Published online Apr 27, 2025. doi: 10.4240/wjgs.v17.i4.100555
Revised: January 7, 2025
Accepted: February 5, 2025
Published online: April 27, 2025
Processing time: 221 Days and 8.1 Hours
Clostridium difficile infection (CDI) is common in patients with inflammatory bowel disease (IBD).
To assess the association of CDI with clinical outcomes of IBD.
PubMed, EMBASE, Web of Science, and the Cochrane Library databases were searched from inception to March 2024. Eligible articles included observational studies that reported on outcomes such as mortality, colectomy, hospitalization, intensive care unit (ICU) admission, complication rates, and length of hospital stay in IBD patients with and without CDI. Data were extracted, and a random-effects model was used to calculate pooled odds ratios (ORs) and mean diffe
As shown in the data from 21 studies with 1249158 participants, CDI significantly increased the risk of mortality in IBD patients [pooled OR = 4.569, 95% confidence intervals (95%CI): 2.584 to 8.079]. Although the pooled OR for colectomy was 1.409 (95%CI: 0.922 to 2.155), it was not statistically significant. Similarly, CDI did not impact hospitalization (pooled OR = 1.056, 95%CI: 0.512 to 2.179) and ICU admission outcomes (pooled OR = 1.970, 95%CI: 0.420 to 9.246) of patients with IBD. The rate of complications was comparable in the two groups (pooled OR = 0.658, 95%CI: 0.378 to 1.147). However, CDI was associated with a significantly more extended hospital stay (pooled MD = 0.349 days, 95%CI: 0.002 to 0.696).
CDI is linked to increased mortality and prolonged hospitalization in IBD patients. These results emphasize the need for early detection and appropriate management. Implementing routine CDI screening during IBD flare-ups and stringent infection control measures could mitigate severe complications and reduce the healthcare burden.
Core Tip: Clostridium difficile infection (CDI) significantly complicates the management of patients with inflammatory bowel disease (IBD), exacerbating disease severity and leading to worse clinical outcomes. This systematic review and meta-analysis demonstrated the association of CDI with increased mortality and prolonged hospital stays in IBD patients. While CDI was not significantly linked to colectomy or other complications, its impact on healthcare resource utilization underscores the need for routine screening, timely treatment, and robust infection control measures. These findings provide critical insights into optimizing care strategies and reducing the burden of CDI in IBD management.
- Citation: Qi HX, Wang Q, Zhou GQ. Association of Clostridium difficile infection with clinical outcomes of patients with inflammatory bowel disease: A meta-analysis. World J Gastrointest Surg 2025; 17(4): 100555
- URL: https://www.wjgnet.com/1948-9366/full/v17/i4/100555.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i4.100555
Inflammatory bowel disease (IBD), which includes Crohn's disease (CD) and ulcerative colitis (UC), is a chronic inflammatory gastrointestinal condition[1] which significantly impacts the quality of life of patients and is associated with a substantial burden on the healthcare system[2].
Despite recent advances in disease management, IBD patients are still at increased risk for various complications, such as strictures, fistulas, and abscesses, which often require surgical intervention and increase morbidity and mortality[3,4]. Colectomy, a surgical procedure to remove part or all of the colon, is a common outcome for patients with severe or re
One of the significant infectious complications of IBD is Clostridium difficile infection (CDI)[6,7], a primary cause of healthcare-associated infections that is particularly prevalent in patients with compromised gut integrity. Research shows that CDI exacerbates the underlying IBD and leads to a more severe disease course[8], higher rates of morbidity, prolonged hospital stays, increased healthcare costs, and greater mortality[9]. Furthermore, exacerbation of IBD symptoms by CDI can also lead to more frequent and severe flare-ups, often resulting in increased rates of colectomy and other surgical interventions[10]. CDI can also complicate the medical management of patients with IBD, limiting therapeutic options and necessitating more aggressive treatment[6,7].
Despite the clear clinical implications, there is still no comprehensive evidence of the full spectrum of CDI-associated outcomes in IBD patients. Previous reviews have primarily focused on individual outcomes, such as colectomy rates[11-14]. However, the association of CDI with a broader range of critical outcomes, including mortality, overall complications, and hospitalization rates in IBD patients, is still unclear.
Nurses are crucial in managing IBD patients, particularly in monitoring symptoms, administering treatments, and providing patient education. Understanding the link between CDI and clinical outcomes of IBD will help nurses develop more effective care plans and improve patient outcomes. This study aimed to systematically evaluate the association of CDI with a wide array of clinical outcomes, including mortality, colectomy rates, complications, and hospitalization outcomes, in patients with IBD.
This study protocol was registered in PROSPERO (CRD42024547255).
The review included studies assessing the association of CDI with outcomes in patients with IBD. The inclusion criteria were as follows: (1) Studies of adult IBD patients (aged 18 years and older), including both CD and UC cases; (2) Studies reporting the association of CDI with clinical outcomes such as mortality, colectomy rates, complications (defined as clinical events related to IBD or its treatment, including bowel obstruction, strictures, fistula, abscess formation, or infections such as sepsis and toxic megacolon), and hospitalization outcomes; (3) Studies providing clear data on the presence and absence of CDI in IBD patients; and (4) Cohort, cross-sectional, and case-control peer-reviewed studies.
The exclusion criteria were as follows: (1) Studies not reporting relevant outcomes specific to CDI in IBD patients; (2) Studies in pediatric populations (younger than 18 years); (3) Case reports, case series, editorials, commentaries, or not peer-reviewed studies; (4) Abstracts or conference presentations.
A systematic search was conducted in PubMed (https://pubmed.ncbi.nlm.nih.gov), EMBASE (https://www.embase.com), Web of Science (https://www.webofscience.com), and the Cochrane Library (https://www.cochranelibrary.com) for studies from inception until March 2024. Additionally, bibliography sections of identified studies were manually searched. The final literature search was performed on May 31, 2024.
The search strategy used free text words and Medical Subject Headings terms. The search terms used included "inflammatory bowel disease", "colitis, ulcerative", "Crohn’s disease", "inflammatory bowel", "IBD", "ulcerative colitis", "Crohn’s", "Clostridium difficile", and "C. difficile". Boolean operators (AND/OR) were used to refine search results. Reference sections of recent reviews and publications from major gastroenterology conferences were also screened. Additionally, authors of identified studies were contacted for further data or clarification of study methodologies when necessary. A detailed search strategy for different databases is shown in Supplementary material.
Two independent reviewers assessed the titles and abstracts of identified papers and reviewed full texts of potentially relevant studies. Any discrepancies regarding study eligibility were resolved by consensus or consulting with a third senior reviewer. Rayyan software was used to identify and remove duplicate reports while maintaining a clear and auditable trail of the review process[15].
Data were independently extracted by two reviewers using a standardized form designed for this study and pre-tested on a subset of studies to ensure its effectiveness in capturing all pertinent data points clearly and concisely. The data collected included study characteristics, patient characteristics, method of CDI diagnosis, and outcome assessment. The reviewers cross-checked each other’s data entries to correct discrepancies and confirm data integrity. In cases where data were unclear or missing, study authors were contacted to provide further details or clarification to ensure the completeness and accuracy of the data set.
The primary outcomes of interest were mortality, colectomy rates, complications, and hospitalization outcomes in patients with IBD who also had CDI. Mortality was defined as death occurring within specified follow-up periods, including in-hospital mortality and long-term mortality. Colectomy rates referred to the percentage of patients undergoing surgical removal of part or all of the colon. Complications included any reported adverse events such as bowel obstruction, strictures, fistula, or infection. Hospitalization outcomes included the length of hospital stay, readmission rates, and the need for intensive care. Data for all measures, time points, and analyses provided in the studies were collected. In cases where multiple time points or analyses were reported, priority was given to the most clinically relevant and commonly reported time points, such as immediate postoperative outcomes and follow-ups at one year.
In addition to the primary outcomes, data on various other variables was collected to provide a comprehensive understanding of the study populations and methodologies. These included: (1) Patient characteristics such as age, gender, IBD type (UC or CD), hospitalization status (inpatient or outpatient), and comorbidity scores; (2) Study characteristics, including the primary author, year of publication, time period of the study, geographic location, study design (cohort, cross-sectional, or case-control), and whether the study was published as a full paper or abstract; (3) Details of CDI diagnosis methods (e.g., enzyme-linked immunosorbent assay, enzyme immunoassay, cytotoxicity assay, and stool polymerase chain reaction); (4) Intervention characteristics, which provided specific details of treatments and interventions used for managing IBD and CDI; and (5) Information regarding funding sources and potential conflicts of interest (to account for the influence of financial biases on study outcomes).
The Newcastle-Ottawa Scale (NOS)[16] was used to assess the risk of bias. The NOS evaluates studies based on three key domains: Selection of study groups (maximum of 4 points), comparability of groups (maximum of 2 points), and ascertainment of the exposure or outcome of interest for case-control or cohort studies, respectively (maximum of 3 points). Each study was carefully rated on these domains, with a maximum possible score of 9 points. Studies scoring 7-9 were categorized as having low risk of bias, 5–6 as having moderate risk, and below 5 as having high risk of bias.
Two independent reviewers conducted the evaluations to ensure a rigorous and reliable assessment process. Pre-specified guidelines were used to assign scores for each domain, ensuring consistency in applying NOS criteria across studies. Any disagreements between reviewers were documented and discussed in detail. A third senior reviewer was consulted to resolve the discord if a consensus could not be reached. This triangulated approach ensured objectivity and minimized the potential for bias during the quality assessment process.
Effect measures used in this meta-analysis included odds ratios (ORs) with 95% confidence intervals (95%CIs) for dichotomous outcomes, such as mortality, and mean differences (MDs) for continuous outcomes, such as the length of hospital stay. Studies were tabulated based on intervention characteristics to determine eligibility for synthesis and compared against predefined criteria. Data preparation involved addressing missing summary statistics and converting data to ensure consistency. Results from individual studies and syntheses were tabulated and visually displayed using forest plots. A random-effects model was employed to synthesize results due to anticipated heterogeneity, assessed by the I² statistic. Meta-analyses were conducted using Stata version 17[17]. The funnel plot was used for publication bias assessment. Sensitivity analysis by excluding studies, one at a time, was done to identify any potential outliers or influential studies that could disproportionately affect the meta-analysis. Meta-regression was performed to explore the sources of heterogeneity for outcomes with at least 10 studies. The factors assessed for meta-regression were the sample size, year of publication, age, gender distribution, IBD duration, and behavioral habits like smoking and alcohol use. A P value less than 0.05 indicates that the factor significantly contribute to the heterogeneity and adjusted R-squared value was used to explain the extent of heterogeneity explained by the factor.
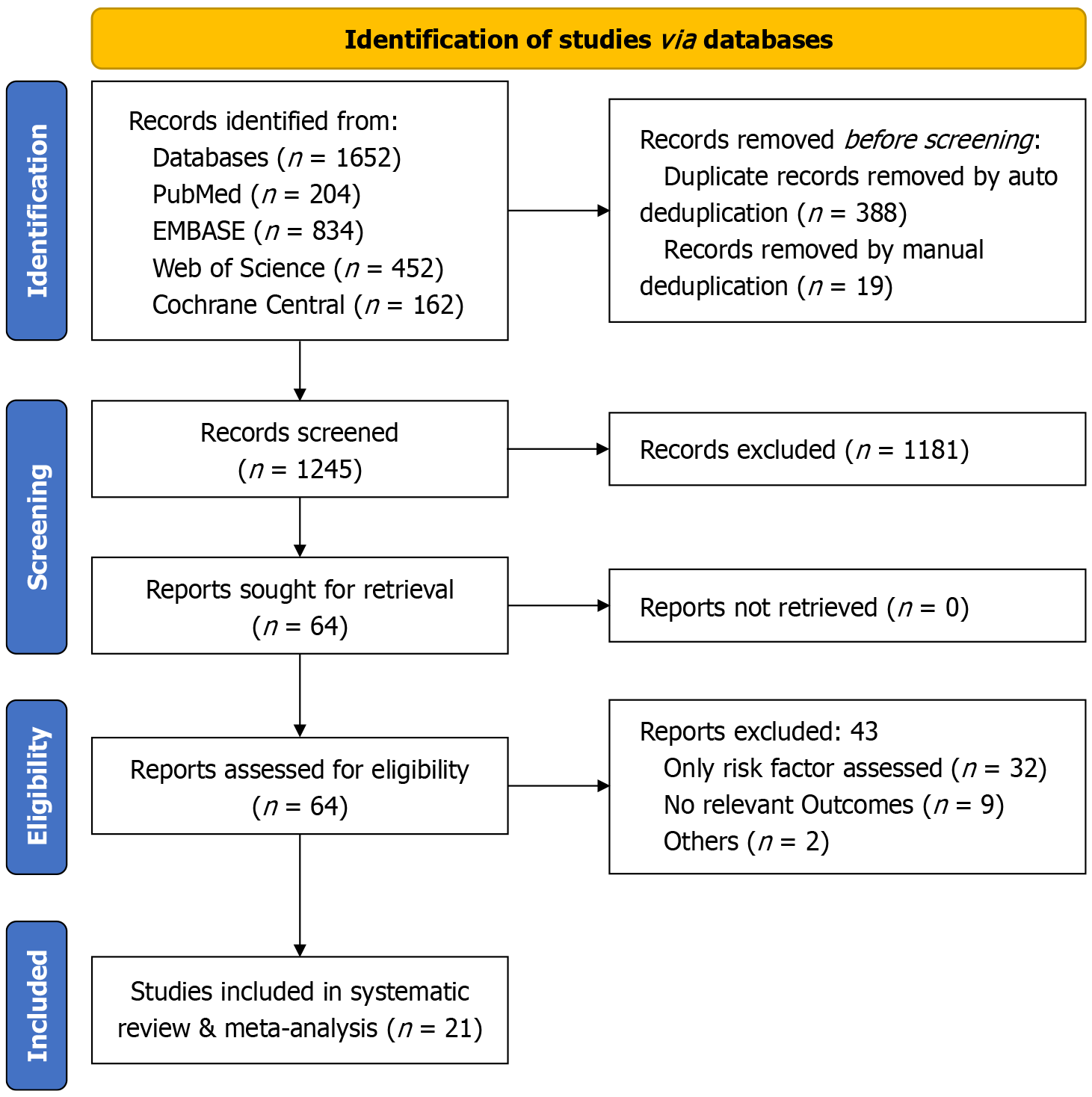
The initial pool of studies contained 1652 records that were retrieved from PubMed (n = 204), Embase (n = 834), Web of Science (n = 452), and Cochrane Central (n = 162) databases. Following an auto-deduplication process, 388 records were removed, and further 19 duplicates were removed manually. Of the remaining 1245 records subjected to the primary title and abstract screening, 1181 were excluded. Subsequently, 64 reports were assessed for eligibility. Of them, 44 were eliminated. The reasons for elimination were as follows: 32 studies assessed only risk factors, 9 did not report relevant outcomes, and 2 were excluded for other reasons. Ultimately, 21 papers were included in the analysis (Figure 1)[18-38].
This review encompassed a diverse range of study designs and geographic locations (Table 1). The studies were published between 2009 and 2023 in the United States, China, Canada, Saudi Arabia, Brazil, South Korea, and Israel. The sample sizes of the studies varied significantly, ranging from 66 to 562000 participants. The number of CDI-positive cases ranged from 8 to 3832 across the studies, and the median age of the cohort ranged from 24 [interquartile range (IQR): 19-33] to 49 (IQR: 34-62) years. The percentage of male patients ranged from 36.3% to 77.1%.
| Ref. | Country | Study design | Sample size | CDI positive cases | CDI negative cases | Age (mean, SD; median, IQR; range), years | Gender (% male) | IBD type | IBD duration (years) | Smoking (% of current smokers) | Location of IBD | IBD severity (remission, mild, moderate, severe) | Co-morbidities | Risk of bias assessment (NOS score) |
| AlKhormi et al[18], 2023 | Saudi Arabia | Case-control study | 95 | 16 | 79 | Median: 24 (19-33) | 47.4 | Both CD and UC | 6 | 11.7 | Colon-dominant CD-10, ileum-dominant CD-55, CD unclassified-3 | - | Diabetes: 9.4, hypertension: 8.3 | 5 |
| Chen et al[19], 2019 | China | Prospective observational cohort study | 230 | 32 | 198 | Mean: 37.7 (14.3) | 66.5 | Both CD and UC | - | - | - | Remission: 3.9, mild: 34.3, moderate: 47.3, severe: 14.3 | - | 5 |
| Garcia et al[20], 2018 | Brazil | Prospective cohort study | 66 | 19 | 47 | Mean: 40.95 (13.2) | 36.3 | Both CD and UC | 6 | 9.1 | Extensive-36.8, left-sided UC-21.1, colonic/ileocolonic CD-42.1 | Mild-to-moderate: 84.2, severe: 15.8 | - | 7 |
| Gros et al[21], 2023 | Spain | Retrospective case-control study | 105 | 35 | 70 | Median: 48 (28-65) | 77.1 | Both CD and UC | - | 11.4 | - | - | Diabetes: 6.6 | 7 |
| Joshi et al[22], 2017 | United Kingdom | Retrospective cohort study | 94 | 47 | 47 | - | - | Both CD and UC | - | - | - | - | - | 7 |
| Kariv et al[23], 2011 | United States | Retrospective case-control study | 78 | 39 | 39 | Median: 39 (33–55) | UC | 7.6 | 30.7 | - | - | Diabetes: 5.12; other comorbidities: 27.8 | 5 | |
| Kim et al[24], 2018 | Korea | Prospective multicentre observational study | 81 | 8 | 73 | Mean: 39.6 (14.5) | 49.3 | UC | 3.4 | - | - | - | - | 3 |
| Li et al[25], 2018 | China | Case-control retrospective study | 197 | 60 | 137 | Median: 38.0 (28.0-49.0) | 60.9 | Both CD and UC | 12 | 16.2 | Proctitis-1, left-sided colitis-24.5, extensive colitis-74.5 | Mild: 16.7, moderate: 44.1, severe: 39.2 | Diabetes: 6.6 | 7 |
| Maharshak et al[26], 2018 | Israel | Retrospective case-control study | 383 | 28 | 355 | Median: 38.1 (30-60) | 42.8 | Both CD and UC | 6 | 20.1 | Small intestine-11.8, colon-29.4, ileocolonic-58.8 | - | Charlson comorbidity index ≥ 2 = 35.7% | 5 |
| Samman et al[27], 2023 | Saudi Arabia | Retrospective cohort study | 602 | 53 | 549 | Mean: 36.58 (14.03) | 48.8 | Both CD and UC | - | 2.4 | - | - | - | 7 |
| Xu et al[28], 2019 | China | Retrospective cohort study | 102 | 34 | 68 | Median: 44.5 (29.0-60.0) | 47.1 | Both CD and UC | 12.7 | - | Remission: 16.2, mild: 45.1, moderate: 38.2 | Diabetes: 9.5 | 5 | |
| Gu et al[35], 2017 | China | Retrospective cohort study | 260 | 13 | 247 | Mean: 35.7 (16.2) | 56.1 | Both CD and UC | - | - | Ileum-34.6, colon-38.8, ileocolon-26.5, upper GI involvement-1.92 | Remission: 15.7, mild: 50, moderate: 34.3 | - | 3 |
| Zhang et al[29], 2016 | China | Retrospective cohort study | 646 | 99 | 547 | Median: 33 (16-76) | 63.7 | Both CD and UC | - | - | - | Remission: 47.9, mild: 36.2, severe: 15.7 | - | 5 |
| Bernard et al[30], 2022 | United States | Retrospective cohort study | 300 | 161 | 139 | Median: 49 (34–62) | 47.7 | Both CD and UC | 9.8 | 16.6 | - | - | - | 7 |
| Negrón et al[31], 2016 | Canada | Cohort study | 1754 | 81 | 1673 | Median: 41 | 50.4 | Both CD and UC | - | - | - | - | - | 7 |
| Rezapour et al[32], 2018 | United States | Retrospective cohort study | 224500 | 3832 | 220668 | Mean: 45 (20.7) | 47 | Both CD and UC | - | - | - | - | - | 5 |
| Saffouri et al[33], 2017 | United States | Prospective cohort study | 562000 | 20798 | 541202 | 47.1 | Both CD and UC | - | - | - | - | - | 5 | |
| Anderson et al[34], 2017 | United States | Propensity scores matched analysis | 198 | 66 | 192 | Mean: 45.4 (15.2) | 43.7 | Both CD and UC | - | - | Ileal-26.5, colonic-31.6, ileocolonic-44.9, upper GI-4.1 | - | - | 5 |
| Navaneethan et al[36], 2012 | United States | Prospective cohort study | 146 | 45 | 101 | Mean: 45.9 (17.2) | 65 | Both CD and UC | 2.3 | - | Rectum-4, left sided-11.5, extensive-84.4 | - | - | 5 |
| Murthy et al[37], 2012 | Canada | Retrospective cohort study | 181 | 1835 | 47 | UC | 2 | - | - | - | - | 7 | ||
| Jodorkovsky et al[38], 2010 | United States | Prospective cohort study | 99 | 47 | 52 | 52.4 | UC | - | - | - | - | - | 5 |
The studies included both CD and UC patients, with 100% of the studies focusing on both conditions simultaneously. Smoking and alcohol use were also documented, although not consistently across all studies. According to studies that reported on the specific segments of the gastrointestinal tract affected, the location of IBD involvement varied. The prevalence of colon-dominant disease ranged from 10% to 55%, and that of ileum-dominant disease from 26.5% to 55%. The severity of IBD was reported in terms of remission, mild, moderate, and severe cases, with some studies providing detailed percentages for each category, with the reported remission rates of 47.9%, mild IBD rates of 50%, moderate IBD rates of 47.3%, and severe IBD rates of 39.2%.
The meta-analysis of mortality outcomes included data from ten studies (Figure 2A). The pooled OR for mortality of patients with IBD and CDI was 4.569 (95%CI: 2.584 to 8.079), indicating a significantly increased risk of mortality in this patient population compared to patients without CDI, with substantial heterogeneity (I² = 91.19%). The Egger test for small-study effects yielded a beta coefficient of -0.75 [standard error (SE): 0.570, P = 0.1879], indicating no significant evidence of publication bias (Supplementary Figure 1A). Meta-regression showed that the sample size was the only significant factor, explaining 90.15% of the total heterogeneity. Although the association of other factors with mortality was non-significant, IBD duration could explain 53.92% of heterogeneity and the year of publication 17.7% of heterogeneity. Sensitivity analysis revealed that the effect sizes did not differ significantly in magnitude/direction after excluding any single study.
The meta-analysis that examined the need for colectomy included 17 studies, with a pooled OR of 1.409 (95%CI: 0.922 to 2.155; Figure 2B). These results indicated a slightly but not significantly elevated risk of requiring colectomy in IBD patients with CDI compared to patients without, with substantial heterogeneity (I² = 84.44%). The Egger test for small-study effects showed a beta coefficient of -0.74 (SE: 0.802, P = 0.3551), indicating no significant publication bias (Supplementary Figure 1B). Meta-regression results indicated that none of the factors showed a significant contribution to the heterogeneity. However, the sample size explained 20.4%, and gender distribution explained 21.5% of the total heterogeneity. Sensitivity analysis revealed that the effect sizes did not differ significantly in magnitude/direction after excluding any single study.
Nine studies reported hospitalization outcomes (Figure 2C). CDI had no significant association with hospitalization risks, with a pooled OR of 1.056 (95%CI: 0.512 to 2.179) and substantial heterogeneity (I² = 82.21%). The Egger test for small-study effects showed a beta coefficient of 0.88 (SE: 1.312, P = 0.5014), indicating no significant evidence of publication bias (Supplementary Figure 1C). Sensitivity analysis revealed that the effect sizes did not differ significantly in terms of magnitude/direction after excluding any single study.
The meta-analysis that examined ICU admission outcomes included four studies, with a pooled OR of 1.970 (95%CI: 0.420 to 9.246), which indicated no significant difference in the risk of ICU admission in patients with or without CDI (Figure 2D). The Egger test for small-study effects showed a beta coefficient of -0.63 (SE: 1.764, P = 0.7197), indicating no significant evidence of publication bias. Sensitivity analysis revealed that the effect sizes did not differ significantly in terms of magnitude/direction after excluding any single study.
The meta-analysis on complication rates incorporated data from three studies (Figure 2E). The pooled OR for complications in patients with IBD and CDI was 0.658 (95%CI: 0.378 to 1.147), suggesting a comparable incidence of complications in the two groups. The Egger test for small-study effects yielded a beta coefficient of 1.67 (SE: 5.161, P = 0.7462), indicating the lack of publication bias. Sensitivity analysis revealed that the effect sizes did not differ significantly in terms of magnitude/direction after excluding any single study.
The meta-analysis that evaluated the mean duration of hospitalization included data from eight studies, with a pooled MD of 0.349 days (95%CI: 0.002 to 0.696; Figure 2F). The result showed that CDI was associated with considerably longer hospital stays in IBD patients. The included studies had extremely high heterogeneity (I² of 99.53%). The Egger test for small-study effects revealed a beta coefficient of -2.34 (SE: 1.485, P = 0.1156), indicating no significant evidence of publication bias (Supplementary Figure 1D). Sensitivity analysis revealed that the effect sizes did not differ significantly in terms of magnitude/direction after excluding any single study.
Our study demonstrated a link between CDI and significantly increased mortality and extended hospital stays in IBD patients.
The study showed that CDI positivity in patients with IBD was associated with significantly increased mortality, with a pooled OR of 4.569 (95%CI: 2.584 to 8.079). This finding confirms the results reported by Tariq et al[13] in 2019, who also observed a heightened mortality risk among IBD patients with CDI (OR = 4.39; 95%CI: 3.56-5.42), particularly in those with UC. The increased mortality risk is attributed to the severe complications associated with CDI, such as toxic megacolon, sepsis, and multi-organ failure[39]. In IBD patients, the compromised intestinal mucosal barrier and the use of immunosuppressive therapies exacerbate these complications, leading to worse clinical outcomes and increased mortality[40]. Our study found a somewhat higher risk of needing colectomy in IBD patients with CDI, with a pooled OR of 1.409 (95%CI: 0.922 to 2.155). However, the lack of statistical significance suggests a potential but inconclusive association. This contrasts with the finding from Chen et al[11] in 2017, who reported a significant increase in colectomy risk (OR = 1.90; 95%CI: 1.23-2.93) among UC patients with CDI. The discrepancy might be due to different study populations or diagnostic criteria for CDI, or variations in medical and surgical management across studies. Additionally, it is plausible that the presence of CDI further exacerbates colonic inflammation and damage, which can lead to treatment-refractory disease and the subsequent need for surgical intervention.
Our study found no significant difference in hospitalization rates between IBD patients with and without CDI, with a pooled OR of 1.056 (95%CI: 0.512 to 2.179). This finding aligns with a study by Law et al[12] in 2017, who also reported no significant increase in short-term hospitalization rates in patients with CDI (OR = 1.35; 95%CI: 0.68-2.67). While the immediate hospitalization rates may not differ significantly, the prolonged duration of hospital stays in CDI patients, as evidenced by our pooled MD of 0.349 days (95%CI: 0.002 to 0.696), underscores the substantial healthcare burden and increased resource use associated with managing CDI in IBD patients. Such prolonged hospitalization can be attributed to the need for more intensive medical management and monitoring of CDI complications.
Our study reported comparable rates of ICU admission and complications in IBD patients with and without CD [a pooled OR of 1.970 (95%CI: 0.420 to 9.246) and 0.658 (95%CI: 0.378 to 1.147), respectively]. These results are in line with the findings of Law et al[12] in 2017. The lack of significant differences may suggest that while CDI contributes to prolonged hospital stays, it does not necessarily increase the immediate severity of the disease to the extent that it would require ICU care or lead to additional complications. Early detection and effective management of CDI, therefore, may mitigate the need for more extensive care levels despite the overall increased burden of the disease.
Research shows that CDI exacerbates IBD outcomes through a combination of toxin-mediated and immune response mechanisms. CDI toxins A and B cause direct cytotoxic effects on colonic epithelial cells, disrupting the mucosal barrier and inducing inflammation[41]. This disruption worsens the underlying pathology of IBD, characterized by a compromised intestinal barrier and dysregulated immune response[42]. The inflammatory cascade triggered by CDI toxins includes upregulation of pro-inflammatory cytokines (IL-1β, IL-6, and tumor necrosis factor-alpha) that may further aggravate mucosal inflammation and damage. Patients with IBD, who often undergo immunosuppressive therapies, have weakened immunity and ability to combat the infection, leading to a prolonged and more severe course of the disease[43]. These mechanisms have profound clinical significance, as they further emphasize the need for aggressive and timely management of CDI in IBD patients. Early intervention with appropriate antibiotics, consideration of immunomodulatory therapy adjustments, and close monitoring for complications are all essential strategies to mitigate the adverse impacts of CDI on this vulnerable population. Nurses are pivotal in implementing these strategies, as they are often the first to detect early signs of CDI in IBD patients and initiate timely interventions. They can significantly reduce the risk of severe outcomes by conducting routine assessments, educating patients on recognizing symptoms, and ensuring their adherence to treatment plans. Furthermore, nursing interventions focused on infection control practices and patient education play a crucial role in minimizing the transmission of CDI within healthcare settings.
This study has several strengths, including a comprehensive systematic review and meta-analysis that integrates data from a diverse range of studies, enhancing the generalizability of our findings. The use of rigorous inclusion criteria and standardized methods for data extraction and analysis ensures the reliability and validity of the results. Additionally, a large, pooled sample size provides robust statistical power to detect significant associations.
However, our study has limitations. The detected heterogeneity in study design, population characteristics, and diagnostic criteria for CDI may have affected the consistency of the findings. Furthermore, the reliance on observational studies may have introduced bias, as some confounding factors were not adequately controlled for in the primary studies. We could not perform subgroup analyses comparing outcomes between UC and CD due to the lack of stratified data in the included studies. Lastly, despite efforts to include unpublished data and grey literature, the potential for publication bias cannot be entirely ruled out. Therefore, our results and their implications for clinical practice must be considered cautiously.
Early detection and timely management are vital for improving outcomes in IBD patients with CDI. Delayed therapy has been shown to result in worsened outcomes, emphasizing the importance of implementing specific strategies to address this issue. For instance, the routine administration of stool testing for Clostridium difficile toxins or PCR-based diagnostic tests should be integrated into standard care protocols for IBD patients, particularly during flare-ups or hospital admissions. Early identification can enable the prompt initiation of evidence-based treatments, such as oral vancomycin or fidaxomicin, which have demonstrated superior efficacy in reducing morbidity and mortality in CDI patients. Additionally, stringent infection control measures, including isolation of infected patients, hand hygiene adherence, and environmental disinfection, are critical to limiting the spread of CDI, especially in immunocompromised IBD populations. Incorporating antimicrobial stewardship programs can further optimize the use of antibiotics, reducing the risk of recurrent CDI and minimizing complications.
Our findings suggest that routine CDI screening protocols should be systematically implemented during IBD exacerbations or before initiating immunosuppressive therapies, given the elevated risk of CDI in these scenarios. For instance, targeted screening during hospital admissions or in cases of symptoms such as diarrhea and abdominal pain can facilitate early diagnosis and mitigate severe outcomes. Furthermore, a multidisciplinary approach involving gastroenterologists, infectious disease specialists, and clinical pharmacists is essential to developing personalized management plans that address the unique challenges of CDI in IBD patients. These recommendations underscore the need for proactive and integrated clinical strategies to reduce CDI-related mortality, complications, and healthcare burden. By emphasizing early detection, effective antimicrobial therapy, and robust infection control measures, clinicians can significantly improve outcomes and quality of care for this vulnerable patient population.
Future research should focus on exploring the differential impact of CDI on CD and UC through well-designed, stratified studies. Longitudinal research is needed to evaluate the effectiveness of routine CDI screening and antimicrobial stewardship programs in improving outcomes in IBD patients. Additionally, studies assessing patient-centered outcomes, such as quality of life and healthcare costs, can provide a more comprehensive understanding of the burden of CDI in IBD.
This study emphasizes the significant association of CDI with mortality in patients with IBD, underscoring the critical need for early detection and targeted treatment. While the association between CDI and colectomy or hospitalization was less conclusive, the extended hospital stays observed in CDI patients are indicative of the increased healthcare burden. The results also underscore the importance of preventive measures, including judicious antibiotic use and infection control practices. Implementing routine CDI screening and prompt management in IBD care can improve patient outcomes and reduce complications.
| 1. | Padoan A, Musso G, Contran N, Basso D. Inflammation, Autoinflammation and Autoimmunity in Inflammatory Bowel Diseases. Curr Issues Mol Biol. 2023;45:5534-5557. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (8)] |
| 2. | GBD 2017 Inflammatory Bowel Disease Collaborators. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5:17-30. |
| 3. | Pal P, Kanaganti S, Banerjee R, Ramchandani M, Nabi Z, Reddy DN, Tandan M. Systematic Review of Endoscopic Management of Stricture, Fistula and Abscess in Inflammatory Bowel Disease. Gastroenterol Insights. 2023;14:45-63. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 4. | Kaur M, Dalal RL, Shaffer S, Schwartz DA, Rubin DT. Inpatient Management of Inflammatory Bowel Disease-Related Complications. Clin Gastroenterol Hepatol. 2020;18:1346-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 5. | Burns L, Kelly ME, Whelan M, O'Riordan J, Neary P, Kavanagh DO. A contemporary series of surgical outcomes following subtotal colectomy and/or completion proctectomy for management of inflammatory bowel disease. Ir J Med Sci. 2022;191:2705-2710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 6. | Sehgal K, Yadav D, Khanna S. The interplay of Clostridioides difficile infection and inflammatory bowel disease. Therap Adv Gastroenterol. 2021;14:17562848211020285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Rodríguez C, Romero E, Garrido-Sanchez L, Alcaín-Martínez G, Andrade RJ, Taminiau B, Daube G, García-Fuentes E. Microbiota insights in Clostridium difficile infection and inflammatory bowel disease. Gut Microbes. 2020;12:1725220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 8. | Axelrad JE, Cadwell KH, Colombel JF, Shah SC. The role of gastrointestinal pathogens in inflammatory bowel disease: a systematic review. Therap Adv Gastroenterol. 2021;14:17562848211004493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 9. | Boeriu A, Roman A, Fofiu C, Dobru D. The Current Knowledge on Clostridioides difficile Infection in Patients with Inflammatory Bowel Diseases. Pathogens. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 10. | Del Vecchio LE, Fiorani M, Tohumcu E, Bibbò S, Porcari S, Mele MC, Pizzoferrato M, Gasbarrini A, Cammarota G, Ianiro G. Risk Factors, Diagnosis, and Management of Clostridioides difficile Infection in Patients with Inflammatory Bowel Disease. Microorganisms. 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 11. | Chen Y, Furuya-Kanamori L, Doi SA, Ananthakrishnan AN, Kirk M. Clostridium difficile Infection and Risk of Colectomy in Patients with Inflammatory Bowel Disease: A Bias-adjusted Meta-analysis. Inflamm Bowel Dis. 2017;23:200-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Law CC, Tariq R, Khanna S, Murthy S, McCurdy JD. Systematic review with meta-analysis: the impact of Clostridium difficile infection on the short- and long-term risks of colectomy in inflammatory bowel disease. Aliment Pharmacol Ther. 2017;45:1011-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 13. | Tariq R, Law CCY, Khanna S, Murthy S, McCurdy JD. The Impact of Clostridium difficile Infection on Mortality in Patients With Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. J Clin Gastroenterol. 2019;53:127-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 14. | Peng JC, Shen J, Zhu Q, Ran ZH. The impact of Clostridum difficile on surgical rate among ulcerative colitis patients: A systemic review and meta-analysis. Saudi J Gastroenterol. 2015;21:208-212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18843] [Cited by in RCA: 15315] [Article Influence: 1531.5] [Reference Citation Analysis (6)] |
| 16. | Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. [cited 13 May 2024]. Available from: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp#:~:text=The%20Newcastle-Ottawa%20Scale%20%28NOS%29%20is%20an%20ongoing%20collaboration,the%20Universities%20of%20Newcastle%2C%20Australia%20and%20Ottawa%2C%20Canada. |
| 17. | Stata. Stata 17 released. [cited 13 May 2024]. Available from: https://blog.stata.com/2021/04/20/stata-17-released/. |
| 18. | AlKhormi A, Altheyabi AM, AlGhamdi SA, Alshahrani O, Alotay AA, Deeb A. The prevalence and clinical characteristics of Clostridium difficile infection in Saudi patients admitted with inflammatory bowel disease: A case-control study. Saudi J Gastroenterol. 2023;29:251-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Chen XL, Deng J, Chen X, Wan SS, Wang Y, Cao Q. High incidence and morbidity of Clostridium difficile infection among hospitalized patients with inflammatory bowel disease: A prospective observational cohort study. J Dig Dis. 2019;20:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 20. | Garcia PG, Chebli LA, da Rocha Ribeiro TC, Gaburri PD, de Lima Pace FH, Barbosa KVBD, Costa LA, de Almeida Cruz W, de Assis IC, Moraes BRM, Zanini A, Chebli JMF. Impact of superimposed Clostridium difficile infection in Crohn's or ulcerative colitis flares in the outpatient setting. Int J Colorectal Dis. 2018;33:1285-1294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 21. | Gros B, Soto P, Causse M, Marín S, Iglesias E, Benítez JM. Impact of Clostridioides difficile infection in patients admitted with ulcerative colitis. Scand J Gastroenterol. 2023;58:232-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 22. | Joshi NM, Marks IH, Crowson R, Ball D, Rampton DS. Incidence and Outcome of Clostridium difficile Infection in Hospitalized Patients with Inflammatory Bowel Disease in the UK. J Crohns Colitis. 2017;11:70-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Kariv R, Navaneethan U, Venkatesh PG, Lopez R, Shen B. Impact of Clostridium difficile infection in patients with ulcerative colitis. J Crohns Colitis. 2011;5:34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 67] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 24. | Kim DB, Lee KM, Park SH, Kim YS, Kim ES, Lee J, Jung SA, Seo GS, Lee JM. Is Clostridium difficile infection a real threat in patients with ulcerative colitis? A prospective, multicenter study in Korea. Intest Res. 2018;16:267-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Li Y, Xu H, Xu T, Xiao M, Tang H, Wu D, Tan B, Li J, Yang H, Lv H, Xu Y, Qian J. Case-Control Study of Inflammatory Bowel Disease Patients with and without Clostridium difficile Infection and Poor Outcomes in Patients Coinfected with C. difficile and Cytomegalovirus. Dig Dis Sci. 2018;63:3074-3083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 26. | Maharshak N, Barzilay I, Zinger H, Hod K, Dotan I. Clostridium difficile infection in hospitalized patients with inflammatory bowel disease: Prevalence, risk factors, and prognosis. Medicine (Baltimore). 2018;97:e9772. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 27. | Samman AA, Alfares M, Bajabur RA, Alnefaie RO, Alwadai LS, Murad WW, Dahal BM. Prevalence and Risk Factors of Clostridium difficile Infection Among Patients Hospitalized for a Flare of Inflammatory Bowel Disease in King Abdulaziz University Hospital. Cureus. 2023;15:e48451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Xu H, Tang H, Xu T, Xiao M, Li J, Tan B, Yang H, Lv H, Li Y, Qian J. Retrospective analysis of Clostridium difficile infection in patients with ulcerative colitis in a tertiary hospital in China. BMC Gastroenterol. 2019;19:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 29. | Zhang T, Lin QY, Fei JX, Zhang Y, Lin MY, Jiang SH, Wang P, Chen Y. Clostridium Difficile Infection Worsen Outcome of Hospitalized Patients with Inflammatory Bowel Disease. Sci Rep. 2016;6:29791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 30. | Bernard R, Hammami MB, Arnold FW, Mcgrath B, Patel A, Wuerth B, Nicholson MR, Rao K, Micic D. Clostridioides difficile toxin is infrequently detected in inflammatory bowel disease and does not associate with clinical outcomes. Gut Pathog. 2022;14:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 31. | Negrón ME, Rezaie A, Barkema HW, Rioux K, De Buck J, Checkley S, Beck PL, Carroll M, Fedorak RN, Dieleman L, Panaccione R, Ghosh S, Kaplan GG. Ulcerative Colitis Patients With Clostridium difficile are at Increased Risk of Death, Colectomy, and Postoperative Complications: A Population-Based Inception Cohort Study. Am J Gastroenterol. 2016;111:691-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 32. | Rezapour M, Galoosian A, Liu B, Bhuket T, Wong RJ. Clostridium difficile co-infection in inflammatory bowel disease is associated with significantly increased in-hospital mortality. Eur J Gastroenterol Hepatol. 2018;30:1041-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 33. | Saffouri G, Gupta A, Loftus EV Jr, Baddour LM, Pardi DS, Khanna S. The incidence and outcomes from Clostridium difficile infection in hospitalized adults with inflammatory bowel disease. Scand J Gastroenterol. 2017;52:1240-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 34. | Anderson A, Click B, Ramos-Rivers C, Cheng D, Babichenko D, Koutroubakis IE, Hashash JG, Schwartz M, Swoger J, Barrie AM 3rd, Dunn MA, Regueiro M, Binion DG. Lasting Impact of Clostridium difficile Infection in Inflammatory Bowel Disease: A Propensity Score Matched Analysis. Inflamm Bowel Dis. 2017;23:2180-2188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 35. | Gu YB, Zhang MC, Sun J, Lv KZ, Zhong J. Risk factors and clinical outcome of Clostridium difficile infection in patients with IBD: A single-center retrospective study of 260 cases in China. J Dig Dis. 2017;18:207-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 36. | Navaneethan U, Mukewar S, Venkatesh PG, Lopez R, Shen B. Clostridium difficile infection is associated with worse long term outcome in patients with ulcerative colitis. J Crohns Colitis. 2012;6:330-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 37. | Murthy SK, Steinhart AH, Tinmouth J, Austin PC, Daneman N, Nguyen GC. Impact of Clostridium difficile colitis on 5-year health outcomes in patients with ulcerative colitis. Aliment Pharmacol Ther. 2012;36:1032-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 38. | Jodorkovsky D, Young Y, Abreu MT. Clinical outcomes of patients with ulcerative colitis and co-existing Clostridium difficile infection. Dig Dis Sci. 2010;55:415-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 113] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 39. | Trunfio M, Scabini S, Rugge W, Bonora S, Di Perri G, Calcagno A. Concurrent and Subsequent Co-Infections of Clostridioides difficile Colitis in the Era of Gut Microbiota and Expanding Treatment Options. Microorganisms. 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 40. | Tang L, Wang J, Lin N, Zhou Y, He W, Liu J, Ma X. Immune Checkpoint Inhibitor-Associated Colitis: From Mechanism to Management. Front Immunol. 2021;12:800879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (4)] |
| 41. | Bassotti G, Fruganti A, Stracci F, Marconi P, Fettucciari K. Cytotoxic synergism of Clostridioides difficile toxin B with proinflammatory cytokines in subjects with inflammatory bowel diseases. World J Gastroenterol. 2023;29:582-596. [PubMed] [DOI] [Full Text] |
| 42. | Zhao M, Chu J, Feng S, Guo C, Xue B, He K, Li L. Immunological mechanisms of inflammatory diseases caused by gut microbiota dysbiosis: A review. Biomed Pharmacother. 2023;164:114985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 259] [Article Influence: 86.3] [Reference Citation Analysis (1)] |
| 43. | Wang L, Villafuerte Gálvez JA, Lee C, Wu S, Kelly CP, Chen X, Cao Y. Understanding host immune responses in Clostridioides difficile infection: Implications for pathogenesis and immunotherapy. Imeta. 2024;3:e200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/