INTRODUCTION
Tacrolimus (FK506) is a calcineurin phosphatase inhibitor that has been used to prevent acute rejection after solid organ transplantation, improving graft survival[1]. However, tacrolimus has a significant ability to induce hyperglycemia and hyperinsulinemia, which suggests that insulin resistance occurs in patients treated with tacrolimus[2,3]. Moreover, it has been observed that the capacity of islet secretion can be negatively influenced by tacrolimus from an early stage after transplantation[4]. Animal experiments also show that tacrolimus contributes to development of diabetes, and promotes expression of intestinal glucose transporter (GLUT) and strengthens glucose absorption[5,6]. Our previous study has demonstrated that islet cell injury occurred in a tacrolimus-treated rat model, but the injury was reversible. During the period of tacrolimus discontinuation, the islet cell injury was improved[7]. Therefore, it has been confirmed that tacrolimus is associated with the development of new-onset diabetes after transplantation (NODAT). NODAT leads to serious deterioration of graft function, and tacrolimus is more diabetogenic than other calcineurin phosphatase inhibitors[8-10].
Many studies have been performed to discover the underlying mechanisms of tacrolimus-induced diabetes. Calcineurin phosphatase activity is critical in glucose-promoted insulin gene transcription and Npas4 expression, which is a cytoprotective factor in islet β-cells[11,12]. However, tacrolimus achieves its immunosuppressive effect through inhibiting calcineurin phosphatase activity[1]. A change from tacrolimus to cyclosporine A can increase insulin gene expression[13]. In addition, there is speculation that FK506 binding protein affects the intracellular calcium signaling pathway when binding tacrolimus, and reduces insulin exocytosis[14].
The mechanisms of tacrolimus-associated insulin resistance are still controversial. Animal experiments indicated that tacrolimus did not suppress insulin signaling in all tissues of rats[15]. Pereira et al[16], in 2013 discovered that tacrolimus enhanced lipolysis and inhibited lipogenesis, which ultimately led to dyslipidemia[16]. Dyslipidemia plays an important role in the development of insulin resistance. The mechanisms by which dyslipidemia causes insulin resistance have not been completely elucidated, but some important factors have been identified, including oxidative and endoplasmic reticulum stress, proinflammatory cytokines and generation of lipid metabolites[17]. Accordingly, tacrolimus may induce insulin resistance through an indirect pathway. Further study has shown that tacrolimus reduces glucose uptake of adipocytes by increasing GLUT4 endocytosis without inhibiting the insulin signaling pathway[18]. These results provide us with a novel insight in the study of tacrolimus-induced insulin resistance.
Liver is the backbone in substance metabolism. In hepatocytes, glycogen synthase and glycogen phosphorylase are the main enzymes in glucose metabolism. In fasting conditions, glucagon promotes glycogenolysis by activating glycogen phosphorylase and inhibiting glycogen synthase[19]. During the postprandial phase, gastrointestinal nutrients are absorbed into the liver prior to entering the blood circulation, and insulin promotes glycogenesis by reversing the action of glucagon to maintain glucose homeostasis[19]. It has been suggested that insulin receptor substrate (IRS) 2 of hepatocytes is mainly expressed in the periportal area of the liver, which dominates glucose metabolism and is suppressed by high levels of insulin induced by excess free fatty acids (FFAs)[20,21]. Therefore, disorder of insulin signaling in hepatocytes is decisive in development of insulin resistance and there is a need to explore the direct effects of tacrolimus on hepatocytes. Our previous study demonstrated that tacrolimus increased phosphorylation at Thr 308 of Akt in HL7702 cells[22]. Accordingly, the effects of tacrolimus on other important sites in insulin signaling of hepatocytes will be further investigated in this study.
MATERIALS AND METHODS
Materials
FK506 was purchased from Cell Signaling Technology (3 Trask Lane, Danvers, MA, United States). The primary antibodies were as follows: Anti-phospho-GSK3α (Ser 21) rabbit monoclonal antibody (mAb), anti-GSK3α rabbit mAb, anti-phospho-GSK3β (Ser 9) rabbit mAb, anti-GSK3β rabbit mAb, anti-phospho-GS (Ser 641) rabbit Ab, anti-GS rabbit Ab, anti-phospho-S6K1 (Thr 389) rabbit mAb, anti-phospho-IRS1 (Ser 307) rabbit mAb, anti-phospho-IRS1 (Ser 323) rabbit mAb, anti-phospho-IRS1 (Ser 1101) rabbit Ab, and anti-IRS1 rabbit Ab (Cell Signaling Technology). An anti-S6K1 rabbit antibody was obtained from Millipore. An anti-glycogen phosphorylase (GP) antibody was obtained from Proteintech Group (Chicago, IL, United States). An anti-actin mouse mAb was obtained from Abcam. The secondary antibodies for western blotting were horseradish-peroxidase-linked anti-mouse/rabbit IgG (Cell Signaling Technology). Other chemicals and reagents were purchased from commercial sources.
Cell culture and treatment
Human liver cell line HL7702 was obtained from Cell Bank of the Chinese Academy of Science. HL7702 cells were cultivated in RPMI-1640 medium (Gibco) supplemented with 10% newborn bovine serum (Zhejiang Tianhang Biotechnology). The atmosphere was humidified with 5% CO2 at 37 °C. Before the medium was replaced with fresh medium that contained different concentrations of FK506 (0.1, 1 and 5 mg/L, dissolved in DMSO), HL7702 cells were allowed to grow for 24 hours in logarithmic phase. The fresh medium was used to treat HL7702 cells in the control group.
Western blotting
HL7702 cells in each group were lysed by RIPA buffer [1% NP-40, 150 mmol/L NaCl, 0.1% SDS, 0.5% deoxycholate, 50 mmol/L Tris (pH 7.4), protease inhibitors] to extract total protein. The concentration of protein was quantified by BCA assay. The protein was heated for 5 minutes in boiled water and denatured. SDS-PAGE was used to separate the denatured protein. The denatured protein was transferred onto nitrocellulose (NC) membranes. NC membranes were blocked by Tris-Buffered Saline with Tween-20 for 3 hours which contained 5% fat-free milk. NC membranes incubated with the corresponding primary antibodies overnight at 4 °C and secondary antibodies at room temperature for 1 hours. The target bands were visualized by the chemiluminescence imaging system and quantified by ImageJ software.
GP activity analysis
GP activity was measured according to the kit instruction of manufacturer (GenMed, Plymouth, MN, United States). Total protein was added to a buffer containing substrate and measured once every 60 seconds for 5 minutes at an absorbance of 340 nm. The difference between the immediate absorbance and the absorbance after 5 minutes represented the activity.
Statistical analysis
The data are shown as the mean ± SD from three independent experiments (n = 3). The significance of the differences between the control and tacrolimus treatment groups was analyzed with SPSS version 17.0 using one-way analysis of variance. P < 0.05 was defined as statistically significant.
RESULTS
Tacrolimus induces phosphorylation of IRS1 at serine residues
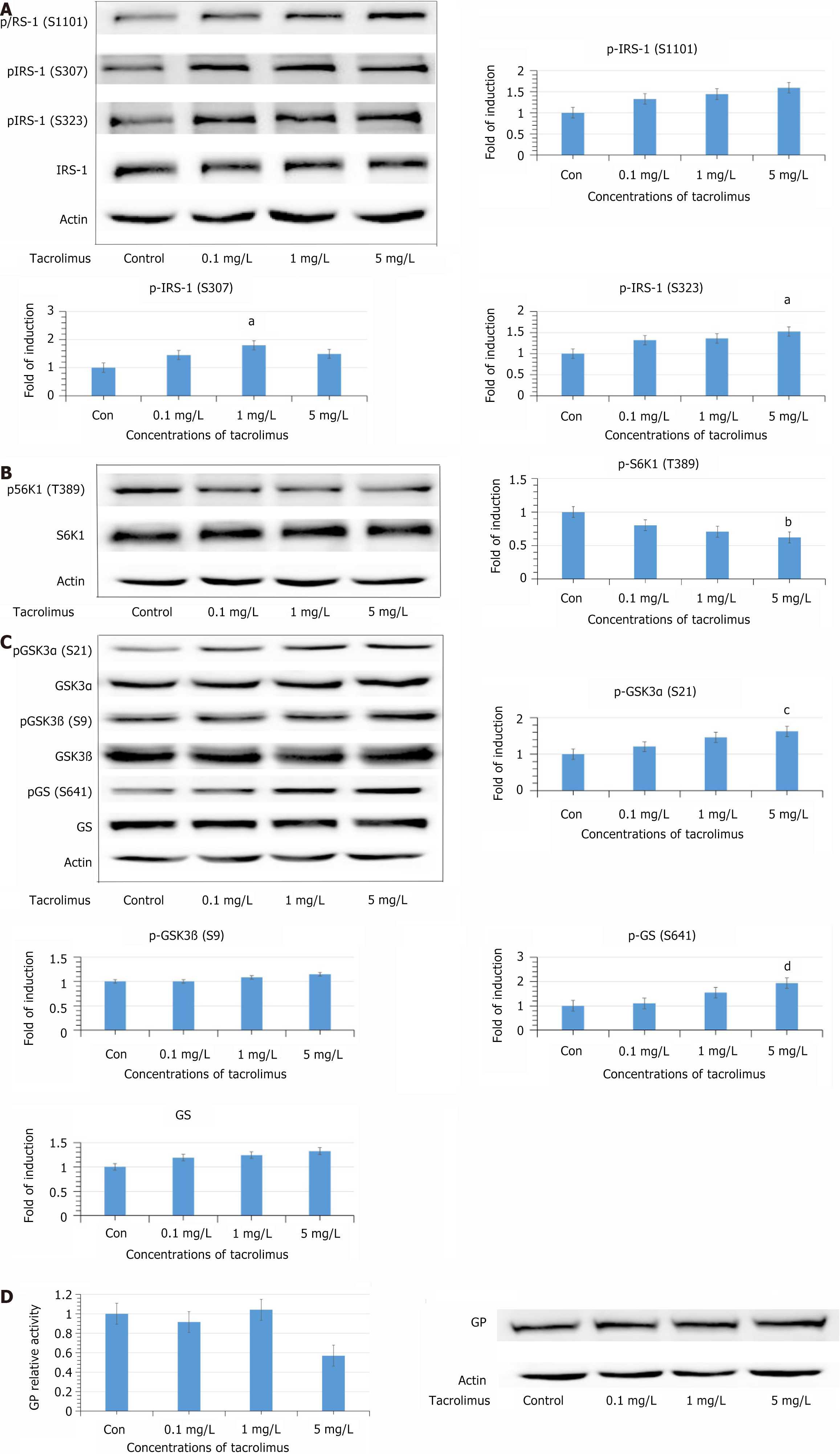
HL7702 cells were exposed to tacrolimus at different concentrations for 24 hours, and we detected phosphorylation at the serine sites of IRS1. Phosphorylation of IRS1 (Ser 307 and Ser 323) was significantly increased at 1 and 5 mg/L (P < 0.05; Figure 1A). Phosphorylation of IRS1 at Ser 1101 was also increased, but there was no significant change. The total levels of IRS-1 did not show any significant change.
Figure 1 After exposure to tacrolimus for 24 hours.
A: Phosphorylation of insulin receptor substrate (IRS) 1 at Ser 307 and Ser 323 was significantly increased at concentrations of 1 and 5 mg/L (aP < 0.05). Phosphorylation of IRS1 at Ser 1101 was also increased, although not significantly; B: Phosphorylation of S6K1 at Thr 389 was decreased in a concentration-dependent manner (bP < 0.05); C: Phosphorylation of glycogen synthase kinase (GSK) 3α at Ser 21 was increased (cP < 0.05). Phosphorylation of GSK3βat Ser 9 was also increased, but not significantly. Unexpectedly, the phosphorylation of glycogen synthase at Ser 641 was increased (dP < 0.05); D: Measurement of glycogen phosphorylase (GP) activity to ascertain the effects of tacrolimus on glycogen synthesis. After exposure to tacrolimus for 24 hours, there was no significant change in total expression of GP. IRS: Insulin receptor substrate; GSK: Glycogen synthase kinase; GP: Glycogen phosphorylase.
Tacrolimus decreases phosphorylation of S6K1
Our previous study showed that tacrolimus increased phosphorylation at Thr 308 of Akt in HL7702 cells after 24 hours treatment. IRS1 serine residues were mediated by the PI3K–Akt–S6K1 negative feedback loop, so the phosphorylation level of S6K1 (Thr 389) was further detected. However, phosphorylation of S6K1 (Thr 389) was decreased significantly (P < 0.05; Figure 1B).
Tacrolimus induces GS hyperphosphorylation, but did not influence activity of GP
Phosphorylation of glycogen synthase kinase (GSK)3α (Ser 21) and GSK3β (Ser 9) was increased, which demonstrated that activity of GSK3 was inhibited (Figure 1C). Surprisingly, the phosphorylation of the GSK3 substrate, GS (Ser 641), was not decreased but increased. Moreover, the total levels of GS were increased after tacrolimus exposure. There was no significant change in the activity of GP (Figure 1D).
DISCUSSION
In this study, tacrolimus decreased the level of p-S6K1 (Thr 389), which indicated that the mTORC/S6K1 pathway was inhibited. Besides, IRS1 was found to be phosphorylated at Ser 323, Ser 307 and Ser 1101. These results demonstrated that the activity of IRS1 was also inhibited. Insulin signaling mediated by IRS1 in hepatocytes is mainly responsible for lipogenesis[21]. Therefore, tacrolimus may inhibit lipid synthesis by disrupting insulin signaling associated with IRS1, and then increasing FFA concentration in the circulation. FFAs have been identified as important signaling molecules in insulin secretion. The experiment proved that FFAs are able to promote insulin secretion stimulated by glucose from pancreatic β-cells[23]. High level of insulin in the fasting phase can inhibit the expression of IRS2 in hepatocytes and induce hyperglycemia.
Our previous study found that tacrolimus increased phosphorylation at Thr 308 of Akt in HL7702 cells, which indicated that the activity of Akt had been promoted. It is well known that the PI3K/Akt pathway is important in substance metabolism. The PI3K/Akt pathway is activated by insulin binding to cell surface receptors, and activated Akt increases expression of GLUT proteins and promotes their translocation[24]. In hepatocytes, glucose transport is equivalent to free diffusion and not regulated by insulin[25]. Our previous study has also shown that insulin cannot mediate GLUT2 expression and translocation in HL7702 cells[26]. Accordingly, the enzymes associated with glucose metabolism are crucial in maintaining blood glucose homeostasis. GS plays a major role in glucose metabolism, and defective glycogen synthesis is an important cause of insulin resistance[27]. A lack of hepatic glycogen synthesis promotes lipid synthesis, and excess lipid impairs insulin signaling in hepatocytes[28]. Therefore, we detected the proteins associated with glycogen synthesis.
GSK3, the main substrate of Akt, is the principal kinase that is responsible for GS (Ser 641) phosphorylation[29]. GSK3 is important in regulating the expression of GS, which can be significantly promoted when the activity of GSK3 is inhibited[30]. In this study, tacrolimus increased phosphorylation of GSK3α at Ser 21 and GSK3β at Ser 9, which suggested that activity of GSK3 was inhibited. However, phosphorylation of GS (Ser 641) was elevated and there was no significant increase in GS expression. An animal study demonstrated that GS activity was strongly related to phosphorylation of Ser 641[31]. Increased phosphorylation at Ser 641 suggested that activity of GS was inhibited. To ascertain the effects of tacrolimus on glycogen synthesis and explore the mechanism by which tacrolimus leads to phosphorylation of GS at Ser 641, GP activity was further measured. GP determines the rate of glycogen degradation, and inhibiting GP activity has been identified as one of therapeutic measures in type 2 diabetes. Phosphorylated GS and phosphorylated GP are both substrates of protein phosphatase (PP)1[32,33]. After 24 h exposure of tacrolimus, the change in GP activity failed to achieve significance and there was no change in its expression. Phosphorylated GP is the active form. Dephosphorylation of GP is a critical action in glycogen synthesis, and this process is catalyzed by PP1[32]. Therefore, this result suggests that tacrolimus does not influence the activity of PP1, and phosphorylation of GS induced by tacrolimus is not achieved by inhibiting PP1.
FFAs have been identified as a critical factor in insulin resistance development. Clinical experiments have shown that insulin resistance can be induced by excess FFAs in the circulation, and endurance training cannot eliminate the negative effect[34]. Moreover, lowering plasma FFAs level can ameliorate insulin resistance and improve glucose tolerance[35]. Pancreatic β-cells are stimulated by FFAs to secrete insulin[36]. In hepatocytes, the insulin signaling pathway mediated by IRS2 is responsible for the regulation of glucose metabolism. The activity of IRS2 is suppressed in hyperinsulinemia[20,21].
Sterol-regulatory-element-binding proteins (SREBPs), which control the expression of lipogenic genes, are important transcription factors in lipogenesis. SREBPs are regulated by mTORC/S6K1 signaling. Inhibiting mTORC/S6K1 signaling can reduce the expression of SREBPs[37]. Triglyceride breakdown is negatively regulated by mTORC/S6K1 signaling. The process of lipolysis is catalyzed by adipose triglyceride lipase (ATGL), and mTORC1 influences the lipolytic process by regulating the expression of ATGL. The expression of ATGL significantly increases when the activity of mTORC1 is inhibited[38].
There were some limitations to this study. Due to the complexity of the regulation of glycogen synthesis, allosteric regulation is also a critical pathway in glycogen synthesis[39]. Therefore, the phosphorylation regulation cannot completely determine glycogen synthesis, and the rate of glycogen synthesis should be detected in future experiments.
CONCLUSION
This study shows that tacrolimus has no direct effect on hepatic glucose metabolism, but inhibits IRS1-mediated insulin signaling. This effect may result in hepatic lipid metabolism disorder and elevated FFAs in the circulation. This may be one of the underlying mechanisms by which tacrolimus induces insulin resistance in the body.
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/License/by-nc/4.0/
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Endocrinology and metabolism
Country of origin: China
Peer-review report’s classification
Scientific Quality: Grade C, Grade C, Grade C, Grade C, Grade E
Novelty: Grade B, Grade B, Grade B, Grade D
Creativity or Innovation: Grade B, Grade B, Grade C, Grade D
Scientific Significance: Grade B, Grade B, Grade C, Grade C
P-Reviewer: Ferreira GSA; Lei P; Tessarin GWL; Wu QN; Zhang ZQ S-Editor: Li L L-Editor: A P-Editor: Zhang XD