Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.101295
Revised: January 18, 2025
Accepted: February 11, 2025
Published online: April 27, 2025
Processing time: 98 Days and 23.3 Hours
Common bile duct stones pose a high risk of recurrence or disease progression if not promptly treated. However, there is still no optimal treatment approach.
To investigate the clinical efficacy of modified pancreatic duct stent drainage in endoscopic retrograde cholangiopancreatography (ERCP) for treating common bile duct stones.
This retrospective study included 175 patients with common bile duct stones treated at Taizhou Fourth People’s Hospital between January 1, 2021, and November 30, 2023. The patients were divided into three groups-the modified pancreatic duct stent drainage group (59 cases), the nasobiliary drainage group (58 cases), and the standard biliary drainage group (58 cases). Preoperative general clinical data, laboratory indicators, and the visual analog scale (VAS) at two time points (24 hours before and after surgery) were compared, along with postoperative complications across the three groups.
Serum levels of aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, gamma-glutamyltransferase, total bilirubin, direct bilirubin, C-reactive protein, and amylase were significantly lower in the modified pancreatic duct stent drainage group and the standard biliary drainage group than those in the nasobiliary drainage group (P < 0.05). However, no statistically significant differences were observed in white blood cells, hemoglobin, or neutrophil levels among the three groups (P > 0.05). The standard biliary drainage group had significantly lower VAS scores [(4.36 ± 1.18) points] than those for the modified pancreatic duct stent drainage group [(4.92 ± 1.68) points] (P = 0.033), and the nasobiliary drainage group [(5.54 ± 1.24) points] (P = 0.017). There were no statistically significant differences in complication rates across the three groups (P > 0.05).
Compared to standard biliary drainage and nasobiliary drainage, the modified pancreatic duct stent used during ERCP for patients with bile duct stones significantly reduced hepatocyte injury, improved liver function parameters, alleviated inflammation and pain, enhanced patient comfort, and demonstrated superior safety.
Core Tip: There is currently a paucity of multidimensional comparative analyses concerning the clinical efficacy of modified pancreatic duct plastic stents for endoscopic retrograde biliary drainage, endoscopic nasobiliary drainage, and Christmas tree-shaped plastic stents for standard biliary drainage following endoscopic retrograde cholangiopancreatography (ERCP) for common bile duct stone removal. This study addresses this gap with a comprehensive analysis. Our findings reveal that compared to standard biliary drainage and nasobiliary drainage, modified pancreatic stents offer significant benefits, including a significant reduction in hepatocyte damage, improvement in liver function parameters, alleviated inflammation and pain, enhanced patient comfort, and increased treatment safety. Therefore, we recommend modified pancreatic plastic stents as the preferred post-ERCP drainage method for patients with common bile duct stones.
- Citation: Qian LJ, Xu C, Wang JR, Quan J. Efficacy of modified pancreatic duct stent drainage during endoscopic retrograde cholangiopancreatography for common bile duct stones. World J Gastrointest Surg 2025; 17(4): 101295
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/101295.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.101295
Cholelithiasis of the common bile duct refers to the presence of stones at the distal end of the common bile duct. These stones are typically a mixture, with bile pigment stones being the most common type. The causes of choledocholithiasis in patients with choledochal cholelithiasis are multifaceted, resulting from factors such as cholangitis, roundworm infestations, and bile stasis. The etiology of the disease is complex, leading to a wide range of clinical manifestations, such as epigastric cramps, chills, and penetrating low back pain[1]. Choledochal stones occur in approximately 5%-20% of patients with cholelithiasis and in about 10% of those who undergo cholecystectomy[2,3]. Relevant epidemiological data showed that cholelithiasis is the most common biliary tract disease in the United States, with a mortality rate of 260.8 deaths per 100000 people. Each year, approximately 180000 patients receive treatment for cholelithiasis, while over 700000 undergo surgical procedures related to the condition[4]. Studies indicate that the incidence of common bile duct stones ranges from approximately 5% to 20%, with an increasing trend over time[5]. More than 20% of affected patients present with clinical symptoms such as fever, abdominal pain, and jaundice. In severe cases, complications, such as acute obstructive suppurative cholangitis and septic shock, may occur[6]. If left untreated, gallstones can lead to recurrent symptoms and disease progression, highlighting the critical need for timely and effective stone removal[7].
Currently, endoscopic retrograde cholangiopancreatography (ERCP) serves as a cornerstone in the clinical treatment of bile duct stones, effectively alleviating symptoms and facilitating their complete removal[8]. Image-guided techniques are essential in enhancing the precision and success of surgical interventions. Since its development in the 1960s, ERCP has evolved into a widely used procedure for managing common bile duct stones. It is an imaging technique involving the insertion of a catheter through a duodenoscope into the pancreaticobiliary duct opening at the ampulla of Vater. By injecting contrast dye through the scope, ERCP allows visualization of the pancreato-biliary system, enabling precise identification of stone count, location, assessment of biliary pressure, and evaluation of the papilla’s function. ERCP offers advantages such as minimal invasiveness and rapid recovery[9]. However, it is a technically demanding procedure with a relatively high risk of complications, ranging from 5% to 10%[10]. Common complications following ERCP include wound infection, pancreatitis, cholangitis, hemorrhage, perforation, and hyperamylasemia, which can significantly affect patients’ postoperative recovery and overall well-being, limiting its clinical use[10-12]. To improve surgical outcomes, clinicians often use biliary stenting to reduce biliary pressure, support wound healing, and facilitate inflammation resolution while aiding in the removal of residual stones from the bile duct.
Stent drainage procedures include both standard biliary drainage and endoscopic retrograde biliary drainage (ERBD)[13]. The Christmas tree-shaped plastic stent is commonly used in clinical practice for biliary drainage due to its low risk of spontaneous detachment. Guidelines[14] recommend removal after 3 to 6 months; however, repeated removals may increase treatment costs, psychological burden, and the risk of complications. Research has shown that endoscopic nasobiliary drainage (ENBD) is a minimally invasive and relatively straightforward endoscopic technique that can achieve bile duct drainage outcomes comparable to traditional surgery, with minimal trauma and endoscopic intervention, while potentially reducing the risk of complications following ERCP[15]. Park et al[16] reported that using pancreatic plastic stents for biliary internal drainage resulted in significant drainage outcomes. Similarly, a study by He et al[17] demonstrated that modified pancreatic stents effectively prevented post-ERCP pancreatitis, with a self-removal rate of 84.21% after 14 days.
Currently, there is no standardized consensus on the optimal surgical approach for biliary drainage in clinical practice, highlighting the need for further research to establish a unified strategy. Additionally, no multidimensional comparative analysis has been conducted to evaluate the clinical efficacy of using a modified pancreatic duct plastic stent for ERBD or ENBD and a Christmas tree-shaped plastic stent for standard biliary drainage following ERCP for bile duct stone removal. Therefore, in this study, we compared the clinical efficacy of the modified pancreatic duct plastic stent for ERBD, ENBD, and Christmas tree-shaped plastic stents in ERCP for treating common bile duct stones. We aim to explore the effectiveness of the improved pancreatic duct plastic stent in patients with ERCP and its impact on postoperative complications, thereby identifying the most suitable drainage technique after ERCP for common bile duct stones and improving patient outcomes.
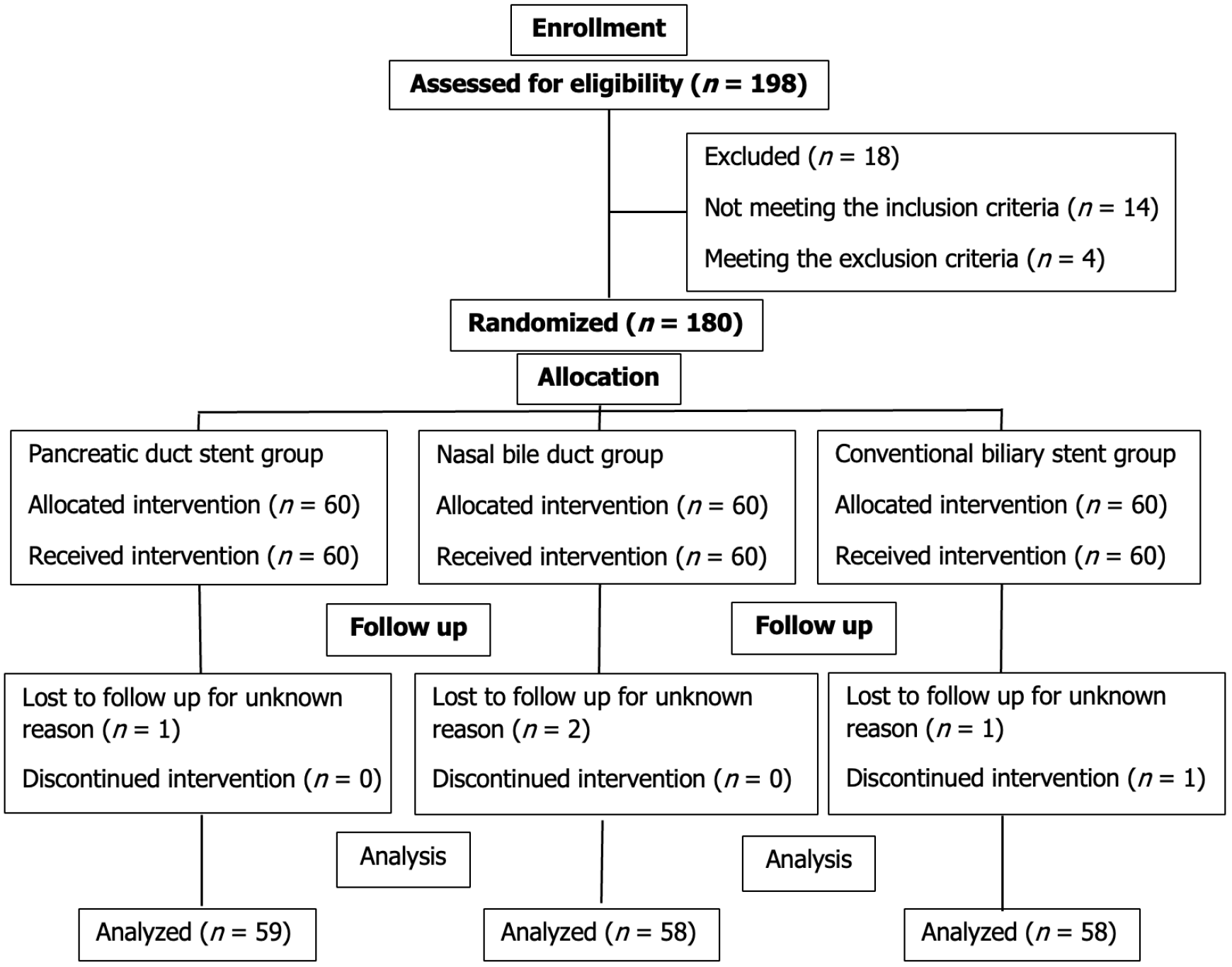
This retrospective study included 175 patients with choledocholithiasis admitted to Taizhou Fourth People’s Hospital between January 1, 2021, and November 30, 2023. The patients were divided into three groups-the modified pancreatic duct stent drainage group (using ERBD technology, n = 59), the nasobiliary drainage group (using ENBD technology, n = 58), and the standard biliary drainage group (using Christmas tree-shaped stent, n = 58). The patient inclusion process is graphically illustrated in Figure 1.
The inclusion criteria were as follows: Diagnosis of choledocholithiasis according to the 2019 European Society of Gastrointestinal Endoscopy clinical practice guidelines[18]; confirmation of gallstones in the biliary ducts through histopathological examination or imaging techniques, such as laparoscopy[19]; pre-treatment abdominal pain of varying degrees, with a visual analog scale (VAS) score of 5 or higher; adherence to treatment protocols; and all surgeries performed by the same surgical team. Patients were excluded if they had any of the following: Significant organ dysfunction (heart, liver, or kidney), not meeting the indications for ERCP, severe digestive system impairment, concurrent malignant tumors, or poor compliance or refusal to participate.
Surgical equipment: The following surgical equipment was utilized during the interventions: An electronic duodenoscope (TJF260V by Olympus Corporation, Japan), guidewire (Yellow Stripe, 0.035 inches or 4500 mm, Nanjing Minimally Invasive Medical Technology Co., Ltd.), papillotome (Medi-Globe GmbH), forceps, balloon catheter (Nanjing Minimally Invasive Medical Technology Co., Ltd.), stone retrieval basket (Cook Medical), Christmas tree-shaped plastic stent (8.5 Fr, Nanjing Minimally Invasive Medical Technology Co., Ltd.), standard nasobiliary drainage tube (NBDS-B-7/250-P, Nanjing Minimally Invasive Medical Technology Co., Ltd.), and a modified pancreatic duct stent (SPSOF-7-5, Cook Medical).
ERCP procedure: All patients underwent routine blood tests, liver and kidney function assessments, coagulation function evaluations, and imaging examinations 24 hours before the operation. They were required to fast and refrain from food and water for at least 6 hours before the operation. After positioning the patients prone, they received routine anesthesia and analgesia. The endoscope was then inserted through the mouth to reach the duodenal papilla. Selective choledochal intubation was performed using a papillotome knife with a guidewire, followed by an injection of a 20% iophedronol contrast. The choledochal ducts were observed using C-arm fluoroscopy. During the procedure, endoscopic sphincterotomy and endoscopic balloon dilatation were performed based on the location and size of the stones, as well as the diameter of the bile duct. After stone extraction using a balloon or mesh basket, re-imaging was performed to confirm the complete clearance of the stones.
Drainage procedure: In the modified pancreatic duct stent drainage group, following cholecystectomy for gallstones and ERBD, a modified pancreatic duct plastic stent (COOK Company) was inserted into the pancreatic duct. Correct placement of the stent was confirmed using intraoperative C-arm fluoroscopy. The papilla and the field of vision were free from active hemorrhage, and the endoscope was removed, marking the completion of the procedure.
In the nasobiliary drainage group, following choledochal stone extraction, an NBDS-B-7/250-P (Nanmicro Medical Technology Co., Ltd.) was inserted into the choledochus under the guidance of a guidewire. ENBD was performed. The tip of the nasobiliary tube was positioned at the openings of the left and right hepatic ducts, allowing bile outflow. C-arm fluoroscopy was used to confirm that the nasobiliary drainage tube was securely placed. The papilla and field of view were examined, revealing no active hemorrhage. The procedure was concluded by retracting the endoscope.
In the standard biliary drainage group, following choledochal stone extraction, a BPDS-13442-0807/22 Christmas tree-shaped stent (Nanjing Microtronics) was inserted into the choledochal duct for biliary drainage. C-arm fluoroscopy was used to confirm that the stent was securely placed. An examination of the papilla and the visual field showed no active hemorrhage. The procedure was concluded with the removal of the endoscope. Postoperatively, the patient’s vital signs and condition should be closely monitored. Additionally, liver function, blood routine, amylase, C-reactive protein (CRP), and other relevant indices should be retested as needed. Supportive treatments, such as anti-infection measures, acid suppression, enzyme suppression, and fluid rehydration, should also be administered.
General information: This study recorded and compared various parameters among the three patient groups, including age, gender, body mass index (BMI), history of alcohol consumption, time of disease onset before surgery, total hospital stay duration, disease duration, American Society of Anesthesiologists (ASA) classification, surgical duration, bile duct diameter, stone diameter, and the number of gallstones in the bile duct.
ASA classification standards[20] are as follows: Class I refers to a physically healthy individual with normal organ function; Class II describes a patient with mild systemic diseases and a surgical condition but with preserved compensatory mechanisms; Class III indicates a patient with severe systemic diseases that limits physical activity but does not preclude daily functioning; Class IV applies to a patient with severe systemic disease that is life-threatening; Class V refers to a critically ill patient who is unlikely to survive without surgical intervention; and Class VI designates a deceased patient whose organs are being prepared for transplantation.
Laboratory indicators, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), total bilirubin (TBIL), direct bilirubin (DBIL), CRP, white blood cells (WBC), hemoglobin (Hb), neutrophils (NEU), and amylase (AMY), were measured and compared across the three patient groups before and 24 hours after drainage surgery. Pain levels were evaluated using a VAS, and postoperative complications, such as pancreatitis, cholangitis, hemorrhage, perforation, wound infection, hyperamylasemia, and mortality, were recorded.
The VAS scoring scale[21]: The scale ranges from 1 to 10, where a score of 3 or below indicates mild or no pain, a score of 4 to 6 reflects significant pain that may impact sleep, and a score of 7 or above represents severe pain with a significant impact on sleep and appetite.
Pancreatitis: This is defined by at least two of the following revised Atlanta criteria[22]: (1) Persistent upper abdominal pain attributable to pancreatitis; (2) A threefold or more significant increase in amylase or lipase levels within 24 hours postsurgery; or (3) Characteristic imaging findings on computed tomography (CT) scans, such as pancreatic enlargement, necrosis, or peripancreatic fluid collection.
Severe pancreatitis: This is characterized by peripancreatic fluid collections and/or pleural and peritoneal effusions on abdominal CT scans[23], requiring management with percutaneous catheter drainage or surgical intervention.
Cholangitis: Fever (temperature > 38 °C) occurring within 24 hours postsurgery, accompanied by elevated WBC count and abdominal pain, with no evidence of infection at another site[24].
Hemorrhage: Symptoms include black stool or hematemesis within 24 hours postsurgery, fresh blood in the nasobiliary drainage tube, and a drop in blood pressure, necessitating endoscopic hemostasis.
Perforation: Following surgery, the patient experienced worsening abdominal pain within 24 hours. Upon imaging confirmation of a perforation, the patient was managed with a medically supervised fasting regimen, gastrointestinal decompression, and antibiotic therapy for infection control. In cases of extensive perforation, prompt surgical intervention was deemed necessary.
Wound infection: Obvious redness and oozing at the surgical incision site within 24 hours postsurgery, necessitating aggressive antibiotic treatment, along with concurrent physiotherapy and other supportive care.
Hyperamylasemia: A postsurgery increase in blood amylase levels exceeding three times the upper normal limit, occurring within 24 hours without fever or abdominal pain symptoms.
Statistical analysis was conducted using IBM Statistics Version 27.0.1 (SPSS Inc., Beijing, China). In the patients’ general information, gender, history of alcohol consumption, ASA classification, number of gallstones in the bile duct, and complication rates were all categorical data presented as n (%). Since the gender, ASA classification, and complication rates had theoretical frequencies > 5 and a total sample size of at least 40, Pearson’s χ2 test was applied. For the number of gallstones in the bile duct, data satisfying 1 ≥ theoretical frequency ≥ 5 and a total sample size of at least 40 were analyzed using the continuity-corrected χ2 test (Yates’ correction). Continuous data were initially analyzed for normal distribution using histograms and the Shapiro-Wilk test for single samples. In the patients’ general information, variables such as age, BMI, time of disease onset before surgery, total hospital stay duration, disease duration, surgical duration, bile duct diameter, stone diameter, and all laboratory indicators, as well as VAS scores, were normally distributed and represented as (mean ± SD). The AST, ALT, ALP, GGT, TBIL, DBIL, CRP, WBC, Hb, NEU, and AMY indicators met the assumption of homoscedasticity. These variables were compared using a one-way analysis of variance (ANOVA) across the three groups. Pairwise comparisons were performed using the least significant difference t-test for all comparisons, and a one-way ANOVA with repeated measures was used to compare data collected 24 hours before and after surgery within each group. Differences were considered statistically significant when P < 0.05.
Between January 1, 2021, and November 30, 2023, 175 patients with common bile duct stones successfully underwent ERCP treatment. Among them, 59 patients (33.71%) used pancreatic stent drainage, 58 patients (33.14%) employed nasobiliary drainage, and 58 patients (33.14%) utilized biliary stent drainage.
The baseline characteristics of all patients, including age, gender, BMI, history of alcohol consumption, time of disease onset before surgery, total hospital stay duration, disease duration, ASA classification, surgical duration, bile duct diameter, stone diameter, and the number of gallstones in the bile duct, are presented in Table 1. There were no statistically significant differences among the three patient groups (P > 0.05) (Table 1).
| Modified pancreatic duct stent drainage group (n = 59) | Nasobiliary drainage group (n = 58) | Standard biliary drainage group | χ2/F value | P value | |
| Gender (male/female) | 35 (59.32)/24 (40.68) | 33 (56.90)/25 (43.10) | 32 (55.17)/26 (44.83) | 0.208 | 0.901 |
| ASA classification (I/II/III) | 22 (37.29)/28 (47.46)/9 (15.25) | 11 (18.97)/36 (62.07)/11 (18.97) | 16 (27.59)/32 (54.24)/10 (17.24) | 4.887 | 0.299 |
| The number of gallstones in the bile duct (1/2/3/≥ 4) | 29 (49.15)/7 (11.86)/5 (8.47)/18 (30.51) | 28 (48.28)/5 (8.62)/5 (8.62)/20 (34.48) | 23 (39.66)/6 (10.34)/5 (8.62)/24 (41.38) | 2 | 0.92 |
| Age (year) | 51.42 ± 15.14 | 52.00 ± 16.08 | 51.84 ± 15.84 | 0.021 | 0.979 |
| BMI (kg/m2) | 23.34 ± 3.82 | 23.29 ± 3.67 | 23.16 ± 3.54 | 0.037 | 0.964 |
| History of drinking (yes/no) | 26 (44.07)/33 (55.93) | 24 (41.38)/34 (58.62) | 23 (39.66)/35 (60.34) | 0.238 | 0.888 |
| Time of onset before surgery (hours) | 35.51 ± 3.25 | 35.94 ± 3.58 | 36.10 ± 4.33 | 0.389 | 0.678 |
| Total hospital stay duration (days) | 9.12 ± 4.34 | 10.62 ± 5.06 | 11.04 ± 5.13 | 2.536 | 0.082 |
| Disease duration (month) | 21.14 ± 4.28 | 22.04 ± 4.05 | 21.67 ± 3.94 | 0.715 | 0.491 |
| Surgical duration (minutes) | 61.04 ± 8.91 | 60.55 ± 6.42 | 60.07 ± 7.49 | 0.233 | 0.792 |
| Diameter of the bile duct (cm) | 1.34 ± 0.43 | 1.31 ± 0.48 | 1.38 ± 0.51 | 0.318 | 0.728 |
| Stone diameter (cm) | 0.86 ± 0.35 | 0.88 ± 0.36 | 0.91 ± 0.43 | 0.254 | 0.776 |
There were no statistically significant differences in serum levels of AST, ALT, ALP, GGT, TBIL, DBIL, CRP, WBC, Hb, NEU, and AMY among the three patient groups 24 hours before surgery (all P > 0.05).
However, serum levels of AST, ALT, ALP, GGT, TBIL, DBIL, CRP, WBC, Hb, NEU, and AMY in all three patient groups significantly decreased 24 hours after surgery compared to 24 hours before surgery (all P < 0.05) (Table 2).
| Modified pancreatic duct stent drainage group (n = 59) | Nasobiliary drainage group (n = 58) | Standard biliary drainage group | F value | P value | P1 value | P2 value | P3 value | |
| AST (U/L) | ||||||||
| 24 hours before the surgery | 58.38 ± 18.49 | 55.71 ± 20.07 | 55.75 ± 20.84 | 0.217 | 0.642 | |||
| 24 hours after the surgery | 29.28 ± 3.47 | 48.64 ± 4.37 | 30.13 ± 3.88 | 449.671 | < 0.001 | < 0.001 | < 0.001 | 0.240 |
| ALT (U/L) | ||||||||
| 24 hours before the surgery | 85.36 ± 6.41 | 83.15 ± 6.74 | 84.28 ± 6.69 | 0.221 | 0.639 | |||
| 24 hours after the surgery | 35.44 ± 4.73 | 68.54 ± 5.58 | 36.49 ± 6.33 | 650.159 | < 0.001 | < 0.001 | < 0.001 | 0.294 |
| ALP (U/L) | ||||||||
| 24 hours before the surgery | 160.42 ± 35.48 | 159.45 ± 34.82 | 158.47 ± 31.82 | 1.153 | 0.284 | |||
| 24 hours after the surgery | 133.09 ± 33.21 | 153.14 ± 36.58 | 134.54 ± 31.25 | 6.390 | 0.002 | 0.002 | 0.003 | 0.837 |
| GGT (U/L) | ||||||||
| 24 hours before the surgery | 383.45 ± 106.82 | 364.58 ± 119.42 | 322.84 ± 128.94 | 0.402 | 0.527 | |||
| 24 hours after the surgery | 252.64 ± 44.52 | 294.59 ± 51.60 | 255.52 ± 55.31 | 12.314 | < 0.001 | < 0.001 | < 0.001 | 0.668 |
| TBIL (mol/L) | ||||||||
| 24 hours before the surgery | 28.65 ± 6.77 | 28.94 ± 6.82 | 28.59 ± 5.82 | 1.603 | 0.207 | |||
| 24 hours after the surgery | 16.08 ± 3.38 | 24.36 ± 4.16 | 16.18 ± 3.67 | 94.115 | < 0.001 | < 0.001 | < 0.001 | 0.878 |
| DBIL (mol/L) | ||||||||
| 24 hours before the surgery | 12.53 ± 3.56 | 13.05 ± 3.81 | 13.34 ± 3.64 | 3.165 | 0.077 | |||
| 24 hours after the surgery | 8.59 ± 2.14 | 9.93 ± 2.42 | 8.96 ± 2.57 | 5.213 | 0.006 | 0.002 | 0.032 | 0.328 |
| CRP (mg/L) | ||||||||
| 24 hours before the surgery | 104.58 ± 5.89 | 102.57 ± 6.68 | 101.53 ± 5.46 | 2.516 | 0.115 | |||
| 24 hours after the surgery | 83.65 ± 6.91 | 96.08 ± 7.04 | 84.53 ± 6.31 | 62.572 | < 0.001 | < 0.001 | < 0.001 | 0.311 |
| WBC (× 109/L) | ||||||||
| 24 hours before the surgery | 11.28 ± 5.94 | 10.64 ± 4.58 | 10.53 ± 5.08 | 0.089 | 0.766 | |||
| 24 hours after the surgery | 9.51 ± 4.41 | 9.46 ± 5.02 | 9.48 ± 4.66 | 0.003 | 0.997 | 0.942 | 0.984 | 0.959 |
| Hb (g/L) | ||||||||
| 24 hours before the surgery | 133.54 ± 20.19 | 134.48 ± 20.25 | 134.82 ± 20.72 | 2.171 | 0.143 | |||
| 24 hours after the surgery | 123.14 ± 19.58 | 124.15 ± 18.24 | 123.31 ± 18.94 | 0.043 | 0.958 | 0.796 | 0.805 | 0.992 |
| NEU (%) | ||||||||
| 24 hours before the surgery | 0.78 ± 0.14 | 0.79 ± 0.13 | 0.78 ± 0.15 | 0.627 | 0.430 | |||
| 24 hours after the surgery | 0.73 ± 0.08 | 0.76 ± 0.09 | 0.74 ± 0.07 | 1.337 | 0.265 | 0.157 | 0.159 | 0.999 |
| AMY (IU/L) | ||||||||
| 24 hours before the surgery | 281.26 ± 94.15 | 274.59 ± 93.08 | 268.42 ± 99.81 | 1.116 | 0.292 | |||
| 24 hours after the surgery | 121.46 ± 44.28 | 175.83 ± 46.82 | 127.43 ± 41.08 | 26.653 | < 0.001 | < 0.001 | < 0.001 | 0.432 |
After adjusting for preoperative variables, different drainage methods significantly affected the changes in serum levels of AST, ALT, ALP, GGT, TBIL, DBIL, CRP, and AMY. Post-treatment comparisons showed that the modified pancreatic duct stent drainage group had significantly lower AST, ALT, ALP, GGT, TBIL, DBIL, CRP, and AMY serum levels than the nasobiliary drainage group (P < 0.05). Similarly, the serum levels of AST, ALT, ALP, GGT, TBIL, DBIL, CRP, and AMY in the standard biliary drainage group were significantly lower than those in the nasobiliary drainage group, with a statistically significant difference (P < 0.05). There were no statistically significant differences in serum levels of AST, ALT, ALP, GGT, TBIL, DBIL, CRP, and AMY between patients in the standard biliary drainage group and those in the nasobiliary drainage group (P < 0.05). Additionally, no statistically significant differences were found in two-by-two comparisons of WBC, Hb, and NEU indicators among the three patient groups (all P > 0.05) (Table 2).
There was no statistically significant difference in VAS scores (F = 0.077, P = 0.781) 24 hours before surgery among the three patient groups. However, statistically significant differences in the VAS scores were observed among the three patient groups 24 hours after surgery (F = 10.416, P < 0.001) (Table 3). Preoperative covariates had no significant impact on the corresponding indicators in the three patient groups (P = 0.781).
| Modified pancreatic duct stent drainage group (n = 59) | Nasobiliary drainage group (n = 58) | Standard biliary drainage group (n = 58) | F value | P value | P1 value | P2 value | P3 value | |
| VAS (point) | ||||||||
| 24 hours before the surgery | 6.25 ± 2.16 | 6.24 ± 2.45 | 6.26 ± 2.54 | 0.077 | 0.781 | |||
| 24 hours after the surgery | 4.36 ± 1.18 | 5.54 ± 1.24 | 4.92 ± 1.68 | 10.416 | < 0.001 | < 0.001 | 0.017 | 0.033 |
VAS scores in all three groups significantly decreased 24 h after surgery compared to 24 hours before surgery (P < 0.05) (Table 3).
After adjusting for preoperative VAS scores, significant differences were found in VAS score changes between different drainage methods (P < 0.001). Post-hoc comparisons revealed that VAS scores in the modified pancreatic duct stent drainage group were significantly lower than those in the nasobiliary drainage group (P < 0.05). Similarly, VAS scores in the modified pancreatic duct stent drainage group were significantly lower than those in the standard biliary drainage group (P = 0.033). Finally, the standard biliary drainage group had significantly lower VAS scores than the nasobiliary drainage group (P = 0.017) (Table 3).
There was no statistically significant difference in complication rates among the three patient groups (P = 0.336) (Table 4).
| Modified pancreatic duct stent drainage group (n = 59) | Nasobiliary drainage group (n = 58) | Standard biliary drainage group (n = 58) | χ2 value | P value | |
| Complications | 14 (23.73) | 18 (31.03) | 21 (36.21) | 2.180 | 0.336 |
| Pancreatitis | 1 (1.69) | 0 (0.00) | 0 (0.00) | ||
| Cholangitis | 0 (0.00) | 1 (1.72) | 0 (0.00) | ||
| Hemorrhage | 0 (0.00) | 2 (3.45) | 2 (3.45) | ||
| Perforation | 1 (1.69) | 0 (0.00) | 1 (1.72) | ||
| Wound infection | 0 (0.00) | 0 (0.00) | 1 (1.72) | ||
| Hyperamylasemia | 12 (20.34) | 15 (25.86) | 17 (29.31) | ||
| Death | 0 (0.00) | 0 (0.00) | 0 (0.00) |
Our study found that the modified pancreatic duct stent in ERCP treatment for patients with bile duct stones effectively reduced hepatocyte injury, improved liver function, alleviated inflammation and pain, enhanced patient comfort, and a higher level of safety compared to standard biliary drainage and nasobiliary drainage methods.
After performing ERCP lithotripsy, both domestic and international units commonly use ENBD and ERBD to reduce the risk of postoperative cholangitis and to lower the rate of postoperative pancreatitis[25-27]. Numerous randomized control trial studies have found no significant difference in bile drainage effectiveness or complications between biliary stents and ENBD[28-31], largely due to the fundamental principles of these two methods. ENBD, as an external drainage method, allows for the objective evaluation of drainage effectiveness through bile output volume and composition, facilitating better patient monitoring and management, particularly for thick bile. Furthermore, it reduces the risk of drainage tube blockage and insufficient drainage[32]. However, ENBD has its limitations. Its insertion involves a tube through the nasal passage, which may cause electrolyte imbalances and digestive issues if left in place for extended periods[33]. The tube can cause discomfort in the nasal and pharyngeal regions, local swelling, and hemorrhage, potentially increasing the risk of respiratory infections[33]. Meanwhile, ERBD, a newer drainage method, achieves bile drainage by inserting a biliary stent. It is generally more comfortable than ENBD. During the treatment of common bile duct stones, ERBD may facilitate stone reduction and improve postoperative outcomes by promoting gut nutrition[34]. Additionally, due to its stable design with cedar-like wings at the head end and the absence of side holes, the Christmas tree-shaped plastic stent enhances drainage patency and reduces the risk of retrograde infection. However, most plastic stents in the common bile duct cannot be dislodged independently and must be pulled out under endoscopy, increasing patients’ discomfort and medical costs[35,36].
Cholelithiasis of the common bile duct can lead to obstruction of the hepatic bile duct circulation, causing liver cell injury and surrounding tissue damage, as evidenced by elevated liver function parameters, such as AST, ALT, ALP, GGT, TBIL, and DBIL[37,38]. Our study findings indicated that the postoperative levels of these markers were significantly lower in all three patient groups than preoperative values. However, both the modified pancreatic duct stent drainage group and the standard biliary drainage group had significantly lower levels of AST, ALT, ALP, GGT, TBIL, and DBIL than the nasobiliary drainage group. This indicates that all three drainage methods effectively alleviate intraductal pressure, facilitate bile flow, and reduce liver function impairment and bile metabolism abnormalities, thereby improving hepatobiliary function. Our results were consistent with Mangiavillano et al’s research[39], which showed that modified pancreatic duct plastic stent drainage and standard biliary stent drainage were more effective in reducing liver cell injury and improving liver function in patients with common bile duct stones undergoing ERCP. This may be because placing the tube through the patient’s nostril enables ENBD to reduce bile duct pressure and facilitates adequate drainage. However, prolonged nasal drainage could lead to bile loss and impair digestive function, hindering liver function recovery. Conversely, the biliary plastic stent placement aligns more closely with human anatomy, causing less discomfort to the throat and respiratory system. It maintains bile duct patency, effectively draining the pus and minimizing liver cell damage, which promotes recovery of liver function[39].
Elevated WBC is a traditional diagnostic indicator for bacterial infections; however, it is not a specific toxicological marker, as it can also be elevated due to granulocyte colony-stimulating factors, cytokines, and, in some inflammatory conditions, may even decrease[40]. NEU plays a crucial role in defending against bacteria by engulfing them, and an increased NEU level indicates bacterial infection and heightened inflammation[41]. CRP, an acute-phase response protein, helps clear pathogens and necrotic cells from the body, aiding the immune response. CRP levels significantly rise in response to tissue injury, inflammation, or postsurgery[42]. AMY activity tests are clinically used to diagnose acute pancreatitis and can also assist in differentiating between other acute abdominal conditions, such as abdominal pain, rigidity, nausea, and vomiting[43]. Our study findings showed that after surgery, all three patient groups had significantly lower levels of WBC, Hb, and NEU than before surgery. CRP and AMY serum levels were significantly lower in the modified pancreatic duct stent drainage and standard biliary drainage groups than in the nasobiliary drainage group. This indicates that all groups experienced a trend toward improved inflammatory markers after cholecystectomy. However, compared to ENBD, both the modified pancreatic duct plastic stent and conventional biliary stent drainage were more effective in alleviating inflammation in patients undergoing ERCP for gallstone disease. This may be because biliary drainage induces autophagy in the intestinal mucosa, reducing the expression of inflammatory factors and thus mitigating inflammation[44]. Conversely, ENBD, with its prolonged nasal bile drainage, can lead to bile loss and intestinal mucosa damage, impairing autophagy and hindering the regulation of inflammatory factor expression.
Our study findings indicated that VAS scores in the modified pancreatic duct stent drainage group were significantly lower than those in the standard biliary drainage group, which had lower VAS scores than the nasobiliary drainage group. This indicates that pain symptoms were alleviated in all three patient groups with common bile duct stones following ECRP treatment. However, the modified pancreatic duct plastic stent offered more effective pain relief and improved comfort than the ENBD and standard biliary stents. This finding aligns with the findings of Longo et al[45]. The likely reason is that the modified pancreatic duct plastic stent better aligns with the human physiological structure, reducing nasopharynx stimulation associated with tube retention and minimizing discomfort related to the throat and respiratory system. The expert consensus on accelerated recovery after hepatobiliary and pancreatic surgery (2015 edition) highlights that the basic principle of accelerated recovery surgery is to reduce the stress response[46]. The earlier introduction of oral intake and fluids in the modified pancreatic duct stent group postsurgery aligns with this concept, promoting gastrointestinal function recovery, reducing intestinal stress, and minimizing fluid overload[47]. Additionally, the placement of the pancreatic stent effectively prevents bile reflux into the pancreas, preventing premature activation of pancreatic enzymes, enabling proper pancreatic fluid drainage, lowering intraductal pressure, and reducing pancreatic fluid infiltration into glandular tissue. This helps prevent self-digestion of the pancreas and alleviates patient pain[48].
Our study findings further showed no statistically significant difference in complication rates among the three groups, indicating that the three drainage methods had a similar impact on complications after ECRP for patients with common bile duct stones. This could be due to the modified pancreatic stent having the same diameter as the nasobiliary tube, enabling effective drainage and reducing the risk of early detachment, which could lead to drainage failure. Additionally, our study demonstrated that ERBD facilitated stone removal by causing the stent to move along with intestinal contractions, potentially breaking the stones into smaller pieces. Some stones are even fractured into multiple parts, making them easier to retrieve during subsequent procedures. Additionally, the single pigtail structure at the end of the modified 7Fr pancreatic stent helped prevent proximal displacement and reduced pressure on the intestinal mucosa, reducing the risk of biliary duct or duodenum perforations. By modifying the bile duct end to remove lateral wings, the study showed a higher likelihood of spontaneous detachment, a lower need for manual intervention, and sufficient drainage without the risk of prolonged stent retention, which could cause biliary obstruction, injury, or cholangitis[49].
Malpositioning and occlusion of the stent are common reasons for failed drainage following stent placement. The stent should be positioned within the dilated bile duct, ideally above the stone, with its proximal end extending at least 1 cm above the rim of the stone and its distal end located 1 cm beyond the papilla of Vater. Temporary narrowing of the biliary tract may occur during stent placement, which, while inevitable, is necessary for a smooth procedure. When selecting a biliary stent, it is important to consider factors such as effective drainage, preferably opting for a larger-diameter stent. In cases of recurrent jaundice or cholecystitis, the stent should be promptly removed and replaced with a new stent[50].
This study found that the modified pancreatic duct plastic stent effectively addresses the limitations of ENBD and standard biliary stent implantation, reducing the risk of displacement during drainage. This might be because it could better conform to the physiological structure of the human body. However, patients with this stent had a higher risk of cholangitis, potentially due to its reduced flexibility and the need for endoscopic resection.
This study has several limitations. First, it is a single-center study with a small sample size, a short follow-up period, and potential confounding factors. Second, the timing of laboratory assessments was not strictly controlled, and there was a lack of both short- and long-term data on patients’ satisfaction with the drainage method. These limitations may introduce bias into the findings, highlighting the need for further multi-center, large-sample, prospective, and long-term follow-up studies to validate the results.
In conclusion, the use of modified pancreatic duct plastic stent drainage as an adjunct to post-ERCP treatment for choledochal stones effectively alleviated patients’ pain, did not significantly affect the occurrence of postoperative complications, and demonstrated a higher safety profile. Compared to ENBD and standard biliary stenting, the modified pancreatic duct plastic stent drainage reduced liver cell injury, improved liver function parameters, alleviated inflammation and pain, enhanced comfort, accelerated recovery, and improved quality of life. Its clinical value is substantial, leading to its recommendation for broader application in ERCP treatment for patients with choledochal stones. We suggest that the modified pancreatic duct plastic stent be considered the first choice for post-ERCP drainage in these patients.
| 1. | Zhu SY, Huang J, Li YJ, Zhou B, Zheng CY, Sun DL, Fu Y. Systematic Appraisal of Guidelines for the Diagnosis and Treatment of Choledocholithiasis. Surg Laparosc Endosc Percutan Tech. 2023;33:673-681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 2. | Dasari BV, Tan CJ, Gurusamy KS, Martin DJ, Kirk G, McKie L, Diamond T, Taylor MA. Surgical versus endoscopic treatment of bile duct stones. Cochrane Database Syst Rev. 2013;2013:CD003327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 96] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 3. | Koc B, Karahan S, Adas G, Tutal F, Guven H, Ozsoy A. Comparison of laparoscopic common bile duct exploration and endoscopic retrograde cholangiopancreatography plus laparoscopic cholecystectomy for choledocholithiasis: a prospective randomized study. Am J Surg. 2013;206:457-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 95] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 4. | Almadi MA, Barkun JS, Barkun AN. Management of suspected stones in the common bile duct. CMAJ. 2012;184:884-892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Cai JS, Qiang S, Bao-Bing Y. Advances of recurrent risk factors and management of choledocholithiasis. Scand J Gastroenterol. 2017;52:34-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 6. | An Z, Braseth AL, Sahar N. Acute Cholangitis: Causes, Diagnosis, and Management. Gastroenterol Clin North Am. 2021;50:403-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 7. | Masuda S, Koizumi K, Shionoya K, Jinushi R, Makazu M, Nishino T, Kimura K, Sumida C, Kubota J, Ichita C, Sasaki A, Kobayashi M, Kako M, Haruki U. Comprehensive review on small common bile duct stones. World J Gastroenterol. 2023;29:1955-1968. [PubMed] [DOI] [Full Text] |
| 8. | Fujita N, Yasuda I, Endo I, Isayama H, Iwashita T, Ueki T, Uemura K, Umezawa A, Katanuma A, Katayose Y, Suzuki Y, Shoda J, Tsuyuguchi T, Wakai T, Inui K, Unno M, Takeyama Y, Itoi T, Koike K, Mochida S. Evidence-based clinical practice guidelines for cholelithiasis 2021. J Gastroenterol. 2023;58:801-833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 86] [Article Influence: 28.7] [Reference Citation Analysis (1)] |
| 9. | Zhang J, Li L, Jiang Y, Li W, Li L. Comparative analysis of laparoscopic choledocholithiasis and ERCP treatment after cholecystectomy. BMC Surg. 2023;23:304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Wang P, Li ZS, Liu F, Ren X, Lu NH, Fan ZN, Huang Q, Zhang X, He LP, Sun WS, Zhao Q, Shi RH, Tian ZB, Li YQ, Li W, Zhi FC. Risk factors for ERCP-related complications: a prospective multicenter study. Am J Gastroenterol. 2009;104:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 334] [Article Influence: 19.6] [Reference Citation Analysis (4)] |
| 11. | Shi Y, Lin J, Zhu J, Gao J, Liu L, Yin M, Yu C, Liu X, Wang Y, Xu C. Predicting the Recurrence of Common Bile Duct Stones After ERCP Treatment with Automated Machine Learning Algorithms. Dig Dis Sci. 2023;68:2866-2877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 12. | Zhou J, Chen Y, Yu S, Wang H, Wang Y, Chen Q. Comparison of 1-stage and 2-stage Managements for Common Bile Duct Stones and Gallstones (CBDS): A Retrospective Study. J Clin Gastroenterol. 2024;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (5)] |
| 13. | Liao YJ, Lin WT, Tsai HJ, Chen CC, Tung CF, Yang SS, Peng YC. Critically-Ill Patients with Biliary Obstruction and Cholangitis: Bedside Fluoroscopic-Free Endoscopic Drainage versus Percutaneous Drainage. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 14. | Dumonceau JM, Tringali A, Papanikolaou IS, Blero D, Mangiavillano B, Schmidt A, Vanbiervliet G, Costamagna G, Devière J, García-Cano J, Gyökeres T, Hassan C, Prat F, Siersema PD, van Hooft JE. Endoscopic biliary stenting: indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline - Updated October 2017. Endoscopy. 2018;50:910-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 554] [Article Influence: 69.3] [Reference Citation Analysis (1)] |
| 15. | Zhang GQ, Li Y, Ren YP, Fu NT, Chen HB, Yang JW, Xiao WD. Outcomes of preoperative endoscopic nasobiliary drainage and endoscopic retrograde biliary drainage for malignant distal biliary obstruction prior to pancreaticoduodenectomy. World J Gastroenterol. 2017;23:5386-5394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Park SY, Park CH, Cho SB, Yoon KW, Lee WS, Kim HS, Choi SK, Rew JS. The safety and effectiveness of endoscopic biliary decompression by plastic stent placement in acute suppurative cholangitis compared with nasobiliary drainage. Gastrointest Endosc. 2008;68:1076-1080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 17. | He Q, Wang L, Peng C, Zou X, Zhan Q, Xu Y, Liu Q, Qian J, Gong L, Shen Y, Chen J. Modified prophylactic 5-fr pancreatic duct stent enhances the rate of spontaneous dislodgement: A multicenter randomized controlled trial. United European Gastroenterol J. 2018;6:1519-1526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 18. | Boni L, Huo B, Alberici L, Ricci C, Tsokani S, Mavridis D, Amer YS, Andreou A, Berriman T, Donatelli G, Forbes N, Kapiris S, Kayaalp C, Kylänpää L, Parra-Membrives P, Siersema PD, Black GF, Antoniou SA. EAES rapid guideline: updated systematic review, network meta-analysis, CINeMA and GRADE assessment, and evidence-informed European recommendations on the management of common bile duct stones. Surg Endosc. 2022;36:7863-7876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 19. | Manes G, Paspatis G, Aabakken L, Anderloni A, Arvanitakis M, Ah-Soune P, Barthet M, Domagk D, Dumonceau JM, Gigot JF, Hritz I, Karamanolis G, Laghi A, Mariani A, Paraskeva K, Pohl J, Ponchon T, Swahn F, Ter Steege RWF, Tringali A, Vezakis A, Williams EJ, van Hooft JE. Endoscopic management of common bile duct stones: European Society of Gastrointestinal Endoscopy (ESGE) guideline. Endoscopy. 2019;51:472-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 454] [Article Influence: 64.9] [Reference Citation Analysis (2)] |
| 20. | Irlbeck T, Zwißler B, Bauer A. [ASA classification : Transition in the course of time and depiction in the literature]. Anaesthesist. 2017;66:5-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 21. | Hjermstad MJ, Fayers PM, Haugen DF, Caraceni A, Hanks GW, Loge JH, Fainsinger R, Aass N, Kaasa S; European Palliative Care Research Collaborative (EPCRC). Studies comparing Numerical Rating Scales, Verbal Rating Scales, and Visual Analogue Scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manage. 2011;41:1073-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2131] [Cited by in RCA: 1853] [Article Influence: 123.5] [Reference Citation Analysis (3)] |
| 22. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5667] [Cited by in RCA: 4781] [Article Influence: 367.8] [Reference Citation Analysis (7)] |
| 23. | Balthazar EJ, Robinson DL, Megibow AJ, Ranson JH. Acute pancreatitis: value of CT in establishing prognosis. Radiology. 1990;174:331-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1255] [Cited by in RCA: 958] [Article Influence: 26.6] [Reference Citation Analysis (2)] |
| 24. | Cotton PB, Eisen GM, Aabakken L, Baron TH, Hutter MM, Jacobson BC, Mergener K, Nemcek A Jr, Petersen BT, Petrini JL, Pike IM, Rabeneck L, Romagnuolo J, Vargo JJ. A lexicon for endoscopic adverse events: report of an ASGE workshop. Gastrointest Endosc. 2010;71:446-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2085] [Cited by in RCA: 2028] [Article Influence: 126.8] [Reference Citation Analysis (8)] |
| 25. | Cianci P, Restini E. Management of cholelithiasis with choledocholithiasis: Endoscopic and surgical approaches. World J Gastroenterol. 2021;27:4536-4554. [PubMed] [DOI] [Full Text] |
| 26. | Kim SB, Kim KH, Kim TN. Safety and Efficacy of Endoscopic Retrograde Cholangiopancreatography for Choledocholithiasis in Long-Term Dialysis: A Propensity Score Analysis. Dig Dis Sci. 2018;63:3141-3146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 27. | Park JS, Kim TN, Kim KH. Endoscopic papillary large balloon dilation for treatment of large bile duct stones does not increase the risk of post-procedure pancreatitis. Dig Dis Sci. 2014;59:3092-3098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Zhang W, Che X. Comparison of effect between nasobiliary drainage and biliary stenting in malignant biliary obstruction: a systematic review and updated meta-analysis. World J Surg Oncol. 2020;18:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 29. | Zhang RL, Cheng L, Cai XB, Zhao H, Zhu F, Wan XJ. Comparison of the safety and effectiveness of endoscopic biliary decompression by nasobiliary catheter and plastic stent placement in acute obstructive cholangitis. Swiss Med Wkly. 2013;143:w13823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Lin H, Li S, Liu X. The safety and efficacy of nasobiliary drainage versus biliary stenting in malignant biliary obstruction: A systematic review and meta-analysis. Medicine (Baltimore). 2016;95:e5253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 31. | Ishiwatari H, Kawabata T, Kawashima H, Nakai Y, Miura S, Kato H, Shiomi H, Fujimori N, Ogura T, Inatomi O, Kubota K, Fujisawa T, Takenaka M, Mori H, Noguchi K, Fujii Y, Sugiura T, Ideno N, Nakafusa T, Masamune A, Isayama H, Sasahira N. Endoscopic nasobiliary drainage versus endoscopic biliary stenting for preoperative biliary drainage in patients with malignant hilar biliary obstruction: Propensity score-matched multicenter comparative study. Dig Endosc. 2024;36:726-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 32. | Mi N, Zhang S, Zhu Z, Yu Y, Li W, Zheng L, Chu L, Li J. Randomized Controlled Trial of Modified Nasobiliary Fixation and Drainage Technique. Front Surg. 2022;9:791945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 33. | Yoo KS, Lehman GA. Endoscopic management of biliary ductal stones. Gastroenterol Clin North Am. 2010;39:209-227, viii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 34. | Zhang DY, Zhai YQ, Zhang GJ, Chen SX, Wu L, Chen DX, Li MY. Risk factors for complications in elderly patients aged 85 years and over undergoing endoscopic biliary stone removal. Front Surg. 2022;9:989061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Di Giorgio P, Manes G, Grimaldi E, Schettino M, D'Alessandro A, Di Giorgio A, Giannattasio F. Endoscopic plastic stenting for bile duct stones: stent changing on demand or every 3 months. A prospective comparison study. Endoscopy. 2013;45:1014-1017. [PubMed] [DOI] [Full Text] |
| 36. | Mohammed N, Pinder M, Harris K, Everett SM. Endoscopic biliary stenting in irretrievable common bile duct stones: stent exchange or expectant management-tertiary-centre experience and systematic review. Frontline Gastroenterol. 2016;7:176-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 37. | Bangaru S, Thiele D, Sreenarasimhaiah J, Agrawal D. Severe Elevation of Liver Tests in Choledocholithiasis: An Uncommon Occurrence With Important Clinical Implications. J Clin Gastroenterol. 2017;51:728-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 38. | Yuan WH, Zhang Z, Pan Q, Mao BN, Yuan T. Risk factors for recurrence of common bile duct stones after surgical treatment and effect of ursodeoxycholic acid intervention. World J Gastrointest Surg. 2024;16:103-112. [PubMed] [DOI] [Full Text] |
| 39. | Mangiavillano B, Pagano N, Baron TH, Luigiano C. Outcome of stenting in biliary and pancreatic benign and malignant diseases: A comprehensive review. World J Gastroenterol. 2015;21:9038-9054. [PubMed] [DOI] [Full Text] |
| 40. | Hutzschenreuter F, Monsef I, Kreuzer KA, Engert A, Skoetz N. Granulocyte and granulocyte-macrophage colony stimulating factors for newly diagnosed patients with myelodysplastic syndromes. Cochrane Database Syst Rev. 2016;2:CD009310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 41. | Qian W, Huang GZ. Neutrophil CD64 as a Marker of Bacterial Infection in Acute Exacerbations of Chronic Obstructive Pulmonary Disease. Immunol Invest. 2016;45:490-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 42. | Guo ZM, Wu JH, Li XY, Yang S, Wang GP, Wu SL, Zhang Q. [The association of high-sensitivity C-reactive protein with new-onset hypertension in different age groups]. Zhonghua Xin Xue Guan Bing Za Zhi. 2022;50:993-999. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 43. | Wang L, Qi X, Tian F, Li H, Zhao H, Lv J, Zhou X, Ba D, Song W, Ma X. Diagnostic value of hematological parameters in acute pancreatitis. Ann Palliat Med. 2020;9:2716-2722. [PubMed] [DOI] [Full Text] |
| 44. | Zhang X, Peng B, Zhang Y, Zhang Y, Lu X, Cao Y. Biliary Drainage Reduces Intestinal Barrier Damage in Obstructive Jaundice by Regulating Autophagy. Contrast Media Mol Imaging. 2022;2022:3301330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 45. | Longo F, Panza E, Rocca L, Biffoni B, Lucinato C, Cintoni M, Mele MC, Papa V, Fiorillo C, Quero G, De Sio D, Menghi R, Alfieri S, Langellotti L. Enhanced Recovery After Surgery (ERAS) in Pancreatic Surgery: The Surgeon's Point of View. J Clin Med. 2024;13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 46. | Melloul E, Hübner M, Scott M, Snowden C, Prentis J, Dejong CH, Garden OJ, Farges O, Kokudo N, Vauthey JN, Clavien PA, Demartines N. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations. World J Surg. 2016;40:2425-2440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 424] [Article Influence: 42.4] [Reference Citation Analysis (1)] |
| 47. | Varadhan KK, Neal KR, Dejong CH, Fearon KC, Ljungqvist O, Lobo DN. The enhanced recovery after surgery (ERAS) pathway for patients undergoing major elective open colorectal surgery: a meta-analysis of randomized controlled trials. Clin Nutr. 2010;29:434-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 934] [Cited by in RCA: 802] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 48. | Yi JH, Li ZS, Hu LH. Pancreatic duct stents. J Dig Dis. 2022;23:675-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 49. | Kehlet H, Slim K. The future of fast-track surgery. Br J Surg. 2012;99:1025-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 94] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 50. | Horiuchi A, Nakayama Y, Kajiyama M, Kato N, Kamijima T, Graham DY, Tanaka N. Biliary stenting in the management of large or multiple common bile duct stones. Gastrointest Endosc. 2010;71:1200-1203.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/