Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.116208
Revised: December 30, 2025
Accepted: March 11, 2026
Published online: April 15, 2026
Processing time: 160 Days and 18.4 Hours
Diabetic retinopathy (DR) is among the most prevalent microvascular complications of diabetes, with its onset and progression largely driven by chronic inflammation and mitochondrial dysfunction. In recent years, the nicotinamide adenine dinucleotide+ dependent deacetylase family, sirtuins (SIRTs), has attracted gro
Core Tip: This review summarizes the emerging roles of sirtuins (SIRT1-SIRT7) in diabetic retinopathy, emphasizing their regulatory functions in inflammation, oxidative stress, and mitochondrial dysfunction. By integrating evidence from molecular, cellular, and pharmacological studies, it highlights how selective activation of SIRT1 and SIRT3 restores retinal homeostasis. The review also outlines therapeutic advances in natural and small-molecule SIRT activators, providing a translational framework for multi-target interventions in diabetic retinopathy.
- Citation: Pan CC, Xie QQ, Lu PY, Shi Z, Li HY, Ma YJ, Ding TY, Zeng MQ, Luo C, Zhuge FY. Targeting sirtuins in diabetic retinopathy: Differential roles in inflammation and mitochondrial dysfunction. World J Diabetes 2026; 17(4): 116208
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/116208.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.116208
Diabetic retinopathy (DR) is one of the most prevalent and vision-threatening microvascular complications of diabetes, with a global prevalence of 22%-40%, affecting approximately 103 million individuals in 2020 and reaching 161 million by 2045[1,2]. It currently ranks as the fifth leading cause of blindness worldwide. The likelihood of developing DR increases markedly with the duration of diabetes[3]. Pathologically, this disease is driven primarily by chronic hyperglycemia-induced injury to the retinal microvasculature. In its early stages, constriction of retinal arterioles leads to ischemia, initiating a cascade of pathological events, including pericyte loss, endothelial dysfunction, vascular leakage, and aberrant neovascularization, that ultimately culminate in severe visual impairment or blindness[4,5].
Despite substantial advances in ophthalmic therapeutics, current frontline interventions, such as laser photocoagulation and intravitreal anti-vascular endothelial growth factor (VEGF) therapy, are largely limited to advanced disease stages[6,7]. These approaches are invasive, require repeated administration, and remain insufficient for a subset of patients who continue to experience progressive retinal damage despite treatment[8]. The absence of effective pharmacological strategies targeting early pathogenic mechanisms underscores the urgent need for novel preventive and disease-modifying therapies for DR. Sirtuins (SIRTs) constitute an evolutionarily conserved family of nicotinamide adenine dinucleotide+ (NAD+) dependent class III histone deacetylases, encompassing seven members (SIRT1-SIRT7) in mammals[9,10]. All members share a highly conserved catalytic core of approximately 275 amino acids, containing N-terminal, C-terminal, and zinc (Zn)-binding domains that mediate protein lysine deacetylation and mono-ADP-ri
In recent years, extensive studies have underscored the pivotal involvement of SIRTs in the onset and progression of diabetic complications[13]. Building upon these findings, the present review delineates the molecular pathogenesis of DR, integrates emerging evidence regarding the diverse roles of the SIRT family under these conditions, and evaluates the therapeutic potential of pharmacological SIRT modulation. Our objective is to provide updated insights into the regulatory landscape of SIRTs in DR and to highlight novel avenues for precise and integrative therapeutic intervention.
Recent studies have elucidated multiple biochemical and molecular mechanisms contributing to the onset and progression of DR[14,15]. Nevertheless, no single definitive pathogenic pathway has been identified. Instead, DR is now understood as the outcome of a complex and tightly interconnected pathological network involving hyperglycemia-induced metabolic dysregulation, chronic inflammation, oxidative stress, and mitochondrial dysfunction[16,17]. The interplay among these processes triggers a cascade of cellular and structural injuries within the retina, culminating in blood-retinal barrier disruption, neuronal degeneration, pericyte loss, and pathological neovascularization[18].
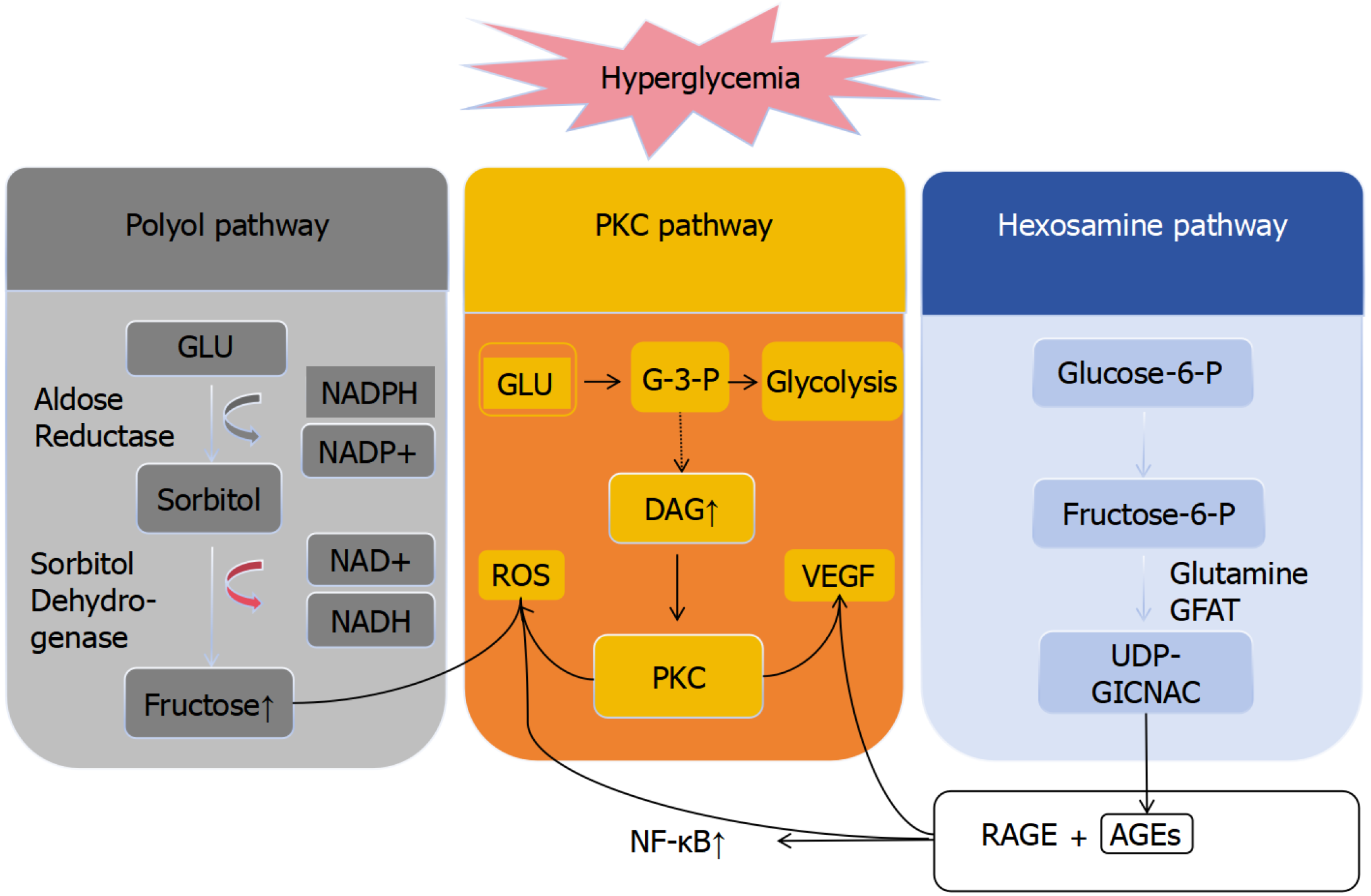
Under hyperglycemic conditions, the polyol pathway becomes abnormally activated[19,20]. Aldose reductase catalyzes the reduction of glucose to sorbitol via nicotinamide adenine nucleotide phosphate (NADPH) as a cofactor, after which sorbitol is oxidized to fructose by sorbitol dehydrogenase in a reaction that converts NAD+ to NADH[21]. This sequence leads to excessive NADPH consumption, thereby weakening the cellular antioxidant defense system and disturbing redox homeostasis. Concurrently, an elevated NADH/NAD+ ratio promotes excessive generation of reactive oxygen species (ROS) within retinal cells[22]. In parallel, excess intracellular glucose flux activates the hexosamine biosynthetic pathway. The rate-limiting enzyme glutamine, fructose-6-phosphate amidotransferase, catalyzes the amination of fructose-6-phosphate to glucosamine-6-phosphate[23]. Subsequent acetylation and isomerization reactions yield UDP-N-acetylglucosamine, which serves as a substrate for O-linked glycosylation and facilitates abnormal protein modification. These processes further promote the formation of advanced glycation end products (AGEs). Upon binding to their receptor RAGE, AGEs not only augment ROS production but also activate proinflammatory signaling cascades, including the nuclear factor-κB (NF-κB) and mitogen-activated protein kinases pathways, thereby intensifying oxidative stress and inflammation and ultimately triggering the apoptosis of retinal pericytes[24]. Furthermore, under high-glucose con
In summary, hyperglycemia drives the progression of DR through the interlinked activation of four principal biochemical pathways, the polyol pathway, the hexosamine biosynthetic pathway, PKC activation, and AGE formation, which act synergistically to perpetuate oxidative stress, inflammation, and microvascular damage (Figure 1).
The human eye is one of the most metabolically active organs, with an oxygen consumption rate comparable to that of the brain and myocardium, rendering it highly vulnerable to oxidative stress and nutritional deficiencies[27]. The retina, in particular, depends critically on a continuous supply of micronutrients, including Zn and copper (Cu), to maintain redox homeostasis[28]. Under conditions of chronic hyperglycemia in diabetes, an imbalance in this delicate system accelerates the pathological cascade leading to DR. Central to this pathogenesis is systemic dyshomeostasis of Zn and Cu, which is clinically characterized by a “low Zn, high Cu” serum profile in DR patients[29]. This elevated Cu/Zn ratio directly impairs the activity of the key antioxidant enzyme Cu/Zn-superoxide dismutase (SOD), crippling a primary cellular defense against ROS[28]. Furthermore, excess Cu synergizes with high glucose to induce mitochondrial dysfunction and endoplasmic reticulum (ER) stress in retinal cells[28,29].
Critically, the dysregulation of these trace elements also undermines other vital protective mechanisms[30]. The functional activity of deacetylases, including SIRT1, is complexly regulated by cell metabolic homeostasis and the availability of essential cofactors. Specifically, Zn acts as a key structural cofactor necessary to maintain the stability and activity of the SIRT1 catalytic core[31]. These findings indicate that the imbalance of trace elements, such as Zn and Cu, in diabetes mellitus may lead to damage to the function of key protective proteins, such as SIRT1. Thus, Zn/Cu imbalance likely contributes to DR not only by weakening direct antioxidant defense but also by impairing the function of key regulatory proteins such as SIRT1[32,33]. This pathophysiological link is supported by large-scale epidemiological data indicating that inadequate dietary intake of Zn and Cu is associated with an increased risk of DR[34]. The resulting compounded oxidative and metabolic stress exacerbates inflammation and capillary damage. Notably, the therapeutic potential of reversing this imbalance is underscored by evidence showing that supplementation with Zn and Cu formulations can mitigate retinopathy progression in diabetic models, likely through attenuating oxidative stress[34]. Therefore, trace element dysregulation acts as a crucial pathogenic interface that amplifies retinal injury, and restoring micronutrient homeostasis represents a promising complementary strategy to support cellular resilience in DR.
Under hyperglycemic conditions, oxidative stress markedly accelerates the progression of DR by promoting lipid peroxidation[35,36]. ROS attack membrane phospholipids, initiating lipid peroxidation that compromises membrane integrity and generates toxic aldehyde byproducts such as 4-hydroxynonenal and 4-hydroxyhexenal[37,38]. These aldehydes accumulate extensively within retinal tissue, where they not only exacerbate mitochondrial ROS leakage and trigger p53-dependent apoptosis but also activate proinflammatory and neurodegenerative signaling cascades, including the Wingless-type MMTV integration site family and NF-κB pathways[37]. Moreover, hyperglycemia-induced mito
Recent studies by Chen et al[41] and Jalgaonkar et al[42] have identified the telomeric protein TERF1-interacting nuclear factor 2 (TIN2) as a pivotal regulator of oxidative stress and apoptosis in retinal pigment epithelial (RPE) cells under high glucose conditions. In the proposed TIN2/FOXO1 mitochondrial shuttling pathway, hyperglycemia upregulates TIN2 expression and leads to its mitochondrial accumulation. This accumulation promotes the phosphorylation of the transcription factor forkhead box O1 (FOXO1), thereby driving its translocation from the nucleus to the mitochondria. Once relocated, FOXO1 binds to the D-loop region of mitochondrial DNA (mtDNA), which impairs respiratory chain activity, diminishes intrinsic antioxidant defenses, and triggers the mitochondrial apoptotic cascade[43]. Under normal conditions, nuclear FOXO1 activates antioxidant gene transcription. Therefore, aberrant mitochondrial relocation under hyperglycemic conditions suppresses this protective response. This dual mechanism, simultaneously inducing mitochondrial dysfunction and disabling nuclear antioxidant defense, exacerbates RPE apoptosis and secondary photoreceptor injury. Collectively, these findings establish the TIN2-FOXO1 axis as a central regulator of metabolic and oxidative stress in DR and highlight its potential as a therapeutic target for early intervention.
In DR, hyperglycemia-induced oxidative stress and chronic inflammation constitute two tightly intertwined pathological processes[44]. Persistent hyperglycemia promotes excessive generation of ROS through multiple mechanisms, including activation of the polyol pathway and accumulation of AGEs, thereby disturbing the cellular redox equilibrium and initiating proinflammatory signaling cascades[45]. ROS and related intermediates act as signaling molecules that activate the NF-κB pathway, resulting in robust upregulation of proinflammatory cytokines such as interleukin (IL)-1β, IL-18, and tumor necrosis factor (TNF)-α, which contribute to pericyte apoptosis and microvascular injury[46]. In parallel, NF-κB signaling provides the essential “priming” signal for NLR family pyrin domain-containing 3 (NLRP3) inflammasome activation, facilitating the subsequent cleavage and release of IL-1β and IL-18, thereby amplifying the inflammatory cascade[3,47]. The synergistic activation of these pathways ultimately drives progressive damage to the retinal neurovascular unit, reinforcing the chronic inflammatory microenvironment that underlies DR pathogenesis[48].
Mitochondria are essential organelles that serve as the primary site of oxidative phosphorylation and ATP synthesis in eukaryotic cells, providing the major source of cellular energy[49,50]. They are also the principal generators of intracellular ROS in human cells[51]. Under hyperglycemic conditions, mitochondrial dysfunction is markedly exa
Physiologically, electrons derived from NADH and FADH2 are transferred through the ETC to oxygen, forming water via oxidative phosphorylation[52]. However, chronic hyperglycemia results in excessive substrate influx and an oversupply of reducing equivalents (NADH/FADH2), which elevates the mitochondrial membrane potential and enhances electron leakage, particularly at complexes I and III, culminating in the formation of superoxide anions (O2•-)[53]. This aberrant electron flux serves as a major trigger of oxidative stress in DR.
Experimental evidence further indicates that exposure of retinal cells to high-glucose conditions (50 mmol/L) leads to elevated ROS generation accompanied by downregulation of PTEN-induced putative kinase 1 (PINK1) and Parkin expression, impaired mitophagy and cellular proliferation, and increased apoptotic activity[54]. Together, these findings underscore that mitochondrial dysfunction is a central driver of oxidative injury and cellular degeneration in DR.
Hyperglycemia directly triggers inflammatory activation in retinal glial cells through the release of damage-associated molecular patterns, such as mtDNA, and persistent activation of the AGE-RAGE signaling axis[55]. Within this context, the NLRP3 inflammasome functions as a central regulatory hub, facilitating the maturation and secretion of proinflammatory cytokines such as IL-1β while simultaneously amplifying inflammatory signaling via the NF-κB pathway[56]. The resulting cytokine surge recruits immune cells, including monocytes and T lymphocytes, ultimately compromising the integrity of the blood-retinal barrier (BRB)[57]. This sustained inflammatory milieu culminates in irreversible injury to the retinal neurovascular unit.
Mitochondrial dysfunction lies at the core of this self-perpetuating cycle. As the predominant endogenous source of ROS, mitochondria under hyperglycemic stress experience ETC overload, disrupted dynamics, and impaired autophagic clearance[58]. The resulting accumulation of damaged mitochondria further intensifies oxidative stress, perpetuating cellular injury and promoting disease progression toward the proliferative phase of DR. Consequently, therapeutic strategies aimed at restoring mitochondrial function, mitigating oxidative stress, and attenuating early inflammatory responses have emerged as promising approaches for halting or reversing the pathogenesis of DR[59].
During the progression of DR, structural and functional disruption of the BRB represents a central pathological hallmark[60,61]. The inner BRB is primarily composed of retinal microvascular endothelial cells that form a selective permeability barrier through tight junction proteins, such as claudin-5 and occludin, and adherens junctions containing VE-cadherin[62]. In contrast, the outer BRB consists of RPE cells interconnected by their own tight junction complexes, maintaining metabolic and ionic homeostasis between the retina and choroid.
Under conditions of sustained hyperglycemia and glycemic variability, angiopoietin-2 (Ang-2) competitively an
Neurodegenerative alterations represent one of the earliest pathological events in the course of DR, occurring well before overt microvascular damage becomes apparent[70]. Advances in retinal imaging, particularly optical coherence tomography, have demonstrated marked thinning of the retinal nerve fiber layer and ganglion cell layer in patients with early-stage diabetes[71-73]. These structural changes closely correlate with functional impairments, including reduced contrast sensitivity and delayed electrophysiological responses. This spectrum of morphological and functional deficits is referred to as diabetic retinal neurodegeneration[74].
The underlying molecular and cellular mechanisms involve multiple interrelated pathways. Under conditions of chronic hyperglycemia, Müller cells experience depletion of their intrinsic antioxidant defenses, characterized by reduced SOD2 activity and suppression of the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway, which transform them from supportive glial elements into pathological mediators that secrete excessive levels of VEGF, Ang-2, and transforming growth factor-β, thereby promoting vascular leakage and fibrotic remodeling[75]. Concurrently, microglia shift from the reparative M2 phenotype to the proinflammatory M1 phenotype, releasing cytokines and neurotoxic molecules and even exhibiting aberrant phagocytosis of retinal ganglion cells (RGCs)[76]. In parallel, as
Together, these glial alterations compromise the metabolic and signaling integrity of the neurovascular unit, initiating a cascade of neurodegenerative events[78], including dysregulated glutamatergic transmission, hyperphosphorylation of tau protein[79], and mitochondrial impairment[80]. The early emergence of neurodegeneration and glial activation before overt microvascular abnormalities highlights their potential as sensitive biomarkers and therapeutic targets for early-stage DR. Nevertheless, whether these neural alterations serve as indispensable initiators of disease onset remains a matter of ongoing debate, as a subset of patients exhibit pronounced vascular pathology in the absence of detectable neurodegenerative changes.
SIRTs constitute a family of evolutionarily conserved, NAD+ dependent histone deacetylases. The seven mammalian isoforms (SIRT1-SIRT7) share a conserved catalytic core domain and are phylogenetically grouped into four classes. Despite their structural similarity, SIRTs exhibit distinct biological functions that are largely determined by their dynamic subcellular localization, primarily within the nucleus, cytoplasm, or mitochondria, which can shift in response to metabolic or oxidative stress.
Among these proteins, SIRT1 is the most extensively studied. It is an NAD+ dependent class III histone deacetylase expressed ubiquitously across mammalian tissues[81]. In humans, the SIRT1 gene, located on chromosome 10q22.1, comprises nine exons and encodes a 747–amino acid protein (737 amino acids in the murine homolog)[82]. Through the deacetylation of both histone and nonhistone substrates, SIRT1 orchestrates a wide range of physiological and patho
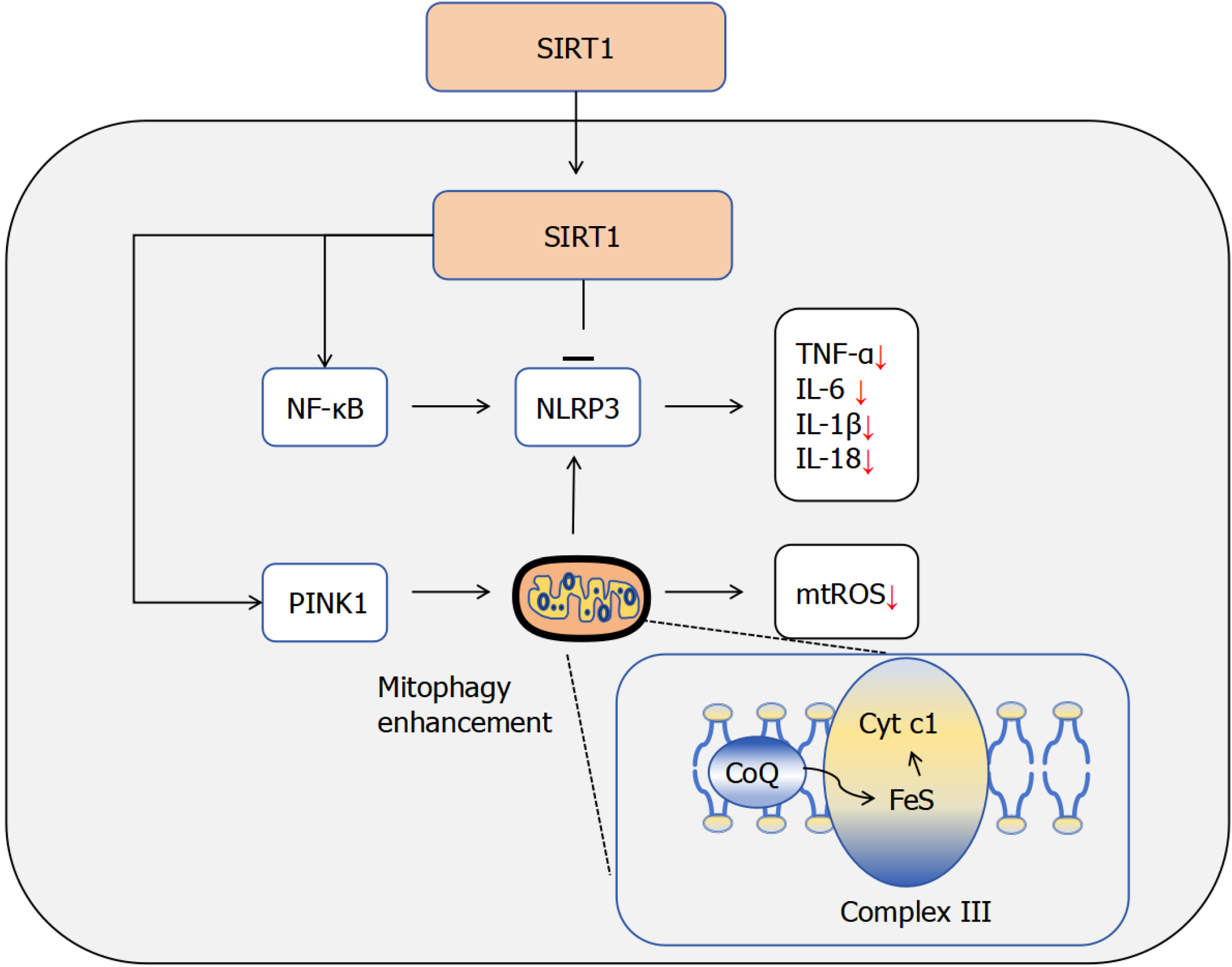
In DR, SIRT1 confers multifaceted protection primarily through the suppression of inflammatory signaling. It inhibits the NF-κB/NLRP3 axis by deacetylating the p65 subunit of NF-κB, thereby attenuating its transcriptional activity and reducing the production of proinflammatory cytokines such as TNF-α and IL-6[87]. In parallel, SIRT1 suppresses NLRP3 inflammasome assembly, resulting in reduced caspase-1-mediated maturation and secretion of IL-1β and IL-18, ultimately alleviating neurovascular injury and retinal inflammation[88,89].
In addition to its anti-inflammatory function, SIRT1 is critically involved in maintaining mitochondrial homeostasis[90]. During DR progression, the activity of mitochondrial complex III is markedly reduced, leading to excessive accumulation of mitochondrial superoxide. SIRT1 counteracts this dysfunction by preserving complex III activity and mitigating oxidative stress-induced damage. By sustaining mitochondrial integrity, SIRT1 indirectly suppresses inflammation, downregulating pro-oxidant proteins and promoting PINK1-dependent mitophagy to eliminate damaged mitochondria that release mtROS and mtDNA, thereby inhibiting NLRP3 activation at its origin[91,92] (the functional roles of SIRT1 are summarized in Figure 2).
Moreover, SIRT1 operates in close concert with the cytoprotective transcription factor Nrf2, forming a central regulatory hub for antioxidant defense[90]. In addition to indirect regulation via microRNAs (miRNAs) such as miR-221 (which inhibits Nrf2 by suppressing SIRT1)[92], SIRT1 directly deacetylates Nrf2, increasing its stability and transcriptional activity. This axis is often embedded in a broader signaling network, where the energy sensor adenosine monophosphate-activated protein kinase (AMPK) activates SIRT1, forming an upstream AMPK/SIRT1/Nrf2 cascade critical for stress adaptation[91]. Conversely, in the diabetic retina, oxidative stress and inflammation can simultaneously inhibit SIRT1 and disrupt Nrf2 signaling, creating a vicious cycle. Dysfunction of this axis leads to an impaired antioxidant response and promotes apoptosis in retinal cells, as evidenced by increased B-cell lymphoma 2 (Bcl-2)-associated X protein and decreased Bcl-2 levels[93]. This multifaceted interplay underscores the pivotal role of the SIRT1-Nrf2 axis in coordinating antioxidative defense and cell survival under diabetic stress conditions.
SIRT2 is an NAD+ dependent deacetylase broadly expressed across multiple tissues and is involved in the regulation of cellular homeostasis through its deacetylation activity[94,95]. It directly targets substrates such as NLRP3 and α-tubulin to inhibit inflammasome activation and the subsequent release of proinflammatory mediators, thereby mitigating chronic inflammation and improving insulin sensitivity[96,97].
At the retinal level, SIRT2 has been shown to inhibit the mammalian target of rapamycin signaling pathway, thereby influencing ciliary formation and cell cycle regulation, suggesting a potential role in maintaining retinal structural and metabolic homeostasis[97]. Fan et al[98] reported significantly decreased SIRT2 levels in the tear fluid of patients with DR, implying that SIRT2 deficiency may contribute to DR progression and that SIRT2 may act as a protective regulator in the retinal defense response. Interestingly, the upregulation of SIRT2 in human retinal microvascular endothelial cells has been demonstrated to attenuate hypoxia-induced migration, invasion, and angiogenesis[99]. Collectively, these findings suggest that SIRT2 may play a protective role in the diabetic retina; however, its precise molecular mechanisms and do
SIRT3, a key NAD+ dependent deacetylase, is synthesized as a 44-kDa nuclear precursor that is subsequently processed within mitochondria into its 28-kDa active form[100,101]. Functionally, SIRT3 safeguards mitochondrial integrity by fine-tuning cellular energy metabolism through the deacetylation of key enzymes involved in the tricar
In the context of DR, particularly during the non-proliferative (NPDR) stage, SIRT3 plays a pivotal role in mitigating oxidative stress and preserving cellular integrity. SIRT3 plays a pivotal role in mitigating oxidative stress in DR by deacetylating key antioxidant enzymes, such as SOD and isocitrate dehydrogenase, thereby enhancing ROS detoxification. Experimental studies in rodent models of DR have demonstrated that SIRT3 expression and enzymatic activity are markedly downregulated; conversely, pharmacological or genetic activation of SIRT3 significantly alleviates oxidative stress and ameliorates retinal pathology[106]. In rat retinal capillary endothelial cells exposed to high-glucose conditions, both SIRT3 and antioxidant enzymes, including SOD and catalase, are suppressed, whereas ROS levels are markedly elevated. Restoration of intracellular NAD+ levels by ginsenoside Rb1 inhibits poly(ADP-ribose) polymerase overac
In addition to exerting direct antioxidant effects, SIRT3 exerts its protective effects in DR by modulating mitochondrial function and angiogenic activity[107]. For example, honokiol (HKL) has been shown to upregulate the mitochondrial fusion protein optic atrophy 1 (OPA1) in a SIRT3-dependent manner, thereby improving mitochondrial dynamics and bioenergetic efficiency[108]. In addition, SIRT3 promotes mitophagy in RPE cells by upregulating FOXO3a, PINK1, Parkin, and LC3B, with PINK1 identified as a critical downstream effector that mediates mitochondrial quality control[109]. Consistent with these findings, Yang et al[109] demonstrated that high-glucose exposure markedly suppresses SIRT3 expression in RPE cells, resulting in impaired mitophagy, excessive ROS accumulation, and increased apoptosis. Conversely, SIRT3 overexpression activates the AMPK/mammalian target of rapamycin/UNC-51-like kinases 1 signaling pathway, enhances mitophagy, and prevents cell death, thereby preserving RPE cell integrity under hyperglycemic stress.
Collectively, these observations underscore the pivotal role of SIRT3 in maintaining mitochondrial quality and maintaining cellular survival during the progression of DR. As DR progresses to the proliferative (PDR) stage, the role of SIRT3 expands to include the regulation of retinal angiogenesis, which is influenced by its impact on cellular metabolism. In the early stage of proliferation, vascular dropout induces retinal ischemia, which triggers aberrant neovascularization characterized by the formation of pathological diabetic retinopathic tufts rather than the desired physiological revascularization[110]. Under normal conditions, tip endothelial cells involved in physiological angiogenesis rely predominantly on glycolysis to sustain their migratory and regenerative activities. In contrast, Cagnone et al[17] demonstrated that endothelial cells within pathological vascular tufts shift their metabolic preference toward FAO, favoring proliferation over migration.
In this PDR context, SIRT3, primarily expressed in astrocytes, modulates angiogenesis by regulating the activity of FAO-associated metabolic enzymes. Notably, SIRT3 deficiency reprograms the retinal microenvironment from FAO dominance to enhanced glycolytic flux, resulting in reduced formation of pathological vascular tufts and increased VEGF expression, thereby promoting physiological revascularization[17]. These findings highlight a stage-specific function of SIRT3 in orchestrating metabolic adaptations within the ischemic retina.
SIRT5 is a conserved NAD+ dependent desuccinylase predominantly localized within mitochondria, although a fraction is also distributed in the cytosol[111]. As a member of the class III SIRT family, SIRT5 is abundantly expressed in metabolically active tissues, including the brain, heart, liver, kidney, and skeletal muscle. While exhibiting relatively weak deacetylase activity, SIRT5 primarily exerts its regulatory functions through the removal of succinyl, malonyl, and glutaryl groups from lysine residues on diverse protein substrates. These substrates are central to the regulation of redox balance and multiple metabolic pathways, such as glycolysis, the pentose phosphate pathway, ketone body synthesis, FAO, glutamine metabolism, and ammonia detoxification[112].
Optineurin (OPTN) is a recently characterized autophagy receptor that is highly and specifically expressed in RGCs[113]. Mutations in OPTN (e.g., M98K and E50K) have been linked to autophagic RGC death in glaucoma. Posttranslational modifications of OPTN, including phosphorylation by TANK-binding kinase 1 and ubiquitination by HECT domain and ankyrin repeat containing E3 ubiquitin protein ligase 1, critically modulate its autophagic functions, governing autophagosome formation and fusion with lysosomes[114]. A study by Zhang et al[115] revealed that in STZ-induced diabetic rats, R28 cells, and primary rat RGCs, hyperglycemia disrupts the tricarboxylic acid cycle, resulting in succinate accumulation. Elevated succinate promotes lysine succinylation of OPTN, particularly at the K108 site, thereby blocking autophagic flux and impairing RGC function under high-glucose conditions. Concurrently, both the expression and activity of SIRT5 are markedly reduced in the diabetic retina, further exacerbating OPTN succinylation. Importantly, overexpression of SIRT5 reverses OPTN K108 succinylation, restores autophagic flux, and preserves RGC viability, underscoring its essential role in maintaining mitochondrial homeostasis and neuronal integrity in DR[116].
SIRT6, a mammalian NAD+ dependent deacylase, exerts pleiotropic effects on genome stability, metabolic regulation, and inflammation, making it a molecule of considerable scientific interest[117]. It protects against metabolic disorders by suppressing hepatic glycolysis and gluconeogenesis[118] and attenuates inflammation by promoting macrophage polarization from the proinflammatory M1 phenotype toward the anti-inflammatory M2 phenotype[119].
In the diabetic retina, early-stage alterations are characterized by reduced expression of SIRT6 and brain-derived neurotrophic factor, accompanied by increased VEGF levels even before overt vascular pathology becomes evident. This pattern suggests a mechanistic link between neurotrophin depletion and early neurodegenerative changes. In support of this notion, high-glucose exposure has been shown to upregulate VEGF and downregulate SIRT6 in Müller cells, while SIRT6 knockout models have confirmed its role in promoting aberrant glycolytic reprogramming and apoptosis within the inner retinal layers. These findings collectively implicate SIRT6 as a critical regulator of the metabolic-neurotrophic balance in the early pathogenesis of DR.
The functions of SIRT4 and SIRT7 in the diabetic retina remain largely unexplored. This gap mirrors the broader research field, where SIRT4 and SIRT7 are less studied than other isoforms in many diseases. However, their known cellular functions suggest potential relevance[120,121]. SIRT4, a mitochondrial SIRT with ADP-ribosyltransferase and deacylase activities, is a key regulator of mitochondrial metabolism and energy homeostasis[121]. Given the central role of mitochondrial dysfunction in DR, future investigations into the impact of SIRT4 on retinal bioenergetics under hyperglycemic conditions are warranted. Notably, Maghbooli et al[122] reported that SIRT4 protein levels are elevated in the peripheral blood mononuclear cells of patients with DR, which may reflect an adaptive or protective response of the body to metabolism- and oxidative stress-related changes associated with DR. Similarly, SIRT7, a nucleolar protein involved in ribosomal biogenesis and the cellular stress response, may influence retinal cell survival and function[123]. Preliminary evidence from other diabetic complications, such as nephropathy, suggests their modulatory roles in disease processes[123]. Therefore, elucidating the contributions of SIRT4 and SIRT7 represents a promising frontier for future research, which will be crucial for achieving a fully integrated understanding of the SIRT family’s regulatory network in DR.
Ginseng, a traditional medicinal herb and functional food, exerts its pharmacological activities primarily through a diverse group of triterpenoid saponins known as ginsenosides[124]. To date, more than 150 distinct ginsenosides have been identified in different parts of the plant. Among them, the predominant and most extensively studied native forms, Ra, Rb1, Rb2, Rc, Rd, Re, Rf, and Rg1, are collectively referred to as the major ginsenosides[125].
Ginsenoside Rd (Rd), a key bioactive compound derived from Panax notoginseng, activates AMPK via the G protein-coupled receptor 30-protein kinase A-liver kinase B1 kinase cascade, leading to an elevated NAD+/NADH ratio and liver kinase B1 deacetylation, thereby increasing AMPK/SIRT1 signaling crosstalk. Through this pathway, Rd restores high glucose-impaired FAO, maintains mitochondrial NADPH regeneration via isocitrate dehydrogenase 2, and reverses NADPH oxidase 2 activation, oxidative stress, mitochondrial dysfunction, and endothelial cell death[126]. Additionally, Rd upregulates SIRT1 expression, increases the NAD+/NADH ratio, and reinforces the AMPK/SIRT1 interplay to sustain mitochondrial function. At the organelle level, it exhibits potent antioxidant activity by increasing mtDNA copy number and increasing the activities of SOD and catalase[127].
In comparison, ginsenoside Rb1 alleviates high glucose-induced oxidative damage through activation of the NAD+-poly(ADP-ribose) polymerase-SIRT axis[102], whereas ginsenoside Rg1 suppresses retinal endothelial cell proliferation, migration, and angiogenesis under hyperglycemic conditions via regulation of the long non-coding RNA small nucleolar RNA host gene 7/miR-2116-5p/SIRT3 signaling axis[128].
Resveratrol (RES), a natural polyphenolic phytoalexin found in berries, peanuts, and red grapes, possesses potent antioxidant, anti-inflammatory, and anti-proliferative properties that contribute to its protective effects against diabetes, cardiovascular disease, and cancer. As an antiangiogenic compound, RES provides cytoprotection against oxidative stress, inflammation, and light-induced retinal degeneration. Mechanistic studies by Kubota et al[129] demonstrated that RES activates AMPK, prevents SIRT1 inactivation, and suppresses NF-κB phosphorylation, thereby attenuating inflammation and oxidative injury.
Recent evidence further indicates that RES mitigates oxidative stress and inflammatory responses in DR by regulating ferroptosis through the SIRT1/high mobility group box 1 signaling pathway. Similarly, Mohammad et al[90] reported that RES upregulates SIRT1 expression while downregulating high mobility group box 1 in the diabetic retina, im
Artesunate (ART) is a semisynthetic derivative of artemisinin, a bioactive compound originally isolated from the traditional Chinese medicinal herb Artemisia annua[131]. In addition to their well-established antimalarial activity, artemisinin and its derivatives exhibit broad pharmacological properties, including the induction of autophagy and the inhibition of fibrosis, and have demonstrated efficacy in the prevention and treatment of hepatitis[132].
Research has reported that ART activates the AMPK signaling pathway during angiogenesis, suggesting that AMPK represents a key molecular target of ART[131]. Similarly, Li et al[133] further demonstrated that ART exerts protective effects in the diabetic retina via activation of the AMPK/SIRT1-dependent autophagy pathway. This activation is characterized by increased Beclin-1 expression and an increased LC3II/I ratio, leading to increased autophagic flux and inhibition of microglial activation.
Carnosic acid (CA), a naturally occurring polyphenolic diterpene abundant in rosemary and several other medicinal plants, has diverse pharmacological activities, including anticancer, anti-inflammatory, and antiviral effects[134]. Previous studies have demonstrated that CA treatment significantly improves glucose tolerance, lowers fasting blood glucose levels, and enhances insulin sensitivity in diabetic mice by reducing ROS accumulation[135]. Mechanistically, CA activates the AMPK signaling pathway, suppresses the expression of gluconeogenic and lipogenic genes, and inhibits the proliferation of human hepatocellular carcinoma G2 cells.
More recently, evidence has shown that CA modulates the SIRT1/p53/SLC7A11 signaling axis, leading to decreased intracellular Fe2+ and total iron content in the retinal tissues of STZ-induced diabetic models. This regulatory action mitigates ferroptosis-related molecular disturbances and prevents pathological iron deposition, thereby exerting a protective effect against retinal oxidative injury in DR[136].
Polydatin (PD), a natural monomeric compound primarily isolated from the dried rhizome of Polygonum cuspidatum, possesses multiple pharmacological properties, including anti-inflammatory, antioxidant, and antiapoptotic activities[137]. In diabetic models, PD has been shown to upregulate casein kinase 2-interacting protein 1 and Nrf2 expression in renal tissues, thereby enhancing antioxidant defenses and improving renal function[138,139].
However, its protective role in the diabetic retina has only recently been elucidated. Emerging evidence indicates that PD suppresses the proliferation and activation of Müller cells, downregulates proangiogenic mediators such as VEGF and hepatocyte growth factor, and reduces the expression of proinflammatory cytokines, including TNF-α, IL-1β, and IL-6, thereby alleviating oxidative stress. Mechanistically, PD upregulates SIRT1 expression to inhibit NLRP3 inflammasome activation, leading to reduced inflammatory and oxidative damage. Notably, the protective effects of PD were abolished upon SIRT1 knockdown or administration of the NLRP3 agonist ATP, confirming the essential role of the SIRT1/NLRP3 axis in mediating PD retinal protection[88].
Plant-derived polyphenols are well recognized for their ability to improve retinal microcirculation and mitigate oxidative stress, inflammation, and apoptosis[140]. Among them, Crataegus (hawthorn) polyphenols exhibit well-established lipid-lowering, antioxidant, and antihypertensive effects, collectively contributing to reduced cardiovascular risk[141]. This broad pharmacological profile suggests that hawthorn polyphenols may also possess therapeutic potential in DR[142].
Research has shown that polyphenols can modulate epigenetic regulation and reverse hyperglycemia-induced retinal injury[143]. However, the specific role of hawthorn polyphenols in the diabetic retina has remained insufficiently explored. Recent work by Liu et al[144] demonstrated that hawthorn polyphenol extract, when used as a potential functional food ingredient, effectively alleviates hyperglycemia-induced retinal damage through multiple mechanisms. These include reducing ROS levels, preventing RPE apoptosis, and suppressing both the AMPK/SIRT1/NF-κB and the miR-34a/SIRT1/p53 signaling pathways. Collectively, these findings suggest that hawthorn polyphenols may serve as promising nutraceutical interventions targeting SIRT1-mediated oxidative and inflammatory cascades in DR.
Baicalin, a flavonoid glycoside derived from Scutellaria baicalensis, has been reported to markedly ameliorate high glucose-induced endothelial injury and suppress angiogenic tube formation in human umbilical vein endothelial cells[145]. Its aglycone form, baicalein, exhibits additional pharmacological activities, including antiplatelet aggregation, enhancement of cerebral blood flow, and vasoactive as well as proangiogenic effects, which collectively contribute to its therapeutic potential in cardiovascular and cerebrovascular diseases[146].
In addition to these systemic effects, baicalein has been shown to improve ischemia-induced retinal dysfunction and attenuate hyperglycemia-driven vascular inflammation. In a study by Wang et al[147], baicalein significantly alleviated high glucose-induced functional impairment in retinal microvascular endothelial cells by activating SIRT3 and modulating ER stress responses. These findings highlight the potential of baicalein as a natural SIRT3 activator that preserves endothelial integrity and mitigates metabolic stress in DR.
HKL, a natural biphenolic compound isolated from the bark of Magnolia species, possesses the remarkable ability to cross the blood-brain barrier and exhibits a wide range of pharmacological activities, including anti-inflammatory, antioxidant, antitumor, analgesic, and neuroprotective effects. HKL, which is traditionally recognized as an antioxidant agent, has long been used to treat inflammatory disorders. Its capacity to penetrate both the blood-brain barrier and the blood-cerebrospinal fluid barrier confers high bioavailability within neural tissues while maintaining low systemic toxicity[148].
A study by Shi et al[149] demonstrated that HKL alleviates high glucose-induced microvascular injury in DR by activating SIRT3, which upregulates the mitochondrial fusion protein OPA1. This increase in mitochondrial fusion reduces mitochondrial fragmentation, suppresses oxidative stress and apoptosis, and ultimately mitigates retinal vascular damage. Further experimental validation in db/db mice and high glucose-treated retinal microvascular endothelial cells confirmed that HKL improves mitochondrial function via the SIRT3-OPA1 signaling axis, underscoring its therapeutic potential as a mitochondrial-protective agent in DR.
Arbutin, a natural compound derived from Arctostaphylos uva-ursi (bearberry) leaves, possesses diverse pharmacological activities, such as anti-inflammatory and antioxidant effects[150]. It has been demonstrated to protect against high glucose-induced cell injury and ameliorate H2O2-mediated damage in RGCs[151]. However, whether arbutin can suppress high glucose-induced retinal injury remains to be fully elucidated. Research indicates that arbutin inhibits lipopolysaccharide-induced effects by activating SIRT1. In a study by Ma et al[152], arbutin was found to directly increase the protein expression level of SIRT1 in adult RPE (ARPE-19) cells and ameliorate high glucose-induced damage in RPE cells.
Gastrodin, the principal bioactive constituent of the traditional Chinese medicinal herb Gastrodia elata, has been reported to exhibit potent anti-inflammatory and antiapoptotic properties across a variety of disease models[153]. In a study by Zhang et al[154], Gastrodin was shown to upregulate SIRT1 expression in human retinal endothelial cells under high-glucose conditions, thereby inhibiting activation of the toll-like receptor 4 (TLR4)/NF-κB p65 signaling pathway, whereas hyperglycemia alone produced the opposite effect. Moreover, SIRT1 overexpression experiments confirmed that activation of this deacetylase effectively suppresses TLR4/NF-κB signaling and reduces apoptosis, highlighting a critical role for the SIRT1-TLR4/NF-κB axis in mediating the cytoprotective effects of Gastrodin against diabetic retinal injury.
Astragalus polysaccharide (APS), a bioactive macromolecule extracted from the roots of Astragalus membranaceus, is widely utilized in clinical practice for its antitumor and antidiabetic properties[155]. Previous in vivo studies have shown that APS treatment can reduce the incidence and delay the onset of both type 1 and type 2 diabetes. It has also been reported to suppress ER stress and subsequent apoptosis. In a type 2 diabetic rat model, APS administration not only restored hepatic glucose homeostasis but also attenuated ER stress, a key contributor to insulin resistance[156]. Collectively, these findings highlight the functional role of APS in regulating glucose metabolism and improving insulin sensitivity.
In a study by Peng et al[156], APS was found to mitigate metabolic memory-induced ER stress and apoptosis in RPE cells by modulating the miRNA-204/SIRT1 signaling axis. Additionally, other evidence indicates that under high-glucose conditions, the upregulation of miR-182 directly targets and suppresses the antiapoptotic protein Bcl-2, resulting in mitochondrial membrane potential loss, cytochrome c release, and activation of the caspase-9/caspase-3 apoptotic cascade. In contrast, APS dose-dependently downregulates miR-182, restores Bcl-2 expression, alleviates mitochondrial dysfunction, and inhibits apoptosis. Notably, this cytoprotective effect of APS was partially abolished by miR-182 overexpression[157]. Together, these findings underscore the potential of APS to preserve retinal cell viability under hy
Rhein, a naturally occurring anthraquinone compound derived from rhubarb (Rheum palmatum), which is used both as a dietary supplement and as a traditional medicinal herb, has shown therapeutic efficacy in a variety of inflammatory diseases, including chronic kidney disease, sepsis, and cancer[158]. In a study by Liu et al[159], rhein was found to attenuate oxidative stress in retinal Müller cells by activating the AMPK/SIRT1/PGC-1α signaling pathway, thereby reducing the production of ROS and malondialdehyde. Moreover, rhein suppressed VEGF expression, downregulated the expression of proinflammatory cytokines such as IL-1β, IL-6, and TNF-α, and decreased the levels of proapoptotic proteins, including Bcl-2-associated X protein and caspase-3. Collectively, these findings highlight rhein as a promising natural compound that ameliorates retinal inflammation, oxidative stress, and apoptosis through activation of the AMPK/SIRT1/PGC-1α axis in DR. Compared with preclinical evidence, natural compounds are promising candidates for targeting SIRTs in DR. However, their translation into clinical ophthalmic practice remains at an early stage, presenting both opportunities and challenges.
Among the discussed compounds, RES stands out for having entered clinical trials for systemic metabolic conditions, with studies confirming its safety and beneficial effects on endothelial function and inflammatory markers[160,161]. Other bioactive compounds from traditional herbs have also been evaluated in clinical settings. For example, gastrodin was assessed in a randomized, double-blind, placebo-controlled trial for postcardiac surgery delirium[162]; baicalein completed a phase I trial against influenza virus[163]; and PD was investigated in a multicenter randomized controlled trial for irritable bowel syndrome[164]. Similarly, hawthorn polyphenols demonstrated antihypertensive benefits in a randomized, double-blind, placebo-controlled crossover study[165], whereas arbutin has been supported by human trials for skin pigmentation[166]. Ginsenoside Rb1 was studied in a randomized placebo-controlled trial for early chronic kidney disease[167], and ginsenoside Rd has been validated in multiple clinical studies for ischemic stroke[168,169]. Additionally, ART, an approved antimalarial, and APS both possess established clinical safety profiles[170,171]. In contrast, many other compounds, such as PD and HKL, still lack direct clinical data, underscoring a significant gap between mechanistic insight and therapeutic application (Table 1).
| Natural products | SIRT | Possible signaling pathways | Function | Clinical evidence/status |
| Ginsenoside Rd | SIRT1 | GPR30-PKA-LKB1-AMPK, AMPK/SIRT1 | (1) Activation of AMPK increases the NAD+/NADH ratio, promotes LKB1 deacetylation, thereby enhancing the AMPK/SIRT1 interaction and restoring FAO; (2) Through IDH2, mitochondrial NADPH regeneration is maintained, thereby suppressing high glucose-induced NOX2 activation, oxidative stress, mitochondrial dysfunction, and endothelial cell death; and (3) Upregulation of SIRT1 expression increases mitochondrial DNA copy number and enhances the activity of antioxidant enzymes SOD and CAT | Many clinical studies have proved the role of ginsenoside RD in ischemic stroke[168,169]. The direct evidence in DR is still mainly preclinical research[128] |
| Ginsenoside Rb1 | SIRT1 | NAD-PARP-SIRT1 | Alleviated high glucose-induced oxidative damage | A randomized, placebo-controlled study (early chronic kidney disease)[167]. The direct evidence in DR is still mainly preclinical research[102] |
| Ginsenoside Rg1 | SIRT3 | lncRNA SNHG7/miR-2116-5p/SIRT3 | Attenuated the pathological processes in retinal endothelial cells induced by high glucose, including proliferation, migration, and angiogenesis | At present, there is no latest evidence of clinical research. The direct evidence in DR is still mainly preclinical research[128] |
| Resveratrol | SIRT1 | SIRT1/HMGB1 | (1) Activation of AMPK, prevention of SIRT1 inactivation, and reduction of NF-κB phosphorylation; (2) Inhibition of high glucose-induced ROS production and apoptosis through the AMPK/SIRT1/PGC-1α pathway; and (3) Upregulation of SIRT1 expression suppresses HMGB1 upregulation, modulates ferroptosis, and reduces oxidative stress, thereby protecting the BRB | Have completed a number of randomized controlled trials for diabetes and its complications, which confirmed that it can improve endothelial function, reduce oxidative stress and inflammatory markers, and promote the expression of SIRT1[160,161]. The direct evidence in DR is still mainly preclinical research[130] |
| Carnosic acid | SIRT1 | SIRT1/p53/SLC7A11, AMPK pathway | Reduces intracellular Fe2+ and total iron content via the SIRT1/p53/SLC7A11 axis, alleviates ferroptosis-related molecular abnormalities, and inhibits iron deposition | At present, there is no latest evidence of clinical research. The direct evidence in DR is still mainly preclinical research[136] |
| Arbutin | SIRT1 | SIRT1/NF-κB pathway | Elevates SIRT1 protein expression, thereby mitigating high glucose-induced damage in RPE cells | Existing human clinical trials (skin pigmentation)[166]. The direct evidence in DR is still mainly preclinical research[144] |
| Hawthorn polyphenols | SIRT1 | MiR-34a/SIRT1, AMPK/SIRT1/NF-κB, miR-34a/SIRT1/p53 | (1) Modulate the miR-34a/SIRT1 axis, reduces acetylation levels and inhibits high glucose-induced inflammation and apoptosis; and (2) Inhibited the AMPK/SIRT1/NF-κB and miR-34a/SIRT1/p53 pathways, it lowers ROS production and attenuates apoptotic cell death | A randomized, double-blind, placebo-controlled, crossover study (hypertension)[161]. The direct evidence in DR is still mainly preclinical research[140] |
| Polydatin | SIRT1 | SIRT1/NLRP3 pathway | Exerts a protective effect on Müller cells through the SIRT1/NLRP3 inflammasome pathway | A multicentric randomized controlled trial (irritable bowel syndrome)[164]. The direct evidence in DR is still mainly preclinical research[88] |
| Baicalein | SIRT3 | Endoplasmic reticulum stress | By activating SIRT3 and modulating endoplasmic reticulum stress, it ameliorates functional impairment in retinal microvascular endothelial cells under | Phase I clinical trial (influenza virus)[163]. The direct evidence in DR is still mainly preclinical research[147] |
| Honokiol | SIRT3 | Mitochondrial fusion | Protects against diabetic retinal microvascular injury by promoting SIRT3-dependent mitochondrial fusion | At present, there is no latest evidence of clinical research. The direct evidence in DR is still mainly preclinical research[149] |
| Rhein | SIRT1 | AMPK/SIRT1/PGC-1α | Ameliorate HG-induced inflammation, oxidative stress, apoptosis and protect against mitochondrial dysfunction by the activation of the AMPK/SIRT1/PGC-1α signaling pathway | The direct evidence in DR is still mainly preclinical research[159] |
| Astragalus polysaccharide | SIRT1 | MiR-204/SIRT1 | (1) Reversed the sustained up-regulation of miR-204 induced by metabolic memory, thereby alleviating its suppression of the protective protein SIRT1; and (2) The subsequent up-regulation of SIRT1 significantly mitigated endoplasmic reticulum stress and ultimately inhibited the ensuing apoptotic process | Randomized, placebo-controlled, phase 2 study (adjuvant chemotherapy-induced)[171]. The direct evidence in DR is still mainly preclinical research[156] |
| Gastrodin | SIRT1 | SIRT1/TLR4/NF-κBp65 | (1) Upregulate SIRT1 expression, which subsequently inhibits the activation TLR4 and the phosphorylation of NF-κBp65; and (2) Alleviates HG-induced oxidative stress and cell apoptosis | Randomized double-blind placebo-controlled trial (delirium after cardiac surgery)[162]. The direct evidence in DR is still mainly preclinical research[154] |
| Artesunate | SIRT1 | AMPK/SIRT1-dependent autophagy pathway | Increased beclin-1 expression and elevated LC3II/I ratio, thereby inhibiting microglial activation | Approved drug (antimalarial)[170]. The direct evidence in DR is still mainly preclinical research[133] |
This translational gap is largely attributable to suboptimal pharmacokinetic (PK) properties, which represent a fundamental bottleneck in development[172]. For example, despite its multimodal activity, RES has low oral bioavailability due to extensive first-pass metabolism and rapid elimination[173]. HKL faces formulation challenges because of its poor aqueous solubility, and its ability to penetrate retinal tissue remains unclear[174]. These PK limitations, including poor solubility, low bioavailability, rapid metabolism, and lack of tissue targeting, collectively contribute to the scarcity of robust clinical trial data and hinder direct ocular efficacy.
Therefore, developing analogs through structural modification of lead compounds such as RES represents a pivotal strategy to overcome these barriers. Modifications such as esterification and glycosylation can increase the stability, solubility, and delivery. In support of this approach, the derivative piceid octanoate has shown superior retinal protection and delayed photoreceptor degeneration in preclinical models, illustrating how analog development can circumvent the inherent PK constraints of natural compounds[175].
Consequently, future progress must extend beyond biological efficacy to address these PK challenges. Success will depend on strategies that improve ocular bioavailability and tissue targeting, particularly through the design and validation of novel analogs with improved pharmacological profiles, as well as on establishing appropriate dosing regimens and conducting rigorous clinical trials. Bridging this translational gap is essential for fully realizing the therapeutic potential of SIRT-targeting natural compounds in DR.
To overcome the limitations of natural compounds, such as their low potency, poor bioavailability, and off-target effects, considerable research efforts have shifted toward developing potent synthetic SIRT-activating compounds. These agents are designed to more directly and specifically target SIRT enzymes (primarily SIRT1) to increase their catalytic activity or achieve specific inhibition, thereby providing precision pharmacological tools with improved potency, selectivity, and PK profiles.
Among the SIRT family members, SIRT1, SIRT2, and SIRT3 have been the primary focus of drug discovery. SRT1720, SRT2104, and SRT1460 rank among the most extensively studied synthetic SIRT1 activators[176]. Unlike RES, which may act through indirect pathways such as AMPK activation, these imidazothiazole derivatives significantly lower the Michaelis constant for NAD+ and acetylated peptides, exhibiting potency up to a thousand times greater than that of RES[177]. SRT1720 has robust protective effects in metabolic disease models[178]. In retinal studies, SRT1720 alleviated hyperglycemia-CoCl2-induced human retinal microvascular endothelial apoptosis by mediating the expression of SIRT1[179]. Additionally, highly selective synthetic inhibitors such as EX-527 serve as critical pharmacological tools. By blocking SIRT1 activity, EX-527 has been instrumental in confirming that the protective effects of various interventions, such as melatonin in attenuating oxidative stress and inflammation in Müller cells, rhein in promoting cell survival under high glucose, and exendin-4 in alleviating retinal damage, are indeed mediated through the activation of the SIRT1 pathway in models of DR[87,159,180].
With respect to SIRT2, inhibitors such as AGK2 and Tenovin have been investigated primarily for their neuroprotective effects and effects on cancer[181]. For SIRT3, activators that mitigate oxidative stress are under active investigation, although their clinical development is less advanced. In contrast, the development of highly selective and potent synthetic modulators for SIRT4, SIRT5, SIRT6, and SIRT7 remains an emerging frontier. However, exploration of their specific effects on ocular diseases, particularly DR, is extremely limited. This disparity underscores the current focus of therapeutic research and highlights a significant avenue for future investigations.
The translational progress of synthetic SIRT modulators, exemplified by SRT2104 entering clinical trials for systemic diseases, underscores their therapeutic potential[182]. However, their direct application in DR remains at the preclinical validation stage[178,179]. Owing to their superior PK profiles, these synthetic agents represent a shift toward precision medicine in DR therapy and are strong candidates for future topical or systemic ophthalmic treatments[183]. Further evaluation of these optimized pharmacological agents in retinal models will be essential to fully assess their therapeutic potential for DR.
This review elucidates the molecular mechanisms underlying the onset and progression of DR, emphasizing the pivotal regulatory roles of the SIRT family, NAD+ dependent deacylases, which coordinate inflammation, mitochondrial function, and metabolic homeostasis. Accumulating evidence indicates that SIRTs, particularly SIRT1 and SIRT3, exert protective effects by suppressing the NF-κB/NLRP3 inflammatory axis, enhancing mitochondrial integrity, and restoring redox balance.
A critical transition in pathological focus characterizes the progression from NPDR to PDR, shifting from a state of chronic inflammation, oxidative stress, and neuronal impairment to one dominated by pathological neovascularization. The SIRT family members examined herein exhibit distinct and stage-specific functions throughout this continuum. During NPDR, SIRT1, SIRT3, and SIRT6 operate predominantly to safeguard retinal cells, including endothelial cells, RPE cells, and ganglion cells, against metabolic and oxidative insults, thereby promoting tissue homeostasis. As the disease progresses to PDR, the functional emphasis of proteins such as SIRT3 shifts toward orchestrating metabolic repro
Promisingly, various natural compounds and small-molecule activators, such as RES, HKL, and ginsenosides, have shown promising efficacy in preclinical models through SIRT-dependent mechanisms. Despite these advances, translation to clinical application remains challenging. Beyond issues of limited subtype selectivity and off-target effects, a critical hurdle is the insufficient and potentially problematic delivery of therapeutics to the retina. Conventional and even some nanocarrier-based strategies face significant limitations: Cationic nanoparticles may induce retinal toxicity and inflammation, whereas widely used polyethylene glycol carriers raise concerns about long-term immunogenicity and accelerated blood clearance.
Therefore, future research should pursue a dual focus: Precise modulation of SIRT activity alongside innovative strategies to overcome existing delivery limitations. Promising next-generation approaches include peptide-mediated systems for increased BRB penetration and cellular targeting, as well as biodegradable polymer carriers, such as those based on poly(lactic-coglycolic acid), engineered for sustained and safer intraocular drug release. Equally important is the development of novel synthetic analogs through structural modifications aimed at optimizing PK profiles. Ultimately, integrating multi-omics profiling with these advanced, retinaoptimized delivery platforms, rather than relying on generic nanocarriers, will be essential to realizing selective, efficacious, and clinically translatable SIRTtargeted therapies. Ultimately, the development of a holistic therapeutic framework that simultaneously addresses inflammation, mi
| 1. | Mishriky BM, Cummings DM, Powell JR. Diabetes-Related Microvascular Complications - A Practical Approach. Prim Care. 2022;49:239-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 2. | Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N, Bikbov MM, Wang YX, Tang Y, Lu Y, Wong IY, Ting DSW, Tan GSW, Jonas JB, Sabanayagam C, Wong TY, Cheng CY. Global Prevalence of Diabetic Retinopathy and Projection of Burden through 2045: Systematic Review and Meta-analysis. Ophthalmology. 2021;128:1580-1591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1948] [Cited by in RCA: 1405] [Article Influence: 281.0] [Reference Citation Analysis (8)] |
| 3. | Seo H, Park SJ, Song M. Diabetic Retinopathy (DR): Mechanisms, Current Therapies, and Emerging Strategies. Cells. 2025;14:376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 45] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 4. | Steens ILM, Schram MT, Houben AJHM, Berendschot TTJM, Jansen JFA, Backes WH, Koster A, Bosma H, Eussen SJPM, de Galan BE, van Sloten TT. Type 2 diabetes and depression via microvascular dysfunction, neurodegeneration, inflammation, advanced glycation end products (AGEs), and arterial stiffness. Diabetes Obes Metab. 2025;27:4847-4858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 5. | Korkmaz HA, Dogan B, Devebacak A, Değirmenci C, Afrashi F. The Relationship of Serum Diabetes Antibodies With the Development of Early Diabetic Retinopathy Findings in Children With Type 1 Diabetes Mellitus. J Pediatr Ophthalmol Strabismus. 2025;62:135-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 6. | Macaron MM, Al Sabbakh N, Shami MZ, Akrobetu D, Bourdakos NE, Abdulsalam FAM, Nakanishi H, Than CA, Bakri SJ. Anti-VEGF Injections vs. Panretinal Photocoagulation Laser Therapy for Proliferative Diabetic Retinopathy: A Systematic Review and Meta-Analysis. Ophthalmol Retina. 2025;9:105-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 7. | Arrigo A, Aragona E, Bandello F. VEGF-targeting drugs for the treatment of retinal neovascularization in diabetic retinopathy. Ann Med. 2022;54:1089-1111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 128] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 8. | Uludag G, Hassan M, Matsumiya W, Pham BH, Chea S, Trong Tuong Than N, Doan HL, Akhavanrezayat A, Halim MS, Do DV, Nguyen QD. Efficacy and safety of intravitreal anti-VEGF therapy in diabetic retinopathy: what we have learned and what should we learn further? Expert Opin Biol Ther. 2022;22:1275-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 67] [Article Influence: 16.8] [Reference Citation Analysis (1)] |
| 9. | Singh CK, Chhabra G, Ndiaye MA, Garcia-Peterson LM, Mack NJ, Ahmad N. The Role of Sirtuins in Antioxidant and Redox Signaling. Antioxid Redox Signal. 2018;28:643-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 718] [Cited by in RCA: 645] [Article Influence: 80.6] [Reference Citation Analysis (1)] |
| 10. | Carafa V, Rotili D, Forgione M, Cuomo F, Serretiello E, Hailu GS, Jarho E, Lahtela-Kakkonen M, Mai A, Altucci L. Sirtuin functions and modulation: from chemistry to the clinic. Clin Epigenetics. 2016;8:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 185] [Cited by in RCA: 295] [Article Influence: 29.5] [Reference Citation Analysis (1)] |
| 11. | Sosnowska B, Mazidi M, Penson P, Gluba-Brzózka A, Rysz J, Banach M. The sirtuin family members SIRT1, SIRT3 and SIRT6: Their role in vascular biology and atherogenesis. Atherosclerosis. 2017;265:275-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 156] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 12. | Mautone N, Zwergel C, Mai A, Rotili D. Sirtuin modulators: where are we now? A review of patents from 2015 to 2019. Expert Opin Ther Pat. 2020;30:389-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 13. | Pang K, Huang J, Zhang S, Guan Y, Zou N, Kang J, Du H, Zhao D, Abramochkin DV, Chen H, Zhang N, Gu Y, Liu N, Niu Y, Xiong Z, Zhang X, Lu F, Fan H, Tian J, Yu B, Li S, Zhang W. Translocation of SIRT6 promotes glycolysis reprogramming to exacerbate diabetic angiopathy. Redox Biol. 2025;85:103736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 14. | Mimura T, Noma H. Oxidative Stress in Diabetic Retinopathy: A Comprehensive Review of Mechanisms, Biomarkers, and Therapeutic Perspectives. Antioxidants (Basel). 2025;14:1204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 15. | Biswas A, Choudhury AD, Agrawal S, Bisen AC, Sanap SN, Verma SK, Kumar M, Mishra A, Kumar S, Chauhan M, Bhatta RS. Recent Insights into the Etiopathogenesis of Diabetic Retinopathy and Its Management. J Ocul Pharmacol Ther. 2024;40:13-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 16. | Cai X, McGinnis JF. Diabetic Retinopathy: Animal Models, Therapies, and Perspectives. J Diabetes Res. 2016;2016:3789217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 17. | Cagnone G, Pundir S, Betus C, Agnihotri T, Ren A, -Kim JS, Harvey NR, Heckel E, Chen MX, Situ A, Gaub P, Kim N, Das A, Leclerc S, Wünnemann F, Berillon L, Andelfinger G, Crespo-Garcia S, Dubrac A, Rezende FA, Clish CB, Maranda B, Rivera JC, Smith LEH, Sapieha P, Joyal JS. Metabolic reprogramming of the neovascular niche promotes regenerative angiogenesis in proliferative retinopathy. Nat Commun. 2025;16:5377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 18. | Abu-El-Asrar AM, Dralands L, Missotten L, Al-Jadaan IA, Geboes K. Expression of apoptosis markers in the retinas of human subjects with diabetes. Invest Ophthalmol Vis Sci. 2004;45:2760-2766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 205] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 19. | Safi SZ, Qvist R, Kumar S, Batumalaie K, Ismail IS. Molecular mechanisms of diabetic retinopathy, general preventive strategies, and novel therapeutic targets. Biomed Res Int. 2014;2014:801269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 168] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 20. | Wang W, Matsukura M, Fujii I, Ito K, Zhao JE, Shinohara M, Wang YQ, Zhang XM. Inhibition of high glucose-induced VEGF and ICAM-1 expression in human retinal pigment epithelium cells by targeting ILK with small interference RNA. Mol Biol Rep. 2012;39:613-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 21. | Adki KM, Kulkarni YA. Paeonol attenuates retinopathy in streptozotocin-induced diabetes in rats by regulating the oxidative stress and polyol pathway. Front Pharmacol. 2022;13:891485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 22. | Kowluru RA. Diabetic Retinopathy and NADPH Oxidase-2: A Sweet Slippery Road. Antioxidants (Basel). 2021;10:783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (2)] |
| 23. | Nerlich AG, Sauer U, Kolm-Litty V, Wagner E, Koch M, Schleicher ED. Expression of glutamine:fructose-6-phosphate amidotransferase in human tissues: evidence for high variability and distinct regulation in diabetes. Diabetes. 1998;47:170-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 61] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 24. | Ono Y, Aoki S, Ohnishi K, Yasuda T, Kawano K, Tsukada Y. Increased serum levels of advanced glycation end-products and diabetic complications. Diabetes Res Clin Pract. 1998;41:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 94] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 25. | Tan J, Xiao A, Yang L, Tao YL, Shao Y, Zhou Q. Diabetes and high-glucose could upregulate the expression of receptor for activated C kinase 1 in retina. World J Diabetes. 2024;15:519-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 26. | Titchenell PM, Antonetti DA. Using the past to inform the future: anti-VEGF therapy as a road map to develop novel therapies for diabetic retinopathy. Diabetes. 2013;62:1808-1815. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 27. | Beatty S, Koh H, Phil M, Henson D, Boulton M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv Ophthalmol. 2000;45:115-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1246] [Cited by in RCA: 1336] [Article Influence: 51.4] [Reference Citation Analysis (1)] |
| 28. | Wróblewska J, Nuszkiewicz J, Wróblewski M, Wróblewska W, Woźniak A. Selected Trace Elements and Their Impact on Redox Homeostasis in Eye Health. Biomolecules. 2024;14:1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 29. | Dascalu AM, Anghelache A, Stana D, Costea AC, Nicolae VA, Tanasescu D, Costea DO, Tribus LC, Zgura A, Serban D, Duta L, Tudosie M, Balasescu SA, Tanasescu C, Tudosie MS. Serum levels of copper and zinc in diabetic retinopathy: Potential new therapeutic targets (Review). Exp Ther Med. 2022;23:324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 30. | Jiang D, Yang X, Ge M, Hu H, Xu C, Wen S, Deng H, Mei X. Zinc defends against Parthanatos and promotes functional recovery after spinal cord injury through SIRT3-mediated anti-oxidative stress and mitophagy. CNS Neurosci Ther. 2023;29:2857-2872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (1)] |
| 31. | Andreini C, Banci L, Bertini I, Rosato A. Counting the zinc-proteins encoded in the human genome. J Proteome Res. 2006;5:196-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 700] [Cited by in RCA: 748] [Article Influence: 37.4] [Reference Citation Analysis (1)] |
| 32. | Alanazi ST, Salama SA, Althobaiti MM, Alotaibi RA, AlAbdullatif AA, Musa A, Harisa GI. Alleviation of Copper-Induced Hepatotoxicity by Bergenin: Diminution of Oxidative Stress, Inflammation, and Apoptosis via Targeting SIRT1/FOXO3a/NF-κB Axes and p38 MAPK Signaling. Biol Trace Elem Res. 2025;203:3195-3207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 33. | Yu X, Chen X, Wu W, Tang H, Su Y, Lian G, Zhang Y, Xie L. Zinc Alleviates Diabetic Muscle Atrophy via Modulation of the SIRT1/FoxO1 Autophagy Pathway Through GPR39. J Cachexia Sarcopenia Muscle. 2025;16:e13771. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 34. | Xu H, Dong X, Wang J, Cheng X, Qu S, Jia T, Liu J, Li Z, Yao Y. Association of Calcium, Magnesium, Zinc, and Copper Intakes with Diabetic Retinopathy in Diabetics: National Health and Nutrition Examination Survey, 2007-2018. Curr Eye Res. 2023;48:485-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 35. | Robinson R, Srinivasan M, Shanmugam A, Ward A, Ganapathy V, Bloom J, Sharma A, Sharma S. Interleukin-6 trans-signaling inhibition prevents oxidative stress in a mouse model of early diabetic retinopathy. Redox Biol. 2020;34:101574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 36. | Zhou T, Zhou KK, Lee K, Gao G, Lyons TJ, Kowluru R, Ma JX. The role of lipid peroxidation products and oxidative stress in activation of the canonical wingless-type MMTV integration site (WNT) pathway in a rat model of diabetic retinopathy. Diabetologia. 2011;54:459-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 5.4] [Reference Citation Analysis (4)] |
| 37. | Chiang YF, Chen HY, Chang YJ, Shih YH, Shieh TM, Wang KL, Hsia SM. Protective Effects of Fucoxanthin on High Glucose- and 4-Hydroxynonenal (4-HNE)-Induced Injury in Human Retinal Pigment Epithelial Cells. Antioxidants (Basel). 2020;9:1176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 38. | Jaganjac M, Zarkovic N. Lipid Peroxidation Linking Diabetes and Cancer: The Importance of 4-Hydroxynonenal. Antioxid Redox Signal. 2022;37:1222-1233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 39. | Chen Q, Tang L, Xin G, Li S, Ma L, Xu Y, Zhuang M, Xiong Q, Wei Z, Xing Z, Niu H, Huang W. Oxidative stress mediated by lipid metabolism contributes to high glucose-induced senescence in retinal pigment epithelium. Free Radic Biol Med. 2019;130:48-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 40. | Yuan F, Han S, Li Y, Li S, Li D, Tian Q, Feng R, Shao Y, Liang X, Wang J, Lei H, Li X, Duan Y. miR-214-3p attenuates ferroptosis-induced cellular damage in a mouse model of diabetic retinopathy through the p53/SLC7A11/GPX4 axis. Exp Eye Res. 2025;253:110299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 41. | Chen S, Sun D, Zhang S, Xu L, Wang N, Li H, Xu X, Wei F. TIN2 modulates FOXO1 mitochondrial shuttling to enhance oxidative stress-induced apoptosis in retinal pigment epithelium under hyperglycemia. Cell Death Differ. 2024;31:1487-1505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 42. | Jalgaonkar MP, Parmar UM, Kulkarni YA, Oza MJ. SIRT1-FOXOs activity regulates diabetic complications. Pharmacol Res. 2022;175:106014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 43. | Shanbagh S, Gadde SG, Shetty R, Heymans S, Abilash VG, Chaurasia SS, Ghosh A. Hyperglycemia-induced miR182-5p drives glycolytic and angiogenic response in Proliferative Diabetic Retinopathy and RPE cells via depleting FoxO1. Exp Eye Res. 2024;238:109713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 44. | Dharmarajan S, Carrillo C, Qi Z, Wilson JM, Baucum AJ 2nd, Sorenson CM, Sheibani N, Belecky-Adams TL. Retinal inflammation in murine models of type 1 and type 2 diabetes with diabetic retinopathy. Diabetologia. 2023;66:2170-2185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 45. | Prud'homme GJ, Wang Q. Anti-Inflammatory Role of the Klotho Protein and Relevance to Aging. Cells. 2024;13:1413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 46. | Mahajan N, Arora P, Sandhir R. Perturbed Biochemical Pathways and Associated Oxidative Stress Lead to Vascular Dysfunctions in Diabetic Retinopathy. Oxid Med Cell Longev. 2019;2019:8458472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 47. | Zheng X, Wan J, Tan G. The mechanisms of NLRP3 inflammasome/pyroptosis activation and their role in diabetic retinopathy. Front Immunol. 2023;14:1151185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 48. | Volpe CMO, Villar-Delfino PH, Dos Anjos PMF, Nogueira-Machado JA. Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death Dis. 2018;9:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 979] [Cited by in RCA: 868] [Article Influence: 108.5] [Reference Citation Analysis (5)] |
| 49. | Liu W, Tong B, Xiong J, Zhu Y, Lu H, Xu H, Yang X, Wang F, Yu P, Hu Y. Identification of macrophage polarisation and mitochondria-related biomarkers in diabetic retinopathy. J Transl Med. 2025;23:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 50. | Hu X, Lv J, Zhao Y, Li X, Qi W, Wang X. Important regulatory role of mitophagy in diabetic microvascular complications. J Transl Med. 2025;23:269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (1)] |
| 51. | McCann MA, Baccouche B, Li Y, Roy P, Sheth N, Lim JI, Mieler WF, Chau FY, Ulanski LJ, Chan RVP, Munro M, Hyde RA, Berek C, Ong A, De S, Siedlecki B, Chee RI, Leiderman YI, Heiferman MJ, Kazlauskas A. Proliferative diabetic retinopathy subtypes defined by immune defense and endothelial mitochondrial dysfunction. Signal Transduct Target Ther. 2025;10:350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 52. | Jiménez-Loygorri JI, Benítez-Fernández R, Viedma-Poyatos Á, Zapata-Muñoz J, Villarejo-Zori B, Gómez-Sintes R, Boya P. Mitophagy in the retina: Viewing mitochondrial homeostasis through a new lens. Prog Retin Eye Res. 2023;96:101205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 59] [Article Influence: 19.7] [Reference Citation Analysis (1)] |
| 53. | Zhang SM, Fan B, Li YL, Zuo ZY, Li GY. Oxidative Stress-Involved Mitophagy of Retinal Pigment Epithelium and Retinal Degenerative Diseases. Cell Mol Neurobiol. 2023;43:3265-3276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 67] [Reference Citation Analysis (0)] |
| 54. | Alka K, Kumar J, Kowluru RA. Impaired mitochondrial dynamics and removal of the damaged mitochondria in diabetic retinopathy. Front Endocrinol (Lausanne). 2023;14:1160155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 55. | Mahaling B, Low SWY, Beck M, Kumar D, Ahmed S, Connor TB, Ahmad B, Chaurasia SS. Damage-Associated Molecular Patterns (DAMPs) in Retinal Disorders. Int J Mol Sci. 2022;23:2591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 56. | Xie J, Cui Y, Chen X, Yu H, Chen J, Huang T, He M, Zhang L, Meng Q. VDAC1 regulates mitophagy in NLRP3 inflammasome activation in retinal capillary endothelial cells under high-glucose conditions. Exp Eye Res. 2021;209:108640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 57. | Lu M, Ye X, Deng X, Jiao F, Ren H, Xing Y. Inhibition of LCP2 in T cells alleviated apoptosis and oxidative stress via PD-1/PD-L1 in diabetic retinopathy. Int Immunopharmacol. 2025;163:115240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 58. | Birtel J, von Landenberg C, Gliem M, Gliem C, Reimann J, Kunz WS, Herrmann P, Betz C, Caswell R, Nesbitt V, Kornblum C, Charbel Issa P. Mitochondrial Retinopathy. Ophthalmol Retina. 2022;6:65-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 59. | Młynarska E, Czarnik W, Dzieża N, Jędraszak W, Majchrowicz G, Prusinowski F, Stabrawa M, Rysz J, Franczyk B. Type 2 Diabetes Mellitus: New Pathogenetic Mechanisms, Treatment and the Most Important Complications. Int J Mol Sci. 2025;26:1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 136] [Article Influence: 136.0] [Reference Citation Analysis (1)] |
| 60. | Antonetti DA, Silva PS, Stitt AW. Current understanding of the molecular and cellular pathology of diabetic retinopathy. Nat Rev Endocrinol. 2021;17:195-206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 498] [Cited by in RCA: 417] [Article Influence: 83.4] [Reference Citation Analysis (1)] |
| 61. | O'Leary F, Campbell M. The blood-retina barrier in health and disease. FEBS J. 2023;290:878-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 213] [Article Influence: 71.0] [Reference Citation Analysis (1)] |
| 62. | Jobling AI, Greferath U, Dixon MA, Quiriconi P, Eyar B, van Koeverden AK, Mills SA, Vessey KA, Bui BV, Fletcher EL. Microglial regulation of the retinal vasculature in health and during the pathology associated with diabetes. Prog Retin Eye Res. 2025;106:101349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 63. | Pang B, Ni Q, Di S, Du LJ, Qin YL, Li QW, Li M, Tong XL. Luo Tong Formula Alleviates Diabetic Retinopathy in Rats Through Micro-200b Target. Front Pharmacol. 2020;11:551766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 64. | van Splunder H, Villacampa P, Martínez-Romero A, Graupera M. Pericytes in the disease spotlight. Trends Cell Biol. 2024;34:58-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 116] [Article Influence: 58.0] [Reference Citation Analysis (1)] |
| 65. | Al Sabaani N. Kaempferol Protects Against Hydrogen Peroxide-Induced Retinal Pigment Epithelium Cell Inflammation and Apoptosis by Activation of SIRT1 and Inhibition of PARP1. J Ocul Pharmacol Ther. 2020;36:563-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 66. | Mazzeo A, Gai C, Trento M, Porta M, Beltramo E. Effects of thiamine and fenofibrate on high glucose and hypoxia-induced damage in cell models of the inner blood-retinal barrier. Acta Diabetol. 2020;57:1423-1433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 67. | Hong H, Lee J. Thyroid-Stimulating Hormone as a Biomarker for Stress After Thyroid Surgery: A Prospective Cohort Study. Med Sci Monit. 2022;28:e937957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 68. | Chaudhary V, Mar F, Amador MJ, Chang A, Gibson K, Joussen AM, Kim JE, Lee J, Margaron P, Saffar I, Wong D, Wykoff C, Sadda S. Emerging clinical evidence of a dual role for Ang-2 and VEGF-A blockade with faricimab in retinal diseases. Graefes Arch Clin Exp Ophthalmol. 2025;263:1239-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 69. | Uemura A, Fruttiger M, D'Amore PA, De Falco S, Joussen AM, Sennlaub F, Brunck LR, Johnson KT, Lambrou GN, Rittenhouse KD, Langmann T. VEGFR1 signaling in retinal angiogenesis and microinflammation. Prog Retin Eye Res. 2021;84:100954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 257] [Article Influence: 51.4] [Reference Citation Analysis (5)] |
| 70. | Ramos H, Hernández C, Simó R, Simó-Servat O. Inflammation: The Link between Neural and Vascular Impairment in the Diabetic Retina and Therapeutic Implications. Int J Mol Sci. 2023;24:8796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (4)] |
| 71. | Simão S, Costa MÂ, Sun JK, Cunha-Vaz J, Simó R; European Consortium for the Early Treatment of Diabetic Retinopathy (EUROCONDOR). Development of a Normative Database for Multifocal Electroretinography in the Context of a Multicenter Clinical Trial. Ophthalmic Res. 2017;57:107-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (3)] |
| 72. | Crincoli E, Sacconi R, Querques L, Querques G. OCT angiography 2023 update: focus on diabetic retinopathy. Acta Diabetol. 2024;61:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 73. | Sohn EH, van Dijk HW, Jiao C, Kok PH, Jeong W, Demirkaya N, Garmager A, Wit F, Kucukevcilioglu M, van Velthoven ME, DeVries JH, Mullins RF, Kuehn MH, Schlingemann RO, Sonka M, Verbraak FD, Abràmoff MD. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci U S A. 2016;113:E2655-E2664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 507] [Article Influence: 50.7] [Reference Citation Analysis (2)] |
| 74. | Obara EA, Hannibal J, Heegaard S, Fahrenkrug J. Loss of Melanopsin-Expressing Retinal Ganglion Cells in Patients With Diabetic Retinopathy. Invest Ophthalmol Vis Sci. 2017;58:2187-2192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 75. | Yu Y, Wang J, Liu Q, Wei F, Xie X, Zhang M. Integrated serum pharmacochemistry and serum pharmacology to investigate the active components and mechanism of Bushen Huoxue Prescription in the treatment of diabetic retinopathy. J Pharm Biomed Anal. 2023;235:115586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 76. | Zhang Z, Peng S, Xu T, Liu J, Zhao L, Xu H, Zhang W, Zhu Y, Yang Z. Retinal Microenvironment-Protected Rhein-GFFYE Nanofibers Attenuate Retinal Ischemia-Reperfusion Injury via Inhibiting Oxidative Stress and Regulating Microglial/Macrophage M1/M2 Polarization. Adv Sci (Weinh). 2023;10:e2302909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 77. | Li X, Lv J, Li J, Ren X. Kir4.1 may represent a novel therapeutic target for diabetic retinopathy (Review). Exp Ther Med. 2021;22:1021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 78. | Xi X, Liu X, Chen Q, Ma J, Wang X, Gui Y, Zhang Y, Li Y. Acteoside relieves diabetic retinopathy through the inhibition of Müller cell reactive hyperplasia by regulating TXNIP and mediating Kir4.1 channels in a PI3K/Akt-dependent manner. PLoS One. 2024;19:e0312565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 79. | Abyadeh M, Gupta V, Paulo JA, Mahmoudabad AG, Shadfar S, Mirshahvaladi S, Gupta V, Nguyen CTO, Finkelstein DI, You Y, Haynes PA, Salekdeh GH, Graham SL, Mirzaei M. Amyloid-beta and tau protein beyond Alzheimer's disease. Neural Regen Res. 2024;19:1262-1276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 65] [Article Influence: 32.5] [Reference Citation Analysis (1)] |
| 80. | Rolev KD, Shu XS, Ying Y. Targeted pharmacotherapy against neurodegeneration and neuroinflammation in early diabetic retinopathy. Neuropharmacology. 2021;187:108498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 81. | Karbasforooshan H, Karimi G. The role of SIRT1 in diabetic retinopathy. Biomed Pharmacother. 2018;97:190-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 96] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 82. | Jin Q, Liu T, Ma F, Fu T, Yang L, Mao H, Wang Y, Peng L, Li P, Zhan Y. Roles of Sirt1 and its modulators in diabetic microangiopathy: A review. Int J Biol Macromol. 2024;264:130761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 83. | Taurone S, De Ponte C, Rotili D, De Santis E, Mai A, Fiorentino F, Scarpa S, Artico M, Micera A. Biochemical Functions and Clinical Characterizations of the Sirtuins in Diabetes-Induced Retinal Pathologies. Int J Mol Sci. 2022;23:4048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 84. | Xu Y, Zou H, Ding Q, Zou Y, Tang C, Lu Y, Xu X. tiRNA-Val promotes angiogenesis via Sirt1-Hif-1α axis in mice with diabetic retinopathy. Biol Res. 2022;55:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 85. | Cui C, Li Y, Liu Y. Down-regulation of miR-377 suppresses high glucose and hypoxia-induced angiogenesis and inflammation in human retinal endothelial cells by direct up-regulation of target gene SIRT1. Hum Cell. 2019;32:260-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 86. | Mishra M, Duraisamy AJ, Kowluru RA. Sirt1: A Guardian of the Development of Diabetic Retinopathy. Diabetes. 2018;67:745-754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 105] [Article Influence: 13.1] [Reference Citation Analysis (5)] |
| 87. | Tu Y, Song E, Wang Z, Ji N, Zhu L, Wang K, Sun H, Zhang Y, Zhu Q, Liu X, Zhu M. Melatonin attenuates oxidative stress and inflammation of Müller cells in diabetic retinopathy via activating the Sirt1 pathway. Biomed Pharmacother. 2021;137:111274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 84] [Article Influence: 16.8] [Reference Citation Analysis (1)] |
| 88. | Wang B, Qu X, Su A, Zhu H. PD protects Müller cells through the SIRT1/NLRP3 inflammasome pathway. Int Ophthalmol. 2024;44:97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 89. | Lian L, Le Z, Wang Z, Chen YA, Jiao X, Qi H, Hejtmancik JF, Ma X, Zheng Q, Ren Y. SIRT1 Inhibits High Glucose-Induced TXNIP/NLRP3 Inflammasome Activation and Cataract Formation. Invest Ophthalmol Vis Sci. 2023;64:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 90. | Mohammad G, Kumar J, Kowluru RA. Mitochondrial Genome-Encoded Long Noncoding RNA Cytochrome B and Mitochondrial Dysfunction in Diabetic Retinopathy. Antioxid Redox Signal. 2023;39:817-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 91. | Yu J, Ke L, Zhou J, Ding C, Yang H, Yan D, Yu C. Stachydrine Relieved the Inflammation and Promoted the Autophagy in Diabetes Retinopathy Through Activating the AMPK/SIRT1 Signaling Pathway. Diabetes Metab Syndr Obes. 2023;16:2593-2604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 92. | Law M, Wang PC, Zhou ZY, Wang Y. From Microcirculation to Aging-Related Diseases: A Focus on Endothelial SIRT1. Pharmaceuticals (Basel). 2024;17:1495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 93. | Tong P, Peng QH, Gu LM, Xie WW, Li WJ. LncRNA-MEG3 alleviates high glucose induced inflammation and apoptosis of retina epithelial cells via regulating miR-34a/SIRT1 axis. Exp Mol Pathol. 2019;107:102-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 82] [Article Influence: 10.3] [Reference Citation Analysis (3)] |
| 94. | Wu B, You S, Qian H, Wu S, Lu S, Zhang Y, Sun Y, Zhang N. The role of SIRT2 in vascular-related and heart-related diseases: A review. J Cell Mol Med. 2021;25:6470-6478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 34] [Reference Citation Analysis (1)] |
| 95. | Lim J, Son J, Ryu J, Kim JE. SIRT2 Affects Primary Cilia Formation by Regulating mTOR Signaling in Retinal Pigmented Epithelial Cells. Int J Mol Sci. 2020;21:2240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 96. | Wortham M, Ramms B, Zeng C, Benthuysen JR, Sai S, Pollow DP, Liu F, Schlichting M, Harrington AR, Liu B, Prakash TP, Pirie EC, Zhu H, Baghdasarian S, Lee ST, Ruthig VA, Wells KL, Auwerx J, Shirihai OS, Sander M. The protein deacetylase SIRT2 exerts metabolic control over adaptive β cell proliferation. J Clin Invest. 2025;135:e187020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 97. | Zhu C, Dong X, Wang X, Zheng Y, Qiu J, Peng Y, Xu J, Chai Z, Liu C. Multiple Roles of SIRT2 in Regulating Physiological and Pathological Signal Transduction. Genet Res (Camb). 2022;2022:9282484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 98. | Fan Z, Hu Y, Chen L, Lu X, Zheng L, Ma D, Li Z, Zhong J, Lin L, Zhang S, Zhang G. Multiplatform tear proteomic profiling reveals novel non-invasive biomarkers for diabetic retinopathy. Eye (Lond). 2024;38:1509-1517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 99. | Wang X, Zhang P, Xie J, Zuo X. USP39 promotes retinal pathological angiogenesis in retinopathy of prematurity by stabilizing SIRT2 expression through deubiquitination. Int Ophthalmol. 2025;45:39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 100. | Chen Y, Wang Y, Yu D, Zhao H, Li P. Sirtuin 3 in diabetic kidney disease: mechanisms and pharmacotherapy. Ren Fail. 2025;47:2543927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 101. | Ji Z, Liu GH, Qu J. Mitochondrial sirtuins, key regulators of aging. Life Med. 2025;4:lnaf019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 102. | Fan C, Ma Q, Xu M, Qiao Y, Zhang Y, Li P, Bi Y, Tang M. Ginsenoside Rb1 Attenuates High Glucose-Induced Oxidative Injury via the NAD-PARP-SIRT Axis in Rat Retinal Capillary Endothelial Cells. Int J Mol Sci. 2019;20:4936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 103. | Wang D, Cao L, Zhou X, Wang G, Ma Y, Hao X, Fan H. Mitigation of honokiol on fluoride-induced mitochondrial oxidative stress, mitochondrial dysfunction, and cognitive deficits through activating AMPK/PGC-1α/Sirt3. J Hazard Mater. 2022;437:129381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 129] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 104. | Salvatori I, Valle C, Ferri A, Carrì MT. SIRT3 and mitochondrial metabolism in neurodegenerative diseases. Neurochem Int. 2017;109:184-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 105. | Mao XB, Cheng YH, Peng KS, You ZP. Sirtuin (Sirt) 3 Overexpression Prevents Retinopathy in Streptozotocin-Induced Diabetic Rats. Med Sci Monit. 2020;26:e920883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 106. | Gao J, Zheng Z, Gu Q, Chen X, Liu X, Xu X. Deacetylation of MnSOD by PARP-regulated SIRT3 protects retinal capillary endothelial cells from hyperglycemia-induced damage. Biochem Biophys Res Commun. 2016;472:425-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 107. | Shi J, Liu M, Zhu H, Jiang C. SIRT3 mitigates high glucose-induced damage in retinal microvascular endothelial cells via OPA1-mediated mitochondrial dynamics. Exp Cell Res. 2025;444:114320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 108. | Huang L, Yao T, Chen J, Zhang Z, Yang W, Gao X, Dan Y, He Y. Effect of Sirt3 on retinal pigment epithelial cells in high glucose through Foxo3a/ PINK1-Parkin pathway mediated mitophagy. Exp Eye Res. 2022;218:109015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (1)] |
| 109. | Yang W, Qiu C, Lv H, Zhang Z, Yao T, Huang L, Wu G, Zhang X, Chen J, He Y. Sirt3 Protects Retinal Pigment Epithelial Cells From High Glucose-Induced Injury by Promoting Mitophagy Through the AMPK/mTOR/ULK1 Pathway. Transl Vis Sci Technol. 2024;13:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 110. | Gaonkar B, Prabhu K, Rao P, Kamat A, Rao Addoor K, Varma M. Plasma angiogenesis and oxidative stress markers in patients with diabetic retinopathy. Biomarkers. 2020;25:397-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (3)] |
| 111. | Zhou Y, Zhang H, He B, Du J, Lin H, Cerione RA, Hao Q. The bicyclic intermediate structure provides insights into the desuccinylation mechanism of human sirtuin 5 (SIRT5). J Biol Chem. 2012;287:28307-28314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 112. | Tang L, Sun Q, Luo J, Peng S. Metformin hydrochloride improves hepatic glucolipid metabolism in diabetes progression through SIRT5-mediated ECHA desuccinylation. Sci Rep. 2025;15:7768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 113. | Rezaie T, Child A, Hitchings R, Brice G, Miller L, Coca-Prados M, Héon E, Krupin T, Ritch R, Kreutzer D, Crick RP, Sarfarazi M. Adult-onset primary open-angle glaucoma caused by mutations in optineurin. Science. 2002;295:1077-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 645] [Cited by in RCA: 717] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 114. | Wild P, Farhan H, McEwan DG, Wagner S, Rogov VV, Brady NR, Richter B, Korac J, Waidmann O, Choudhary C, Dötsch V, Bumann D, Dikic I. Phosphorylation of the autophagy receptor optineurin restricts Salmonella growth. Science. 2011;333:228-233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1127] [Cited by in RCA: 1075] [Article Influence: 71.7] [Reference Citation Analysis (1)] |
| 115. | Zhang Y, Li T, Cai X, Long D, Wang X, Liu C, Wu Q. Sirt5-mediated desuccinylation of OPTN protects retinal ganglion cells from autophagic flux blockade in diabetic retinopathy. Cell Death Discov. 2022;8:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 116. | Cheng J, Keuthan CJ, Esumi N. The many faces of SIRT6 in the retina and retinal pigment epithelium. Front Cell Dev Biol. 2023;11:1244765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 117. | Chang AR, Ferrer CM, Mostoslavsky R. SIRT6, a Mammalian Deacylase with Multitasking Abilities. Physiol Rev. 2020;100:145-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 189] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 118. | Zou Y, Zhang J, Xu J, Fu L, Xu Y, Wang X, Li Z, Zhu L, Sun H, Zheng H, Guo J. SIRT6 inhibition delays peripheral nerve recovery by suppressing migration, phagocytosis and M2-polarization of macrophages. Cell Biosci. 2021;11:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 119. | Elkhwanky MS, Hakkola J. Extranuclear Sirtuins and Metabolic Stress. Antioxid Redox Signal. 2018;28:662-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 120. | Yamagata K, Mizumoto T, Yoshizawa T. The Emerging Role of SIRT7 in Glucose and Lipid Metabolism. Cells. 2023;13:48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 121. | Betsinger CN, Cristea IM. Mitochondrial Function, Metabolic Regulation, and Human Disease Viewed through the Prism of Sirtuin 4 (SIRT4) Functions. J Proteome Res. 2019;18:1929-1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 122. | Maghbooli Z, Emamgholipour S, Aliakbar S, Amini M, Gorgani-Firuzjaee S, Hossein-Nezhad A. Differential expressions of SIRT1, SIRT3, and SIRT4 in peripheral blood mononuclear cells from patients with type 2 diabetic retinopathy. Arch Physiol Biochem. 2020;126:363-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 123. | Li X, Liu J, Lu L, Huang T, Hou W, Wang F, Yu L, Wu F, Qi J, Chen X, Meng Z, Zhu M. Sirt7 associates with ELK1 to participate in hyperglycemia memory and diabetic nephropathy via modulation of DAPK3 expression and endothelial inflammation. Transl Res. 2022;247:99-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 124. | Fan S, Zhang Z, Su H, Xu P, Qi H, Zhao D, Li X. Panax ginseng clinical trials: Current status and future perspectives. Biomed Pharmacother. 2020;132:110832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 125. | Fan W, Fan L, Wang Z, Mei Y, Liu L, Li L, Yang L, Wang Z. Rare ginsenosides: A unique perspective of ginseng research. J Adv Res. 2024;66:303-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 83] [Article Influence: 41.5] [Reference Citation Analysis (1)] |
| 126. | Tang K, Huang C, Huang Z, Wang Z, Tan N. GPR30-driven fatty acid oxidation targeted by ginsenoside Rd maintains mitochondrial redox homeostasis to restore vascular barrier in diabetic retinopathy. Cardiovasc Diabetol. 2025;24:121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 127. | Tang K, Qin W, Wei R, Jiang Y, Fan L, Wang Z, Tan N. Ginsenoside Rd ameliorates high glucose-induced retinal endothelial injury through AMPK-STRT1 interdependence. Pharmacol Res. 2022;179:106123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (4)] |
| 128. | Xue L, Hu M, Li J, Li Y, Zhu Q, Zhou G, Zhang X, Zhou Y, Zhang J, Ding P. Ginsenoside Rg1 Inhibits High Glucose-Induced Proliferation, Migration, and Angiogenesis in Retinal Endothelial Cells by Regulating the lncRNA SNHG7/miR-2116-5p/SIRT3 Axis. J Oncol. 2022;2022:6184631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 129. | Kubota S, Ozawa Y, Kurihara T, Sasaki M, Yuki K, Miyake S, Noda K, Ishida S, Tsubota K. Roles of AMP-activated protein kinase in diabetes-induced retinal inflammation. Invest Ophthalmol Vis Sci. 2011;52:9142-9148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 107] [Article Influence: 7.1] [Reference Citation Analysis (3)] |
| 130. | Peng Y, Hu L, Xu H, Fang J, Zhong H. Resveratrol alleviates reactive oxygen species and inflammation in diabetic retinopathy via SIRT1/HMGB1 pathway-mediated ferroptosis. Toxicol Appl Pharmacol. 2025;495:117214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 131. | Chang Y, Lyu T, Luan X, Yang Y, Cao Y, Qiu Y, Feng H. Artesunate-multiple pharmacological effects beyond treating malaria. Eur J Med Chem. 2025;286:117292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 132. | Liu X, Cai Y, Zhang Y, Zhang H, Tian S, Gong Y, Song Q, Chen X, Ma X, Wen Y, Chen Y, Zeng J. Artesunate: A potential drug for the prevention and treatment from hepatitis to hepatocellular carcinoma. Pharmacol Res. 2024;210:107526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (1)] |
| 133. | Li L, Chen J, Zhou Y, Zhang J, Chen L. Artesunate alleviates diabetic retinopathy by activating autophagy via the regulation of AMPK/SIRT1 pathway. Arch Physiol Biochem. 2023;129:943-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 134. | Vallverdú-Queralt A, Regueiro J, Martínez-Huélamo M, Rinaldi Alvarenga JF, Leal LN, Lamuela-Raventos RM. A comprehensive study on the phenolic profile of widely used culinary herbs and spices: rosemary, thyme, oregano, cinnamon, cumin and bay. Food Chem. 2014;154:299-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 238] [Article Influence: 19.8] [Reference Citation Analysis (1)] |
| 135. | Xia G, Wang X, Sun H, Qin Y, Fu M. Carnosic acid (CA) attenuates collagen-induced arthritis in db/db mice via inflammation suppression by regulating ROS-dependent p38 pathway. Free Radic Biol Med. 2017;108:418-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 136. | Hasei S, Yamamotoya T, Nakatsu Y, Ohata Y, Itoga S, Nonaka Y, Matsunaga Y, Sakoda H, Fujishiro M, Kushiyama A, Asano T. Carnosic Acid and Carnosol Activate AMPK, Suppress Expressions of Gluconeogenic and Lipogenic Genes, and Inhibit Proliferation of HepG2 Cells. Int J Mol Sci. 2021;22:4040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 137. | Ke J, Li MT, Xu S, Ma J, Liu MY, Han Y. Advances for pharmacological activities of Polygonum cuspidatum - A review. Pharm Biol. 2023;61:177-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 57] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 138. | Yasmin F, Tang KS. Potential role of polydatin in treating diabetes mellitus and diabetes-related chronic complications. Biosci Rep. 2025;45:303-323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 139. | Gong W, Li J, Chen Z, Huang J, Chen Q, Cai W, Liu P, Huang H. Polydatin promotes Nrf2-ARE anti-oxidative pathway through activating CKIP-1 to resist HG-induced up-regulation of FN and ICAM-1 in GMCs and diabetic mice kidneys. Free Radic Biol Med. 2017;106:393-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 66] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 140. | Wei H, Rui J, Yan X, Xu R, Chen S, Zhang B, Wang L, Zhang Z, Zhu C, Ma M, Xiao H. Plant polyphenols as natural bioactives for alleviating lipid metabolism disorder: Mechanisms and application challenges. Food Res Int. 2025;203:115682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 141. | Li R, Luan F, Zhao Y, Wu M, Lu Y, Tao C, Zhu L, Zhang C, Wan L. Crataegus pinnatifida: A botanical, ethnopharmacological, phytochemical, and pharmacological overview. J Ethnopharmacol. 2023;301:115819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 142. | Wang S, Du Q, Meng X, Zhang Y. Natural polyphenols: a potential prevention and treatment strategy for metabolic syndrome. Food Funct. 2022;13:9734-9753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 49] [Reference Citation Analysis (0)] |
| 143. | Gómez-Jiménez V, Burggraaf-Sánchez de Las Matas R, Ortega ÁL. Modulation of Oxidative Stress in Diabetic Retinopathy: Therapeutic Role of Natural Polyphenols. Antioxidants (Basel). 2025;14:875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 144. | Liu S, Fang Y, Yu J, Chang X. Hawthorn polyphenols reduce high glucose-induced inflammation and apoptosis in ARPE-19 cells by regulating miR-34a/SIRT1 to reduce acetylation. J Food Biochem. 2021;45:e13623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 145. | Chen J, Ren Z, Sun X, Wang L, Liu D, Chen Y, Cao X. Baicalein Inhibited Amino-modified Polystyrene Nanoplastics Induced Human Umbilical Vein Endothelial Cells Pyroptosis by Reducing the Expression of NLRP3/Caspase-1/Gasdermin D Pathway-related Proteins. Chem Biodivers. 2025;22:e202500085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 146. | Magne Nde CB, Zingue S, Winter E, Creczynski-Pasa TB, Michel T, Fernandez X, Njamen D, Clyne C. Flavonoids, Breast Cancer Chemopreventive and/or Chemotherapeutic Agents. Curr Med Chem. 2015;22:3434-3446. [PubMed] |
| 147. | Wang SM, Xia Y. Baicalein modulates endoplasmic reticulum stress by activating SIRT3 to attenuate the dysfunction of retinal microvascular endothelial cells under high glucose conditions. Exp Eye Res. 2025;254:110250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 148. | Rauf A, Patel S, Imran M, Maalik A, Arshad MU, Saeed F, Mabkhot YN, Al-Showiman SS, Ahmad N, Elsharkawy E. Honokiol: An anticancer lignan. Biomed Pharmacother. 2018;107:555-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (4)] |
| 149. | Shi J, Liu M, Zhao J, Tan Y, Jiang C. Honokiol protects against diabetic retinal microvascular injury via sirtuin 3-mediated mitochondrial fusion. Front Pharmacol. 2024;15:1485831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 150. | Zhou H, Zhao J, Li A, Reetz MT. Chemical and Biocatalytic Routes to Arbutin (†). Molecules. 2019;24:3303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 151. | Abduh MS, Alzoghaibi MA, Alzoghaibi AM, Bin-Ammar A, Alotaibi MF, Kamel EM, Mahmoud AM. Arbutin ameliorates hyperglycemia, dyslipidemia and oxidative stress and modulates adipocytokines and PPARγ in high-fat diet/streptozotocin-induced diabetic rats. Life Sci. 2023;321:121612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 152. | Ma C, Zhang D, Ma Q, Liu Y, Yang Y. Arbutin inhibits inflammation and apoptosis by enhancing autophagy via SIRT1. Adv Clin Exp Med. 2021;30:535-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 153. | Xiao G, Tang R, Yang N, Chen Y. Review on pharmacological effects of gastrodin. Arch Pharm Res. 2023;46:744-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 79] [Article Influence: 26.3] [Reference Citation Analysis (2)] |
| 154. | Zhang TH, Huang CM, Gao X, Wang JW, Hao LL, Ji Q. Gastrodin inhibits high glucoseinduced human retinal endothelial cell apoptosis by regulating the SIRT1/TLR4/NFκBp65 signaling pathway. Mol Med Rep. 2018;17:7774-7780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 155. | Liu S, Wang L, Zhang Z, Leng Y, Yang Y, Fu X, Xie H, Gao H, Xie C. The potential of astragalus polysaccharide for treating diabetes and its action mechanism. Front Pharmacol. 2024;15:1339406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (1)] |
| 156. | Peng QH, Tong P, Gu LM, Li WJ. Astragalus polysaccharide attenuates metabolic memory-triggered ER stress and apoptosis via regulation of miR-204/SIRT1 axis in retinal pigment epithelial cells. Biosci Rep. 2020;40:BSR20192121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 157. | Gao LM, Fu S, Liu F, Wu HB, Li WJ. Astragalus Polysaccharide Regulates miR-182/Bcl-2 Axis to Relieve Metabolic Memory through Suppressing Mitochondrial Damage-Mediated Apoptosis in Retinal Pigment Epithelial Cells. Pharmacology. 2021;106:520-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 158. | Fu Y, Yang L, Liu L, Kong L, Sun H, Sun Y, Yin F, Yan G, Wang X. Rhein: An Updated Review Concerning Its Biological Activity, Pharmacokinetics, Structure Optimization, and Future Pharmaceutical Applications. Pharmaceuticals (Basel). 2024;17:1665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 159. | Liu C, Cao Q, Chen Y, Chen X, Zhu Y, Zhang Z, Wei W. Rhein protects retinal Müller cells from high glucose-induced injury via activating the AMPK/Sirt1/PGC-1α pathway. J Recept Signal Transduct Res. 2023;43:62-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 160. | Gimblet CJ, Kruse NT, Geasland K, Michelson J, Sun M, Mandukhail SR, Wendt LH, Eyck PT, Pierce GL, Jalal DI. Effect of Resveratrol on Endothelial Function in Patients with CKD and Diabetes: A Randomized Controlled Trial. Clin J Am Soc Nephrol. 2024;19:161-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 161. | García-Martínez BI, Ruiz-Ramos M, Pedraza-Chaverri J, Santiago-Osorio E, Mendoza-Núñez VM. Effect of Resveratrol on Markers of Oxidative Stress and Sirtuin 1 in Elderly Adults with Type 2 Diabetes. Int J Mol Sci. 2023;24:7422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 49] [Reference Citation Analysis (0)] |
| 162. | Bai YX, Wu HL, Xie WL, Li X, Han JJ, Liu J, Chen SQ, Yin P, Dong NG, Wu QP. Efficacy and safety of gastrodin in preventing postoperative delirium following cardiac surgery: a randomized placebo controlled clinical trial. Crit Care. 2025;29:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 163. | Li L, Gao H, Lou K, Luo H, Hao S, Yuan J, Liu Z, Dong R. Safety, tolerability, and pharmacokinetics of oral baicalein tablets in healthy Chinese subjects: A single-center, randomized, double-blind, placebo-controlled multiple-ascending-dose study. Clin Transl Sci. 2021;14:2017-2024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 164. | Di Nardo G, Bernardo L, Cremon C, Barbara G, Felici E, Evangelisti M, Ferretti A, Furio S, Piccirillo M, Coluzzi F, Parisi P, Mauro A, Di Mari C, D'Angelo F, Mennini M. Palmitoylethanolamide and polydatin in pediatric irritable bowel syndrome: A multicentric randomized controlled trial. Nutrition. 2024;122:112397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 165. | Zeng W, Chu TTW, Fok BSP, Ho WKK, Chan JCN, Tomlinson B. Effects of Hawthorn Fruit Extract Drink in Chinese Patients With Mild Hypertension and/or Hyperlipidaemia: A Randomized, Double-Blind, Placebo-Controlled, Crossover Study. Dose Response. 2024;22:15593258241303136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 166. | Gabhane M, Patil R, Dharmadhikari S, Shah P, Khandhedia C, Mehta S. Efficacy and Safety of a Topical Formulation Containing Trihydroxybenzoic Acid Glucoside and α-Arbutin, Applied Along With a Sunscreen: A Noncomparative, Prospective, Interventional Study in Indian Females With Facial Melasma or Dark Spots. J Cosmet Dermatol. 2025;24:e70017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 167. | Xu X, Lu Q, Wu J, Li Y, Sun J. Impact of extended ginsenoside Rb1 on early chronic kidney disease: a randomized, placebo-controlled study. Inflammopharmacology. 2017;25:33-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 168. | Zhang G, Xia F, Zhang Y, Zhang X, Cao Y, Wang L, Liu X, Zhao G, Shi M. Ginsenoside Rd Is Efficacious Against Acute Ischemic Stroke by Suppressing Microglial Proteasome-Mediated Inflammation. Mol Neurobiol. 2016;53:2529-2540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 169. | Liu X, Wang L, Wen A, Yang J, Yan Y, Song Y, Liu X, Ren H, Wu Y, Li Z, Chen W, Xu Y, Li L, Xia J, Zhao G. Ginsenoside-Rd improves outcome of acute ischaemic stroke - a randomized, double-blind, placebo-controlled, multicenter trial. Eur J Neurol. 2012;19:855-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 170. | Keating C. Artesunate versus quinine: the controlled trials watershed. Lancet. 2023;401:1329-1331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 171. | Shen WC, Chen SC, Wang CH, Hung CM, Peng MT, Liu CT, Chang YS, Kuo WL, Chou HH, Yeh KY, Wu TH, Wu CF, Chang PH, Huang YM, Yu CC, Lee CH, Rau KM. Astragalus polysaccharides improve adjuvant chemotherapy-induced fatigue for patients with early breast cancer. Sci Rep. 2024;14:25690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 172. | Fontana G, Innamorati G, Giacomello L. Nanoparticle-Based Oral Insulin Delivery: Challenges, Advances, and Future Directions. Pharmaceutics. 2025;17:1563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 173. | Walle T, Hsieh F, DeLegge MH, Oatis JE Jr, Walle UK. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab Dispos. 2004;32:1377-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1589] [Cited by in RCA: 1371] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 174. | Shahid A, Bhatt P, Miller A, Sutariya V. Honokiol-Loaded Methoxy Poly (Ethylene Glycol) Polycaprolactone Micelles for the Treatment of Age-Related Macular Degeneration. Assay Drug Dev Technol. 2021;19:350-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 175. | Valdés-Sánchez L, Moshtaghion SM, Caballano-Infantes E, Peñalver P, Rodríguez-Ruiz R, González-Alfonso JL, Plou FJ, Desmet T, Morales JC, Díaz-Corrales FJ. Synthesis and Evaluation of Glucosyl-, Acyl- and Silyl- Resveratrol Derivatives as Retinoprotective Agents: Piceid Octanoate Notably Delays Photoreceptor Degeneration in a Retinitis Pigmentosa Mouse Model. Pharmaceuticals (Basel). 2024;17:1482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 176. | Bai X, Yao L, Ma X, Xu X. Small Molecules as SIRT Modulators. Mini Rev Med Chem. 2018;18:1151-1157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 177. | Pacholec M, Bleasdale JE, Chrunyk B, Cunningham D, Flynn D, Garofalo RS, Griffith D, Griffor M, Loulakis P, Pabst B, Qiu X, Stockman B, Thanabal V, Varghese A, Ward J, Withka J, Ahn K. SRT1720, SRT2183, SRT1460, and resveratrol are not direct activators of SIRT1. J Biol Chem. 2010;285:8340-8351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 780] [Cited by in RCA: 713] [Article Influence: 44.6] [Reference Citation Analysis (4)] |
| 178. | Hammer SS, Vieira CP, McFarland D, Sandler M, Levitsky Y, Dorweiler TF, Lydic TA, Asare-Bediako B, Adu-Agyeiwaah Y, Sielski MS, Dupont M, Longhini AL, Li Calzi S, Chakraborty D, Seigel GM, Proshlyakov DA, Grant MB, Busik JV. Fasting and fasting-mimicking treatment activate SIRT1/LXRα and alleviate diabetes-induced systemic and microvascular dysfunction. Diabetologia. 2021;64:1674-1689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (1)] |
| 179. | Zeng Y, Cui Z, Liu J, Chen J, Tang S. MicroRNA-29b-3p Promotes Human Retinal Microvascular Endothelial Cell Apoptosis via Blocking SIRT1 in Diabetic Retinopathy. Front Physiol. 2019;10:1621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 180. | Liu J, Zhang Y, Tang J. Exendin-4 alleviates diabetic retinopathy by activating autophagy via regulation of the adenosine monophosphate-activated protein kinase/sirtuin 1 pathway. J Mol Histol. 2025;56:210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 181. | Kaya SG, Eren G. Selective inhibition of SIRT2: A disputable therapeutic approach in cancer therapy. Bioorg Chem. 2024;143:107038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 182. | van der Meer AJ, Scicluna BP, Moerland PD, Lin J, Jacobson EW, Vlasuk GP, van der Poll T. The Selective Sirtuin 1 Activator SRT2104 Reduces Endotoxin-Induced Cytokine Release and Coagulation Activation in Humans. Crit Care Med. 2015;43:e199-e202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 183. | Kamal S, Babar S, Ali W, Rehman K, Hussain A, Akash MSH. Sirtuin insights: bridging the gap between cellular processes and therapeutic applications. Naunyn Schmiedebergs Arch Pharmacol. 2024;397:9315-9344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |