Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.114679
Revised: November 1, 2025
Accepted: February 5, 2026
Published online: April 15, 2026
Processing time: 200 Days and 18.2 Hours
Diabetic nephropathy (DN) is a major cause of end-stage renal disease. Despite their ability to delay the progression of DN, current therapies do not halt renal injury. Ferroptosis, a regulated form of cell death characterized by iron-dependent lipid peroxidation, contributes to DN pathogenesis.
To test the hypothesis that hyperoside—a natural flavonoid—protects against DN by activating the Nrf2/SLC7A11/GPX4 antioxidant signaling pathway, thereby reducing ferroptosis and oxidative stress-related renal injury.
High-glucose-treated HK-2 cells and streptozotocin-induced DN rats were used to evaluate the effects of hyperoside. Cell viability, apoptosis, oxidative stress, and ferroptosis markers were measured using Cell Counting Kit-8, terminal deo
Hyperoside significantly improved HK-2 cell viability under high-glucose con
Hyperoside alleviates diabetic renal injury by activating the Nrf2/SLC7A11/GPX4 pathway and suppressing ferroptosis-driven oxidative damage. These findings provide mechanistic and experimental evidence supporting hyperoside as a potential therapeutic candidate for DN.
Core Tip: This study, for the first time, demonstrates that hyperoside, a natural flavonoid, protects against diabetic nephropathy by suppressing ferroptosis via activation of the Nrf2/SLC7A11/GPX4 antioxidant axis. In high-glucose-challenged HK-2 cells, hyperoside restored glutathione, reduced lipid reactive oxygen species and Fe2+ accumulation, and improved cell viability, effects comparable to ferrostatin-1. In streptozotocin-induced diabetic nephropathy rats, hyperoside ameliorated renal dysfunction and histologic injury while upregulating Nrf2/SLC7A11/GPX4. These findings provide mechanistic and experimental support for hyperoside as a potential therapeutic candidate for diabetic nephropathy.
- Citation: Liu C, Li Y, Zhang Y, Gao M, Yang SF. Hyperoside attenuates diabetic nephropathy by activating the Nrf2/SLC7A11/GPX4 axis to restrain ferroptosis. World J Diabetes 2026; 17(4): 114679
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/114679.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.114679
Diabetic nephropathy (DN) is one of the most common and severe chronic complications of diabetes mellitus and remains a leading cause of end-stage renal disease[1,2]. With the global prevalence of diabetes continuing to rise, the incidence, disability, and mortality related to DN are steadily increasing, imposing a substantial health and so
Ferroptosis, an iron-dependent and lipid peroxidation-driven form of regulated cell death, has emerged as a key contributor to kidney diseases[6,7]. Increasing evidence indicates that hyperglycemia promotes excessive reactive oxygen species (ROS) generation, disrupts iron metabolism, and induces lipid peroxidation, thereby triggering ferroptosis in renal tubular epithelial cells and accelerating DN progression[8]. Targeting ferroptosis has thus become a promising strategy for DN therapy. Accumulating evidence further positions ferroptosis as a central driver of DN pathogenesis, offering a tractable therapeutic entry point that integrates aberrant iron handling, lipid peroxidation, and ROS dysregulation[9,10].
Among multiple ferroptosis-regulatory pathways, the Nrf2/SLC7A11/GPX4 axis plays a central role in maintaining redox homeostasis and preventing ferroptosis-related injury[11]. Nrf2 is a master antioxidant transcription factor that induces the expression of genes involved in glutathione (GSH) synthesis and detoxification[12]. SLC7A11 mediates cystine import to support GSH biosynthesis, while GPX4 detoxifies lipid peroxides to suppress ferroptotic cell death[13]. Impairment of this axis has been implicated in oxidative stress and renal tubular injury in DN, highlighting it as a potential therapeutic target[14]. In parallel, recent syntheses have highlighted ferroptosis-oriented interventions as promising strategies for DN, further underscoring the clinical relevance of the Nrf2/SLC7A11/GPX4 axis[15].
Hyperoside, a naturally occurring flavonoid, possesses antioxidant, anti-inflammatory, antifibrotic, and anti-apoptotic activities[16,17], and has shown protective effects in cardiovascular, neural, hepatic, and renal diseases. Recent studies also suggest that hyperoside can modulate oxidative stress-related signaling pathways, including Nrf2 activation, yet whether hyperoside can inhibit ferroptosis—particularly through the Nrf2/SLC7A11/GPX4 pathway in the context of DN—remains unknown. To date, no studies have systematically evaluated the role of hyperoside in ferroptosis-mediated renal injury in DN.
Taken together, ferroptosis represents a critical driver of DN, and modulation of the Nrf2/SLC7A11/GPX4 axis may offer a promising therapeutic approach. The existing gap lies in whether hyperoside can regulate this ferroptotic pathway to alleviate DN. Therefore, the present study aimed to elucidate the protective effects and mechanistic basis of hyperoside in DN, focusing on ferroptosis regulation via the Nrf2/SLC7A11/GPX4 antioxidant axis. We hypothesized that hyperoside ameliorates diabetic renal injury by activating this pathway, thereby suppressing ferroptosis and oxidative stress-induced damage.
HK-2 cells were obtained from the Chinese Academy of Sciences Cell Bank (Shanghai, China) and cultured in Dulbecco’s Modified Eagle Medium/F12 (Gibco, United States) supplemented with 10% fetal bovine serum (Gibco, United States), 100 U/mL penicillin, and 100 μg/mL streptomycin in a humidified incubator at 37 °C with 5% CO2. Cells were sub
Male Sprague-Dawley rats (200-220 g) were obtained from the Experimental Animal Center of Xi’an Jiaotong University (license No. SCXK-2022-0012). All procedures conformed to the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University, approval No. XJTUAE2025-3549. Rats were housed under specific pathogen-free conditions (22 °C ± 2 °C, 55% ± 10% relative humidity, 12 hours light/dark cycle) with free access to standard chow and water. To induce DN, rats were fed a high-fat diet (60% of calories from fat) for 4 weeks to establish insulin resistance, followed by a single intraperitoneal injection of low-dose streptozotocin (STZ; 35 mg/kg, dissolved in 0.1 mol/L citrate buffer, pH 4.5). This high-fat diet combined with low-dose STZ protocol is a well-established method to induce type 2 diabetic nephropathy (T2DN) in rodents, which recapitulates key metabolic features of human T2DN, including insulin resistance, dyslipidemia, moderate hyperglycemia, and progressive renal injury. Compared with high-dose STZ-induced type 1 diabetes models, this approach better reflects the pathophysiological characteristics of T2DN in clinical settings. Fasting blood glucose levels were measured 72 hours after STZ injection, and values ≥ 16.7 mmol/L were considered indicative of successful model establishment. Rats in the control group received vehicle injection and standard chow. Successfully modeled diabetic rats were randomly divided using a random-number table (allocation ratio 1:1:1:1:1) into four DN-related groups, in addition to a non-diabetic control group, with n = 6 per group: Control, DN model, DN + hyperoside 25 mg/kg, DN + hyperoside 50 mg/kg, and DN + ferrostatin-1 (Fer-1) 1 mg/kg. Hyperoside (25 mg/kg or 50 mg/kg) and Fer-1 (1 mg/kg) were administered intraperitoneally once daily for 8 consecutive weeks after confirmation of diabetes. During the treatment period, body weight and general health were monitored regularly. Humane endpoints were implemented in accordance with institutional guidelines. Prespecified exclusion criteria included severe illness unrelated to DN, injection failure, or sample hemolysis; all exclusions and their reasons were prospectively recorded. Investigators performing biochemical, histological, and immunoblotting assessments were blinded to group allocation.
Hyperoside (purity ≥ 98%) was purchased from China National Pharmaceutical Group Chemical Reagent Co., Ltd. Fer-1 and erastin were obtained from Sigma-Aldrich (United States). For in vitro experiments, hyperoside was used at 1-100 μmol/L, a range selected based on previous pharmacological studies demonstrating effective antioxidant and cytoprotective properties without evident cytotoxicity. For in vivo experiments, hyperoside doses of 25 mg/kg and 50 mg/kg were chosen according to published reports of renal protection and preliminary toxicity tests indicating good tolerability at these levels. Fer-1 (1 μmol/L in vitro and 1 mg/kg in vivo) and erastin (5 μmol/L) were used as a classical ferroptosis inhibitor and inducer, respectively, at doses commonly adopted to modulate ferroptosis in kidney injury models. Commercial kits were used to determine cell viability [Cell Counting Kit-8 (CCK-8); Dojindo, Japan], apoptosis [terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL); Roche, Switzerland], and ROS levels [2’,7’-dichlorodihydrofluorescein diacetate (DCFH-DA); Beyotime, China]. Enzyme-linked immunosorbent assay (ELISA) kits for lactate dehydrogenase (LDH), malondialdehyde (MDA), Fe2+, GSH, total cholesterol (TC), triglycerides (TG), serum creatinine (Scr), blood urea nitrogen (BUN), urinary albumin excretion (UAE), superoxide dismutase (SOD), and catalase (CAT) were purchased from Nanjing Jiancheng Bioengineering Institute (China). Quantitative reverse transcription-polymerase chain reaction (PCR) (qRT-PCR) reagents were obtained from Takara (Japan), and protein extraction reagents, bi
The main instruments used in this study included a flow cytometer (BD FACSCalibur, United States), a fluorescence microscope (Leica DMi8, Germany), a microplate reader (Bio-Rad, United States), a real-time PCR system (ABI StepOnePlus, United States), electrophoresis and protein transfer apparatus (Bio-Rad, United States), a biochemical analyzer (Hitachi 7020, Japan), and ImageJ software (NIH, United States) for image acquisition and densitometric analysis.
HK-2 cells were divided into the following experimental groups: NG (5.5 mmol/L glucose), HG (30 mmol/L glucose), HG combined with hyperoside at different concentrations (1 μmol/L, 10 μmol/L, 50 μmol/L, and 100 μmol/L), HG combined with Fer-1 (1 μmol/L), and HG combined with erastin (5 μmol/L). In general, cells were pretreated with hyperoside or Fer-1 for 2 hours, followed by HG exposure for 24 hours, or co-treated with HG and drugs for the indicated periods, in line with commonly used protocols in studies of ferroptosis and oxidative injury in renal tubular cells. For mechanistic experiments, cells were transfected with negative-control siRNA or Nrf2-specific siRNA (si-Nrf2) using Lipofectamine 2000 at a final siRNA concentration of 50 nM. After 6 hours, the transfection medium was replaced with fresh complete medium, and subsequent treatments with HG and hyperoside were initiated 24 hours post-transfection. Additional groups included si-Nrf2 combined with hyperoside (50 μmol/L) under HG conditions to verify the involvement of Nrf2 in hyperoside-mediated protection.
Rat body weights were recorded weekly throughout the experiment. At the end of the 8-week treatment period, rats were fasted overnight, anesthetized with isoflurane, and sacrificed by exsanguination. Both kidneys were promptly removed, decapsulated, and weighed. The kidney weight index (KWI) was calculated according to the following formula: KWI = [kidney wet weight (g)/body weight (g)] × 1000, which reflects renal hypertrophy relative to body size in DN.
HK-2 cells were seeded at a density of 5 × 103 cells per well in 96-well plates, with five replicate wells per group. After the indicated treatments, 10 μL of CCK-8 reagent was added to each well and incubated for 1 hour at 37 °C. Absorbance at 450 nm was measured using a microplate reader, and the cell survival rate was calculated as (OD_experimental/OD_control) × 100%. For time-course analyses, CCK-8 measurements were performed at multiple time points (e.g., 0-48 hours) to assess dynamic changes in cell viability in response to HG, erastin, and hyperoside.
Apoptosis in HK-2 cells was assessed by TUNEL staining and Annexin V-fluorescein isothiocyanate/propidium iodide flow cytometry. For TUNEL staining, cells were fixed with 4% paraformaldehyde for 30 minutes at room temperature, permeabilized with 0.1% Triton X-100 for 10 minutes, and incubated with TUNEL reaction mixture according to the manufacturer’s instructions. Nuclei were counterstained with 4’,6-diamidino-2-phenylindole, and five random mi
Intracellular ROS levels were detected using the DCFH-DA fluorescent probe (Beyotime, China) at a final concentration of 10 μM. After treatment, cells were incubated with DCFH-DA for 30 minutes at 37 °C, washed three times with PBS to remove excess dye, and analyzed by flow cytometry (excitation 488 nm, emission 525 nm). The mean fluorescence in
Cells and kidney tissues were homogenized in ice-cold PBS and centrifuged at 12000 rpm for 10 minutes at 4 °C to obtain supernatants for biochemical analysis. LDH activity was determined from cell-culture supernatants, whereas MDA, Fe2+, and GSH levels were measured from cell and tissue lysates using ELISA kits (Nanjing Jiancheng Bioengineering Institute, China). These indicators were selected because MDA and lipid ROS represent lipid peroxidation, Fe2+ reflects iron overload, and GSH depletion is a hallmark of ferroptotic susceptibility, while LDH release is associated with membrane damage during cell death. Data were normalized to total protein concentration determined by the BCA method and expressed as U/mg or μmol/mg protein. For animal samples, additional assays for SOD and CAT were conducted to assess antioxidant enzyme activities and overall oxidative status. All measurements were performed strictly according to the manufacturers’ instructions, and each assay was repeated at least three times to ensure consistency.
At the end of the 8-week treatment period, rats were placed in metabolic cages for 24-hour urine collection to determine UAE. After anesthesia, blood samples were collected from the heart and centrifuged at 3000 rpm for 15 minutes to obtain serum. Scr, BUN, TC, and TG levels were measured using an automatic biochemical analyzer (Hitachi 7020; Japan). Oxidative stress parameters, including MDA, GSH, SOD, CAT, and Fe2+, were determined in kidney tissue homogenates normalized to total protein content. These indicators comprehensively reflect renal function, metabolic status, and oxidative injury in DN.
After euthanasia, one kidney from each rat was fixed in 10% neutral-buffered formalin for 24 hours. The fixed tissues were then processed using standard histological procedures: Dehydration through graded ethanol, clearing in xylene, embedding in paraffin, and sectioning at 3-5 μm thickness. Paraffin sections were stained with hematoxylin and eosin for general morphology, Masson’s trichrome for collagen deposition, and periodic acid-Schiff for basement membrane thickening and mesangial expansion. All slides were evaluated under a Leica DMi8 microscope by two independent observers blinded to group allocation. Lesions of the glomeruli, tubules, and interstitium were semi-quantitatively scored on a scale of 0-4 based on the extent of structural damage and fibrosis.
Total RNA was extracted from HK-2 cells and kidney tissues using TRIzol reagent (Invitrogen, United States) according to the manufacturer’s instructions. RNA purity and concentration were assessed by measuring the OD260/OD280 ratio, and samples with ratios between 1.8 and 2.0 were used for subsequent experiments. One microgram of total RNA was reverse transcribed into cDNA using a commercial reverse transcription kit (Takara, Japan). qRT-PCR was performed using SYBR Green Master Mix on an ABI StepOnePlus real-time PCR system. The amplification protocol consisted of an initial denaturation at 95 °C for 30 seconds, followed by 40 cycles of 95 °C for 5 seconds and 60 °C for 30 seconds. β-Actin was used as the internal reference gene, and relative gene expression was calculated using the 2-ΔΔCt method. Primer sequences for Nrf2, SLC7A11, GPX4, and β-actin are listed in Supplementary Table 1.
Total protein from cells or kidney tissues was extracted using radioimmunoprecipitation assay lysis buffer containing protease inhibitors and quantified by the BCA method. Equal amounts of protein (30 μg per lane) were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes (0.22 μm pore size). Membranes were blocked with 5% skim milk in Tris-buffered saline with 0.1% Tween-20 (TBST) for 1 hour at room temperature and then incubated overnight at 4 °C with primary antibodies against Nrf2 (CST #12721), SLC7A11 (Abcam #37185), GPX4 (Abcam #125066), and β-actin (CST #3700), all at 1:1000 dilution. After washing with Tris-buffered saline with 0.1% Tween-20, membranes were incubated with horseradish peroxidase-conjugated secondary antibody (CST #7074, 1:5000) for 1 hour at room temperature. Protein bands were visualized using enhanced chemiluminescence reagents and imaged, and band intensities were quantified using ImageJ software. Target protein levels were normalized to β-actin.
To assess Nrf2 subcellular localization and nuclear translocation, immunofluorescence staining was performed. HK-2 cells were seeded on glass coverslips in 24-well plates at a density of 2 × 104 cells per well. After the indicated treatments, cells were washed three times with PBS and fixed with 4% paraformaldehyde for 15 minutes at room temperature. Following fixation, cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes and blocked with 5% bovine serum albumin in PBS for 1 hour at room temperature. Cells were then incubated with primary antibody against Nrf2 (1:200, CST #12721) overnight at 4 °C. After washing three times with PBS, cells were incubated with Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody (1:500) for 1 hour at room temperature in the dark. Nuclei were counterstained with 4’,6-diamidino-2-phenylindole for 5 minutes. Coverslips were mounted with antifade mounting medium and images were captured using a fluorescence microscope (Leica DMi8, Germany).
For quantitative analysis of Nrf2 nuclear translocation, the nuclear and cytoplasmic fluorescence intensities were measured using ImageJ software (NIH, United States). The nuclear region was defined by 4’,6-diamidino-2-phenylindole staining, and the cytoplasmic region was defined as the area between the nuclear boundary and the cell membrane. The nuclear/cytoplasmic Nrf2 ratio was calculated by dividing the mean nuclear fluorescence intensity by the mean cyto
All experiments were performed with at least three independent replicates for in vitro studies and six animals per group for in vivo studies. Data are expressed as the mean ± SD. Statistical analyses were performed using SPSS version 22.0. Differences between two groups were evaluated using the independent-samples t-test, whereas multiple-group comparisons were analyzed using one-way analysis of variance followed by Tukey’s post hoc test. A P value < 0.05 was considered statistically significant.
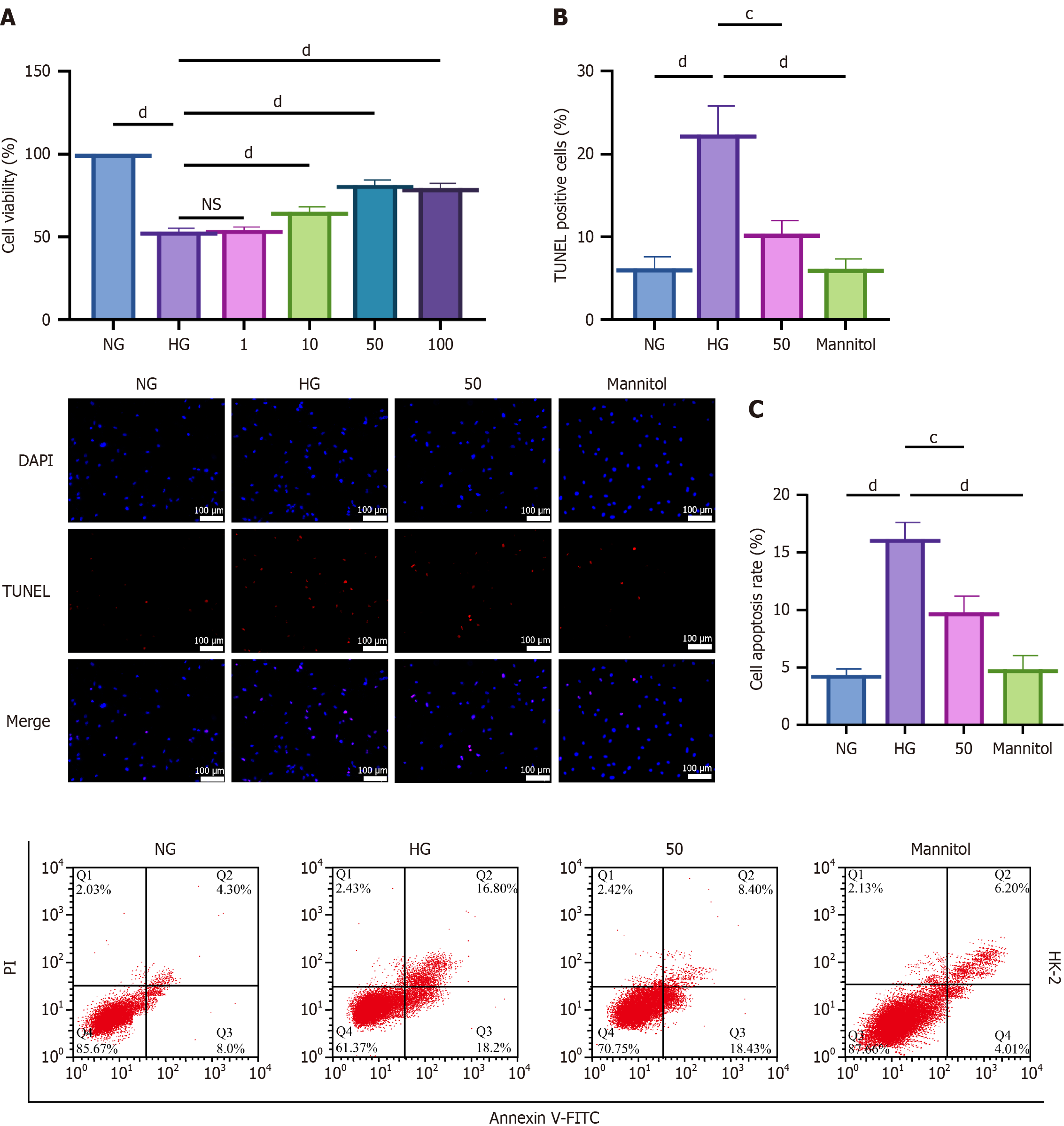
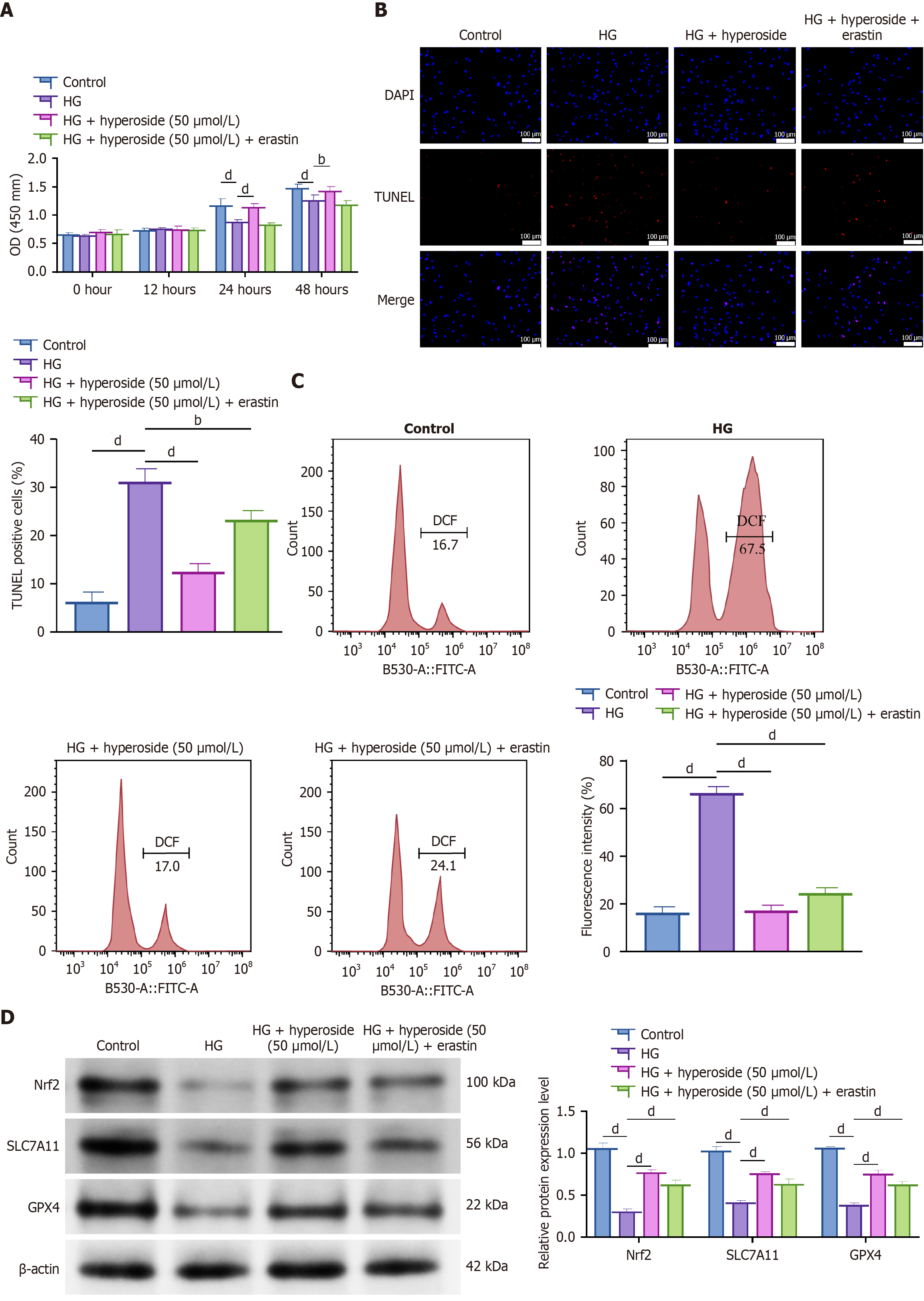
To investigate the protective effects of hyperoside on HK-2 cells under HG conditions, we used CCK-8, TUNEL staining, and flow cytometry. Mannitol was used as an osmotic control to exclude the potential contribution of hyperosmolarity. The results demonstrated that hyperoside significantly ameliorated the HG-induced decline in HK-2 cell viability and apoptosis. CCK-8 detection results indicated that, compared to the NG group, HG significantly reduced cell viability, whereas intervention with different concentrations of hyperoside showed dose-dependent improvement in cell viability, particularly at 50 μmol/L and 100 μmol/L, where cellular proliferation capacity markedly recovered. Notably, mannitol treatment did not significantly affect cell viability compared with the NG group (P > 0.05), indicating that the HG-induced cytotoxicity was attributable to glucose metabolism rather than osmotic stress (Figure 1A). TUNEL detection results revealed that HG significantly increased HK-2 cell apoptosis rates, while 50 μmol/L hyperoside treatment significantly reduced TUNEL-positive cell numbers, suggesting protective effects against apoptosis. In contrast, the mannitol group showed comparable TUNEL-positive rates to the NG group, further confirming that HG-induced apoptosis was not due to hyperosmolarity (Figure 1B). Further flow cytometry analysis of apoptosis rates showed results consistent with TUNEL detection: HG groups showed significantly increased apoptotic cells, while 50 μmol/L hy
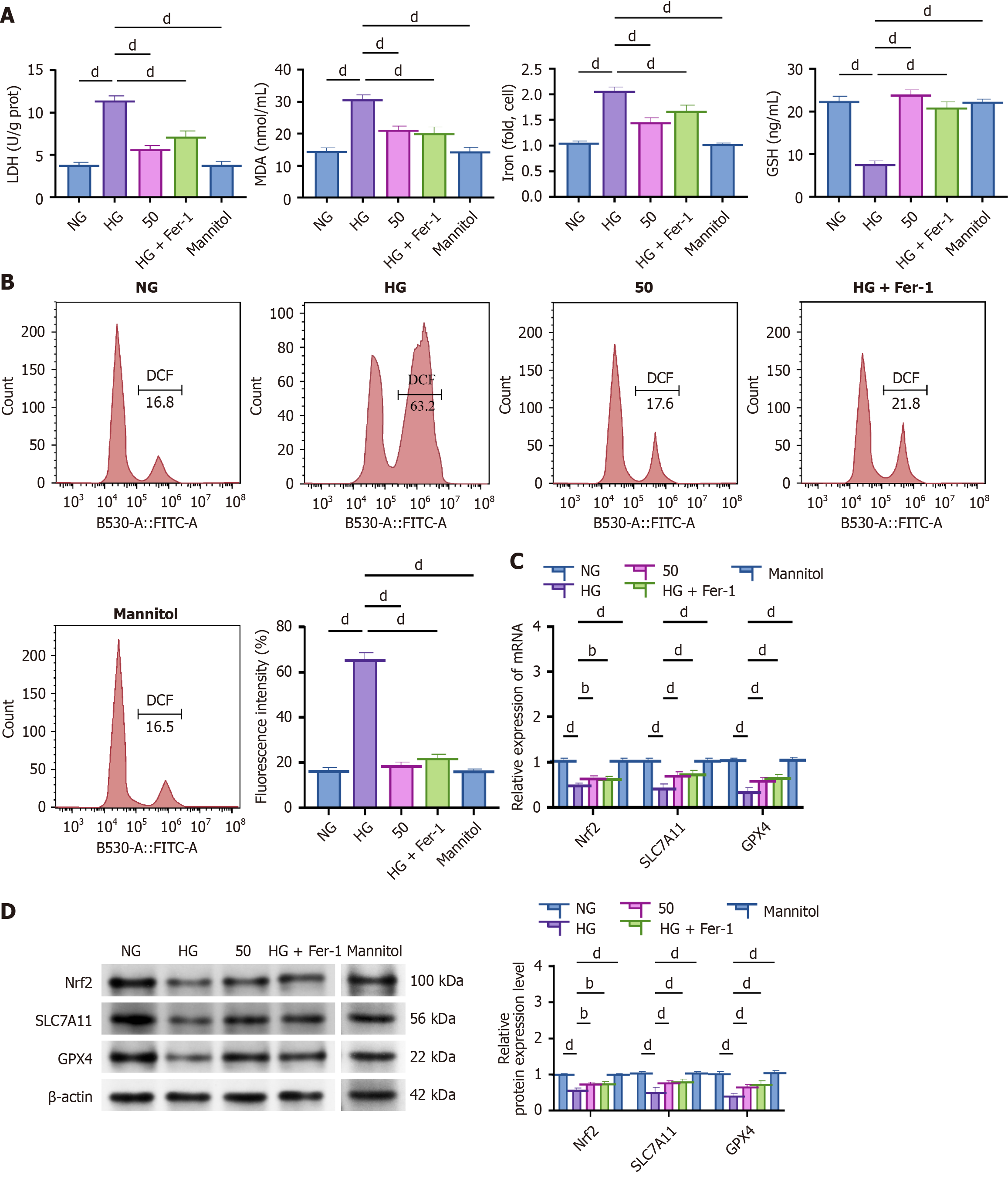
To explore the effects of hyperoside on HG-induced oxidative stress damage in HK-2 cells, we examined the cellular oxidative stress markers, lipid peroxidation levels, and changes in the Nrf2/SLC7A11/GPX4 signaling axis. ELISA results showed that, compared to the NG group, the HG group demonstrated significantly elevated levels of LDH, MDA, and iron, while GSH levels were significantly decreased. Hyperoside (50 μmol/L) intervention markedly reversed these changes, with effects similar to those of the ferroptosis inhibitor Fer-1. Importantly, the mannitol group showed no significant differences in LDH, MDA, Fe²+, or GSH levels compared with the NG group (all P > 0.05), confirming that these oxidative stress markers were altered by glucose metabolism rather than hyperosmotic conditions (Figure 2A, P < 0.05). The flow cytometry results indicated that HG significantly increased intracellular lipid ROS levels, whereas both hyperoside and Fer-1 significantly reduced lipid ROS accumulation. Mannitol treatment did not induce lipid ROS accumulation, with levels remaining comparable to the NG group (Figure 2B, P < 0.05). At the gene level, qRT-PCR results showed that HG treatment significantly downregulated Nrf2, SLC7A11, and GPX4 expression, whereas both hyperoside and Fer-1 treatments restored their transcription levels. The mannitol group maintained mRNA expression levels similar to those of the NG group (Figure 2C, P < 0.05). This trend was verified using western blotting, with hy
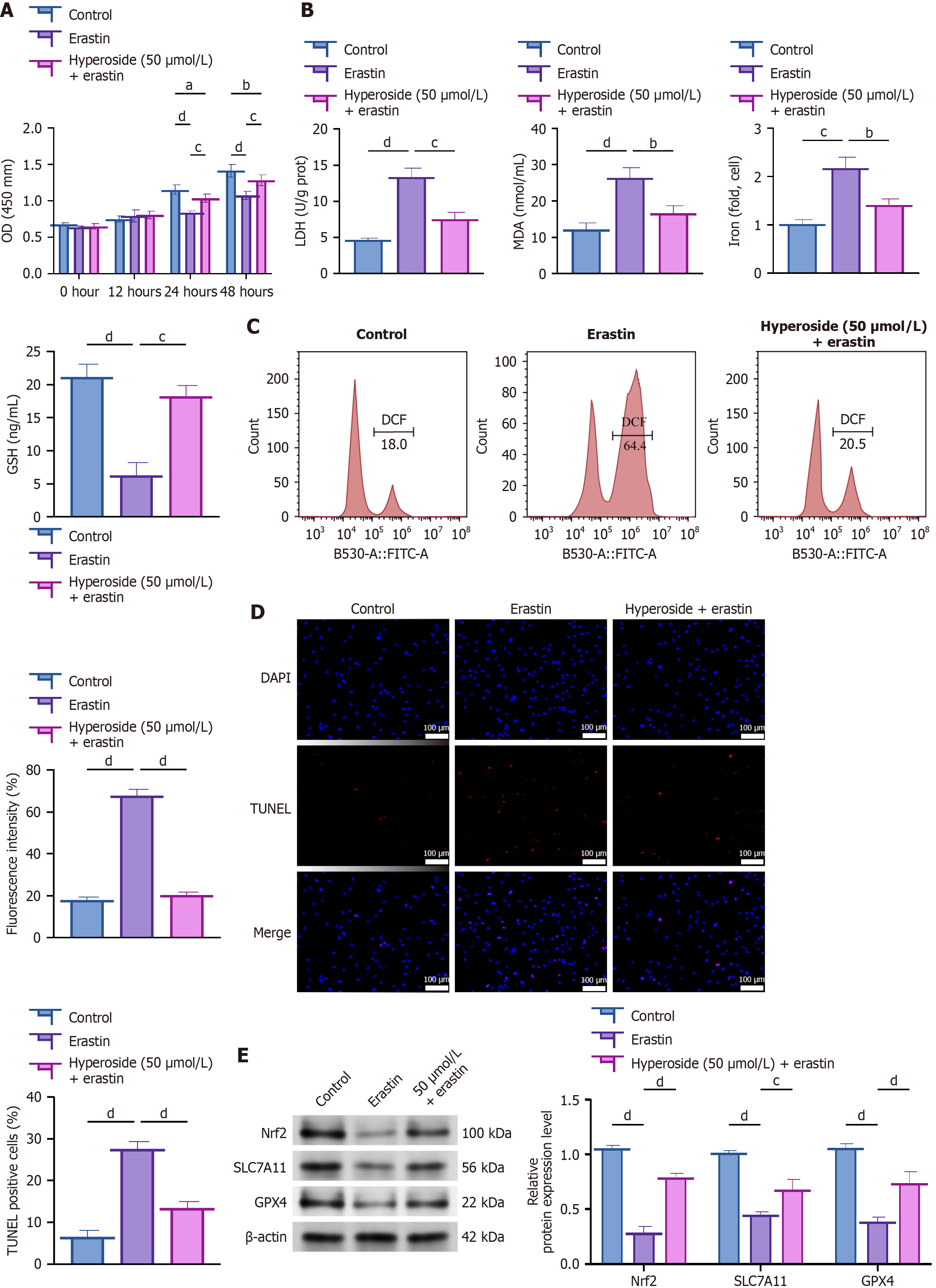
To further verify hyperoside’s mechanism of action in cellular damage, we used the ferroptosis inducer erastin to assess whether hyperoside (50 μmol/L) could ameliorate ferroptosis-related damage. CCK-8 detection results showed that with time extension, erastin significantly inhibited HK-2 cell proliferation, while hyperoside (50 μmol/L) treatment partially restored cellular proliferation capacity (Figure 3A; P < 0.05). ELISA results indicated that erastin-treated cells showed markedly elevated LDH, MDA, and iron levels and significantly decreased GSH levels, whereas hyperoside treatment significantly improved these indicators (Figure 3B; P < 0.05). Flow cytometry revealed that erastin induction significantly increased cellular lipid ROS levels, whereas hyperoside treatment significantly reduced ROS accumulation (Figure 3C; P < 0.05). The TUNEL staining results further confirmed that the erastin treatment group showed significantly elevated cell apoptosis rates, while hyperoside effectively reduced the number of TUNEL-positive cells (Figure 3D; P < 0.05).
Western blotting also showed that erastin significantly downregulated Nrf2, SLC7A11, and GPX4 protein expression, whereas hyperoside treatment partially reversed these changes (Figure 3E; P < 0.05). These results demonstrate that hyperoside exerts protective effects by inhibiting ferroptosis and modulating the Nrf2/SLC7A11/GPX4 signaling axis.
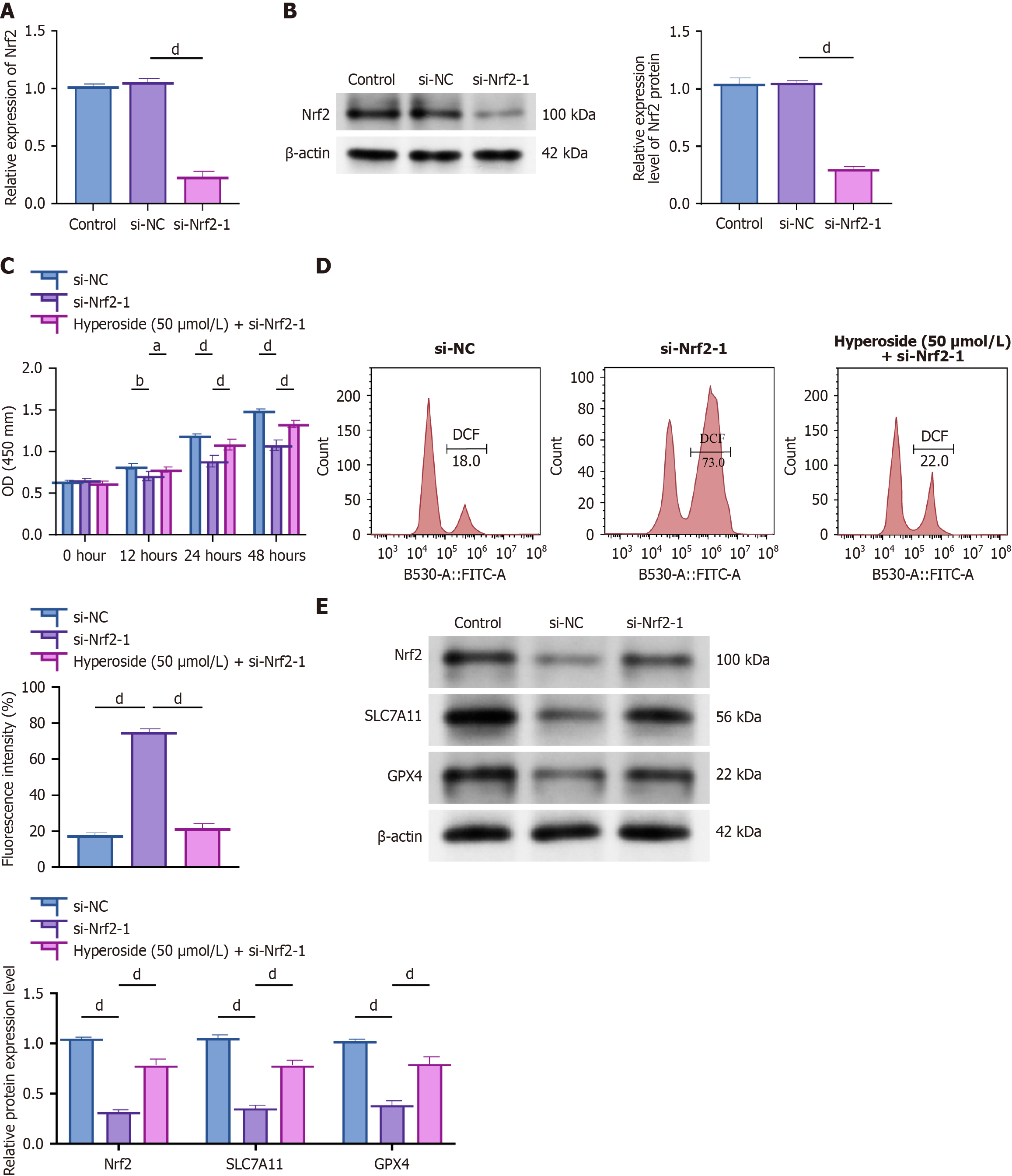
To determine whether hyperoside exerts its effects via the Nrf2 signaling pathway, we used siRNA to inhibit Nrf2 expression and observed functional changes following hyperoside treatment. qRT-PCR results showed that si-Nrf2 significantly reduced Nrf2 mRNA levels, verifying the transfection efficiency (Figure 4A; P < 0.05). Western blotting results further confirmed that si-Nrf2 significantly downregulated Nrf2 protein levels (Figure 4B; P < 0.05). In cell proliferation experiments, CCK-8 detection results indicated that the si-Nrf2 group showed markedly decreased cell proliferation capacity, while hyperoside (50 μmol/L) treatment partially restored proliferation capacity (Figure 4C; P < 0.05). Flow cytometry showed that si-Nrf2 significantly increased cellular lipid ROS levels, whereas hyperoside combined with si-Nrf2 significantly reduced ROS accumulation (Figure 4D; P < 0.05). Western blotting results also indicated that si-Nrf2 treatment significantly downregulated Nrf2, SLC7A11, and GPX4 protein expression, while hyperoside combined with si-Nrf2 treatment partially reversed these changes (Figure 4E; P < 0.05). These results suggest that hyperoside primarily targets Nrf2 to modulate the Nrf2/SLC7A11/GPX4 signaling axis, thereby inhibiting ferroptosis and exerting protective effects.
To further clarify whether the protective effects of hyperoside under HG conditions are mediated through ferroptosis inhibition, we introduced the ferroptosis inducer erastin for mechanistic validation. CCK-8 assays showed that HG significantly reduced HK-2 cell viability compared with the NG group (P < 0.01), whereas hyperoside (50 μmol/L) markedly restored proliferation (P < 0.05 vs HG). When erastin was co-administered, the recovery of cell viability was significantly attenuated compared with hyperoside alone (P < 0.05), indicating that ferroptosis induction counteracts the protective effect of hyperoside (Figure 5A). Consistently, TUNEL staining demonstrated that HG significantly increased apoptosis rates (P < 0.01), hyperoside significantly reduced the proportion of TUNEL-positive cells (P < 0.05), and erastin partially reversed this anti-apoptotic effect (P < 0.05 vs HG + hyperoside) (Figure 5B). Flow cytometry further showed that HG markedly elevated lipid ROS levels (P < 0.001), hyperoside significantly blunted ROS accumulation (P < 0.01 vs HG), and erastin co-treatment diminished the inhibitory effect of hyperoside (P < 0.05 vs HG + hyperoside) (Figure 5C). Western blot analysis revealed that HG significantly downregulated the expression of Nrf2, SLC7A11, and GPX4 (all P < 0.01), while hyperoside significantly restored their protein levels (P < 0.05 vs HG). Co-treatment with erastin significantly weakened the hyperoside-induced upregulation of these ferroptosis-related proteins (P < 0.05) (Figure 5D). Collectively, these results indicate that hyperoside alleviates HG-induced oxidative stress, lipid peroxidation, and apoptosis primarily by inhibiting ferroptosis through activation of the Nrf2/SLC7A11/GPX4 axis.
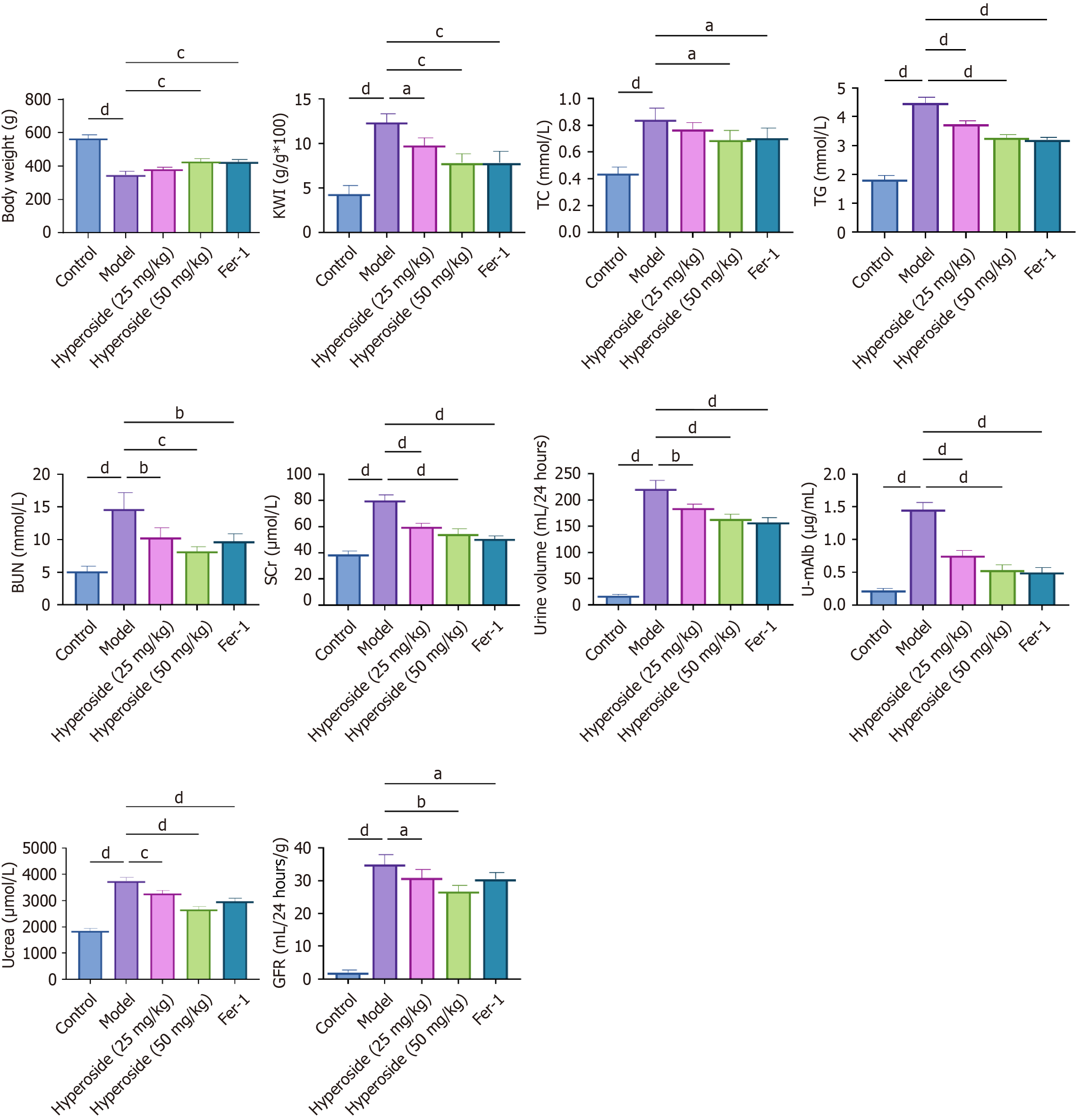
To further investigate the effects of hyperoside in vivo, we administered 25 mg/kg and 50 mg/kg hyperoside to DN rat models, with the Fer-1 group serving as a ferroptosis inhibition control. Rats in the model group demonstrated significantly higher body weights and KWI than those in the control group. Hyperoside treatment at 50 mg/kg markedly reduced body weight and KWI, whereas the 25 mg/kg group showed no significant difference from the model group for these two indicators (Figure 6; P > 0.05). Regarding blood lipids and renal function-related indicators, the model group rats showed significantly elevated serum TC, TG, Scr, and BUN levels, increased UAE, and significantly decreased GSH levels. Compared with the model group, both 25 mg/kg and 50 mg/kg hyperoside interventions significantly improved these abnormal indicators, with the 50 mg/kg group showing more pronounced effects. The Fer-1 group demonstrated protective effects similar to those of the 50 mg/kg hyperoside group (Figure 6, P < 0.05).
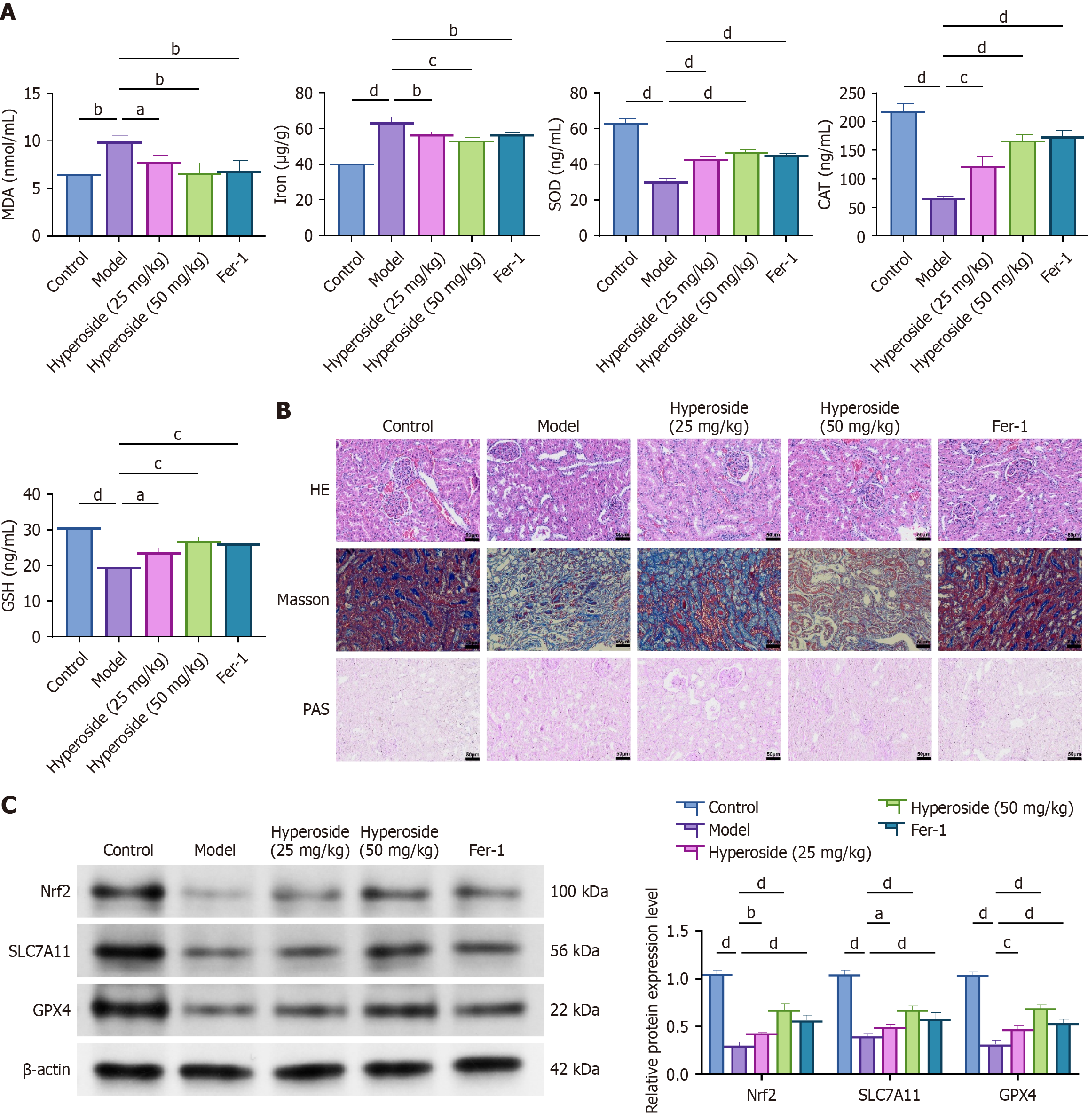
To further explore the effects of hyperoside in DN rats in vivo, we measured oxidative stress indicators and renal histopathological changes by analyzing ferroptosis-related protein expression levels. ELISA results showed that kidney tissues from the model group exhibited significantly increased levels of MDA and iron, accompanied by markedly decreased levels of SOD, CAT, and GSH. Hyperoside (25 mg/kg and 50 mg/kg) markedly improved these abnormalities, with 50 mg/kg showing the most significant effect. Fer-1 group’s protective effects were similar to those of the high-dose hyperoside (Figure 7A; P < 0.05). Hematoxylin and eosin staining revealed disordered glomerular structure and obvious renal tubular epithelial cell damage in the model group, Masson staining indicated massive collagen deposition, and periodic acid-Schiff staining showed basement membrane thickening and mesangial area expansion.
Both the hyperoside intervention groups (25 mg/kg and 50 mg/kg) and the Fer-1 group showed improved renal histopathological changes, with the 50 mg/kg group showing the most obvious improvement (Figure 7B; P < 0.05). Western blotting results indicated that rat kidney tissues from the model group showed significantly downregulated Nrf2, SLC7A11, and GPX4 protein expression, whereas both hyperoside and Fer-1 interventions restored their expression levels, suggesting that hyperoside ameliorates DN-related kidney damage by modulating the Nrf2/SLC7A11/GPX4 signaling axis (Figure 7C; P < 0.05).
DN is a serious chronic complication of diabetes mellitus, characterized by complex, interacting pathological processes, including metabolic dysregulation, hemodynamic stress, inflammation, oxidative damage, and maladaptive repair. It remains a leading cause of end-stage renal disease worldwide[18,19]. Despite advances in glycemic and blood-pressure control, the residual risk of progressive renal decline remains substantial, necessitating mechanistically informed adjunctive strategies. Ferroptosis, a regulated, iron-dependent mode of cell death driven by lipid peroxidation, is a major contributor to kidney injury, specifically to DN progression[20]. Building on this paradigm, our study demonstrates that hyperoside attenuates DN-associated injury by activating the Nrf2/SLC7A11/GPX4 axis and limiting ferroptosis-driven oxidative stress in tubular cells and in the kidneys of diabetic rats.
Across the complementary models, we observed a consistent protective signature of hyperoside. In HK-2 tubular epithelial cells exposed to HG (30 mmol/L), hyperoside improved cell viability and reduced apoptosis in a dose-responsive manner, with 50-100 μM yielding the most consistent restoration of cellular function. These phenotypic benefits were accompanied by coordinated biochemical changes: Decreases in LDH (membrane damage), MDA (lipid peroxidation), Fe2+ (labile iron), and ROS, alongside an increase in GSH, indicating an improved redox state and dampened ferroptotic pressure. These findings are consistent with those of previous reports that hyperoside ameliorates inflammation and apoptosis in diabetic conditions and kidney injury[17,21]. In STZ-induced DN rats, daily hyperoside (25-50 mg/kg) improved Scr, BUN, and UAE and mitigated glomerular and tubulointerstitial histological abnormalities, supporting translational relevance. The 50 mg/kg dose consistently outperformed the 25 mg/kg dose in functional and structural readouts, including trends in body weight and KWI, suggesting a pharmacodynamic dose-response that may guide future dose selection and exposure-response modeling[22,23]. Importantly, these effects were not observed in a single readout layer. In vivo renal homogenates mirrored the cellular antioxidant pattern, with lower MDA and iron content and higher SOD/CAT/GSH, indicating the restoration of enzymatic and non-enzymatic antioxidant defenses. Collectively, the convergence of functional indices, histopathology, and oxidative stress biomarkers across models strengthens the inference that hyperoside favorably remodels the redox and ferroptosis landscapes in the diabetic kidney. These findings also align with the view that ferroptosis can amplify inflammatory signaling in DN and that alleviating lipid peroxidation and iron overload may concurrently dampen inflammation-driven injury[9].
The Nrf2/SLC7A11/GPX4 axis integrates several nodes that counteract ferroptosis. Nrf2 transcriptionally coordinates antioxidant and detoxification programs and maintains redox homeostasis under stress[24]. SLC7A11 imports cystine for GSH synthesis, thereby supplying the substrate required by GPX4, a phospholipid hydroperoxidase that removes lipid peroxides and prevents ferroptotic execution[25]. GPX4 levels are also associated with DN progression, highlighting their clinical relevance[26]. In our study, HG reduced the mRNA and protein levels of Nrf2, SLC7A11, and GPX4; hyperoside restored this axis both in vitro and in vivo. These patterns are consistent with studies showing that hyperoside confers renoprotection against toxicant-induced kidney injury and alleviates oxidative stress and inflammation[27,28], and with broader anti-inflammatory and antioxidant effects linked to Nrf2 modulation[29]. The loss-of-function experiments provided mechanistic support: SiRNA-mediated Nrf2 knockdown dampened the biochemical rescue by hyperoside (elevated ROS, reduced viability) and blunted the upregulation of SLC7A11/GPX4, indicating that Nrf2 activity is functionally required for the complete protective phenotype. Interestingly, we observed partial restoration of SLC7A11/GPX4 under Nrf2 knockdown, suggesting either residual Nrf2 activity or Nrf2-independent inputs, consistent with the multitarget properties of natural products and possible crosstalk with PI3K/Akt or AMPK nodes that can influence redox and metabolic signaling[25,27-29]. From a systems standpoint, the coordinated improvement in cystine import (SLC7A11), GSH availability, and lipid peroxide clearance (GPX4) offers a physiologically coherent explanation for the reduction in ferroptotic stress under hyperoside.
Ferroptosis has been increasingly implicated in kidney diseases, specifically DN, in which hyperglycemia-induced oxidative stress, altered iron handling, and lipid peroxidation can amplify tubular damage and propagate interstitial fibrosis[30]. Ferroptosis profiling was performed using biochemical markers and pharmacological benchmarks. HG elevated MDA and Fe2+ and depleted GSH, a pattern reversed by hyperoside and closely resembling the profile seen with Fer-1, a canonical ferroptosis inhibitor. The erastin challenge served as a mechanistic stress test: By inhibiting SLC7A11, erastin reinstated ferroptotic vulnerability. Hyperoside partially countered erastin-induced injury and lipid ROS accumulation, consistent with its action on the SLC7A11-GSH-GPX4 axis, but also indicated that hyperoside is not a complete substitute for direct ferroptosis blockade. This nuance is biologically plausible and consistent with previous studies showing that agents restoring GPX4 activity or reducing lipid peroxidation mitigate DN progression in preclinical models[31,32]. Recent mechanistic studies implicating PRMT6/STAT1/ACSL1 transcriptional wiring and STING-FPN1-driven iron handling in DN-related ferroptosis further highlight the broader network into which the effects of hyperoside may be integrated[33,34]. Within this network, our data suggest that targeting the nodal antioxidant axis (Nrf2/SLC7A11/GPX4) offers practical leverage.
The pleiotropic nature of hyperoside, spanning antioxidant, anti-inflammatory, anti-fibrotic, and anti-apoptotic actions, confers attractive developmental properties in a syndrome as multifactorial as DN[32]. From a therapeutic positioning standpoint, the mechanism of action of hyperoside complements current standard treatments (e.g., ACEI/ARB), which primarily modulate the hemodynamic/RAAS axes but only indirectly address oxidative and ferroptotic stress. Prior work suggests that hyperoside influences additional pathways pertinent to DN, including ROS-ERK signaling and pyroptosis, broadening the mechanistic base for renal protection[29]; it also exerts systemic benefits in diabetic models through tumor necrosis factor alpha/nuclear factor kappa B/caspase-3 modulation[35]. Consistent with recent therapeutic syntheses, positioning ferroptosis as a druggable vulnerability in DN provides a mechanistic rationale for adjunctive strategies that complement current standards of care[15]. Biomarker strategies are important for translation. Elements of the axis (Nrf2/SLC7A11/GPX4) and ferroptosis markers (MDA, Fe2+, and GSH) can serve as pharmacodynamic readouts to track target engagement and dose adequacy. In addition, composite panels incorporating lipid peroxidation surrogates, iron indices, and antioxidant enzyme activities (SOD/CAT) could improve the sensitivity to changes and help stratify patients with a high ferroptosis signature. Such a framework can also facilitate early go/no-go decisions in dose-finding studies and enable hypothesis-driven combinations (e.g., with ACEI/ARB) while monitoring overlapping toxicities and redox balance.
First, the in vitro experiments relied on a single tubular epithelial cell line (HK-2). Although tubular cells are central to oxidative stress and ferroptosis in DN, validation in podocytes and mesangial cells would strengthen cell-type generalizability and clarify potential glomerular-tubular cross-talk[20,23]. Second, although the high-fat diet combined with low-dose streptozotocin model is widely used to mimic T2DN, it does not fully recapitulate the heterogeneity of human disease. Moreover, fasting insulin levels and insulin resistance indices (e.g., HOMA-IR) were not assessed; direct metabolic characterization would further strengthen model validation[19]. Third, while a mannitol osmotic control was included to exclude hyperosmolar effects, the in vitro model did not incorporate palmitic acid. Given that glucolipotoxicity is a key driver of tubular injury in T2DN, future studies combining high glucose and lipid overload would better reflect the diabetic metabolic milieu and further define the anti-ferroptotic effects of hyperoside. Fourth, our mechanistic analysis focused on the Nrf2/SLC7A11/GPX4 axis. Upstream regulation of Nrf2, including Keap1 modification and potential cross-talk with PI3K/Akt or AMPK signaling, warrants further investigation[25,27-29]. Fifth, although multiple biochemical and pharmacological approaches were used to assess ferroptosis, additional orthogonal validation—such as mitochondrial ultrastructural analysis and alternative ferroptosis inhibitors—would strengthen causal inference[10,29-32]. Finally, translational development will require pharmacokinetic, toxicological, and drug-drug interaction studies, particularly in the context of combination therapy with established renoprotective agents. Advanced multi-omics and single-cell approaches may further elucidate target specificity and identify patient subgroups most likely to benefit[29,35-38].
Collectively, the evidence presented herein places Nrf2/SLC7A11/GPX4 at the core of the renoprotective action of hyperoside, operationalizing a biologically coherent antiferroptotic program in the diabetic kidney. The axis-level improvements observed—enhanced cystine import and GSH availability (SLC7A11), increased lipid peroxide detoxification (GPX4), and upstream stress-response activation (Nrf2)—map onto reduced lipid peroxidation and iron-driven oxidative load, which in turn align with improvements in tubular viability, renal function, and histopathology. These signals are consistent with the contemporary literature implicating ferroptosis in DN pathogenesis and support the broader concept that redox and iron handling represent actionable vulnerabilities in diabetic kidney disease[20,30-34]. Within this framework, hyperoside has emerged as a biologically plausible candidate to complement standard-of-care therapies. It acts through a verifiable mechanism, can be monitored via pharmacodynamic markers, and exhibits generalizability to support combination therapeutic strategies.
Our data indicate that hyperoside mitigates diabetic renal injury by activating the Nrf2/SLC7A11/GPX4 axis and suppressing ferroptosis-related oxidative damage in HG-challenged HK-2 cells and in STZ-induced DN rats. Hyperoside increases Nrf2, SLC7A11, and GPX4 expression, reduces lipid ROS, MDA, and Fe2+, restores GSH, and improves renal functional indices and histopathology. The protective effects are weakened by erastin and by Nrf2 knockdown, supporting a mechanistic role for this pathway. These preclinical findings support hyperoside as a candidate modulator of ferroptosis in DN; future studies are required to define upstream regulation, cell-type specificity, pharmacokinetics/safety, and efficacy across additional disease models.
| 1. | Tanase DM, Gosav EM, Anton MI, Floria M, Seritean Isac PN, Hurjui LL, Tarniceriu CC, Costea CF, Ciocoiu M, Rezus C. Oxidative Stress and NRF2/KEAP1/ARE Pathway in Diabetic Kidney Disease (DKD): New Perspectives. Biomolecules. 2022;12:1227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 138] [Reference Citation Analysis (1)] |
| 2. | Al-Karawi AS, Kadhim AS. Correlation of autoimmune response and immune system components in the progression of IgA nephropathy: A comparative study. Hum Immunol. 2024;85:111181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 3. | Wang W, Ji Q, Ran X, Li C, Kuang H, Yu X, Fang H, Yang J, Liu J, Xue Y, Feng B, Lei M, Zhu D. Prevalence and risk factors of diabetic peripheral neuropathy: A population-based cross-sectional study in China. Diabetes Metab Res Rev. 2023;39:e3702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 61] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 4. | Asaad LA, Al-Shimary AA, AL-Azzawi MA, Al-Karawi AS, Rasool KH. The relationship between renal impairment and specific laboratory markers: a comprehensive investigation focusing on athletes. J Reattach Ther Develop Diversit. 2023;6:452-458. |
| 5. | Jiang S, Fang J, Yu T, Li W. Angiotensin-converting enzyme inhibitors versus angiotensin II receptor blockers for renal outcomes and mortality in diabetic kidney disease. Eur J Intern Med. 2021;85:127-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 6. | Saimoto Y, Kusakabe D, Morimoto K, Matsuoka Y, Kozakura E, Kato N, Tsunematsu K, Umeno T, Kiyotani T, Matsumoto S, Tsuji M, Hirayama T, Nagasawa H, Uchida K, Karasawa S, Jutanom M, Yamada KI. Lysosomal lipid peroxidation contributes to ferroptosis induction via lysosomal membrane permeabilization. Nat Commun. 2025;16:3554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 66] [Reference Citation Analysis (1)] |
| 7. | Lei G, Mao C, Horbath AD, Yan Y, Cai S, Yao J, Jiang Y, Sun M, Liu X, Cheng J, Xu Z, Lee H, Li Q, Lu Z, Zhuang L, Chen MK, Alapati A, Yap TA, Hung MC, You MJ, Piwnica-Worms H, Gan B. BRCA1-Mediated Dual Regulation of Ferroptosis Exposes a Vulnerability to GPX4 and PARP Co-Inhibition in BRCA1-Deficient Cancers. Cancer Discov. 2024;14:1476-1495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 8. | Liu S, Chen JH, Li LC, Ye ZP, Liu JN, Chen YH, Hu BX, Tang JH, Feng GK, Li ZM, Deng CX, Deng R, Zhu XF, Zhang HL. Susceptibility of Mitophagy-Deficient Tumors to Ferroptosis Induction by Relieving the Suppression of Lipid Peroxidation. Adv Sci (Weinh). 2025;12:e2412593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 9. | Li J, Li L, Zhang Z, Chen P, Shu H, Yang C, Chu Y, Liu J. Ferroptosis: an important player in the inflammatory response in diabetic nephropathy. Front Immunol. 2023;14:1294317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 60] [Reference Citation Analysis (1)] |
| 10. | Mengstie MA, Seid MA, Gebeyehu NA, Adella GA, Kassie GA, Bayih WA, Gesese MM, Anley DT, Feleke SF, Zemene MA, Dessie AM, Solomon Y, Bantie B, Dejenie TA, Teshome AA, Abebe EC. Ferroptosis in diabetic nephropathy: Mechanisms and therapeutic implications. Metabol Open. 2023;18:100243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (1)] |
| 11. | Yuan Y, Zhai Y, Chen J, Xu X, Wang H. Kaempferol Ameliorates Oxygen-Glucose Deprivation/Reoxygenation-Induced Neuronal Ferroptosis by Activating Nrf2/SLC7A11/GPX4 Axis. Biomolecules. 2021;11:923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 375] [Article Influence: 75.0] [Reference Citation Analysis (5)] |
| 12. | Su S, Ma Z, Wu H, Xu Z, Yi H. Oxidative stress as a culprit in diabetic kidney disease. Life Sci. 2023;322:121661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 43] [Reference Citation Analysis (1)] |
| 13. | Liang D, Feng Y, Zandkarimi F, Wang H, Zhang Z, Kim J, Cai Y, Gu W, Stockwell BR, Jiang X. Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones. Cell. 2023;186:2748-2764.e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 554] [Article Influence: 184.7] [Reference Citation Analysis (5)] |
| 14. | Wu Q, Huang F. Targeting ferroptosis as a prospective therapeutic approach for diabetic nephropathy. Ann Med. 2024;56:2346543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 36] [Reference Citation Analysis (1)] |
| 15. | Tripathi S, Kharkwal G, Mishra R, Singh G. Nuclear factor erythroid 2-related factor 2 (Nrf2) signaling in heavy metals-induced oxidative stress. Heliyon. 2024;10:e37545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 49] [Reference Citation Analysis (1)] |
| 16. | Wang S, Sheng F, Zou L, Xiao J, Li P. Hyperoside attenuates non-alcoholic fatty liver disease in rats via cholesterol metabolism and bile acid metabolism. J Adv Res. 2021;34:109-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 128] [Article Influence: 25.6] [Reference Citation Analysis (1)] |
| 17. | Liu J, Zhang Y, Sheng H, Liang C, Liu H, Moran Guerrero JA, Lu Z, Mao W, Dai Z, Liu X, Zhang L. Hyperoside Suppresses Renal Inflammation by Regulating Macrophage Polarization in Mice With Type 2 Diabetes Mellitus. Front Immunol. 2021;12:733808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 18. | Upadhyay A, Haider L. Mineralocorticoid Receptor Antagonists in Diabetic Kidney Disease: Clinical Evidence and Potential Adverse Events. Clin Diabetes. 2025;43:43-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 19. | Wang T, Zhang J, Wang Y, Zhao L, Wu Y, Ren H, Zou Y, Zhang R, Xu H, Chai Z, Cooper ME, Zhang J, Liu F. Whether Renal Pathology Is an Independent Predictor for End-Stage Renal Disease in Diabetic Kidney Disease Patients with Nephrotic Range Proteinuria: A Biopsy-Based Study. J Clin Med. 2022;12:88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 20. | Mu F, Luo P, Zhu Y, Nie P, Li B, Bai X. Iron Metabolism and Ferroptosis in Diabetic Kidney Disease. Cell Biochem Funct. 2025;43:e70067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 21. | Zhou L, An XF, Teng SC, Liu JS, Shang WB, Zhang AH, Yuan YG, Yu JY. Pretreatment with the total flavone glycosides of Flos Abelmoschus manihot and hyperoside prevents glomerular podocyte apoptosis in streptozotocin-induced diabetic nephropathy. J Med Food. 2012;15:461-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 22. | Zhou J, Zhang S, Sun X, Lou Y, Bao J, Yu J. Hyperoside ameliorates diabetic nephropathy induced by STZ via targeting the miR-499-5p/APC axis. J Pharmacol Sci. 2021;146:10-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 23. | An X, Zhang L, Yuan Y, Wang B, Yao Q, Li L, Zhang J, He M, Zhang J. Hyperoside pre-treatment prevents glomerular basement membrane damage in diabetic nephropathy by inhibiting podocyte heparanase expression. Sci Rep. 2017;7:6413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 24. | Sies H, Jones DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020;21:363-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4366] [Cited by in RCA: 3473] [Article Influence: 578.8] [Reference Citation Analysis (1)] |
| 25. | He R, Wei Y, Peng Z, Yang J, Zhou Z, Li A, Wu Y, Wang M, Li X, Zhao D, Liu Z, Dong H, Leng X. α-Ketoglutarate alleviates osteoarthritis by inhibiting ferroptosis via the ETV4/SLC7A11/GPX4 signaling pathway. Cell Mol Biol Lett. 2024;29:88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 43] [Reference Citation Analysis (1)] |
| 26. | Wang YH, Chang DY, Zhao MH, Chen M. Glutathione Peroxidase 4 Is a Predictor of Diabetic Kidney Disease Progression in Type 2 Diabetes Mellitus. Oxid Med Cell Longev. 2022;2022:2948248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 27. | Lucky IO, Aisuhuehien II, Adejoke ME. Renoprotective effect of hyperin against CdCl(2) prompted renal damage by activation of Nrf-2/Keap-1 ARE pathway in male mice. Toxicol Mech Methods. 2024;34:717-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 28. | Li Z, Liao W, Yin X, Liu L, Zhao Z, Lu X, Xu F, Lin X, Chen Y, Song J, He Z, Wei Q, Wu W, Wu Y, Yang X. Hyperoside attenuates Cd-induced kidney injury via inhibiting NLRP3 inflammasome activation and ROS/MAPK/NF-κB signaling pathway in vivo and in vitro. Food Chem Toxicol. 2023;172:113601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (1)] |
| 29. | Zhang K, Li M, Yin K, Wang M, Dong Q, Miao Z, Guan Y, Wu Q, Zhou Y. Hyperoside mediates protection from diabetes kidney disease by regulating ROS-ERK signaling pathway and pyroptosis. Phytother Res. 2023;37:5871-5882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 30. | Chu J, Wang K, Lu L, Zhao H, Hu J, Xiao W, Wu Q. Advances of Iron and Ferroptosis in Diabetic Kidney Disease. Kidney Int Rep. 2024;9:1972-1985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 31. | Xue M, Tian Y, Zhang H, Dai S, Wu Y, Jin J, Chen J. Curcumin nanocrystals ameliorate ferroptosis of diabetic nephropathy through glutathione peroxidase 4. Front Pharmacol. 2024;15:1508312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 32. | Huang J, Chen G, Wang J, Liu S, Su J. Platycodin D regulates high glucose-induced ferroptosis of HK-2 cells through glutathione peroxidase 4 (GPX4). Bioengineered. 2022;13:6627-6637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 33. | Hong J, Li X, Hao Y, Xu H, Yu L, Meng Z, Zhang J, Zhu M. The PRMT6/STAT1/ACSL1 axis promotes ferroptosis in diabetic nephropathy. Cell Death Differ. 2024;31:1561-1575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 34. | Zhao QX, Yan SB, Wang F, Li XX, Shang GK, Zheng ZJ, Xiao J, Lin ZW, Li CB, Ji XP. STING deficiency alleviates ferroptosis through FPN1 stabilization in diabetic kidney disease. Biochem Pharmacol. 2024;222:116102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 35. | Zhang W, Wang R, Guo R, Yi Z, Wang Y, Wang H, Li Y, Li X, Song J. The multiple biological activities of hyperoside: from molecular mechanisms to therapeutic perspectives in neoplastic and non-neoplastic diseases. Front Pharmacol. 2025;16:1538601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 36. | Chen X, Famurewa AC, Tang J, Olatunde OO, Olatunji OJ. Hyperoside attenuates neuroinflammation, cognitive impairment and oxidative stress via suppressing TNF-α/NF-κB/caspase-3 signaling in type 2 diabetes rats. Nutr Neurosci. 2022;25:1774-1784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 37. | Abdelhameed RFA, Ibrahim AK, Elfaky MA, Habib ES, Mahamed MI, Mehanna ET, Darwish KM, Khodeer DM, Ahmed SA, Elhady SS. Antioxidant and Anti-Inflammatory Activity of Cynanchum acutum L. Isolated Flavonoids Using Experimentally Induced Type 2 Diabetes Mellitus: Biological and In Silico Investigation for NF-κB Pathway/miR-146a Expression Modulation. Antioxidants (Basel). 2021;10:1713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 38. | Wu W, Xie Z, Zhang Q, Ma Y, Bi X, Yang X, Li B, Chen J. Hyperoside Ameliorates Diabetic Retinopathy via Anti-Oxidation, Inhibiting Cell Damage and Apoptosis Induced by High Glucose. Front Pharmacol. 2020;11:797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |