Copyright: ©Author(s) 2026.
World J Diabetes. Apr 15, 2026; 17(4): 114679
Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.114679
Published online Apr 15, 2026. doi: 10.4239/wjd.v17.i4.114679
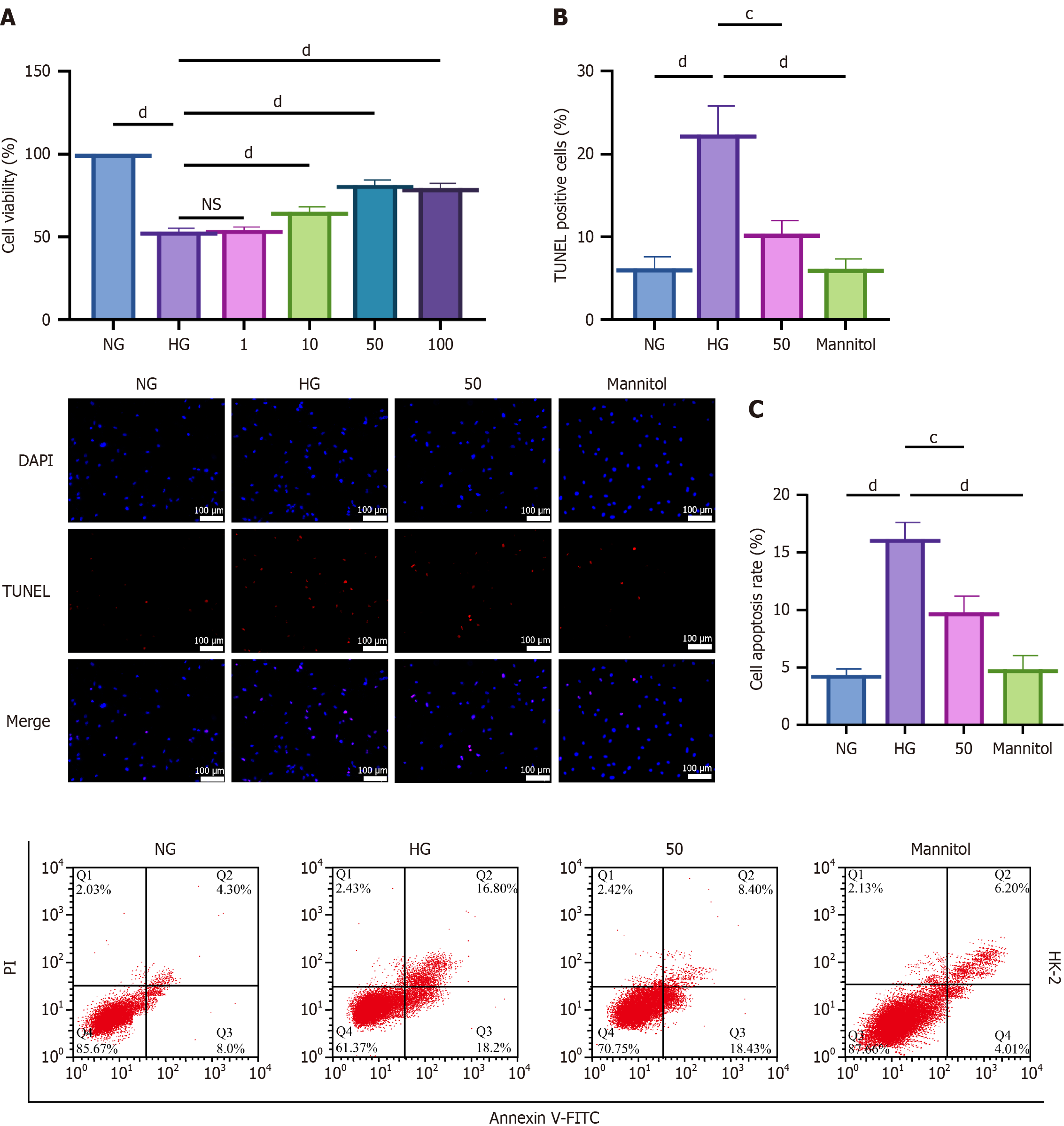
Figure 1 Effects of hyperoside on HK-2 cell viability under high-glucose conditions.
cP < 0.001, dP < 0.001. One-way analyses of variance with Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 3 independent experiments. A: Cell Counting Kit-8 assay showing viability with hyperoside (1 μM, 10 μM, 50 μM, and 100 μM) under high glucose (HG) (30 mmol/L); B: Terminal deoxynucleotidyl transferase dUTP nick-end labeling: Normal glucose, HG, and HG + hyperoside (50 μM); quantification of terminal deoxynucleotidyl transferase dUTP nick-end labeling-positive cells; C: Flow cytometry apoptosis rates in the same groups. NG: Normal glucose; HG: High glucose; DAPI: 4’,6-diamidino-2-phenylindole; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick-end labeling; FITC: Fluorescein isothiocyanate.
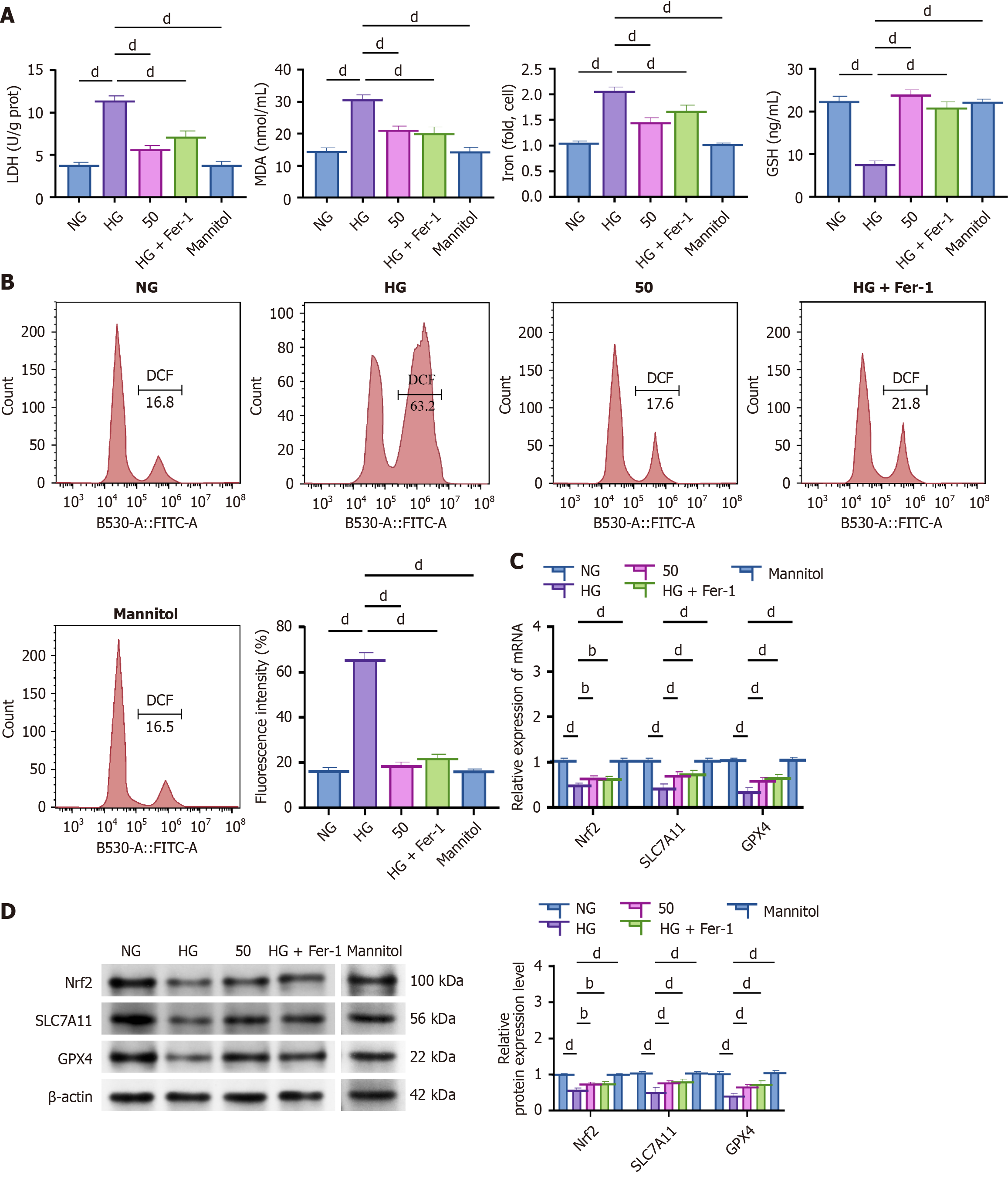
Figure 2 Hyperoside ameliorates high glucose-induced oxidative injury in HK-2 cells.
bP < 0.01, cP < 0.001, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 3 independent experiments. A: Lactate dehydrogenase, malondialdehyde, Fe2+, and glutathione in normal glucose, high glucose (HG), HG + hyperoside (50 μM), and HG + ferrostatin-1 (1 μM); B: Lipid reactive oxygen species using flow cytometry; C: MRNA expression of Nrf2, SLC7A11, and GPX4 detected using quantitative reverse transcription-polymerase chain reaction; D: Protein levels measured using Western blot. NG: Normal glucose; HG: High glucose; Fer-1: Ferrostatin-1; LDH: Lactate dehydrogenase; MAD: Malondialdehyde; FITC-A: Fluorescein isothiocyanate-area; DCF: 2’,7’-dichlorofluorescein.
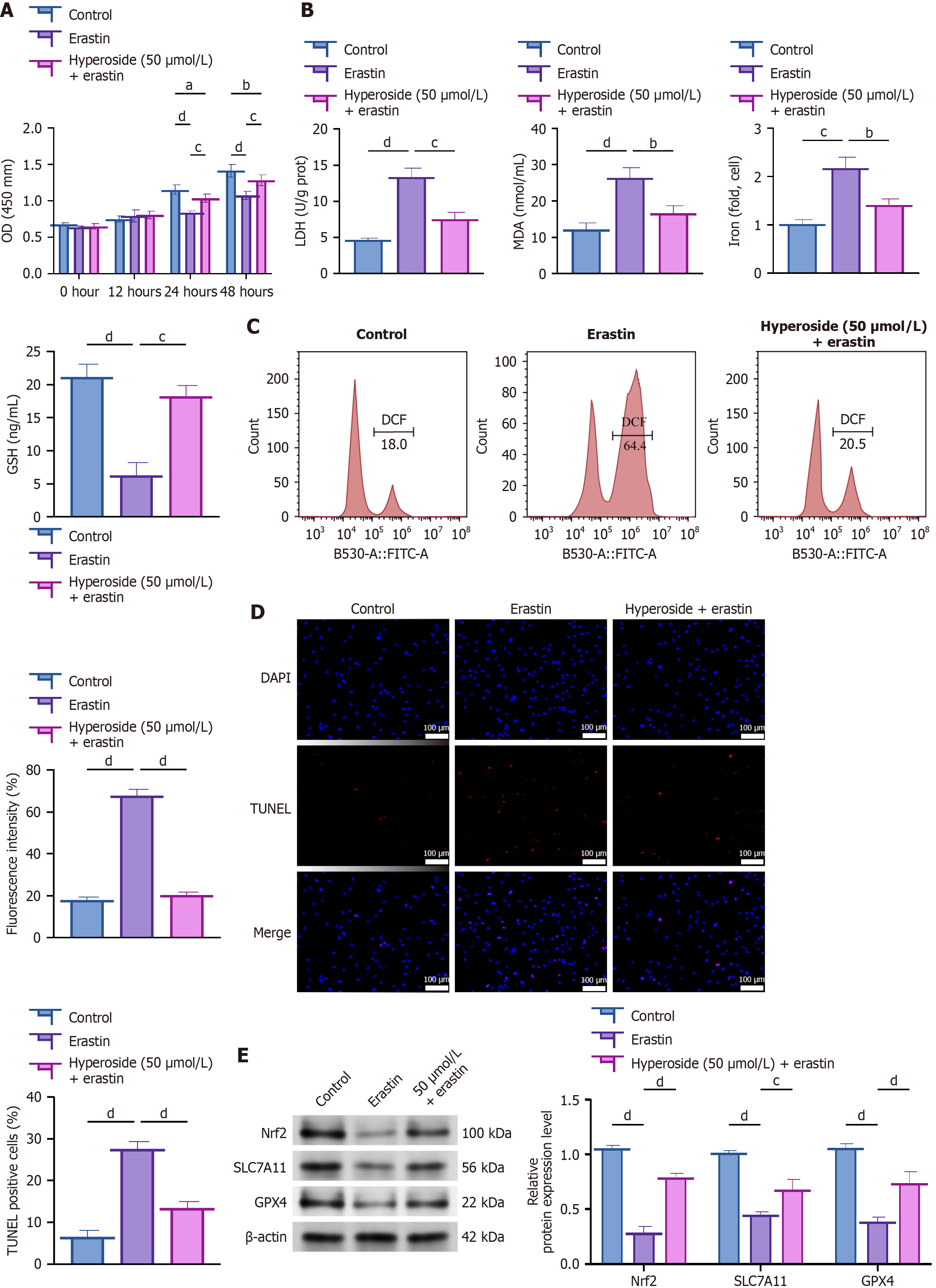
Figure 3 Hyperoside confers protection against erastin-induced ferroptotic injury.
aP < 0.05, bP < 0.01, cP < 0.001, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 3 independent experiments. A: Cell Counting Kit-8 times course (0-48 hours): Control, erastin (5 μM), and hyperoside (50 μM) + erastin; B: Lactate dehydrogenase, malondialdehyde, Fe2+, and glutathione; C: Lipid reactive oxygen species; D: Terminal deoxynucleotidyl transferase dUTP nick-end labeling apoptosis; E: Western blot of Nrf2, SLC7A11, and GPX4. LDH: Lactate dehydrogenase; MAD: Malondialdehyde; OD: Optical density; GSH: Glutathione; FITC-A: Fluorescein isothiocyanate-area; DAPI: 4’,6-diamidino-2-phenylindole; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick-end labeling.
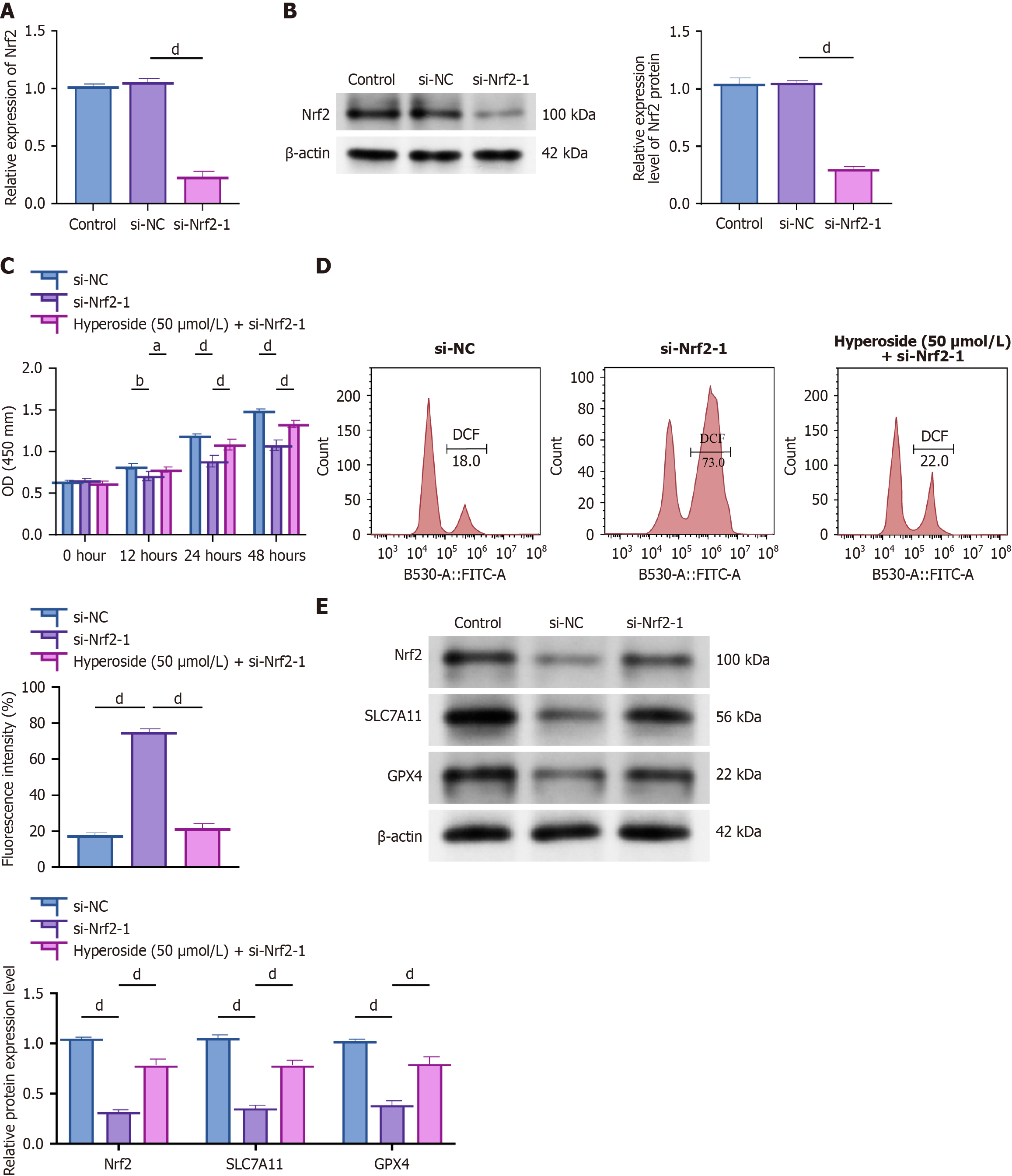
Figure 4 Hyperoside acts via Nrf2 to restrain ferroptosis (siRNA validation).
aP < 0.05, bP < 0.01, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 3 independent experiments. A: Quantitative reverse transcription-polymerase chain reaction confirming Nrf2 knockdown; B: Western blot confirming Nrf2 knockdown; C: Cell Counting Kit-8 time course: Small interfering RNA negative control, small interfering-Nrf2, and small interfering-Nrf2 + hyperoside (50 μM); D: Lipid reactive oxygen species detected using flow cytometry; E: Western blot analysis of Nrf2, SLC7A11, and GPX4 expression. si-NC: Small interfering RNA negative control; OD: Optical density; DCF: 2,7’-dichlorofluorescein; FITC-A: Fluorescein isothiocyanate-area.
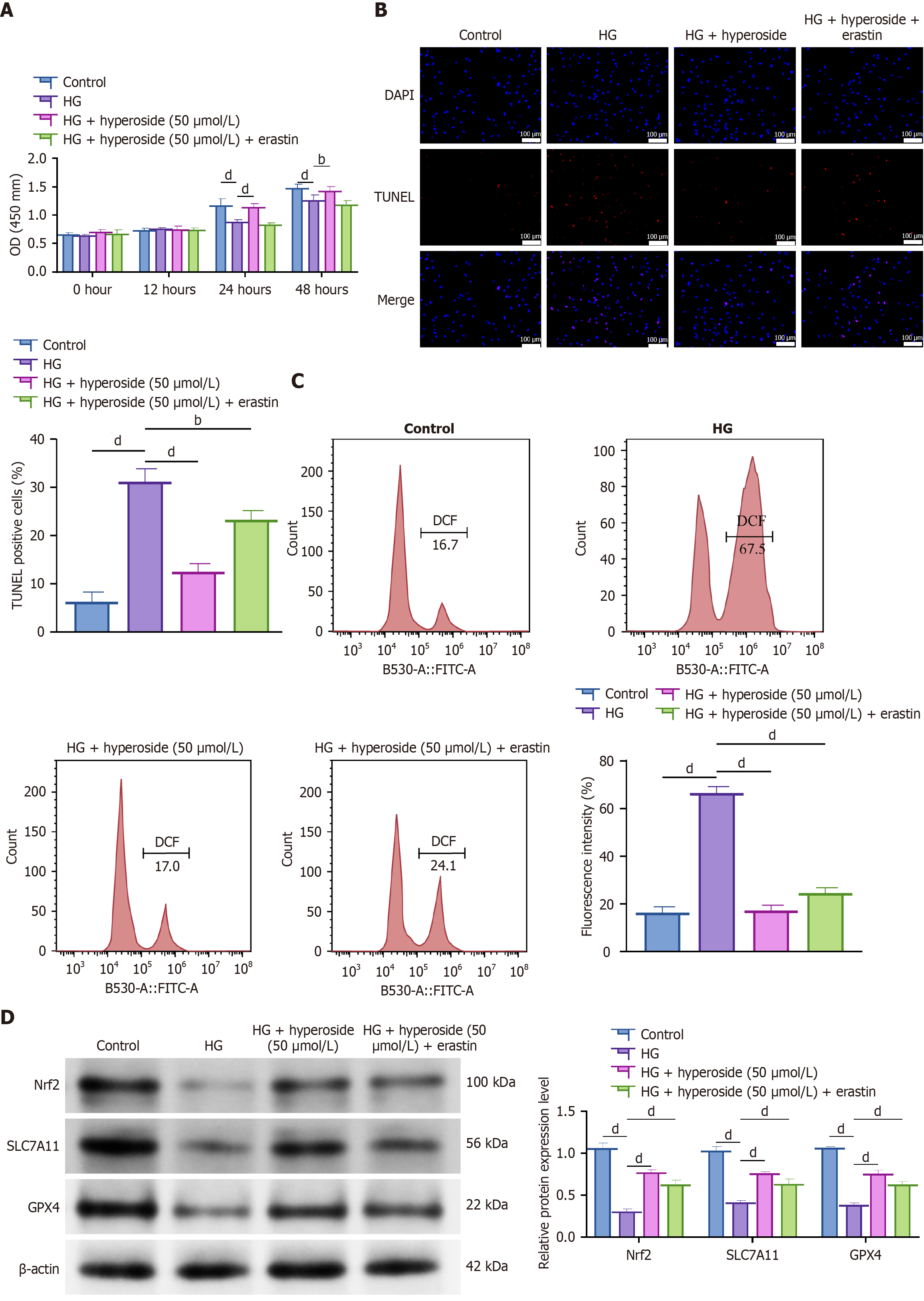
Figure 5 Hyperoside improves viability and reduces high glucose-induced apoptosis by inhibiting ferroptosis.
bP < 0.01, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 3 independent experiments. A: Cell Counting Kit-8 time course: Control, high glucose (HG) (30 mmol/L), HG + hyperoside (50 μM), and HG + hyperoside (50 μM) + erastin (5 μM); B: Terminal deoxynucleotidyl transferase dUTP nick-end labeling apoptosis; C: Lipid reactive oxygen species; D: Western blot analysis of Nrf2, SLC7A11, and GPX4 expression. HG: High glucose; OD: Optical density; DAPI: 4’,6-diamidino-2-phenylindole; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick-end labeling; DCF: 2,7’-dichlorofluorescein; FITC-A: Fluorescein isothiocyanate-area.
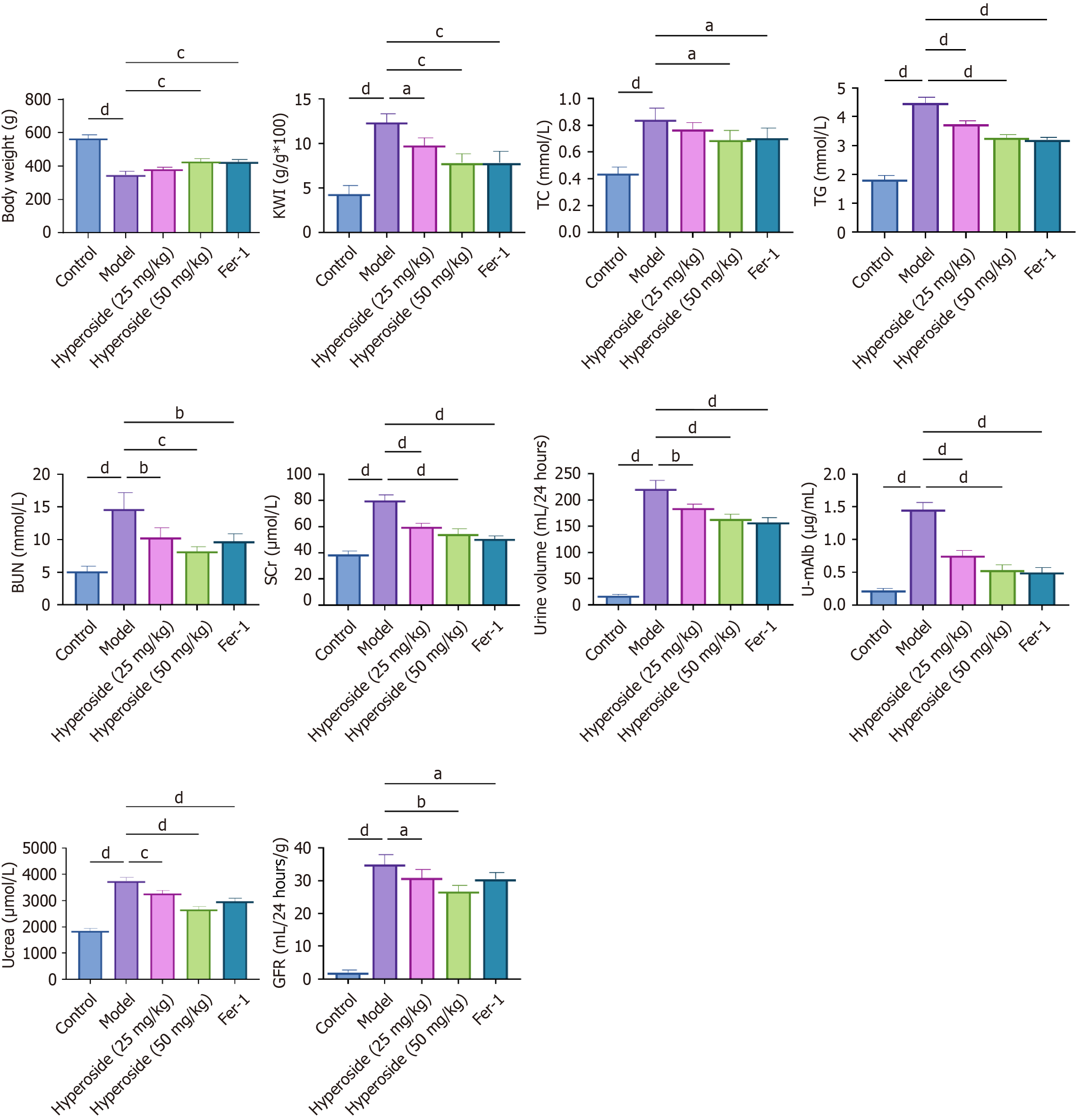
Figure 6 Hyperoside improves dyslipidemia and renal function in diabetic nephropathy rats.
aP < 0.05, bP < 0.01, cP < 0.001, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 6 rats per group. Body weight, kidney weight index, serum total cholesterol, triglycerides, serum creatinine, blood urea nitrogen, 24-hour urinary albumin excretion, and renal glutathione in the control, diabetic nephropathy (DN) model, DN + hyperoside (25 mg/kg or 50 mg/kg), and DN + ferrostatin-1 (1 mg/kg) groups. KWI: Kidney weight index; TC: Total cholesterol; TG: Triglycerides; Scr: Serum creatinine; BUN: Blood urea nitrogen; UAE: Urinary albumin excretion; Fer-1: Ferrostatin-1; U-mAlb: Urinary microalbumin; GFR: Glomerular filtration rate.
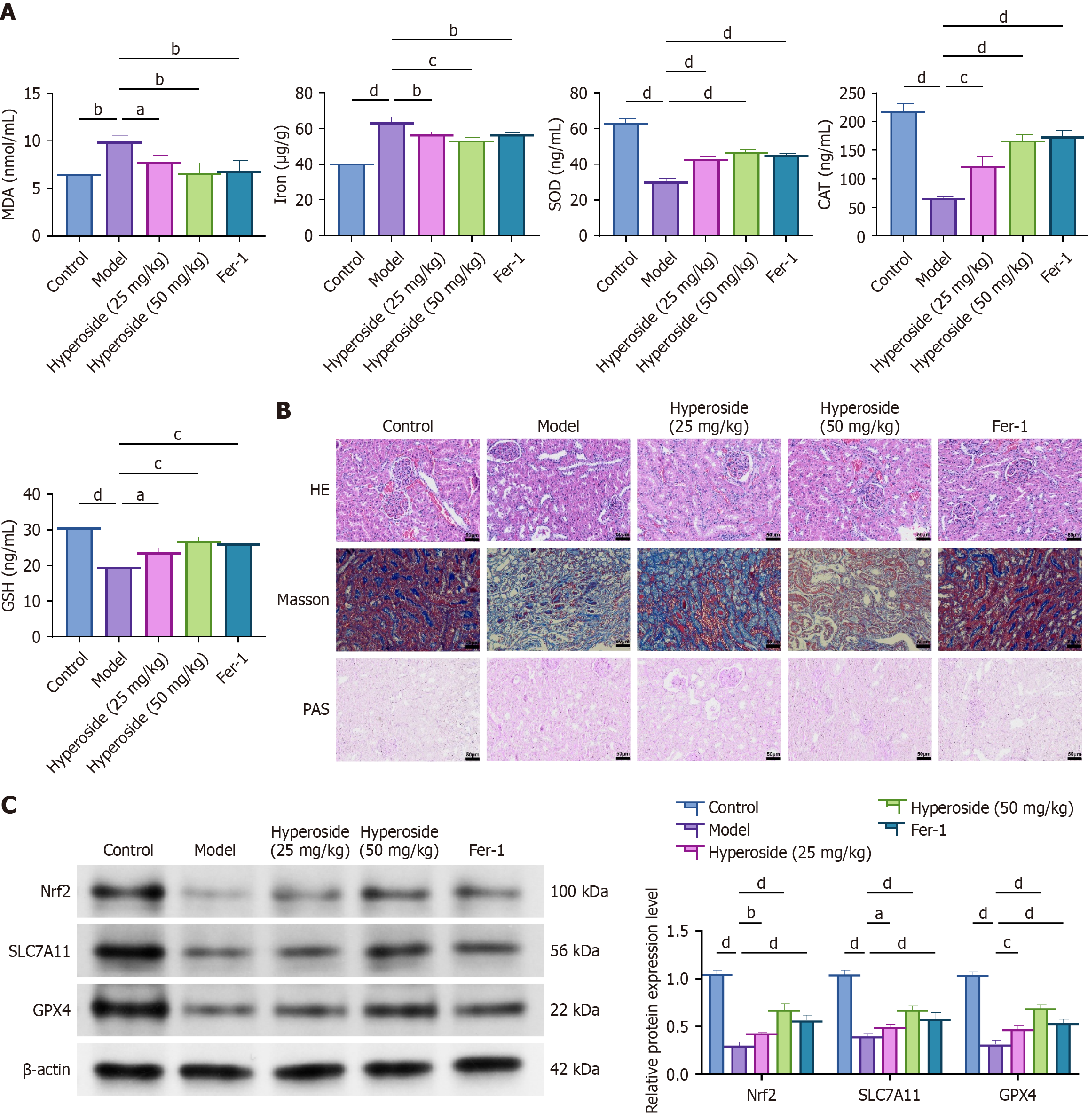
Figure 7 Hyperoside mitigates oxidative stress and glomerular structural damage in diabetic nephropathy rats.
aP < 0.05, bP < 0.01, cP < 0.001, dP < 0.001. One-way analysis of variance + Tukey’s post hoc test; two-sided. Data are expressed as the mean ± SD; n = 6 rats per group. A: Renal malondialdehyde, Fe, superoxide dismutase, catalase, and glutathione; B: Representative hematoxylin and eosin, Masson, and periodic acid-Schiff staining with semi-quantitative scores calculated by two blinded observers; C: Western blot analysis of renal Nrf2, SLC7A11, and GPX4 expression. Fer-1: Ferrostatin-1; MDA: Malondialdehyde; SOD: Superoxide dismutase; CAT: Catalase; GSH: Glutathione; PAS: Periodic acid-Schiff; HE: Hematoxylin and eosin.
- Citation: Liu C, Li Y, Zhang Y, Gao M, Yang SF. Hyperoside attenuates diabetic nephropathy by activating the Nrf2/SLC7A11/GPX4 axis to restrain ferroptosis. World J Diabetes 2026; 17(4): 114679
- URL: https://www.wjgnet.com/1948-9358/full/v17/i4/114679.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i4.114679