Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.117094
Revised: December 30, 2025
Accepted: February 10, 2026
Published online: April 15, 2026
Processing time: 137 Days and 21.9 Hours
Artificial intelligence (AI) is transforming the landscape of diabetic care, bridging the gap between early detection and personalized disease management. In ophthalmology, AI-driven algorithms have demonstrated remarkable accuracy in identifying diabetic retinopathy and diabetic macular edema from retinal fundus and optical coherence tomography images, rivaling expert graders while offering scalable, cost-effective solutions for population-level screening. Beyond image analysis, AI is emerging as a powerful decision-support tool that integrates systemic data, glycemic control metrics, and imaging biomarkers to predict disease progression and treatment response. This narrative opinion review explores how AI can reshape diabetic patient management through predictive analytics, remote monitoring, and automated triage systems, enabling timely referrals and individualized follow-up. We also discuss the ethical, regulatory, and practical barriers to clinical translation, emphasizing the need for explainable algorithms, real-world validation, and interdisciplinary collaboration. By shifting from reactive treatment to proactive prediction, AI promises to redefine the continuum of diabetic eye care – from screening to precision-guided management – ultimately reducing blindness and healthcare burden worldwide.
Core Tip: Artificial intelligence (AI) is transforming diabetic eye care through automated detection, risk assessment, and precision management. Deep learning algorithms utilized in fundus photography and optical coherence tomography imaging now equal or surpass experienced graders in the detection of diabetic retinopathy and macular edema. In addition to screening, AI incorporates multimodal data – such as systemic parameters, imaging biomarkers, and treatment histories – to predict disease progression and therapeutic outcomes. This review articulates contemporary clinical uses, technical underpinnings, and translational obstacles, while delineating potential trajectories for the development of explainable, morally robust, and globally scalable AI systems that can revolutionize the management of diabetic eye disease.
- Citation: Cappellani F, Capobianco M, Visalli F, Khouyyi M, Musa M, Avitabile A, Leandro I, Giglio R, Tognetto D, Gagliano C, D’Esposito F, Zeppieri M. From pixels to precision: Artificial intelligence in diabetic eye disease screening and management. World J Diabetes 2026; 17(4): 117094
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/117094.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.117094
Diabetic retinopathy (DR) and diabetic macular edema (DME) are among the most common microvascular consequences of diabetes mellitus, constituting a primary cause of avoidable vision impairment in working-age individuals globally. Notwithstanding the existence of efficacious therapies like anti-vascular endothelial growth factor (VEGF) medication and laser photocoagulation, late presentation persists frequently because of inadequate screening coverage and a worldwide deficit of skilled graders. The manual analysis of retinal images is resource-demanding, subjective, and susceptible to inter-observer variability, especially when screening large-scale diabetic populations. In this context, artificial intelligence (AI) has emerged as a transformative method that enhances diagnostic capabilities, facilitates early detection, and standardizes clinical decision-making in ophthalmology[1,2].
Gulshan et al[1] conducted the initial significant demonstration of AI’s clinical capabilities in ophthalmic imaging, developing and validating a deep-learning algorithm using over 128000 retinal fundus images, achieving performance levels comparable to those of board-certified ophthalmologists in identifying referable DR. Subsequently, Ting et al[2] expanded this notion to a multi-ethnic cohort, thereby enhancing the validation of algorithmic resilience and generalizability across various imaging devices and populations. These foundational investigations initiated AI-assisted diabetic eye screening. Since then, convolutional neural networks (CNNs) and transformer-based architectures have demon
Following the initial development of fundus-based models, research focus shifted toward multimodal imaging and prognostic forecasting. Optical coherence tomography (OCT) and OCT angiography (OCTA) offer volumetric and microvascular biomarkers that not only facilitate lesion diagnosis but also assess disease severity, macular fluid dynamics, and vascular perfusion status. Zafar et al[6] highlighted the clinical significance of AI algorithms in detecting modest retinal alterations prior to the manifestation of clinically apparent DME, indicating their potential utility in personalized follow-up scheduling and early treatment intervention. The integration of imaging data with systemic variables, including glycemic control, lipid levels, and renal function, significantly improves predictive accuracy, enabling algorithms to categorize patients by their likelihood of progression or recurrence[7,8].
As AI technology advances, its function in diabetic eye disease is transitioning from screening to holistic disease management. Recent research investigated predictive analytics that can project DR progression over multiple years[9], thereby facilitating a proactive clinical approach rather than a reactive one. Concurrently, precision medicine frameworks integrate ocular imaging with electronic health records and wearable data, providing a comprehensive perspective on disease dynamics[10]. Notwithstanding these significant advancements, substantial translational obstacles remain. Algorithmic performance may diminish when applied to datasets with varying demographic or imaging attributes, underscoring the need for external validation and established assessment measures. Issues pertaining to interpretability, accountability, and data privacy exacerbate the complexities of clinical integration, especially within publicly funded healthcare systems[11].
AI is transforming diabetic eye care by enabling early detection, disease forecasting, and personalized management. The amalgamation of automated image analysis, systemic risk modeling, and real-time clinical decision support could transform the continuum of care from population screening to individualized treatment planning. However, actualizing this promise requires thorough validation, transparent model development, and close coordination among physicians, engineers, and politicians. This review aims to critically assess the current state of AI in DR and DME, analyze its clinical implications and limitations, and suggest future pathways for the explainable, ethically responsible integration across various healthcare systems.
This narrative opinion review aims to synthesize and critically evaluate the growing body of evidence regarding the use of AI in the detection and management of DR and DME. Despite not meeting the criteria for a systematic review, the technique was designed to ensure transparency, repeatability, and scientific rigor. A thorough search was conducted in PubMed, Scopus, and Web of Science for peer-reviewed works published from January 2016 to November 2025, covering the contemporary period of deep learning in ophthalmology. The preliminary search approach integrated the terms “artificial intelligence”, “deep learning”, “machine learning”, “diabetic retinopathy”, “diabetic macular edema”, “fundus photography”, “optical coherence tomography”, and “OCT angiography”. Only papers published in English and indexed in PubMed were incorporated to guarantee traceability and methodological rigor. Reference lists from significant reviews and meta-analyses were examined to discover supplementary pertinent studies, ensuring comprehensive coverage across clinical, translational, and regulatory domains.
Studies were included if they used AI or machine learning techniques to detect, classify, predict, or monitor DR or DME using ophthalmic imaging modalities. This encompassed fundus photography, OCT, and OCTA-based investigations, as well as multimodal methodologies that integrate systemic or clinical data. Papers focused solely on preclinical or technical algorithm development were eliminated unless they demonstrated clinically validated performance measures or evident potential for translational application. Two independent reviewers evaluated the titles and abstracts, and thereafter conducted a full-text analysis to confirm methodological relevance. Discrepancies were reconciled through consensus. Each selected study was examined for dataset composition, sample size, training-validation split, use of external test sets, and assessment metrics, including sensitivity, specificity, and the area under the receiver operating characteristic curve. Priority was assigned to studies exhibiting repeatability, generalizability, and the inclusion of multi-ethnic or real-world clinical cohorts.
This research seeks to deliver an interpretive synthesis rather than a quantitative meta-analysis; hence, no explicit bias rating tool was used. The methodological quality was evaluated qualitatively, focusing on the transparency of algo
To guarantee comprehensive coverage, upcoming research from 2024 to 2025 that concentrates on explainable models, multimodal transformers, and anti-VEGF response prediction was incorporated, contingent upon their indexing in PubMed and adherence to established relevant criteria.
The diagnostic use of AI in diabetic eye disease has advanced swiftly over the last decade, moving from algorithmic proof-of-concept to practical clinical implementation. Initial advancements focused on CNNs trained on large collections of retinal fundus images to autonomously detect lesions indicative of DR, including microaneurysms, hemorrhages, and exudates. These models attained performance equivalent to expert graders regarding sensitivity and specificity, and in several instances exceeded manual grading in consistency and throughput. Among the earliest landmark contributions, Gulshan et al[1] developed and validated a deep-learning algorithm on more than 128000 fundus photographs, achieving a sensitivity of 97.5% and specificity of 93.4% for referable DR. This study established the feasibility of high-performance automated DR detection and catalyzed subsequent large-scale AI development.
Ting et al[2] assessed a deep-learning system across 10 datasets comprising nearly 500000 retinal images from mul
Following these advancements, the research focus shifted to extensive real-world validation studies. Xie et al[4] conducted an economic modeling analysis indicating that integrating AI into national teleophthalmology programs could significantly reduce healthcare costs while preserving diagnostic accuracy comparable to manual grading. The study highlighted that AI integration enhances operational efficiency and enables early detection of referable diseases, thereby enabling prompt referrals to ophthalmologists and averting vision-threatening complications. Complementing these findings, Bellemo et al[12] conducted one of the first real-world primary-care evaluations of an AI system for the detection of referable DR. In more than 30000 screened patients, the model sustained high diagnostic accuracy while reducing the clinical burden on ophthalmologists, demonstrating the practical feasibility of AI-assisted screening workflows. Wang et al[5] subsequently developed a semi-automated deep learning approach that integrates algorithmic detection with human verification, attaining high reproducibility while upholding clinical safety criteria. Hybrid methodologies have been prominent in healthcare systems when complete automation remains impractical, facilitating quality assurance while using AI’s computational benefits.
In addition to detecting DR, AI algorithms have remarkable accuracy in diagnosing DME. This condition poses a direct risk to central vision and frequently necessitates anti-VEGF therapy. OCT has become an essential technique, offering high-resolution cross-sectional imaging of the retina and facilitating objective measurement of macular thickness and fluid buildup. Early work by Lee et al[13] demonstrated that CNNs could automatically segment intraretinal fluid and macular thickening on OCT with accuracy comparable to expert graders, establishing the feasibility of OCT-based deep learning for edema assessment. Building on this foundation, Schlegl et al[14] developed a fully automated deep-learning system capable of detecting and quantifying intraretinal and subretinal fluid, a hallmark of DME, achieving specialist-level performance and validating OCT-derived fluid biomarkers as robust diagnostic indicators.
Further expanding clinical applicability, De Fauw et al[15] introduced an end-to-end OCT interpretation framework that provided diagnostic and referral recommendations – including for diabetic maculopathy – with accuracy comparable to retina specialists, supporting the integration of OCT-based AI into triage and population-level screening pathways. As AI-based screening programs for diabetic eye disease scale up with larger and more diverse datasets, their clinical performance is promising. In their review, Poschkamp and Stahl[16] noted that deployment of deep-learning algorithms in real-world screening is influenced by factors such as image quality, camera type/setting, and demographic characteristics of the screened population – emphasizing the need for robust validation across diverse settings. Their conclusion indicates that although algorithmic performance is consistently strong, real-world heterogeneity underscores the necessity for external validation and algorithm retraining across diverse geographic and ethnic populations.
From a translational outlook, several national screening programs are now piloting large-scale AI systems, demon
Rodríguez-Miguel et al[19] developed a hybrid deep-learning system – stacking multiple CNNs with a bidirectional recurrent neural network – to screen for DME using full OCT cubes in a real-world screening program, achieving an area under the receiver operating characteristic of approximately 0.94 and demonstrating how AI can extend beyond fundus-based detection to structural OCT-based automated triage.
A recent large-scale study by Midena et al[20] applied AI-based clustering of OCT-derived biomarkers in over 2300 eyes with DME, delineating four distinct morphological phenotypes characterized by differing fluid distribution, hyperreflective foci counts and ellipsoid-zone integrity. Their results underscore the potential of structural imaging biomarkers – when extracted automatically – to stratify patients beyond traditional diagnostic categories and support tailored therapeutic strategies.
AI has evolved from a nascent concept to a clinically validated element of diabetic eye care. The incorporation of autonomous and semi-autonomous systems into national screening programs illustrates both technological advancement and increasing regulatory approval. Nevertheless, extensive deployment requires substantial evidence of generalizability, seamless integration with current clinical operations, and cost-effectiveness in practical settings. As algorithms advance to integrate multimodal data, such as OCT, OCTA, and systemic factors, AI is set to improve diagnosis precision and radically transform the management approach for DR and macular edema. The forthcoming frontier involves integrating diagnostic automation with tailored therapeutic assistance, necessitating interdisciplinary collaboration among data scientists, doctors, and health policy stakeholders.
The predictive utilization of AI in diabetic eye disease signifies a highly promising avenue for precision ophthalmology. Initially, AI algorithms were developed mainly for binary disease classification (e.g., referable vs non-referable DR); however, current research has shifted towards predicting disease progression, treatment efficacy, and long-term consequences. The justification for this development is evident: Recognizing those at the greatest risk of advancing from non-proliferative to proliferative DR may facilitate earlier intervention, customized follow-up protocols, and individualized treatment plans. In a landmark study, Cao et al[21] developed a machine-learning model based on pre-therapeutic OCT-derived features in 712 patients with DME that predicted individual anatomical response to intravitreal anti-VEGF treatment [area under the curve (AUC) = 0.92] – exemplifying how artificial-intelligence methods may extend beyond disease detection to guide therapeutic decision-making. These findings indicate that AI techniques may extend beyond screening to support disease monitoring and therapy stratification.
In 2022, Alryalat et al[22] developed the “Treatment Response Analyzer System”. This deep-learning framework combines segmentation of pretreatment OCT features with classification of the anatomical response to intravitreal anti-VEGF in DME. Their model matched ophthalmologist-level accuracy in stratifying patients as good or poor responders, illustrating how AI may extend automated diagnostics into precision-guided therapeutic decision-making[22]. These models have established a foundation for individualized ophthalmic care, enabling physicians to predict treatment responses and adjust follow-up accordingly.
In a landmark study, Dai et al[23] developed the DeepDR Plus deep-learning system, trained on 717308 fundus images from 179327 participants and validated on 118868 images from 29868 participants, to predict individualized time-to-progression of DR over a five-year horizon using baseline fundus photographs alone. The model achieved concordance indexes (C-index) of approximately 0.754-0.846 and integrated Brier scores of 0.153-0.241 in internal and external cohorts. Notably, in real-world deployment, the system could extend the mean screening interval from 12 months to about 32 months while maintaining a delayed detection rate of vision-threatening DR at 0.18%[23]. This work exemplifies how AI is transitioning from static detection to dynamic prognostication. This transition from static to dynamic prediction corresponds to growing recognition that DR is a heterogeneous and multifaceted condition affected by systemic, genetic, and metabolic factors.
For therapeutic response prediction in DME, Fang et al[24] developed an ensemble machine-learning model that integrates three-dimensional OCT features with clinical variables (e.g., VA, systemic risk factors) to predict good vs poor visual response at 1 month after 3 anti-VEGF injections. In 170 eyes, the ensemble model (combining AdaBoost, Gradient Boosting, and LightGBM) achieved an AUC of 0.976, with sensitivity of 0.94 and specificity of 0.88 on the test set, demonstrating that three-dimensional structural biomarkers and systemic data can be combined to support treatment decisions[24]. These developments signify a shift toward data-driven therapy stratification, improving resource allocation and reducing exposure to ineffective or invasive interventions. Moreover, an expanding body of work is exploring OCT “radiomics” and multimodal feature engineering to refine prognostic stratification. Meng et al[25] used machine learning on a rich set of OCT-derived radiomic features and clinical parameters to predict persistent DME after anti-VEGF therapy; their model successfully identified eyes at risk of incomplete anatomical resolution after multiple injections, highlighting the value of high-dimensional OCT descriptors for forecasting refractoriness.
Liang et al[26] extended this concept using an unsupervised machine-learning approach on radiomic features extracted from pre-treatment OCT volumes, clustering 234 DME eyes into four distinct phenotypes with significantly different anti-VEGF outcomes. This work suggests that AI-driven phenotyping may reveal latent disease subtypes beyond conventional morphological labels, with direct implications for individualized therapy intensity and agent selection. Furthermore, Lu et al[27] reported a machine-learning nomogram that combines OCT biomarkers with systemic blood-based metabolic and hematologic markers to predict anatomical response in DME treated with anti-VEGF. In parallel, generative and longitudinal modeling approaches are emerging. Baek et al[28] trained a generative adversarial network to predict long-term post-treatment OCT B-scans and visual outcomes for DME patients undergoing anti-VEGF therapy. By synthesizing future OCT morphology conditioned on baseline scans and early treatment response, their system provides a visual, patient-specific forecast of fluid resolution and changes in retinal architecture, potentially supporting shared decision-making about continued injections or treatment switching.
The rapid proliferation of these models has been systematically evaluated by Al-Harbi et al[29], who performed a systematic review and meta-analysis of AI tools predicting anti-VEGF response in DME. Across 23 eligible studies, they reported pooled sensitivity and specificity around the mid-80% range and an overall AUC close to 0.90, while also highlighting heterogeneity in outcome definitions, data quality, and validation strategies[29].
Collectively, these studies indicate that AI in diabetic eye disease is moving from descriptive lesion detection to predictive, therapy-guiding analytics. Fundus-based systems such as DeepDR Plus provide individualized time-to-progression estimates for DR. At the same time, OCT-based and radiomics-based models stratify DME patients by likely anatomical and functional response to anti-VEGF treatment. Meta-analytic evidence suggests that these tools achieve high diagnostic accuracy, but also reveals methodological heterogeneity and a lack of prospective impact studies. As these models are integrated with systemic data, electronic health records, and teleophthalmology platforms, they have the potential to support adaptive, data-driven care pathways – extending screening intervals for low-risk patients, in
The integration of systemic metabolic data with ophthalmic imaging represents a crucial advancement in personalized diabetic eye care. Traditional approaches to DR assessment rely primarily on visualizing retinal lesions and estimating macular thickness, but such localized information often fails to capture the multifactorial nature of diabetic complications. Advances in AI have enabled the integration of fundus and OCT imaging with systemic biomarkers such as glycated hemoglobin, serum lipids, renal function indices, and blood pressure profiles, thereby creating a multidimensional framework for individualized disease modeling. Alshammari et al[30] demonstrated in a comprehensive meta-analysis that AI-driven systems integrating systemic and ocular data outperformed single-modality models in both diagnostic precision and progression prediction, suggesting that cross-domain data fusion improves both sensitivity and specificity in real-world settings. These integrative approaches lay the foundation for precision ophthalmology, in which each patient’s risk profile and therapeutic plan can be tailored using dynamic, data-driven insights rather than static categorical classifications.
A prominent example of such multimodal integration is provided by Nderitu et al[31], who developed deep learning systems to predict the 1-year, 2-year, and 3-year risk of emergent referable DR and maculopathy using either clinical risk-factor characteristics, color fundus photographs, or a combination of both. In a cohort of more than 110000 eyes from the United Kingdom diabetic eye screening program, their multimodal model, which jointly ingested fundus images and clinicodemographic variables, consistently outperformed a tabular-only risk-factor model, achieving AUC values up to 0.95 for 1-year emergent referable DR and maintaining good performance across external validation datasets[31].
Beyond fundus photography, Sandhu et al[32] combined structural OCT, OCTA and common systemic biomarkers (age, sex, hypertension, hyperlipidemia, glycated hemoglobin) in a random-forest-based computer-aided diagnosis system for non-proliferative DR. When OCT data were used alone, the model achieved an AUC around 0.78; adding OCTA increased AUC to 0.92, and the full multimodal model – including clinical and demographic variables – reached an overall accuracy of 96%, sensitivity of 100%, specificity of 94%, and AUC of 0.96 for differentiating no DR vs mild vs moderate non-proliferative DR (NPDR)[32]. The bidirectional relationship between ocular findings and systemic microangiopathy has also been leveraged to predict extraocular outcomes. Zhao et al[33] showed that DR severity graded by a lesion-aware deep learning system on fundus photographs was a strong predictor of progression to end-stage renal disease in Chinese patients with biopsy-proven diabetic nephropathy.
In a complementary direction, Sabanayagam et al[34] developed deep learning algorithms to detect chronic kidney disease from retinal photographs in community populations; a hybrid model combining retinal images with clinical risk factors (age, sex, ethnicity, diabetes, hypertension) achieved AUCs up to 0.94 in internal validation and approximately 0.86-0.89 in external datasets, consistently outperforming image-only or risk-factor-only models. Together, these studies exemplify the “oculomics” paradigm, in which AI-extracted retinal features, contextualized by systemic variables, act as non-invasive biomarkers of kidney function and microvascular health. The clinical implications of these integrative frameworks are far-reaching. By correlating retinal phenotypes with systemic metabolic metrics, AI-driven models may help identify patients in whom retinal findings serve as early biomarkers of systemic microangiopathy. This concept is particularly relevant for endocrinologists and primary-care physicians, as it expands the role of ophthalmic imaging beyond eye health to encompass systemic disease surveillance. Studies conducted across European healthcare systems have begun to explore integrated electronic health records that allow seamless data exchange between diabetic clinics and ophthalmology departments, enabling real-time risk assessment. Such interoperability will be essential for the successful implementation of AI-based predictive platforms in routine care. As algorithms become capable of learning from both retinal and systemic information, their clinical utility will extend from diagnosing ocular disease to anticipating broader complications such as nephropathy or cardiovascular events.
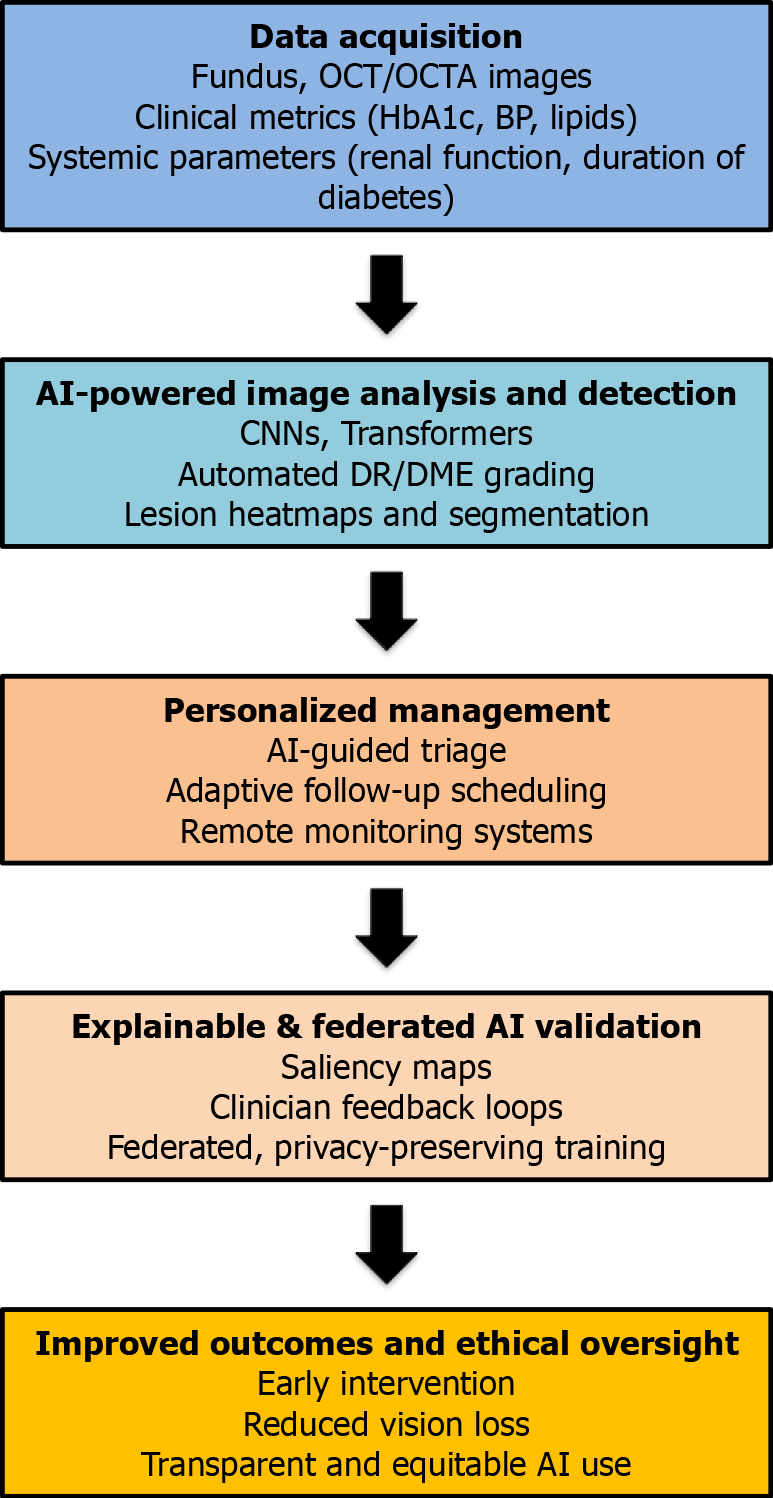
The integration of ocular and systemic data requires rigorous attention to data governance, privacy, and interpretability. The sensitive nature of medical records and imaging data necessitates secure frameworks for data storage, de-identification, and model transparency. Transparent modeling not only enhances clinician trust but also facilitates regulatory approval and ethical compliance in multicenter deployments. As this integrative ecosystem continues to expand, collaboration between data scientists, ophthalmologists, diabetologists, and policy experts will be vital to realize its full potential. When implemented responsibly, multimodal AI can evolve from a diagnostic adjunct to a true precision-medicine instrument, linking ocular biomarkers to systemic health outcomes in a unified predictive continuum. Figure 1 exemplifies how AI synthesizes multimodal ocular and systemic data to revolutionize the continuum of diabetic eye care. The diagram illustrates the sequential progression of AI from picture recognition to predictive analytics and personalized disease treatment. Incorporating explainable and federated AI methodologies enhances diagnostic accuracy while promoting physician trust, transparency, and patient-centered precision care.
Teleophthalmology has emerged as a powerful means of expanding diabetic eye care beyond specialist clinics, and AI is increasingly central to its scalability. By integrating deep-learning systems into remote screening workflows, teleophthalmology programs can achieve rapid, accurate triage of DR in settings where ophthalmologists are scarce. In a landmark example, nine primary care sites under Thailand’s national DR screening, where Ruamviboonsuk et al[35] demonstrated that a real-time AI system deployed across community clinics achieved sensitivity and specificity comparable to retina specialists, enabling immediate referral decisions without the need for onsite expertise. This development illustrates the democratization of diabetic eye screening, in which a simple image captured by a technician or nurse can initiate an automated clinical decision-making process, ultimately increasing coverage and reducing preventable blindness. In pri
In a complementary study, Liu et al[37] found that implementing automated retinal image analysis in community-based primary-care settings significantly increased adherence to follow-up among patients with positive screening results, suggesting that AI may enhance patient engagement by delivering immediate, comprehensible feedback. These findings underscore that the impact of AI extends beyond detection, influencing patient behavior and care continuity.
AI-enabled teleophthalmology has also shown particular value in low-resource regions. In the RAIDERS randomized trial in Rwanda, Mathenge et al[38] demonstrated that AI-based assessment of DR, coupled with on-site counselling, led to significantly higher referral uptake among patients identified with treatable disease – an important step toward reducing disparities in access to specialist care. A large Indian study by Nunez do Rio et al[39] further showed that deep-learning systems can accurately interpret handheld, non-mydriatic fundus images captured by field workers in community settings for referable DR/DME. These results illustrate the potential of portable imaging devices paired with cloud-based AI to support distributed screening models in underserved populations.
Beyond screening, remote monitoring systems are beginning to incorporate AI-driven longitudinal analysis. In the multicenter ACCESS randomized controlled trial, Wolf et al[40] demonstrated that autonomous AI delivered at the point of diabetes care dramatically increased completion of annual eye exams among adolescents and young adults and improved follow-through on positive referrals. Advances in imaging technologies and cloud infrastructure have further accelerated this evolution. Recent developments in handheld fundus cameras, smartphone-based retinal imaging, and portable OCT have enabled high-quality image acquisition in non-specialist environments. Cloud-based systems such as LuxIA – validated across multiple European centers – demonstrate that centralized AI interpretation can be consistently deployed across heterogeneous cameras and clinical settings, supporting large-scale teleophthalmology networks[41,42]. These platforms enable a “hub-and-spoke” model in which image capture is decentralized but diagnosis and quality control remain centrally coordinated, preserving accuracy while maximizing reach.
Despite these advances, the widespread adoption of AI-enabled teleophthalmology faces notable challenges. Privacy and data-security concerns are prominent, especially in cloud-based systems operating across jurisdictions with varying regulatory requirements. Interoperability barriers – including inconsistent imaging standards, variable internet connectivity, and fragmented electronic health record infrastructure – limit seamless scaling. Additionally, ensuring algorithmic fairness remains critical; models trained predominantly on high-income or homogeneous populations may underperform in diverse or underserved communities, potentially exacerbating existing inequities. Addressing these issues will require harmonized standards, transparent reporting of AI performance, and sustained international collaboration.
Nonetheless, the cumulative evidence indicates that AI-driven teleophthalmology can transform diabetic eye care by shifting the paradigm from episodic, specialist-centered evaluation to continuous, accessible, and data-driven monitoring. By combining remote image acquisition, automated analysis, and predictive risk modelling, these systems can create adaptive care pathways that facilitate early intervention, reduce preventable blindness, and optimize healthcare resources. As digital health infrastructure expands, AI-enabled teleophthalmology is poised to become a central component of global DR screening and management strategies.
With the growing integration of AI in diabetic eye care, the demand for openness and interpretability has emerged as a critical issue. Initial deep learning models, albeit exhibiting exceptional diagnostic precision, functioned as “black boxes”, delivering predictions without elucidating the underlying reasoning mechanisms. This opacity has generated concern among physicians and regulators, especially in critical medical decision-making where explainability is essential for trust and responsibility. Explainable AI (XAI) has developed into a crucial subdiscipline, dedicated to guaranteeing that machine-learning systems can deliver human-comprehensible justifications for their predictions while preserving performance integrity.
Sayres et al[43] developed an interpretable, attention-based system where heatmaps were reviewed by retinal specialists to assess whether the model’s highlighted regions aligned with human diagnostic reasoning, demonstrating that interpretability can directly enhance clinician trust. Complementing this approach, Ayhan et al[44] performed the first clinical validation of saliency maps in ophthalmology, showing that although these visualizations can help reveal model reasoning, they require careful interpretation due to variability across methods. Beyond clinical governance, XAI also shapes patient trust and acceptance. In a recent prospective study, Krogh et al[45] found that patients undergoing AI-assisted DR screening were more comfortable with automated assessments when confidence scores and visual explanations accompanied the results. Such transparency helps patients understand why they are being referred – or not referred – and supports shared decision-making in chronic disease management. As AI increasingly contributes not only to screening but also to prognostication and treatment stratification, the need for patient-facing explainability will continue to grow.
Recent work in diabetic eye care provides concrete examples of how explainability can be embedded into model design rather than added as an afterthought. Sharma and Lalwani[46] proposed a multi-model deep network for DR seg
Ikram and Imran[47] introduced the ResViT FusionNet model for automated DR grading from fundus photographs, fusing ResNet-50 with a Vision Transformer backbone and coupling it with Layer-wise Interpretable Model-agnostic Explanations and Grad-CAM to interrogate predictions. Their model achieved an accuracy of approximately 93% with high precision and an F1 score. At the same time, explanation maps are systematically localized to clinically plausible regions, such as areas of venous beading, cotton-wool spots, and neovascular complexes[47]. Explainable methods are also beginning to address data-scarce modalities such as macular OCTA.
Movassagh et al[48] developed a few-shot learning framework using pre-trained CNN backbones combined with a self-attention “Match-Them-Up” network to classify OCTA scans into NPDR and PDR. Their ResNet-50 few-shot model reached an AUC of 0.82 with an accuracy of 76.2%, and the attention-based XAI module produced class-specific maps that emphasized non-perfusion zones in NPDR and neovascular tufts in PDR[48].
Beyond pure classification, XAI is also being used to discover and validate novel structural correlates of visual function. Yoshida et al[49] fine-tuned a RETFound-OCT foundation model to infer VA from macular OCT scans in DR patients and then applied layer-wise relevance propagation to generate attention maps. The model discriminated eyes with poor vision (decimal VA ≤ 0.5) from those with good vision (VA ≥ 1.0) with an AUC of 0.947, and XAI visualizations systematically highlighted the ganglion cell/inner plexiform layer, outer nuclear layer, ellipsoid zone, and choroid as key contributors[49].
Explainability is not limited to imaging. In a metabolomics-based framework, Yagin et al[50] applied a hybrid ensemble model (support vector machine classifier + multi-layer perceptron) to targeted serum metabolomics from patients with no DR, NPDR and PDR. They used SHapley Additive exPlanations (SHAP) to quantify the contribution of each metabolite to the predicted stage. Their model achieved an accuracy close to 90%, and SHAP analysis consistently ranked glucose, glycine, and age as key features across DR classes. At the same time, creatinine and several phosphatidylcholines emerged as particularly important in PDR[50]. At the level of fundus-based screening pipelines, newer architectures such as HIRD-Net further underscore that explainability can be tightly intertwined with image enhancement and attention mechanisms.
Ashraf et al[51] proposed a hierarchical inception-residual-dense CNN for DR diagnosis that incorporates contrast-limited adaptive histogram equalization with a dilated Difference-of-Gaussian filter to enhance lesion visibility and uses squeeze-and-excitation channel attention to focus on salient features. Grad-CAM is then applied to generate heatmaps over CLAHE- and D-DoG-enhanced images, allowing clinicians to visually relate the network’s focus to microaneurysms, hemorrhages and exudates across multiple scales[51].
Taken together, these studies suggest that explainability in ophthalmic AI is evolving from isolated saliency maps to integrated frameworks in which visualization, feature attribution and clinician feedback are co-designed with the underlying model. At the same time, they reinforce that explanation quality itself must be rigorously evaluated: (1) Attention maps should be tested against independent clinical labels or expert expectations; (2) Attribution methods such as SHAP require careful handling to avoid spurious importance rankings; and (3) Human – AI interaction studies are needed to ensure that visually compelling heatmaps do not create over-trust. As regulatory bodies begin to consider requirements for algorithmic transparency in screening and treatment planning, these XAI-driven DR systems offer a template for how high-performing models can justify their decisions in ways that are scientifically meaningful, clinically verifiable and, crucially, intelligible to both physicians and patients. Despite these advancements, major challenges remain. Visualization techniques can be inconsistent or misleading if not rigorously validated, and efforts to simplify models to enhance interpretability may compromise diagnostic accuracy.
AI represents the ethical and scientific advancement of the discipline. It converts AI from a computational curiosity into a reliable element of medical reasoning. The incorporation of interpretability into model design is consistent with the core tenets of evidence-based medicine: (1) Reproducibility; (2) Transparency; and (3) Accountability. The evolution of AI systems will determine their validity in clinical settings based on their ability to justify decisions, reveal biases, and deliver verifiable outputs. XAI fosters clarity and mutual comprehension among developers, physicians, and patients, hence improving diagnostic precision and bolstering the essential trust integral to good healthcare.
The increasing use of AI in diabetic eye care has presented various ethical, legal, and practical challenges that surpass mere algorithmic efficacy. Although AI has unparalleled prospects for early identification and personalized management, it simultaneously engenders essential inquiries around accountability, patient autonomy, and equity in healthcare provision. Ethical analyses have highlighted that the introduction of autonomous or semi-autonomous DR algorithms fundamentally reshapes traditional notions of consent, responsibility and fairness in screening programs.
Ursin et al[52] proposed a detailed checklist of information that should be disclosed to patients when DR is diagnosed using AI in primary care, including the autonomous nature of the system, its validated accuracy, possible error modes, and procedures for escalation, underscoring that conventional generic consent is insufficient in this context. More broadly, Evans et al[53] identified three central ethical concerns for ophthalmic AI: (1) The need for transparency and interpretability; (2) The attribution of responsibility when harm occurs; and (3) The scalable and equitable deployment of screening infrastructures. Complementing these general frameworks, recent DR-specific narrative reviews have catalogued concrete ethical risks, including opacity of proprietary algorithms, the potential for automation bias among non-specialists, and the risk that commercially driven deployments may prioritize high-income settings and further marginalize under-screened populations[54].
Data governance and privacy constitute another critical axis of concern. Retinal imaging datasets used to train DR algorithms are often assembled from multiple institutions and jurisdictions, raising questions about secondary data use, cross-border transfer, and compliance with data-protection legislation such as the GDPR. Recent ophthalmology-focused ethics reviews note that retinal images and multimodal biomarker profiles can be indirectly identifiable and might reveal systemic health information beyond eye disease, strengthening the obligation to implement robust de-identification, access control and cybersecurity measures[55]. Furthermore, stakeholder interviews in DR programs have pointed out that patients and clinicians often have limited understanding of how images are stored, reused or shared with com
From a regulatory perspective, most clinically deployed systems have been evaluated and approved as software-as-a-medical-device for a narrowly defined indication, typically the detection of more-than-mild DR in adults with diabetes. The first autonomous AI system in any field of medicine to receive Food and Drug Administration authorization was IDx-DR, which demonstrated high sensitivity and specificity for more-than-mild DR and DME in a pivotal multicenter trial and was subsequently cleared for use without immediate clinician over-read[3,57]. Similar evidence-based pathways have supported the deployment of the EyeArt automated DR detection system, with prospective studies in screening populations showing clinically acceptable sensitivity for referable and vision-threatening DR and paving the way for regulatory approvals in the United States and other jurisdictions[58].
In Europe, AI-based ophthalmic tools, including screening algorithms for DR , are regulated under a dual framework. On one side, the EU Medical Device Regulation (MDR 2017/745) requires conformity assessment, clinical evaluation and post-market surveillance, with most DR-detection software classified as class IIa (or higher) medical devices[59]. On the other side, with the entry into force of the EU AI Act in August 2024, medical-device AI is considered “high-risk” AI and must comply with additional obligations – including risk management, data governance, technical documentation, transparency and human oversight[60]. As a result, developers and deployers must navigate overlapping and sometimes redundant compliance requirements, raising the bar for conformity assessment and potentially slowing down innovation or limiting market access especially for smaller research-driven teams[61].
Regulatory, ethical and implementation questions are not external constraints but constitutive elements of safe AI-enabled diabetic eye care, determining whether algorithms translate into durable improvements in vision outcomes, patient experience and health-system resilience. Successful growth necessitates a resilient digital infrastructure, encompassing secure data pipelines, interoperability across electronic medical records, and ongoing technical assistance. In the absence of these elements, even thoroughly proven algorithms are likely to remain as pilot projects instead of realizing systemic impact. Concurrently addressing ethical, regulatory, and logistical problems will ascertain whether AI realizes its potential as a transformative instrument for equitable and sustainable diabetic eye care.
On the innovation side, foundation models and self-supervised learning are poised to redefine how retinal AI is developed. The RETFound model, trained with masked autoencoding on 1.6 million unlabeled retinal images, has demonstrated that a single large vision transformer can be efficiently adapted to a variety of downstream tasks – including DR detection – with relatively small labelled datasets, achieving state-of-the-art performance and improved generalizability compared with task-specific networks[62]. Subsequent work has shown that RETFound-derived representations can support not only diagnosis but also prognosis across common ocular diseases, suggesting a path towards multi-task platforms that combine screening, risk stratification and treatment planning within a unified architecture[63].
Multimodal foundation models that integrate fundus photographs, OCT/OCTA, clinical variables and even language-based clinical notes are being explored, with early prototypes indicating that joint visual–language representations could enable more nuanced decision support and patient-specific explanations[64]. Parallel advances are occurring in privacy-preserving model development. Federated learning (FL) has been proposed as a strategy to train high-performing DR detectors without centralizing images, thereby reducing legal and ethical concerns about cross-border data transfer. In a simulation based on nearly 700000 fundus photographs from the French OPHDIAT screening network, Matta et al[65] showed that an FL framework could achieve performance comparable to centrally trained models for DR screening. Other FL implementations using data from multiple institutions have confirmed the feasibility of distributed training, including transformer-based architectures and more recent privacy-preserving models such as FedDRNet[66]. These approaches illustrate how future DR algorithms may be routinely trained or updated across international networks while keeping patient data within local jurisdictions, an especially relevant direction in the context of GDPR and the EU AI Act.
Fairness and bias mitigation represent another crucial frontier. Early work in retinal AI showed that performance can vary by race, sex or ethnicity, raising concerns that naïvely deployed systems could exacerbate existing inequities in diabetic eye care[67,68]. More recently, Tan et al[69] proposed FairerOPTH, an “implicit fairness learning” framework that reduces sex-related and age-related performance gaps in ophthalmic diagnosis without substantially sacrificing overall accuracy. At the same time, a 2025 analysis of sociodemographic reporting in ophthalmic AI highlighted that many studies fail to report basic demographic breakdowns of training and test cohorts, making it difficult to assess or correct for bias[11].
Future DR models will therefore need to incorporate fairness-aware objectives, systematic subgroup analyses and transparent reporting as standard components of development and validation pipelines. From a health-economic and safety point of view, evaluations now suggest that, when embedded into structured pathways, AI-assisted screening can be cost-effective and safe, but also stress that local prevalence, infrastructure, and referral capacity critically influence value[70]. Future work will need to extend these analyses to low-income and middle-income countries, where resource constraints, camera availability and follow-up capacity may differ profoundly from the contexts in which most algo
Despite the accelerating sophistication of AI-driven ophthalmic tools, several persistent limitations must be acknowledged. First, the majority of DR algorithms have been trained and validated on curated datasets that may not reflect the heterogeneity of real-world populations, comorbid eye diseases, media opacities, or acquisition conditions. A recent evaluation of machine-learning and deep-learning algorithms for DR detection highlighted substantial variation in reported performance, limited external validation, and frequent absence of head-to-head comparison across methods, making it difficult to draw firm conclusions about comparative effectiveness[71].
Generalizability across devices, ethnic groups and healthcare settings remains uncertain. This limitation is particularly relevant for DR care in under-represented regions, where fundus cameras, image quality, and disease spectra may diverge from those in large tertiary datasets.
Current systems often lack robust mechanisms for detecting domain shift and silent failure. Most DR algorithms do not continuously monitor their own performance after deployment, nor do they routinely flag out-of-distribution inputs beyond simple image-quality checks, few implementations systematically track post-market performance, re-calibrate thresholds, or retrain models in response to changing populations or hardware, raising concerns about gradual degradation over time[17,72].
Beyond explainability, ethical deployment of AI in diabetic eye care requires systematic algorithmic and equity audits. While many published models report excellent performance metrics (often with AUC values exceeding 0.90), these results predominantly derive from curated datasets collected in high-income settings with controlled imaging conditions. Evidence from low-resource environments and underrepresented populations – including African and Indigenous communities – remains comparatively sparse, raising concerns about generalizability and algorithmic fairness. The absence of standardized bias-risk or equity assessments across studies introduces the possibility of selection and publication bias, whereby high-performing algorithms are preferentially reported, while models that underperform in heterogeneous or resource-limited settings remain underrepresented. Consequently, reported accuracy metrics should be interpreted cautiously and not conflated with clinical readiness for universal deployment. Equity audits – assessing performance across race, ethnicity, sex, socioeconomic status, and imaging context – should become a routine component of AI validation pipelines, alongside post-deployment monitoring to detect performance drift and unintended disparities.
Finally, there is an ongoing mismatch between what is technically feasible and what is practically implemented, despite hundreds of published models, relatively few have been evaluated in prospective multicenter trials, embedded into national screening programs, or compared head-to-head with human graders across multiple health systems[73]. Barriers include regulatory complexity, integration with electronic health records, reimbursement uncertainty, limited clinician training, and resistance to workflow changes. Emerging risks associated with next-generation AI architectures also warrant consideration. Multimodal models that integrate imaging, clinical variables, and free-text data – particularly those incorporating large language model components – may be susceptible to so-called “hallucinations”, whereby plausible but incorrect inferences or explanations are generated. In clinical contexts, such errors could lead to inappropriate risk stratification or misplaced clinician confidence if outputs are not carefully constrained and validated.
In parallel, the increasing reliance on cloud-based infrastructures for image storage and AI inference introduces cybersecurity and data-integrity risks, including unauthorized access, model tampering, and disruption of service availability. These vulnerabilities are particularly relevant for large-scale screening programs and cross-border deployments. Mitigation strategies – including secure encryption, continuous system auditing, human-in-the-loop oversight, and fail-safe clinical workflows – should therefore be considered integral to responsible AI implementation rather than ancillary technical concerns. In this sense, the “future direction” for AI in diabetic eye care is not only more sophisticated algorithms, but also more robust clinical trials, standardized reporting, participatory design with patients and clinicians, and infrastructure investments that allow high-performing models to be deployed safely and equitably across diverse health systems. Taken together, these trends suggest a dual trajectory. On one hand, methodological innovations – foundation models, multimodal learning, federated training and fairness-aware optimization – will likely continue to push technical performance and versatility. On the other, the field must confront enduring limitations related to generalizability, bias, monitoring, and health-system integration. Closing this gap will determine whether AI in diabetic eye care evolves from an impressive diagnostic technology into a mature, trustworthy component of chronic disease management that measurably reduces visual impairment at the population level.
Many of the limitations observed in current AI applications for DR and DME stem from structural constraints in data acquisition, annotation, and deployment rather than from algorithmic shortcomings alone. Most existing models are trained on curated datasets because real-world retinal imaging is characterized by substantial variability in image quality, illumination, field definition, and coexisting ocular pathology. Additionally, the creation of large, representative datasets is hindered by the high cost and time burden of expert annotation, limited availability of retinal specialists, and the absence of universally adopted labeling standards. Further obstacles arise from data governance and privacy regulations, which complicate multicenter data sharing and limit the aggregation of diverse real-world cohorts. As a result, many datasets overrepresent well-resourced healthcare systems and underrepresent low-resource settings, contributing to reduced generalizability and performance degradation when models are deployed outside their training domain.
Addressing these limitations requires a systematic, multi-level strategy. At the data level, solutions include standardized image acquisition protocols, semi-supervised and self-supervised learning to reduce annotation burden, FL frameworks to enable cross-institutional training without centralized data sharing, and deliberate inclusion of underrepresented populations and imaging conditions. At the model level, robustness can be enhanced through domain adaptation techniques, uncertainty estimation, explicit domain-shift detection mechanisms, and continuous post-deployment monitoring with recalibration strategies. At the regulatory and clinical application level, successful translation depends on prospective multicenter validation, transparent reporting of subgroup performance, integration of human-in-the-loop safeguards, and clear accountability structures aligned with evolving regulatory frameworks. Taken together, this layered approach highlights that improving AI readiness for real-world diabetic eye care is not solely a technical challenge, but a socio-technical one that requires coordinated advances in data infrastructure, model design, clinical workflow integration, and regulatory governance.
In the future, international consortia and public-private partnerships will be crucial in establishing standards for validation, interoperability, and ethical compliance. As AI advances, its development will progressively embody not only technical innovation but also societal values – transparency, inclusion, and patient-centeredness. The forthcoming generation of AI systems must consequently reconcile performance with accountability, amalgamating powerful analytics with human empathy. When adhering to these principles, AI will genuinely fulfill its disruptive promise, revolutionizing diabetic eye care into a predictive, preventive, and personalized field.
AI has evolved from a phase of experimental potential to a reliable clinical partner in the treatment of diabetic eye disease; yet, its most significant influence is probably still to come. The integration of retinal imaging, systemic data, and predictive analytics is revolutionizing DR from a condition identified too late into one that can be foreseen, treated, and controlled with accuracy. The essential message is unequivocal: AI must not be perceived as a substitute for clinical competence, but rather as a catalyst for transforming the thought processes, decision-making, and delivery of treatment by ophthalmologists. The persistent challenges – data bias, interpretability deficiencies, and unequal global access – necessitate that the forthcoming phase of innovation be directed by ethics and equity alongside computational capability.
Ironically, as AI nears human-level diagnostic proficiency, the necessity for human oversight intensifies, underscoring that transparency and empathy should underpin every algorithmic choice. The challenge lies in the notion that the genuine revolution may not be technological, but rather philosophical: A transition from perceiving AI as a mere instrument to acknowledging it as a partner in clinical reasoning. The forthcoming revelation will not be that machines possess visual capabilities, but rather that they may enable us to perceive disease – and the patient – more comprehensively than ever before.
| 1. | Gulshan V, Peng L, Coram M, Stumpe MC, Wu D, Narayanaswamy A, Venugopalan S, Widner K, Madams T, Cuadros J, Kim R, Raman R, Nelson PC, Mega JL, Webster DR. Development and Validation of a Deep Learning Algorithm for Detection of Diabetic Retinopathy in Retinal Fundus Photographs. JAMA. 2016;316:2402-2410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6165] [Cited by in RCA: 3556] [Article Influence: 355.6] [Reference Citation Analysis (4)] |
| 2. | Ting DSW, Cheung CY, Lim G, Tan GSW, Quang ND, Gan A, Hamzah H, Garcia-Franco R, San Yeo IY, Lee SY, Wong EYM, Sabanayagam C, Baskaran M, Ibrahim F, Tan NC, Finkelstein EA, Lamoureux EL, Wong IY, Bressler NM, Sivaprasad S, Varma R, Jonas JB, He MG, Cheng CY, Cheung GCM, Aung T, Hsu W, Lee ML, Wong TY. Development and Validation of a Deep Learning System for Diabetic Retinopathy and Related Eye Diseases Using Retinal Images From Multiethnic Populations With Diabetes. JAMA. 2017;318:2211-2223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1963] [Cited by in RCA: 1378] [Article Influence: 153.1] [Reference Citation Analysis (5)] |
| 3. | Abràmoff MD, Lavin PT, Birch M, Shah N, Folk JC. Pivotal trial of an autonomous AI-based diagnostic system for detection of diabetic retinopathy in primary care offices. NPJ Digit Med. 2018;1:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1257] [Cited by in RCA: 882] [Article Influence: 110.3] [Reference Citation Analysis (5)] |
| 4. | Xie Y, Nguyen QD, Hamzah H, Lim G, Bellemo V, Gunasekeran DV, Yip MYT, Qi Lee X, Hsu W, Li Lee M, Tan CS, Tym Wong H, Lamoureux EL, Tan GSW, Wong TY, Finkelstein EA, Ting DSW. Artificial intelligence for teleophthalmology-based diabetic retinopathy screening in a national programme: an economic analysis modelling study. Lancet Digit Health. 2020;2:e240-e249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 185] [Article Influence: 30.8] [Reference Citation Analysis (1)] |
| 5. | Wang Y, Shi D, Tan Z, Niu Y, Jiang Y, Xiong R, Peng G, He M. Screening Referable Diabetic Retinopathy Using a Semi-automated Deep Learning Algorithm Assisted Approach. Front Med (Lausanne). 2021;8:740987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 6. | Zafar S, Mahjoub H, Mehta N, Domalpally A, Channa R. Artificial Intelligence Algorithms in Diabetic Retinopathy Screening. Curr Diab Rep. 2022;22:267-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 7. | Yang P, Yang B. Development and validation of predictive models for diabetic retinopathy using machine learning. PLoS One. 2025;20:e0318226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 8. | Huang X, Wang H, She C, Feng J, Liu X, Hu X, Chen L, Tao Y. Artificial intelligence promotes the diagnosis and screening of diabetic retinopathy. Front Endocrinol (Lausanne). 2022;13:946915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 9. | Kim IM, Radgoudarzi N, Chen EM, Liu J, Moussa K, Shantha JG, Tsui E, Gore C, Baxter SL, Porco TC, Arnold BF, Sun CQ. Time to Progression to Proliferative Diabetic Retinopathy in Patients With Type 2 Diabetes. JAMA Netw Open. 2025;8:e2521150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 10. | Mohsen F, Ali H, El Hajj N, Shah Z. Artificial intelligence-based methods for fusion of electronic health records and imaging data. Sci Rep. 2022;12:17981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 99] [Article Influence: 24.8] [Reference Citation Analysis (2)] |
| 11. | Bondok M, Selvakumar R, Asdo A, Naderi B, Zhang C, Wong C, Felfeli T. Sociodemographic Reporting in Artificial Intelligence Studies of Retinal Diseases: A Critical Appraisal of the Literature. Ophthalmol Retina. 2025;9:1133-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 12. | Bellemo V, Lim ZW, Lim G, Nguyen QD, Xie Y, Yip MYT, Hamzah H, Ho J, Lee XQ, Hsu W, Lee ML, Musonda L, Chandran M, Chipalo-Mutati G, Muma M, Tan GSW, Sivaprasad S, Menon G, Wong TY, Ting DSW. Artificial intelligence using deep learning to screen for referable and vision-threatening diabetic retinopathy in Africa: a clinical validation study. Lancet Digit Health. 2019;1:e35-e44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 213] [Article Influence: 30.4] [Reference Citation Analysis (4)] |
| 13. | Lee CS, Tyring AJ, Deruyter NP, Wu Y, Rokem A, Lee AY. Deep-learning based, automated segmentation of macular edema in optical coherence tomography. Biomed Opt Express. 2017;8:3440-3448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 212] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 14. | Schlegl T, Waldstein SM, Bogunovic H, Endstraßer F, Sadeghipour A, Philip AM, Podkowinski D, Gerendas BS, Langs G, Schmidt-Erfurth U. Fully Automated Detection and Quantification of Macular Fluid in OCT Using Deep Learning. Ophthalmology. 2018;125:549-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 376] [Article Influence: 47.0] [Reference Citation Analysis (1)] |
| 15. | De Fauw J, Ledsam JR, Romera-Paredes B, Nikolov S, Tomasev N, Blackwell S, Askham H, Glorot X, O'Donoghue B, Visentin D, van den Driessche G, Lakshminarayanan B, Meyer C, Mackinder F, Bouton S, Ayoub K, Chopra R, King D, Karthikesalingam A, Hughes CO, Raine R, Hughes J, Sim DA, Egan C, Tufail A, Montgomery H, Hassabis D, Rees G, Back T, Khaw PT, Suleyman M, Cornebise J, Keane PA, Ronneberger O. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med. 2018;24:1342-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2083] [Cited by in RCA: 1315] [Article Influence: 164.4] [Reference Citation Analysis (5)] |
| 16. | Poschkamp B, Stahl A. Application of deep learning algorithms for diabetic retinopathy screening. Ann Transl Med. 2022;10:1298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 17. | Kong M, Song SJ. Artificial Intelligence Applications in Diabetic Retinopathy: What We Have Now and What to Expect in the Future. Endocrinol Metab (Seoul). 2024;39:416-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 18. | Ye X, Gao K, He S, Zhong X, Shen Y, Wang Y, Shao H, Shen L. Artificial Intelligence-Based Quantification of Central Macular Fluid Volume and VA Prediction for Diabetic Macular Edema Using OCT Images. Ophthalmol Ther. 2023;12:2441-2452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 19. | Rodríguez-Miguel A, Arruabarrena C, Allendes G, Olivera M, Zarranz-Ventura J, Teus MA. Hybrid deep learning models for the screening of Diabetic Macular Edema in optical coherence tomography volumes. Sci Rep. 2024;14:17633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 20. | Midena E, Lupidi M, Toto L, Covello G, Veritti D, Pilotto E, Cicinelli MV, Lattanzio R, Figus M, Midena G, Danieli L, Borrelli E, Reibaldi M, Tognetto D, Inferrera L, Donati S, Rossi S, Melillo P, Lanzetta P, Sarao V, Gregori G, Cagini C, Eandi CM, Carnevali A, Scorcia V, Maggio E, Pertile G, Costagliola C, Cennamo G, Mora P, Dell'Omo R, Affatato M, Passamonti M, Parravano M, Lassandro NV, Nassisi M, Viola F, Castellino N, Cappellani F, Giannaccare G, Boscia F, Grassi MO, Musetti D, Folegani V, Invernizzi A, Rossetti L, Bacci T, Ricci F, Lombardo M, Romano M, Valsecchi N, Coppola M, Cavarzeran F, Frizziero L. AI-Assisted OCT Clinical Phenotypes of Diabetic Macular Edema: A Large Cohort Clustering Study. J Clin Med. 2025;14:7893. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 21. | Cao J, You K, Jin K, Lou L, Wang Y, Chen M, Pan X, Shao J, Su Z, Wu J, Ye J. Prediction of response to anti-vascular endothelial growth factor treatment in diabetic macular oedema using an optical coherence tomography-based machine learning method. Acta Ophthalmol. 2021;99:e19-e27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 22. | Alryalat SA, Al-Antary M, Arafa Y, Azad B, Boldyreff C, Ghnaimat T, Al-Antary N, Alfegi S, Elfalah M, Abu-Ameerh M. Deep Learning Prediction of Response to Anti-VEGF among Diabetic Macular Edema Patients: Treatment Response Analyzer System (TRAS). Diagnostics (Basel). 2022;12:312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 23. | Dai L, Sheng B, Chen T, Wu Q, Liu R, Cai C, Wu L, Yang D, Hamzah H, Liu Y, Wang X, Guan Z, Yu S, Li T, Tang Z, Ran A, Che H, Chen H, Zheng Y, Shu J, Huang S, Wu C, Lin S, Liu D, Li J, Wang Z, Meng Z, Shen J, Hou X, Deng C, Ruan L, Lu F, Chee M, Quek TC, Srinivasan R, Raman R, Sun X, Wang YX, Wu J, Jin H, Dai R, Shen D, Yang X, Guo M, Zhang C, Cheung CY, Tan GSW, Tham YC, Cheng CY, Li H, Wong TY, Jia W. A deep learning system for predicting time to progression of diabetic retinopathy. Nat Med. 2024;30:584-594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 186] [Cited by in RCA: 101] [Article Influence: 50.5] [Reference Citation Analysis (1)] |
| 24. | Fang Y, Lin J, Xie P, Zhu H, Ng TK, Zhang G. Ensemble machine learning algorithm for anti-VEGF treatment efficacy prediction in diabetic macular edema. BMC Ophthalmol. 2025;25:352. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 25. | Meng Z, Chen Y, Li H, Zhang Y, Yao X, Meng Y, Shi W, Liang Y, Hu Y, Liu D, Xie M, Yan B, Luo J. Machine learning and optical coherence tomography-derived radiomics analysis to predict persistent diabetic macular edema in patients undergoing anti-VEGF intravitreal therapy. J Transl Med. 2024;22:358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 26. | Liang X, Luo S, Liu Z, Liu Y, Luo S, Zhang K, Li L. Unsupervised machine learning analysis of optical coherence tomography radiomics features for predicting treatment outcomes in diabetic macular edema. Sci Rep. 2025;15:13389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 27. | Lu W, Xiao K, Zhang X, Wang Y, Chen W, Wang X, Ye Y, Lou Y, Li L. A machine learning model for predicting anatomical response to Anti-VEGF therapy in diabetic macular edema. Front Cell Dev Biol. 2025;13:1603958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 28. | Baek J, He Y, Emamverdi M, Mahmoudi A, Nittala MG, Corradetti G, Ip M, Sadda SR. Prediction of Long-Term Treatment Outcomes for Diabetic Macular Edema Using a Generative Adversarial Network. Transl Vis Sci Technol. 2024;13:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 29. | Al-Harbi FA, Alkuwaiti MA, Alharbi MA, Alessa AA, Alhassan AA, Aleidan EA, Al-Theyab FY, Alfalah M, AlHaddad SM, Azzam AY. Diagnostic Accuracy of Artificial Intelligence in Predicting Anti-VEGF Treatment Response in Diabetic Macular Edema: A Systematic Review and Meta-Analysis. J Clin Med. 2025;14:8177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 30. | Alshammari RD, Haliqa WMA, Majzoub RY. Diagnostic accuracy of AI or DL-enhanced technologies in the diagnosis of diabetic retinopathy: a systematic review. J Med Imaging Intervent Radiol. 2025;12:39. [DOI] [Full Text] |
| 31. | Nderitu P, Nunez do Rio JM, Webster L, Mann S, Cardoso MJ, Modat M, Hopkins D, Bergeles C, Jackson TL. Predicting 1, 2 and 3 year emergent referable diabetic retinopathy and maculopathy using deep learning. Commun Med (Lond). 2024;4:167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 32. | Sandhu HS, Elmogy M, Taher Sharafeldeen A, Elsharkawy M, El-Adawy N, Eltanboly A, Shalaby A, Keynton R, El-Baz A. Automated Diagnosis of Diabetic Retinopathy Using Clinical Biomarkers, Optical Coherence Tomography, and Optical Coherence Tomography Angiography. Am J Ophthalmol. 2020;216:201-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 33. | Zhao L, Ren H, Zhang J, Cao Y, Wang Y, Meng D, Wu Y, Zhang R, Zou Y, Xu H, Li L, Zhang J, Cooper ME, Tong N, Liu F. Diabetic Retinopathy, Classified Using the Lesion-Aware Deep Learning System, Predicts Diabetic End-Stage Renal Disease in Chinese Patients. Endocr Pract. 2020;26:429-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (5)] |
| 34. | Sabanayagam C, Xu D, Ting DSW, Nusinovici S, Banu R, Hamzah H, Lim C, Tham YC, Cheung CY, Tai ES, Wang YX, Jonas JB, Cheng CY, Lee ML, Hsu W, Wong TY. A deep learning algorithm to detect chronic kidney disease from retinal photographs in community-based populations. Lancet Digit Health. 2020;2:e295-e302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 144] [Article Influence: 24.0] [Reference Citation Analysis (1)] |
| 35. | Ruamviboonsuk P, Tiwari R, Sayres R, Nganthavee V, Hemarat K, Kongprayoon A, Raman R, Levinstein B, Liu Y, Schaekermann M, Lee R, Virmani S, Widner K, Chambers J, Hersch F, Peng L, Webster DR. Real-time diabetic retinopathy screening by deep learning in a multisite national screening programme: a prospective interventional cohort study. Lancet Digit Health. 2022;4:e235-e244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 120] [Article Influence: 30.0] [Reference Citation Analysis (1)] |
| 36. | Dow ER, Khan NC, Chen KM, Mishra K, Perera C, Narala R, Basina M, Dang J, Kim M, Levine M, Phadke A, Tan M, Weng K, Do DV, Moshfeghi DM, Mahajan VB, Mruthyunjaya P, Leng T, Myung D. AI-Human Hybrid Workflow Enhances Teleophthalmology for the Detection of Diabetic Retinopathy. Ophthalmol Sci. 2023;3:100330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 37. | Liu J, Gibson E, Ramchal S, Shankar V, Piggott K, Sychev Y, Li AS, Rao PK, Margolis TP, Fondahn E, Bhaskaranand M, Solanki K, Rajagopal R. Diabetic Retinopathy Screening with Automated Retinal Image Analysis in a Primary Care Setting Improves Adherence to Ophthalmic Care. Ophthalmol Retina. 2021;5:71-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 38. | Mathenge W, Whitestone N, Nkurikiye J, Patnaik JL, Piyasena P, Uwaliraye P, Lanouette G, Kahook MY, Cherwek DH, Congdon N, Jaccard N. Impact of Artificial Intelligence Assessment of Diabetic Retinopathy on Referral Service Uptake in a Low-Resource Setting: The RAIDERS Randomized Trial. Ophthalmol Sci. 2022;2:100168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 39. | Nunez do Rio JM, Nderitu P, Bergeles C, Sivaprasad S, Tan GSW, Raman R. Evaluating a Deep Learning Diabetic Retinopathy Grading System Developed on Mydriatic Retinal Images When Applied to Non-Mydriatic Community Screening. J Clin Med. 2022;11:614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 40. | Wolf RM, Channa R, Liu TYA, Zehra A, Bromberger L, Patel D, Ananthakrishnan A, Brown EA, Prichett L, Lehmann HP, Abramoff MD. Autonomous artificial intelligence increases screening and follow-up for diabetic retinopathy in youth: the ACCESS randomized control trial. Nat Commun. 2024;15:421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 70] [Article Influence: 35.0] [Reference Citation Analysis (1)] |
| 41. | Blair JPM, Rodriguez JN, Lasagni Vitar RM, Stadelmann MA, Abreu-González R, Donate J, Ciller C, Apostolopoulos S, Bermudez C, De Zanet S. Development of LuxIA, a Cloud-Based AI Diabetic Retinopathy Screening Tool Using a Single Color Fundus Image. Transl Vis Sci Technol. 2023;12:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 42. | Abreu-Gonzalez R, Susanna-González G, Blair JPM, Lasagni Vitar RM, Ciller C, Apostolopoulos S, De Zanet S, Rodríguez Martín JN, Bermúdez C, Calle Pascual AL, Rigo E, Cervera Taulet E, Escobar-Barranco JJ, Cobo-Soriano R, Donate-Lopez J. Validation of artificial intelligence algorithm LuxIA for screening of diabetic retinopathy from a single 45° retinal colour fundus images: the CARDS study. BMJ Open Ophthalmol. 2025;10:e002109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 43. | Sayres R, Taly A, Rahimy E, Blumer K, Coz D, Hammel N, Krause J, Narayanaswamy A, Rastegar Z, Wu D, Xu S, Barb S, Joseph A, Shumski M, Smith J, Sood AB, Corrado GS, Peng L, Webster DR. Using a Deep Learning Algorithm and Integrated Gradients Explanation to Assist Grading for Diabetic Retinopathy. Ophthalmology. 2019;126:552-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 175] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 44. | Ayhan MS, Kümmerle LB, Kühlewein L, Inhoffen W, Aliyeva G, Ziemssen F, Berens P. Clinical validation of saliency maps for understanding deep neural networks in ophthalmology. Med Image Anal. 2022;77:102364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 45. | Krogh M, Germund Nielsen M, Byskov Petersen G, Jensen MSA, Jensen MB, Vorum H, Bruun NH, Kolding Kristensen J. Patient acceptance of AI-assisted diabetic retinopathy screening in primary care: findings from a questionnaire-based feasibility study. Front Med (Lausanne). 2025;12:1610114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 46. | Sharma N, Lalwani P. A multi model deep net with an explainable AI based framework for diabetic retinopathy segmentation and classification. Sci Rep. 2025;15:8777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 47. | Ikram A, Imran A. ResViT FusionNet Model: An explainable AI-driven approach for automated grading of diabetic retinopathy in retinal images. Comput Biol Med. 2025;186:109656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 48. | Movassagh AA, Jajroudi M, Homayoun Jafari A, Khalili Pour E, Farrokhpour H, Faghihi H, Riazi H, ArabAlibeik H. Quantifying the Characteristics of Diabetic Retinopathy in Macular Optical Coherence Tomography Angiography Images: A Few-Shot Learning and Explainable Artificial Intelligence Approach. Cureus. 2025;17:e76746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 49. | Yoshida M, Murakami T, Ishihara K, Mori Y, Tsujikawa A. Explainable Artificial Intelligence-Assisted Exploration of Clinically Significant Diabetic Retinal Neurodegeneration on OCT Images. Ophthalmol Sci. 2025;5:100804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 50. | Yagin FH, Colak C, Algarni A, Gormez Y, Guldogan E, Ardigò LP. Hybrid Explainable Artificial Intelligence Models for Targeted Metabolomics Analysis of Diabetic Retinopathy. Diagnostics (Basel). 2024;14:1364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 51. | Ashraf MH, Mehmood MN, Ahmed M, Hussain D, Khan J, Jung Y, Zakariah M, AlSekait DM. HIRD-Net: An Explainable CNN-Based Framework with Attention Mechanism for Diabetic Retinopathy Diagnosis Using CLAHE-D-DoG Enhanced Fundus Images. Life (Basel). 2025;15:1411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 52. | Ursin F, Timmermann C, Orzechowski M, Steger F. Diagnosing Diabetic Retinopathy With Artificial Intelligence: What Information Should Be Included to Ensure Ethical Informed Consent? Front Med (Lausanne). 2021;8:695217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 53. | Evans NG, Wenner DM, Cohen IG, Purves D, Chiang MF, Ting DSW, Lee AY. Emerging Ethical Considerations for the Use of Artificial Intelligence in Ophthalmology. Ophthalmol Sci. 2022;2:100141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 23] [Reference Citation Analysis (1)] |
| 54. | Crew A, Reidy C, van der Westhuizen HM, Graham M. A Narrative Review of Ethical Issues in the Use of Artificial Intelligence Enabled Diagnostics for Diabetic Retinopathy. J Eval Clin Pract. 2025;31:e14237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 55. | Savastano MC, Rizzo C, Fossataro C, Bacherini D, Giansanti F, Savastano A, Arcuri G, Rizzo S, Faraldi F. Artificial intelligence in ophthalmology: Progress, challenges, and ethical implications. Prog Retin Eye Res. 2025;107:101374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 56. | Chauhan A, Sarkar D, Verma GS, Rastogi H, Adapa K, Duggal M. Evaluating trustworthiness in AI-Based diabetic retinopathy screening: addressing transparency, consent, and privacy challenges. BMC Med Ethics. 2025;26:140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 57. | Rajesh AE, Davidson OQ, Lee CS, Lee AY. Artificial Intelligence and Diabetic Retinopathy: AI Framework, Prospective Studies, Head-to-head Validation, and Cost-effectiveness. Diabetes Care. 2023;46:1728-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 74] [Article Influence: 24.7] [Reference Citation Analysis (2)] |
| 58. | Ipp E, Liljenquist D, Bode B, Shah VN, Silverstein S, Regillo CD, Lim JI, Sadda S, Domalpally A, Gray G, Bhaskaranand M, Ramachandra C, Solanki K; EyeArt Study Group. Pivotal Evaluation of an Artificial Intelligence System for Autonomous Detection of Referrable and Vision-Threatening Diabetic Retinopathy. JAMA Netw Open. 2021;4:e2134254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 153] [Article Influence: 30.6] [Reference Citation Analysis (1)] |
| 59. | Grzybowski A, Brona P. Approval and Certification of Ophthalmic AI Devices in the European Union. Ophthalmol Ther. 2023;12:633-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 60. | van Kolfschooten H, van Oirschot J. The EU Artificial Intelligence Act (2024): Implications for healthcare. Health Policy. 2024;149:105152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 101] [Reference Citation Analysis (1)] |
| 61. | Busch F, Kather JN, Johner C, Moser M, Truhn D, Adams LC, Bressem KK. Navigating the European Union Artificial Intelligence Act for Healthcare. NPJ Digit Med. 2024;7:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 54] [Reference Citation Analysis (1)] |
| 62. | Zhou Y, Chia MA, Wagner SK, Ayhan MS, Williamson DJ, Struyven RR, Liu T, Xu M, Lozano MG, Woodward-Court P, Kihara Y; UK Biobank Eye & Vision Consortium, Altmann A, Lee AY, Topol EJ, Denniston AK, Alexander DC, Keane PA. A foundation model for generalizable disease detection from retinal images. Nature. 2023;622:156-163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 701] [Cited by in RCA: 458] [Article Influence: 152.7] [Reference Citation Analysis (1)] |
| 63. | Fu Y, Ma L, Wan S, Ge S, Yang Z. A novel clinical artificial intelligence model for disease detection via retinal imaging. Innovation (Camb). 2024;5:100575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 64. | Shi D, Zhang W, Yang J, Huang S, Chen X, Xu P, Jin K, Lin S, Wei J, Yusufu M, Liu S, Zhang Q, Ge Z, Xu X, He M. A multimodal visual-language foundation model for computational ophthalmology. NPJ Digit Med. 2025;8:381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (1)] |
| 65. | Matta S, Hassine MB, Lecat C, Borderie L, Guilcher AL, Massin P, Cochener B, Lamard M, Quellec G. Federated Learning for Diabetic Retinopathy Detection in a Multi-center Fundus Screening Network. Annu Int Conf IEEE Eng Med Biol Soc. 2023;2023:1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 66. | Jayalakshmi R, Tamilvizhi T. Privacy preservation in diabetic disease prediction using federated learning based on efficient cross stage recurrent model. Sci Rep. 2025;15:37258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 67. | Nakayama LF, Mitchell WG, Ribeiro LZ, Dychiao RG, Phanphruk W, Celi LA, Kalua K, Santiago APD, Regatieri CVS, Moraes NSB. Fairness and generalisability in deep learning of retinopathy of prematurity screening algorithms: a literature review. BMJ Open Ophthalmol. 2023;8:e001216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 68. | Burlina P, Joshi N, Paul W, Pacheco KD, Bressler NM. Addressing Artificial Intelligence Bias in Retinal Diagnostics. Transl Vis Sci Technol. 2021;10:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 69. | Tan W, Wei Q, Xing Z, Fu H, Kong H, Lu Y, Yan B, Zhao C. Fairer AI in ophthalmology via implicit fairness learning for mitigating sexism and ageism. Nat Commun. 2024;15:4750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 70. | Sobhi N, Sadeghi-Bazargani Y, Mirzaei M, Abdollahi M, Jafarizadeh A, Pedrammehr S, Alizadehsani R, Tan RS, Islam SMS, Acharya UR. Artificial intelligence for early detection of diabetes mellitus complications via retinal imaging. J Diabetes Metab Disord. 2025;24:104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 71. | Moannaei M, Jadidian F, Doustmohammadi T, Kiapasha AM, Bayani R, Rahmani M, Jahanbazy MR, Sohrabivafa F, Asadi Anar M, Magsudy A, Sadat Rafiei SK, Khakpour Y. Performance and limitation of machine learning algorithms for diabetic retinopathy screening and its application in health management: a meta-analysis. Biomed Eng Online. 2025;24:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 72. | Ansari A, Ansari N, Khalid U, Markov D, Bechev K, Aleksiev V, Markov G, Poryazova E. The Role of Artificial Intelligence in the Diagnosis and Management of Diabetic Retinopathy. J Clin Med. 2025;14:5150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 73. | Nderitu P, Keane PA. Real-World Adoption of Artificial Intelligence in Diabetic Retinopathy Screening: What Is the Current Status? Clin Exp Ophthalmol. 2025;53:741-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |