Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.114042
Revised: March 10, 2026
Accepted: March 26, 2026
Published online: May 15, 2026
Processing time: 243 Days and 16.3 Hours
Lipohypertrophy (LH) is the most common complication of insulin injection therapy but remains underrecognized in routine care. Repeated injections into the same area promote subcutaneous fat hypertrophy and fibrosis, which can create insulin depot behavior and lead to delayed, blunted, and highly variable insulin absorption. As a result, patients may develop unexplained glycemic instability, including persistent postprandial hyperglycemia and late, unpredictable hypogly
Core Tip: Lipohypertrophy is a common but often overlooked cause of unstable insulin activity. Tissue hypertrophy and fibrosis at injection sites can create insulin depot behavior, leading to delayed and unpredictable absorption with both hyperglycemia and late hypoglycemia. A practical management pathway combines routine injection-site inspection with palpation (and ultrasound when needed), structured rotation and technique education, and close monitoring with individualized dose adjustment when patients are switched away from affected areas. Embedding injection-site care into routine insulin titration can reduce avoidable glycemic variability and improve insulin safety.
- Citation: Nguyen NTY, Tran HP, Luong TV, Nguyen DX, Tran DH, Hoai PHT, Lee MC, Dang HNN. From lipodystrophy to subcutaneous insulin depots: Overlooked mechanisms of glycemic variability in patients with diabetes. World J Diabetes 2026; 17(5): 114042
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/114042.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.114042
Insulin therapy remains essential for many people with diabetes; however, its effectiveness depends on reasonably predictable absorption from subcutaneous tissue. A common and largely preventable cutaneous complication of repeated injections is insulin injection-related lipodystrophy, most frequently lipohypertrophy (LH), characterized by localized adipose hypertrophy with varying degrees of fibrosis and tissue remodeling at injection sites[1,2]. Injecting into LH has been associated with delayed and more variable insulin absorption and action, which can undermine glycemic control and complicate dose titration in routine practice[3,4]. Clinically, this is often recognized as insulin depot formation (local retention with erratic release), manifesting as persistent hyperglycemia prompting dose escalation interspersed with unexpected or prolonged hypoglycemia when absorption becomes unpredictable[3-5].
LH is reported across diverse insulin-treated populations and is linked to higher insulin requirements and poorer glycemic outcomes[2,6,7]. However, LH is frequently underrecognized because patients may repeatedly inject into affected areas (often perceived as less painful), and subtle or deep lesions can be missed on brief clinical inspection and palpation[2,8]. In such cases, glycemic instability may be misattributed to systemic insulin resistance, dietary factors, or nonadherence, leading to avoidable treatment intensification without addressing the underlying absorption problem[1,2].
High-frequency ultrasound can improve detection and phenotyping by visualizing subcutaneous structural changes and delineating lesion extent, thereby supporting targeted site avoidance and individualized management in selected patients with unstable glycemia[9-12]. In parallel, continuous glucose monitoring (CGM) can provide supportive clinical clues when glucose patterns suggest erratic absorption and late hypoglycemia related to injection site dysfunction[13]. Despite these advances, clinical pathways rarely integrate systematic injection-site assessment, ultrasound support when appropriate, structured injection-technique education, and safe dose adjustment when the affected tissue is transitioned to unaffected tissue, as emphasized in expert recommendations[2,8,14].
Therefore, this mini-review aims to: (1) Summarize how LH-related tissue remodeling can contribute to unpredictable insulin absorption and clinically meaningful glycemic variability; (2) Review pragmatic diagnostic approaches with an emphasis on ultrasound-supported lesion characterization; and (3) Outline prevention and management strategies particularly structured injection education, monitoring, and individualized dose adjustment to improve insulin safety and treatment outcomes.
Insulin injection-related lipodystrophy includes two opposing clinical phenotypes: Lipoatrophy and LH. In the era of highly purified insulin analogs, lipoatrophy is now uncommon but is still reported in selected populations (including children), and it may coexist with LH[15]. In routine practice, LH remains the predominant injection-site phenotype and is most frequently implicated in the clinically relevant impairment of insulin absorption[1,2].
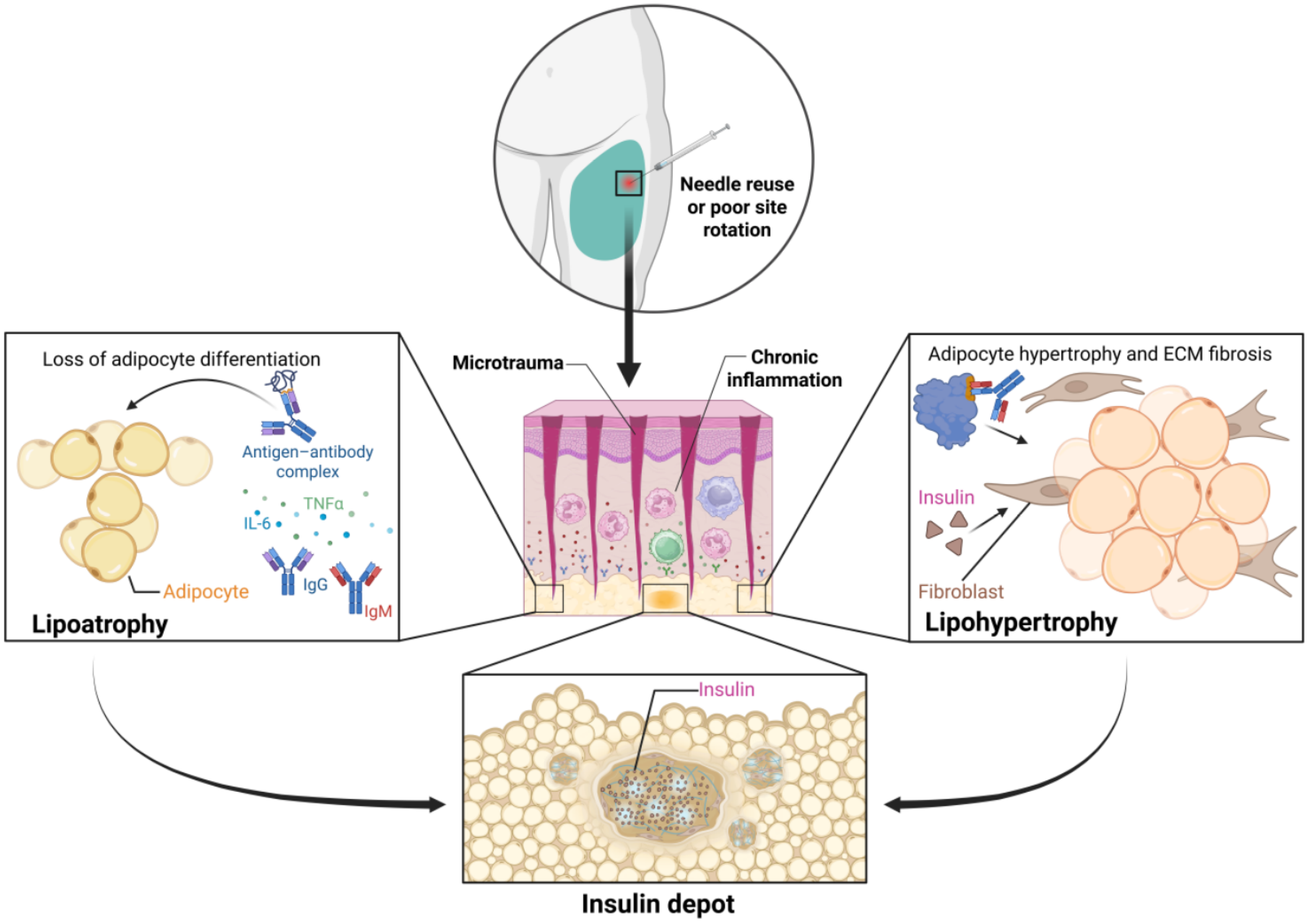
Insulin injection-related LH is not merely localized to adipose tissue enlargement. It represents a process of subcutaneous microenvironment remodeling. Repeated insulin exposure combined with microtrauma at the same injection site promotes adipocyte hypertrophy, activates low-grade inflammatory signaling, and increases extracellular matrix deposition[1,2]. Collagen accumulation, together with microvascular structural changes, increases tissue stiffness and reduces functional capillary density, whereas impaired perfusion and interstitial fluid drainage may alter local insulin transport and diffusion[1]. As a result, the subcutaneous environment within LH areas becomes heterogeneous in both structure and physiological properties, providing a substrate in which insulin depot formation and variable absorption are more likely to occur[2,3]. Collectively, these processes provide the mechanistic basis for insulin depot formation within lipohypertrophic tissue (Figure 1).
The transition from normal tissue to LH does not simply create a physical barrier. It remodels the local tissue architecture and perfusion such that insulin absorption no longer follows consistent kinetics. In this setting, insulin depots may form because insulin dispersion and clearance are impeded by fibrosis and hypoperfusion, increasing the variability in release kinetics and systemic exposure[2-5]. In addition, formulation-specific processes that govern subcutaneous dissolution and release may be sensitive to local conditions (e.g., precipitation-dissolution behavior of some basal insulin analogs), further contributing to unpredictable profiles when injected into markedly remodeled tissue[16].
Changes in the local buffering capacity and microenvironment may also influence depot behavior. Reduced perfusion and lymphatic drainage in fibrotic tissue can plausibly contribute to local potential of hydrogen heterogeneity, which may alter the dissolution kinetics of insulin preparations whose release depends on local physicochemical conditions, including insulin glargine formulations[16]. Although direct imaging of insulin depots is limited in routine practice, the combined effects of tissue remodeling and formulation-dependent release mechanisms provide a plausible explanation for the loss of a stable release profile and greater variability in insulin action when injections are administered into LH[3-5,16].
Importantly, these mechanisms may act synergistically rather than independently, resulting in delayed, blunted, and erratic absorption. Clinically, this helps explain why circulating insulin exposure and glucose-lowering effects can fluctuate despite the unchanged prescribed dose and timing when injections continue in LH areas[3-5].
The effectiveness of insulin therapy depends on the stable absorption of exogenous insulin from subcutaneous tissue into the circulation. In LH tissue, drug transport is impaired by the combined effects of fibrosis and reduced local perfusion, which can reduce bioavailability and increase glycemic variability[2-4]. An insulin depot within LH tissue can therefore be conceptualized as a functional kinetic system in which insulin is temporarily retained within remodeled tissue regions and released into the circulation with greater delay and unpredictability than in unaffected tissue. Numerical modeling supports that subcutaneous depot geometry and effective dissolution surface area influence release kinetics, particularly in environments that limit dispersion and transport[17].
Insulin absorption from subcutaneous tissue depends on two major determinants: Capillary perfusion and the effective dissolution/dispersion characteristics of the injected formulation[17,18]. In healthy tissue, these parameters remain relatively stable, allowing reasonably predictable absorption. By contrast, fibrotic architecture and hypoperfusion in the LH reduce perfusion-dependent transport and prolong diffusion pathways, increasing intraindividual variability in systemic insulin exposure[4,18]. Consistent with these findings, clinical studies and clamp data have shown that compared with injections into normal tissue, injections into LH are associated with reduced and more variable insulin exposure and glucose-lowering action[3,4].
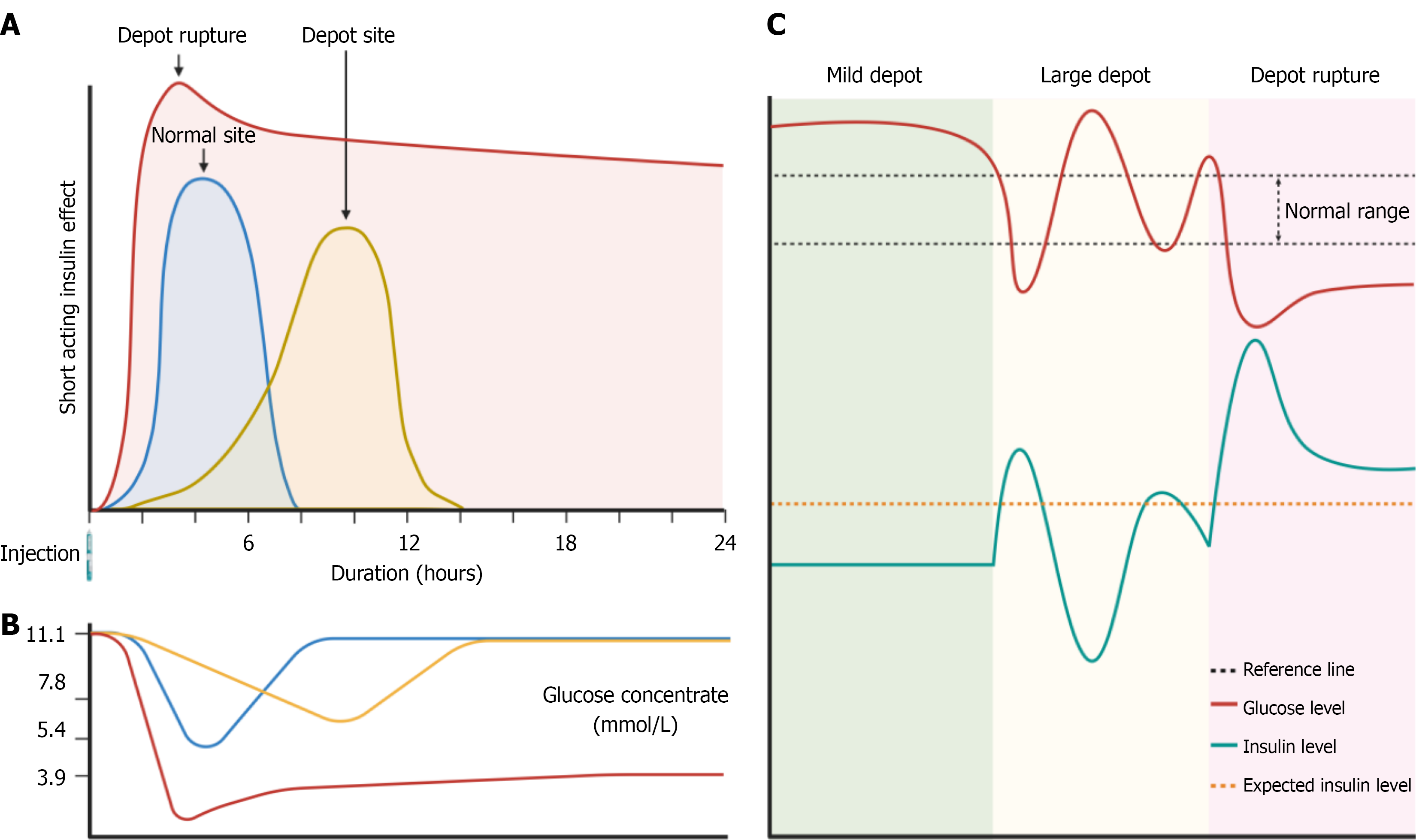
A key principle for clinical interpretation is that insulin depots behave as dynamic entities rather than static structures. Their behavior can be sensitive to changes in local hemodynamics and external factors. Because transport depends on concentration gradients and perfusion, transient triggers such as localized mechanical pressure, temperature changes, or perfusion fluctuations during physical activity may alter depot dissolution and insulin transfer into the circulation[18]. This phenomenon offers a plausible mechanistic explanation for delayed or unexpected hypoglycemia in susceptible patients who are injected into LH areas. Accordingly, switching injection sites to unaffected tissue is a strategic shift from an unstable absorption system to a more predictable pharmacokinetic environment and should be paired with careful monitoring and appropriate dose adjustment[3,4,19]. The resulting differences in insulin action and glucose profiles when insulin is injected into normal vs depot-affected tissue are conceptually summarized in Figure 2.
Euglycemic clamp data from the study by Famulla et al[4] were used to quantify the clinical relevance of LH as a site associated with insulin depot behavior and unpredictable insulin action. Injection into LH areas reduced the peak insulin concentration (Cmax), delayed the time to peak (Tmax), and markedly increased intraindividual variability compared with injections into healthy tissue. Ultrasound findings of heterogeneous subcutaneous abnormalities (including hyperechoic regions consistent with tissue remodeling) provide a structural context for these pharmacokinetic changes and can assist in lesion identification and mapping in clinical practice[10,11]. From a mechanistic perspective, remodeling within the LH plausibly impairs perfusion-dependent transport and prolongs diffusion through a denser extracellular matrix, which is consistent with the greater variability in insulin action observed in experimental studies[4,18].
Pharmacokinetic instability in LH may manifest as characteristic patterns of glycemic fluctuation: Delayed early absorption contributes to prolonged hyperglycemia, and later release of retained insulin increases the risk of un
On the basis of current evidence, the impact of LH on glycemic control can be conceptualized as a three-tier me
This framework integrates structural, pharmacokinetic, and CGM-informed observations into a unified interpretative model. From this perspective, LH should be regarded as a disorder of the injection-site microenvironment rather than a simple dermatologic complication, shifting clinical focus toward correcting injection-site factors that drive avoidable glycemic instability[2,8,14].
Given the predominance and clinical relevance of LH, diagnosis should prioritize systematic detection of LH in patients with unstable glycaemia or escalating insulin requirements[3,4,6]. Accurate identification of LH is therefore more than a technical examination step; it is a practical intervention to address potential insulin depot behavior and otherwise unexplained glycemic events. Modern diagnosis requires the integration of targeted medical information, careful pal
The diagnostic process usually begins with clinical suspicion based on characteristic “red flags,” after systemic factors such as carbohydrate miscalculation, infection, or poor adherence are excluded. A biphasic glucose fluctuation pattern characterized by prolonged postprandial hyperglycemia followed by recurrent late hypoglycemia on CGM serves as an important clinical clue for erratic insulin action potentially related to LH and insulin depot behavior[15]. When patients develop “apparent insulin resistance,” defined as an increase in daily insulin doses without improvement in glycated hemoglobin levels, clinicians should prioritize local injection-site evaluation to identify absorption barriers rather than focusing solely on systemic metabolic changes[2].
Although diagnostic tools continue to advance, trained healthcare providers performing systematic palpation remain the foundation of screening. Examination requires a structured technique that combines inspection with controlled deep palpation to detect changes in tissue density and homogeneity. Clinically, injection-related lesions potentially associated with insulin depot behavior may present in four main patterns: (1) Visibly apparent and clearly palpable lesions; (2) Flat, mildly elastic, subtle nodules detected by skin-fold pinching; (3) Nonvisible lesions identifiable only with deep pressure; and (4) Flat subcutaneous abnormalities that are easily missed during routine examination but are best detected by ultrasound[2].
Because manual examination has limited sensitivity for deep or early lesions, high-frequency ultrasound has become an increasingly valuable adjunct modality. Ultrasound can confirm subcutaneous abnormalities (e.g., diffuse hyperechogenicity, nodular lesions, and heterogeneous echotexture) and delineate lesion extent, supporting more accurate detection and mapping of LH in patients with unstable glycemia[9-12]. On the basis of ultrasound morphology, grading fra
| Grade | Ultrasound imaging characteristics | Suggested clinical considerations |
| Grade 0 | Normal (no nodules or diffuse areas) | Continue routine site rotation education and periodic site checks |
| Grade 1 | Diffuse hyperechogenic areas without clear nodules | Avoid repeated injections into the same area; reinforce rotation and needle single-use; consider ultrasound mapping if glycemia remains unstable |
| Grade 2 | Nodules 1-5.9 mm (width at widest point) | Avoid injecting into lesion; monitor closely when switching to unaffected tissue because absorption may increase; reinforce education and rotation |
| Grade 3 | Nodules 6-9.9 mm | As Grade 2, with heightened vigilance and closer monitoring during site change in patients with prior hypoglycemia/Large dose requirements |
| Grade 4 | Nodules ≥ 10 mm | Strictly avoid injections into lesion; lesions may persist longer and require sustained site avoidance and monitoring; consider follow-up ultrasound in selected patients |
The prognosis for tissue recovery in LH depends on the initial degree of fibrosis and lesion burden. Mild lesions may regress over months after cessation of injections at the affected site, whereas larger or more fibrotic lesions may persist longer or recover only partially in function[2,12]. Overall, systematic diagnosis of LH coupled with targeted injection technique education can yield clinically meaningful improvements in glycemic stability and reduce avoidable treatment intensification[2,8,14]. Therefore, injection-site assessment should be considered a core step in insulin titration workflows and in the evaluation of unexplained glycemic variability.
The primary objective of LH management is to break the cycle between injection-site tissue remodeling, insulin depot behavior, and unstable glycemia. Management relies on discontinuing insulin delivery into LH lesions, reinforcing standardized injection techniques with systematic site rotation, and cautiously adjusting insulin doses with close monitoring when transitioning from affected to unaffected tissue to restore more predictable absorption[2-4].
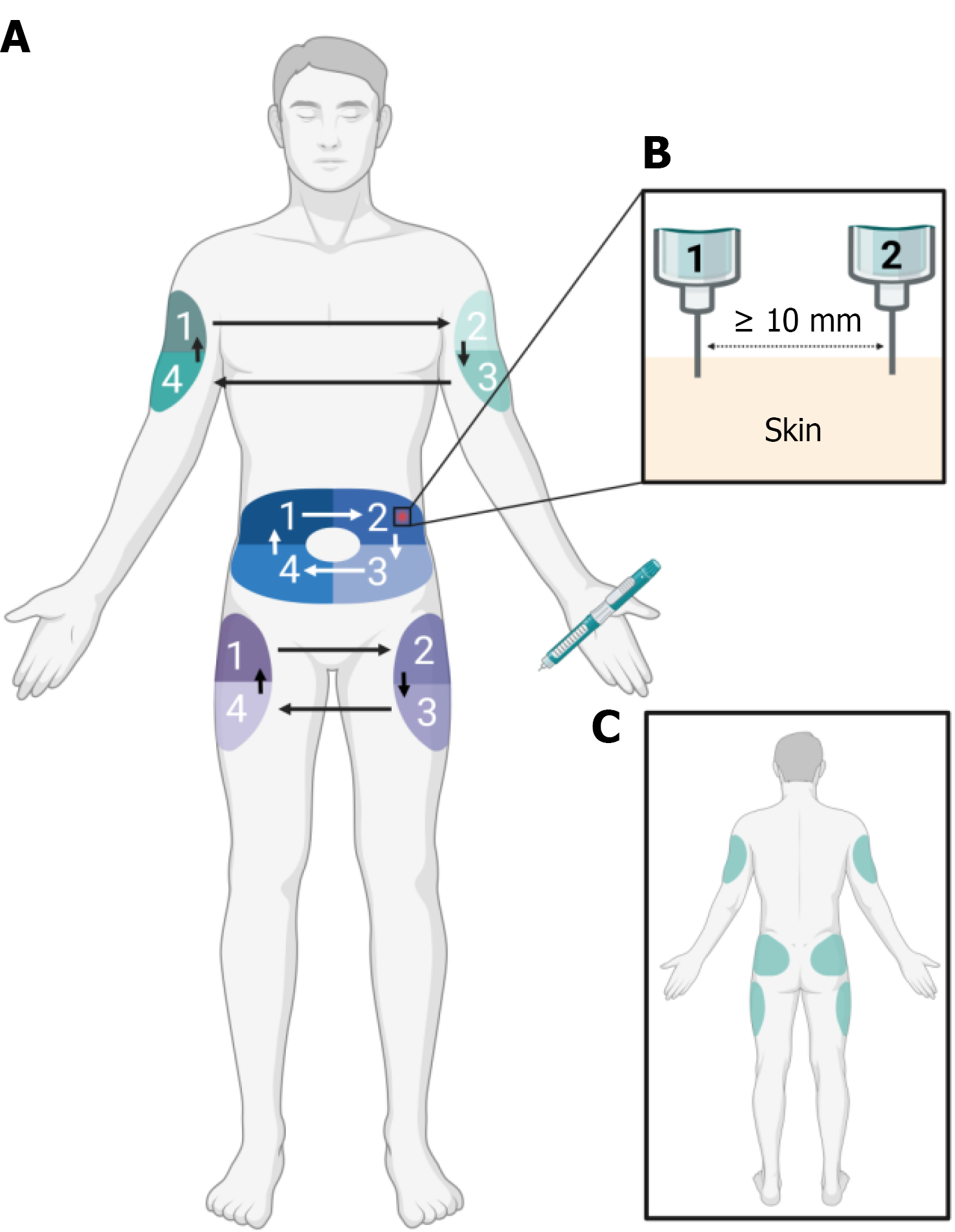
Structured site rotation and correct injection techniques are central to both prevention and treatment[14]. The recommended injection regions and rotation patterns are illustrated in Figure 3. A step-by-step checklist of practical insulin handling, injection technique, site rotation (including spacing and zone-based strategies), and routine site inspection/palpation is summarized in Table 2[13,14]. Implementing these measures can reduce continued injection into affected tissue and mitigate absorption variability linked to LH[2,14].
| Phase | Key actions | Core principles |
| Preparation and insulin handling | Storage and temperature | If insulin has been refrigerated, allow it to reach room temperature by leaving it out of the refrigerator for 30 to 60 minutes before injection; injecting cold insulin can be painful and may contribute to lipodystrophy |
| Resuspension of cloudy insulin | For insulin suspensions (e.g., NPH), gently roll/tip the vial or pen to resuspend the insulin; avoid vigorous shaking to reduce air bubbles | |
| Priming (pen devices) | Prime the pen before injection and confirm flow by seeing insulin at the needle tip to help ensure accurate dose delivery | |
| Needle length | Prefer 4-mm pen needles for most people to support reliable subcutaneous delivery and reduce intramuscular injection risk | |
| Injection technique | Choose appropriate skin area | Use clean, intact skin and avoid injecting into areas with lipodystrophy, scars, bruising, infection, inflammation, edema, or ulceration |
| Angle and skinfold | With a 4-mm pen needle, inject perpendicularly (90°). A skinfold may be required for very young children and very thin adults, particularly when longer needles are used | |
| Delivery and needle dwell time | Inject insulin steadily and, after completing the injection, wait 10 seconds before withdrawing the needle to reduce leakage and ensure full dose delivery | |
| Avoid injection through clothing | Do not inject through clothing, because it prevents appropriate site inspection and can interfere with correct technique | |
| Needle reuse | Use needles once only; avoid reuse to reduce tissue trauma and the risk of lipohypertrophy | |
| Site rotation | Spacing between injections | Inject at least 1 cm from the previous injection site (approximately one adult finger width) and rotate in a consistent direction |
| Separate sites for concurrent injections | If two injections are given at the same time (e.g., basal and prandial insulin), administer them at separate injection sites | |
| Zone-based rotation | Change the body zone used for injections regularly (e.g., use one quadrant/half per week) to avoid overuse of a single area | |
| Monitoring and early detection | Routine inspection and palpation | Visual inspection and palpation of injection sites should be performed routinely as part of patient self-check and/or by healthcare professionals; review site rotation practices at least annually |
| Triggered assessment (warning signs) | Injection sites should be examined carefully when warning signs occur, including unusual pain, insulin leakage, bleeding/bruising, increased force required to deliver insulin, or glucose becoming unstable/unpredictable despite the usual regimen |
When injections are switched from LH lesions to unaffected tissue, hypoglycemia risk increases because insulin absorption may become faster and more predictable; dose reduction is therefore commonly required and should be guided by glucose measurements[3,4]. FITTER Forward Expert recommendations emphasize that the magnitude of dose decrease varies across individuals and that these reductions often exceed 20% of the original dose in some patients after switching away from LH[13]. Larger reductions may be required in selected individuals with greater lesion burden and marked pre-switch glycemic instability, underscoring the need for close monitoring and stepwise titration rather than fixed-dose changes[2,8,12,22].
Individuals with type 1 diabetes and those using continuous subcutaneous insulin infusion can be particularly sensitive to changes in absorption kinetics; conservative, closely supervised titration is warranted after site change[2,13]. In pump users, infusion sets should be replaced according to product instructions (commonly every 2-3 days) because prolonged wear increases local tissue reactions and may contribute to erratic absorption; extended-wear sets (up to 7 days in selected products) do not replace the need for systematic rotation and routine site assessment[23,24].
During the first 48 to 72 hours after switching injection sites, intensified monitoring (CGM where available, or frequent self-monitoring of blood glucose) is recommended to identify early hypoglycemia and guide prompt adjustment[13,14]. Thereafter, titration can follow a structured sequence, stabilizing basal insulin first using fasting and overnight glucose patterns and then adjusting insulin doses based on pre- and post-meal glucose patterns[25]. Therefore, incorporating routine injection-site assessments into insulin titration workflows is a practical, patient-centered strategy to improve glycemic stability and reduce avoidable treatment intensification in insulin-treated patients[2,13,14].
In summary, LH management requires systematic site avoidance, a standardized injection technique with structured rotation, and individualized dose adjustment supported by close glucose monitoring to reduce insulin depot-related variability and improve treatment safety.
Future work should move from lesion-focused care to patient-centered insulin delivery management because real-world behaviors (fear of pain, convenience-driven repeated injections, and needle reuse) can sustain LH and perpetuate insulin depot behavior. Contemporary expert recommendations emphasize structured technique education and routine injection-site assessment (including periodic visual inspection and palpation, with intensified review when warning signs or unexplained glycemic instability occur); the next step is to test how best to deliver these elements at scale through multidisciplinary models, teach-back competency checks, and telehealth reinforcement[14].
Research priorities should focus on patient-relevant outcomes. Given the established variability in insulin exposure and action when injected into lipohypertrophic tissue and the evidence that lipodystrophic areas distort absorption, prospective site-switching cohorts and pragmatic trials should quantify severe hypoglycemia, CGM metrics (time in range and glycemic variability), insulin dose requirements, and treatment burden, with healthcare utilization assessed where feasible[3,4]. In parallel, ultrasound phenotyping/grading should be harmonized with reproducibility testing to enable comparable reporting across settings and facilitate evidence synthesis[11,21]. Finally, technology-enabled pathways (digital rotation aids, CGM-triggered prompts for site reassessment, and automated ultrasound interpretation) and embedded health-economic analyses could improve the scalability and equity of care.
Insulin depot formation within lipohypertrophic tissue reflects disruption of the injection-site microenvironment rather than a minor local complication, and it can translate directly into delayed, blunted, and unpredictable insulin action that drives clinically meaningful glycemic variability and avoidable hypoglycemia. Accordingly, injection-site pathology should be considered whenever glucose patterns become unstable or insulin requirements rise unexpectedly despite apparently appropriate prescribing and adherence. A practical, patient-centered approach integrates structured technique education, systematic site rotation, and routine visual inspection with palpation, with high-frequency ultrasound used when clinical examination is inconclusive or glycemic instability persists, to map lesions and guide site avoidance. When injections are switched away from affected areas, clinicians should anticipate faster and more predictable absorption and therefore pair site changes with intensified glucose monitoring and individualized, stepwise dose adjustments to reduce hypoglycemia risk. Moving forward, priorities include scalable implementation pathways that embed injection-site care into routine insulin management, harmonized ultrasound phenotyping with reproducibility testing and prospective pragmatic studies that quantify patient-relevant outcomes such as severe hypoglycemia, time in range, glycemic variability, insulin dose requirements, treatment burden and healthcare utilization.
| 1. | Strollo F, Guarino G, Gentile S. A Neglected Complication of Insulin Therapy Due to Errors in Injection Technique: Skin Lipohypertrophies: A Narrative Review. Diabetology. 2025;6:22. [DOI] [Full Text] |
| 2. | Tian T, Aaron RE, Huang J, Yeung AM, Svensson J, Gentile S, Forbes A, Heinemann L, Seley JJ, Kerr D, Klonoff DC. Lipohypertrophy and Insulin: An Update From the Diabetes Technology Society. J Diabetes Sci Technol. 2023;17:1711-1721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 3. | Young RJ, Hannan WJ, Frier BM, Steel JM, Duncan LJ. Diabetic lipohypertrophy delays insulin absorption. Diabetes Care. 1984;7:479-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 87] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 4. | Famulla S, Hövelmann U, Fischer A, Coester HV, Hermanski L, Kaltheuner M, Kaltheuner L, Heinemann L, Heise T, Hirsch L. Insulin Injection Into Lipohypertrophic Tissue: Blunted and More Variable Insulin Absorption and Action and Impaired Postprandial Glucose Control. Diabetes Care. 2016;39:1486-1492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 118] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 5. | Jockel JP, Roebrock P, Shergold OA. Insulin depot formation in subcutaneous tissue. J Diabetes Sci Technol. 2013;7:227-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Mader JK, Fornengo R, Hassoun A, Heinemann L, Kulzer B, Monica M, Nguyen T, Sieber J, Renard E, Reznik Y, Ryś P, Stożek-Tutro A, Wilmot EG. Relationship Between Lipohypertrophy, Glycemic Control, and Insulin Dosing: A Systematic Meta-Analysis. Diabetes Technol Ther. 2024;26:351-362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 7. | Gentile S, Guarino G, Corte TD, Marino G, Fusco A, Corigliano G, Colarusso S, Piscopo M, Improta MR, Corigliano M, MartedÌ E, Oliva D, Russo V, Simonetti R, Satta E, Romano C, Alfarone C, Vetrano A, Martino C, Lamberti C, Vecchiato A, Cozzolino G, Brancario C, Strollo F; AMD-OSDI Study Group on Injection Techniques and Nefrocenter Research & Nyx Start-up Study Group. Insulin-Induced Skin Lipohypertrophy in Type 2 Diabetes: a Multicenter Regional Survey in Southern Italy. Diabetes Ther. 2020;11:2001-2017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 8. | Hirsch LJ, Strauss KW. The Injection Technique Factor: What You Don't Know or Teach Can Make a Difference. Clin Diabetes. 2019;37:227-233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 9. | Wang W, Huang R, Chen Y, Tu M. Values of ultrasound for diagnosis and management of insulin-induced lipohypertrophy: A prospective cohort study in China. Medicine (Baltimore). 2021;100:e26743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Kapeluto JE, Paty BW, Chang SD, Meneilly GS. Ultrasound detection of insulin-induced lipohypertrophy in Type 1 and Type 2 diabetes. Diabet Med. 2018;35:1383-1390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Hashem R, Mulnier H, Abu Ghazaleh H, Halson-Brown S, Duaso M, Rogers R, Karalliedde J, Forbes A. Characteristics and morphology of lipohypertrophic lesions in adults with type 1 diabetes with ultrasound screening: an exploratory observational study. BMJ Open Diabetes Res Care. 2021;9:e002553. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Yang H, Zhang L, Dong Q, She Y, Zhang Y, Cao M, Xu M, Chu S. The Distinct Ultrasound Characteristics and Prognostic Features of Insulin-Induced Lipohypertrophy: A Systematic Review. Diabetes Metab Syndr Obes. 2025;18:941-954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Frid AH, Kreugel G, Grassi G, Halimi S, Hicks D, Hirsch LJ, Smith MJ, Wellhoener R, Bode BW, Hirsch IB, Kalra S, Ji L, Strauss KW. New Insulin Delivery Recommendations. Mayo Clin Proc. 2016;91:1231-1255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 194] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 14. | Klonoff DC, Berard L, Franco DR, Gentile S, Gomez OV, Hussein Z, Jain AB, Kalra S, Anhalt H, Mader JK, Miller E, O'Meara MA, Robins M, Strollo F, Watada H, Heinemann L. Advance Insulin Injection Technique and Education With FITTER Forward Expert Recommendations. Mayo Clin Proc. 2025;100:682-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 15. | Singha A, Bhattarcharjee R, Ghosh S, Chakrabarti SK, Baidya A, Chowdhury S. Concurrence of Lipoatrophy and Lipohypertrophy in Children With Type 1 Diabetes Using Recombinant Human Insulin: Two Case Reports. Clin Diabetes. 2016;34:51-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 16. | Heise T, Mathieu C. Impact of the mode of protraction of basal insulin therapies on their pharmacokinetic and pharmacodynamic properties and resulting clinical outcomes. Diabetes Obes Metab. 2017;19:3-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 79] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 17. | Zedelmair M, Mukherjee A. Numerical Simulation of Insulin Depot Formation in Subcutaneous Tissue Modeled as a Homogeneous Anisotropic Porous Media. J Biomech Eng. 2021;143:051002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Gradel AKJ, Porsgaard T, Lykkesfeldt J, Seested T, Gram-Nielsen S, Kristensen NR, Refsgaard HHF. Factors Affecting the Absorption of Subcutaneously Administered Insulin: Effect on Variability. J Diabetes Res. 2018;2018:1205121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 128] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 19. | Bahendeka S, Kaushik R, Swai AB, Otieno F, Bajaj S, Kalra S, Bavuma CM, Karigire C. EADSG Guidelines: Insulin Storage and Optimisation of Injection Technique in Diabetes Management. Diabetes Ther. 2019;10:341-366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 20. | Korkmaz FN, Gökçay Canpolat A, Güllü S. Determination of insulin-related lipohypertrophy frequency and risk factors in patients with diabetes. Endocrinol Diabetes Nutr (Engl Ed). 2022;69:354-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Yu J, Wang H, Zhou M, Zhu M, Hang J, Shen M, Jin X, Shi Y, Xu J, Yang T. A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients. Diagnostics (Basel). 2023;13:1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 22. | Yu J, Wang H, Zhou M, Zhu M, Hang J, Yang T, Shi Y, Xu J. Impact of ultrasound-diagnosed lipohypertrophy subtypes on insulin regimen adjustments in patients with T1DM. Diabet Med. 2025;42:e70034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Zhang G, Romo-Anselmo E, Kwa T, Cohen O, Vigersky R, Chattaraj S. Advances in Insulin Infusion Set in the New Era of Automated Insulin Delivery: A Systematic Review. J Diabetes Sci Technol. 2023;17:302-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Pfützner A, Sachsenheimer D, Grenningloh M. Longer Usage Time for CSII Catheters: An Underestimated Challenge. J Diabetes Sci Technol. 2016;10:987-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 25. | Chun J, Strong J, Urquhart S. Insulin Initiation and Titration in Patients With Type 2 Diabetes. Diabetes Spectr. 2019;32:104-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (0)] |