Copyright: ©Author(s) 2026.
World J Diabetes. Apr 15, 2026; 17(4): 116208
Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.116208
Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.116208
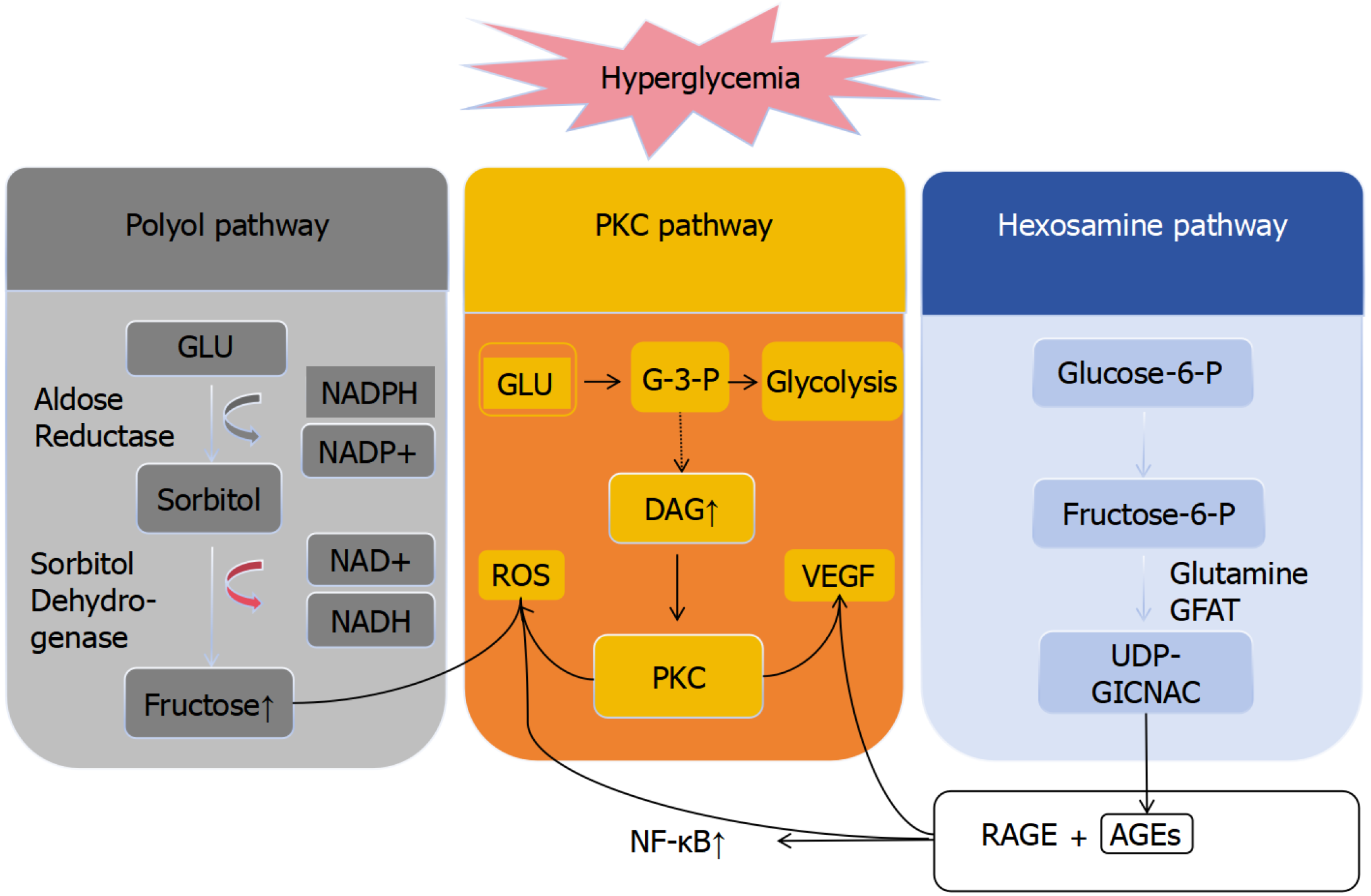
Figure 1 Schematic representation of core metabolic disturbances in diabetic retinopathy.
Under hyperglycemic conditions, the polyol pathway, hexosamine biosynthetic pathway, protein kinase C activation, and advanced glycation end-product (AGE) formation act synergistically to trigger oxidative stress, inflammation, aberrant angiogenesis, and pericyte apoptosis, thereby driving the progression of diabetic retinopathy. In the polyol pathway (gray panel), glucose is converted to sorbitol by aldose reductase, consuming NADPH, and subsequently oxidized to fructose, generating NADH and depleting the NADPH pool, which compromises cellular antioxidant defense. In parallel, hyperglycemia enhances glycolytic flux via glucose transporter 1 (orange panel), leading to the accumulation of glyceraldehyde-3-phosphate and diacylglycerol, which activate protein kinase C and subsequently upregulate vascular endothelial growth factor and reactive oxygen species production. Additionally, glucose entering the hexosamine biosynthetic pathway (blue panel) is converted by the rate-limiting enzyme glutamine fructose-6-phosphate amidotransferase to uridine diphosphate N-acetylglucosamine, promoting abnormal protein glycosylation. Meanwhile, excessive glucose accelerates AGE formation, and the binding of AGEs to receptor for AGE further enhances reactive oxygen species generation and activates nuclear factor-κB-mediated inflammatory signaling, amplifying vascular injury and metabolic stress. PKC: Protein kinase C; GLU: Glucose; NAD+: Oxidized nicotinamide adenine dinucleotide; NADH: Reduced nicotinamide adenine dinucleotide; NADP+: Oxidized nicotinamide adenine dinucleotide phosphate; NADPH: Reduced nicotinamide adenine dinucleotide phosphate; G-3-P: Glyceraldehyde-3-phosphate; DAG: Diacylglycerol; ROS: Reactive oxygen species; VEGF: Vascular endothelial growth factor; NF-κB: Nuclear factor-κB; AGE: Advanced glycation end-product; RAGE: Receptor for advanced glycation end-products; GFAT: Glutamine fructose-6-phosphate amidotransferase; UDP-GLCNAC: Uridine diphosphate N-acetylglucosamine.
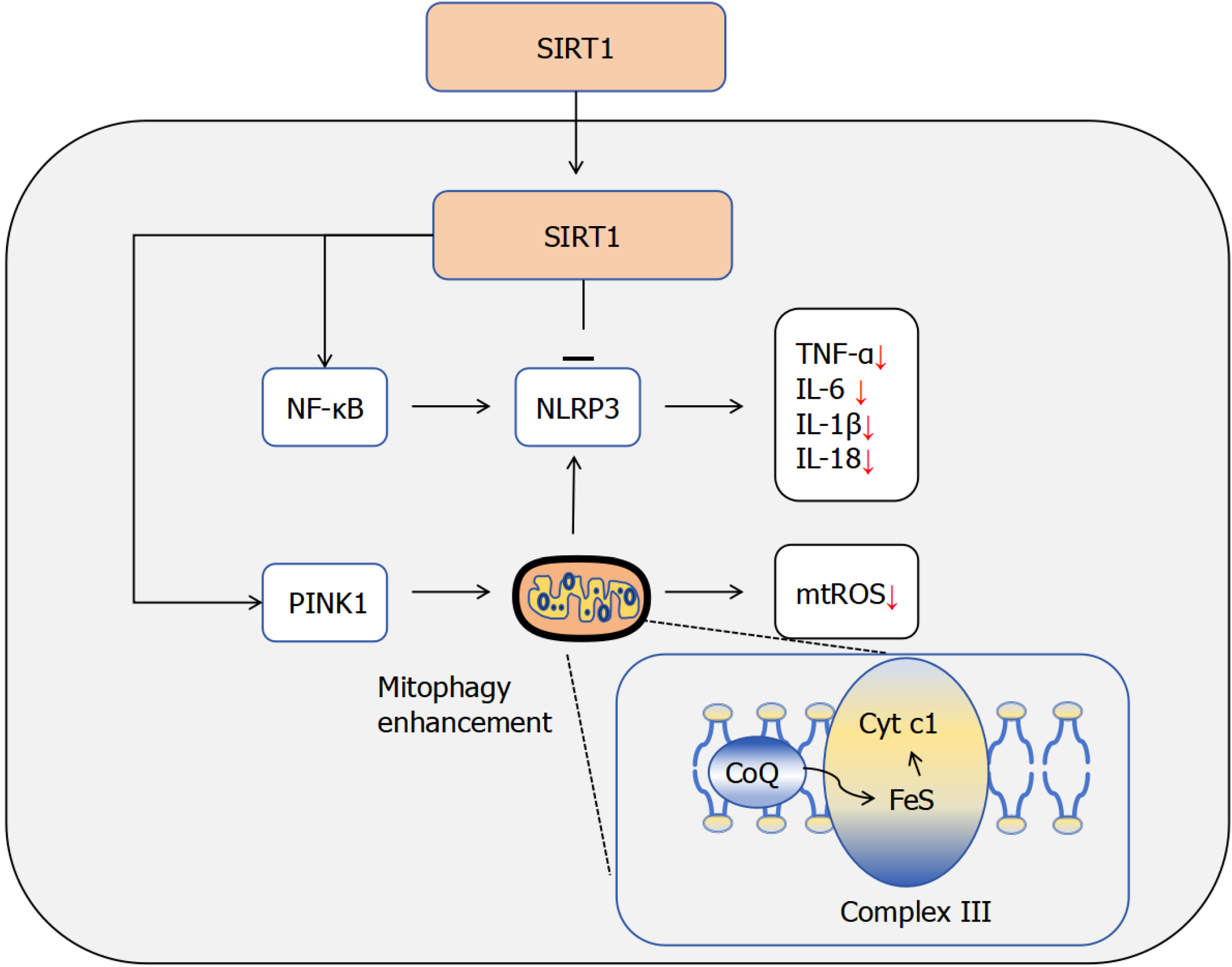
Figure 2 Mechanistic schematic of the sirtuin 1 signaling pathway in diabetic retinopathy.
This diagram illustrates the protective mechanisms of sirtuin 1 (SIRT1) in diabetic retinopathy. SIRT1 activation orchestrates three major protective pathways. First, it suppresses the nuclear factor-κB-NLR family pyrin domain-containing 3 inflammatory axis, thereby reducing the production of pro-inflammatory cytokines such as tumor necrosis factor-α, interleukin-6, and interleukin-1β. Second, it activates the transcription factor nuclear factor erythroid 2-related factor 2, enhancing the cellular antioxidant defense system to counteract oxidative stress. Third, SIRT1 promotes mitophagy via the PTEN-induced putative kinase 1 pathway, facilitating the removal of damaged mitochondria and consequently decreasing mitochondrial reactive oxygen species generation. Through these integrated actions, SIRT1 preserves mitochondrial integrity, alleviates inflammation, and maintains retinal homeostasis under hyperglycemic stress. SIRT1: Sirtuin 1; NF-κB: Nuclear factor-κB; NLRP3: NLR family pyrin domain-containing 3; TNF-α: Tumor necrosis factor-α; IL-1β: Interleukin-1β; IL-6: Interleukin-6; IL-18: Interleukin-18; PINK1: PTEN-induced putative kinase 1; mtROS: Mitochondrial reactive oxygen species; Cyt c1: Cytochrome c1; CoQ: Coenzyme Q.
- Citation: Pan CC, Xie QQ, Lu PY, Shi Z, Li HY, Ma YJ, Ding TY, Zeng MQ, Luo C, Zhuge FY. Targeting sirtuins in diabetic retinopathy: Differential roles in inflammation and mitochondrial dysfunction. World J Diabetes 2026; 17(4): 116208
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/116208.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.116208