cases.
- Citation: Yang QY, Xu J, Hu JW, Huang XD. Gastric adenocarcinoma of fundic gland mucosa arising in heterotopic gastric mucosa of the duodenum: A case report. World J Gastrointest Surg 2025; 17(4): 102730
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/102730.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.102730
Heterotopic gastric mucosa (HGM) has histological characteristics of gastric mucosa, but it is located outside the gastric border and may occur anywhere from the oral cavity to the rectum[1]. The esophagus is the most common site of HGM, followed by the duodenum. The incidence of HGM in the duodenum is reported to range from 0.51% to 8.9%[1-3]. HGM in the duodenum can present as polyps, lateral growth, submucosal lesions, and other growth patterns[4]. It is believed that HGM in the duodenum mainly presents as polypoid lesions[2,5]. Oftentimes, polypoid ectopic gastric mucosa found incidentally during endoscopy is asymptomatic in patients[6]. Primary duodenal cancer is one of the rarest cancers in the gastrointestinal tract, and malignant transformation caused by HGM is extremely rare[7]. Gastric fundic gland neoplasms represent a rare form of gastric neoplasia, distinguished by their differentiation towards the fundic glands. Based on infiltration and cellular differentiation characteristics, it can be classified into oxyntic gland adenoma, gastric adenocarcinoma of the fundic gland, and gastric adenocarcinoma of fundic gland mucosa (GA-FGM). Our case represents the first reported of GA-FGM in the duodenum arising from HGM.
A 55-year-old male was admitted to the hospital for intermittent abdominal pain lasting 5 years.
For the past 5 years, the patient had recurrent mild abdominal pain, with the main location being the upper abdomen and around the navel, without nausea or vomiting, without acid reflux or heartburn, without a feeling of food blockage, occasionally with small amount of bloody stools.
The patient had hypertension for 6 years and was undergoing medication treatment.
The patient denied smoking and drinking habits, and all family members were in good health.
On examination, his body temperature was 36.7 °C, pulse rate 68 beats/minute, respiratory rate 18 breaths/minute, blood pressure 134/92 mmHg, and oxygen saturation 100% on room air.
All blood test results, including tumor markers, were within normal limits. Routine stool examination and occult blood test showed no abnormalities. Enhanced computed tomography scan of the whole abdomen and colonoscopy showed no significant abnormalities.
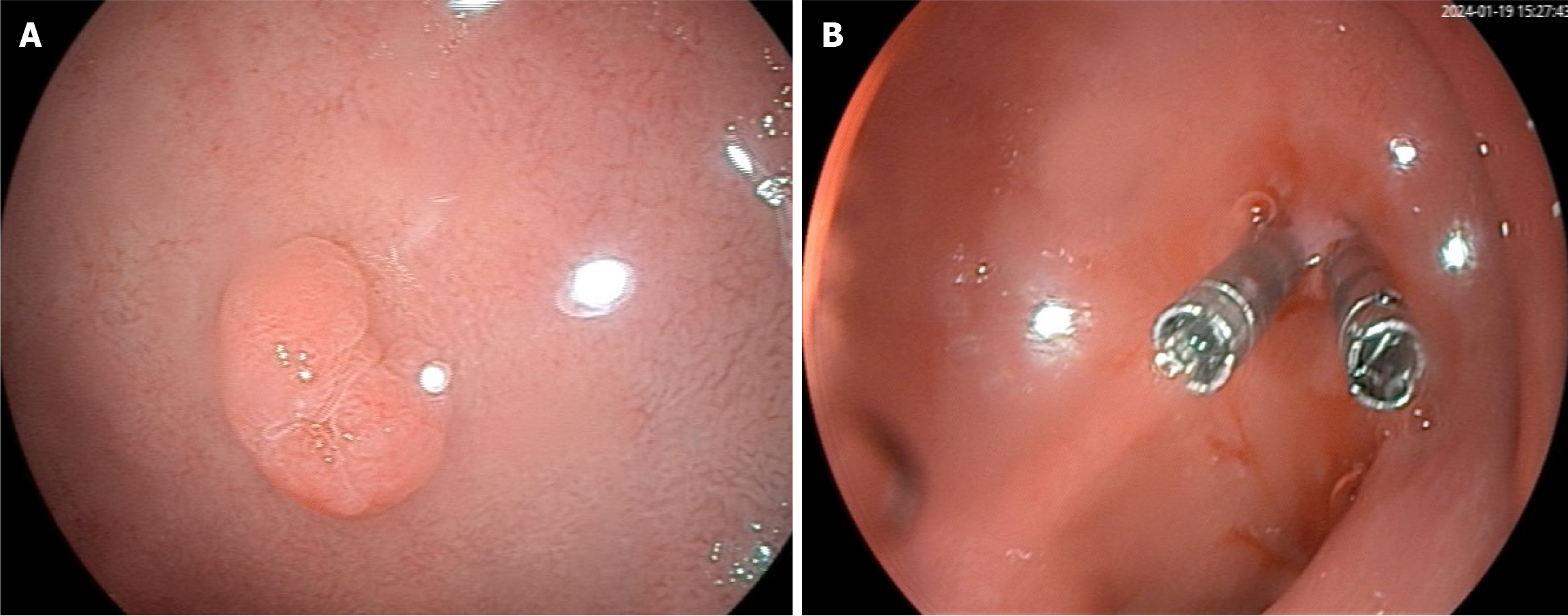
Upper gastrointestinal endoscopy revealed chronic non-atrophic gastritis and a 0.8 cm × 1.2 cm polypoid uplift in the duodenal bulb (Figure 1A). The attending physician directly removed the lesion using a snare, and the wound was closed with two titanium clips (Figure 1B).
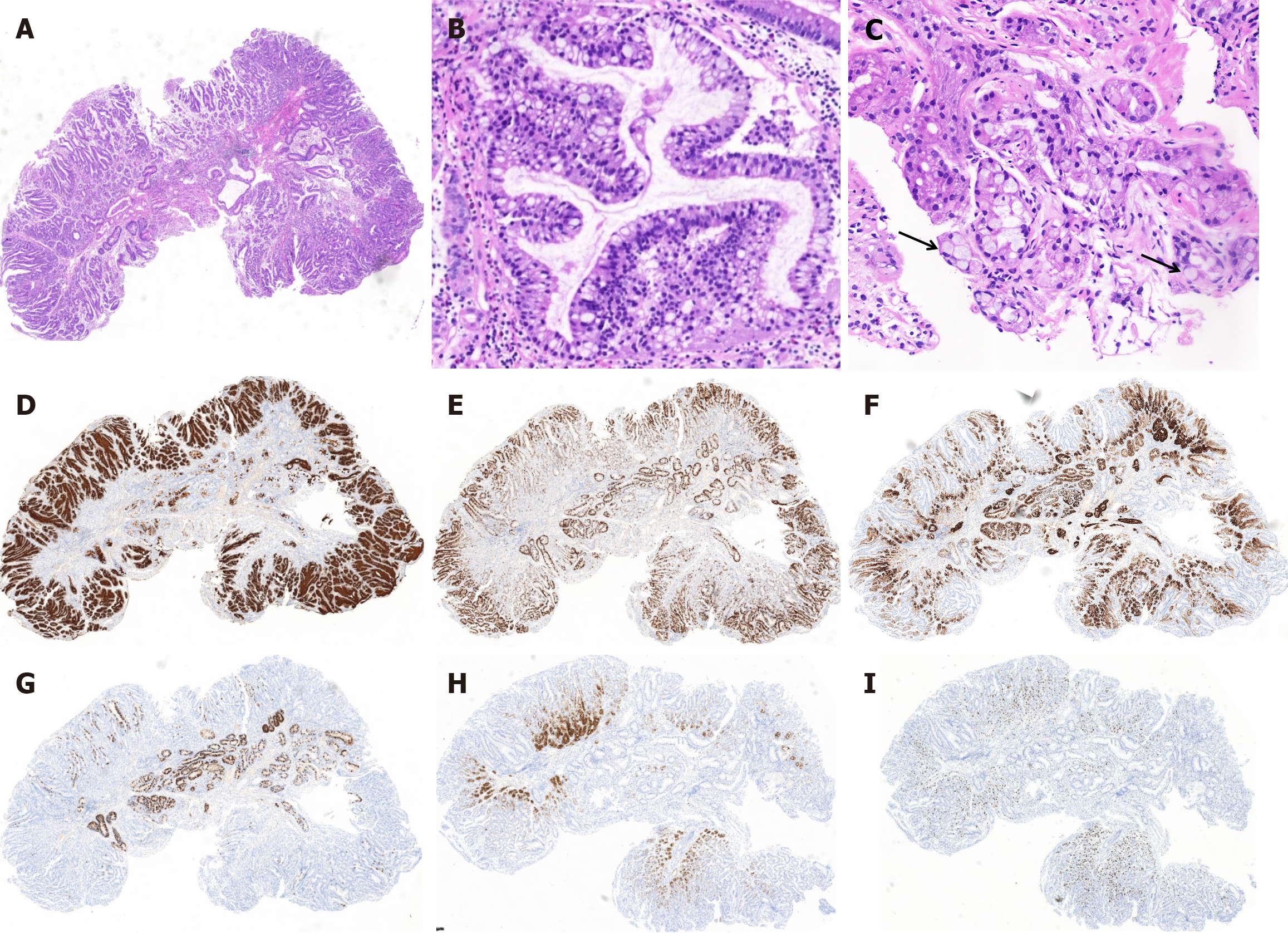
In the presented case, hematoxylin and eosin staining and immunohistochemical analysis demonstrated obvious gastric fundus gland structure and multidirectional cellular differentiation, which were predominantly composed of three distinct components (Figure 2). Firstly, the superficial mucosa exhibited gastric foveolar epithelium, which was extensively positive for MUC5AC (Figure 2A and D), with certain regions demonstrating intraluminal papillary growth, branching, serration, and bicolored cytoplasm, notably lacking mucous caps. The nuclei appeared round and deeply stained. Coupled with the high proliferative activity indicated by Ki-67 (Figure 2E), these findings are entirely consistent with foveolar epithelium high-grade intraepithelial neoplasia. Secondly, except for focal residual normal oxyntic glands, numerous mucous glands were observed. Immunohistochemical staining of these mucous glands revealing MUC6 (Figure 2F), positivity, partial positivity for pepsinogen I (Figure 2H), and H+/K+ ATPase (Figure 2I), and scattered Ki67 positivity. These findings are consistent with a tumor exhibiting differentiation towards oxyntic glands. Thirdly, in the deepest region of the mucosa adjacent to the muscularis mucosae, scattered dilated glands were observed (Figure 2B), which exhibiting disrupted glandular polarity, occasional intraglandular papillary growth, and nuclei arranged in a double layer. The cytoplasm contained small vacuoles, reminiscent of immature goblet cells. Additionally, focal areas displayed several signet-ring cell changes (Figure 2C), dispersed throughout the tissue. Regrettably, the glands initially observed in the hematoxylin and eosin stains were no longer detectable following deeper sectioning for immunohistochemistry. Only a few transversely elongated glands remained, which were partially positive for MUC5AC, MUC6, and MUC2 (Figure 2G), and high proliferative Ki67 activity. Although direct evidence is lacking, we hypothesize that this region of the tumor has undergone enterotype differentiation and may also contain a poorly differentiated component.
These poorly differentiated small mucinous vacuoles in the glands need to be distinguished from the cytoplasmic vacuolization caused by proton pump inhibitors, according to some pathological experts. However, considering the poor prognosis of signet ring cell carcinoma, surgical resection is recommended for patients. The patient rejected surgery and underwent endoscopic mucosal dissection (ESD) 2 months later.
The patient was ultimately diagnosed with GA-FGM arising from HGM of the duodenum, which also had included intestinal-type and a few suspected low-differentiation components.
The patient underwent ESD 2 months later. Duodenal mucosa was observed under the postoperative specimen microscope, part of the surface intestinal epithelium was replaced by gastric foveolar epithelium, with papillary growth, some retaining the mucous cap of gastric concave and some missing the mucous cap, and the nuclei were arranged in pen-shaped pseudolamellar arrangement. Therefore, these residuary glands were considered to be low-grade intraepithelial neoplasia. No signet-ring cell change were observed in the postoperative pathological sections.
A follow-up gastroscopy around 2 months post-ESD did not reveal any significant abnormalities. The follow-up is ongoing.
HGM is frequently identified in duodenal elevated lesions, especially in the duodenal bulb[2]. HGM is defined as the presence of gastric mucosa outside of the stomach, which is documented by histologic finding[8]. Histologically, it resembles gastric body mucosa, with gastric foveolas and oxyntic glands. The presence of oxyntic glands facilitates the secretion of gastric acid and pepsin, which may result in clinical symptoms. However, due to the neutralization of gastric acid and pepsin by duodenal fluid, clinical symptoms of HGM in the duodenum are often atypical, only occasionally discovered during gastroscopy[1,2]. But cases of duodenal HGM have been reported due to symptoms such as bleeding, narrowing, and even malignancy[9-11].
A report showed that among the 137 patients with duodenal ulcer, 76 cases (55%) had HGM in the obtained specimens, suggesting that HGM was strongly associated with concurrent duodenal ulcer[10]. In clinical practice, small polyps in the duodenum are mostly considered benign and rarely biopsied. Duodenal ulcers are rarely biopsied unless malignancy is suspected. Therefore, we believe that the incidence of HGM in the duodenum may be underestimated. Therefore, early biopsies for HGM in the duodenum should be performed for accurate diagnosis and further management to prevent the risk of malignant transformation.
HGM is essentially a benign entity, with the occurrence of malignant transformation being exceedingly rare. However, it is a potential precursor to pyloric gland adenoma and gastric phenotype adenocarcinoma[12]. A Japanese report showed that 3.4% of early carcinomas of the duodenal bulb arise from HGM[13]. The mechanism of carcinomatosis is considered the metaplasia-dysplasia-carcinoma sequence or adenoma-carcinoma sequence, as in other epithelial tissues. Helicobacter pylori infection is the greatest risk factor for gastric cancer (GC)[14].
But GA-FGM may be a subtype of Helicobacter pylori-negative GC[15]. Some studies proposed naming tumors showing differentiation toward fundic glands and foveolar epithelium-like differentiation as GA-FGM[16,17]. Although GA-FGM is no longer a rare case in the stomach, there have been no documented cases involving the duodenum. This case represents the first reported of GA-FGM in the duodenum arising from HGM. The preferred treatment modality com
The distinctiveness of this case is attributable not only to the presence of gastric fundic gland-type adenocarcinoma but also to the tendency of its transformation into enterotype. This phenomenon may elucidate why fundic gland-type tumors are typically identified in early-stage GC, with no documented occurrences in advanced stages. This phenomenon is likely attributable to the highly benign nature of fundic gland differentiation. However, as the tumor progresses and undergoes transformation into an intestinal or common type, it acquires the capacity for rapid advancement, thereby supplanting the most benign component.
In short, our case represents the first reported of GA-FGM in the duodenum arising from HGM. The growth pattern of ectopic gastric mucosa in the duodenum is diverse, with polyps being a common presentation. Therefore, the possibility of HGM should be considered in duodenal polypoid lesions. Although most HGMs are asymptomatic and benign, there is a possibility of malignant transformation. Early biopsy and appropriate treatment are crucial for reducing the risks of complications and malignant transformation.
Special thanks to the pathological experts for their assistance in diagnosing the case.
| 1. | Yu L, Yang Y, Cui L, Peng L, Sun G. Heterotopic gastric mucosa of the gastrointestinal tract: prevalence, histological features, and clinical characteristics. Scand J Gastroenterol. 2014;49:138-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 2. | Terada T. Heterotopic gastric mucosa of the gastrointestinal tract: a histopathologic study of 158 cases. Pathol Res Pract. 2011;207:148-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 3. | Genta RM, Kinsey RS, Singhal A, Suterwala S. Gastric foveolar metaplasia and gastric heterotopia in the duodenum: no evidence of an etiologic role for Helicobacter pylori. Hum Pathol. 2010;41:1593-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 4. | Guo LJ, Ye L, Huang ZY, Yin X, Hu B. Magnetic beads-assisted endoscopic submucosal dissection of duodenal heterotopic gastric mucosa with fibrosis. Endoscopy. 2019;51:E113-E115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Arévalo Suarez F, Barreda C, Portugal S, Cordoba S, Rayme S, Vergara G, Guerra E, Salazar S, Monge E. [Heterotopic gastric mucosa in duodenum: endoscopic and histological features]. Rev Gastroenterol Peru. 2017;37:231-234. [PubMed] |
| 6. | Tibesar EE. Bleeding Gastric Heterotopic Polyp in the Duodenum of a Teenager with Severe Factor VII Deficiency. Case Rep Gastroenterol. 2020;14:467-471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 7. | Fukushima R, Kubo N, Suzuki S, Yagi N, Furuke S, Ooki T, Aihara R, Mogi A, Hosouchi Y, Shirabe K. Primary duodenal carcinoma suspected to arise from ectopic gastric mucosa: a case report. Surg Case Rep. 2023;9:23. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 8. | Lindoso L, Ballengee CR, Patel KP, Romero R, Caltharp S, Alazraki AL, Kugathasan S. Multilocus Heterotopic Gastric Mucosa of Ileum Masquerading as VEOIBD in a Newborn. Pediatrics. 2019;143:e20182398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Gómez Espejo SM, Fernández Pérez F, Gutiérrez Hernández P, Pereda Salgado T. An unusual endoscopic presentation of diffuse duodenal gastric heterotopia. Rev Esp Enferm Dig. 2020;112:414-415. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 10. | Noguchi H, Kumamoto K, Harada Y, Sato N, Nawata A, Tasaki T, Kimura S, Shimajiri S, Nakayama T. Prevalence of Helicobacter pylori infection rate in heterotopic gastric mucosa in histological analysis of duodenal specimens from patients with duodenal ulcer. Histol Histopathol. 2020;35:169-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Gubatan J, Raines N, Khosravi H, Challies TL, Berzin TM. Hemorrhage from Extra-Antral Gastric Antral Vascular Ectasia in a Patient with Duodenal Heterotopic Gastric Mucosa. Case Rep Gastrointest Med. 2016;2016:4325302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Kitagawa D, Yamasaki T, Ikeda T, Sakata Y, Hirata N, Suekane T, Sugimori S, Ishii N, Sakurai K, Nebiki H. A case of pyloric gland adenoma with high-grade dysplasia in the duodenum arising from heterotopic gastric mucosa observed over 5 years. Clin J Gastroenterol. 2023;16:26-31. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 13. | Kawamoto K, Ushio K, Ushio A, Seiku H, Makoto U, Takashi Y, Hitoshi O, Shinji K, Yuichiro S, Tetsuro K, Hideyuki W, Hiroko O, Masanobu M, Shin M, Yoichi Y, Hirotoshi Y. Diagnosis of Neoplastic and Neoplastic-like Duodenal Small Lesions, Adenoma and Cancer. Stomach Intestine. 2001;36:1507-1527. [DOI] [Full Text] |
| 14. | Geng H, Dong Z, Zhang L, Yang C, Li T, Lin Y, Ke S, Xia X, Zhang Z, Zhao G, Zhu C. An Immune Signature for Risk Stratification and Therapeutic Prediction in Helicobacter pylori-Infected Gastric Cancer. Cancers (Basel). 2022;14:3276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Imamura K, Yao K, Nimura S, Tanabe H, Kanemitsu T, Miyaoka M, Ono Y, Ueki T, Iwashita A. Characteristic endoscopic findings of gastric adenocarcinoma of fundic-gland mucosa type. Gastric Cancer. 2021;24:1307-1319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 16. | Iwashita A, Tanabe H. Diagnosis of low grade gastric differentiated adenocarcinoma. Stomach Intestine. 2010;45:1057-1060. [DOI] [Full Text] |
| 17. | Tanabe H, Iwashita A, Ikeda K, Ota A, Yao K. Histopathological Characteristics of Gastric Adenocarcinoma of Fundic Gland Type. Stomach Intestine. 2015;50:1469-1479. [DOI] [Full Text] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/