Published online May 16, 2026. doi: 10.4253/wjge.v18.i5.120628
Revised: April 2, 2026
Accepted: April 29, 2026
Published online: May 16, 2026
Processing time: 69 Days and 14.5 Hours
Current guidelines have not reached consensus on hemostatic management for large polyps (10-20 mm) removed by cold snare polypectomy (CSP). In light of the low complication rate of CSP and the potential hypercoagulability in colorectal cancer (CRC) patients, prophylactic hemoclip use after CSP for polyps ≥ 10 mm appears unlikely to reduce bleeding risk in patients with suspected colorectal cancer.
To evaluate the necessity of prophylactic hemoclip use after CSP for polyps ≥ 10 mm in patients with CRC.
This prospective cohort study with a historical control was conducted in Zhong
Before weighting, the non-clipping group showed significantly shorter procedural time (25.31 ± 16.75 minutes vs 29.31 ± 14.97 minutes; Z = 2.49, P = 0.01) and lower cost (4604.75 ± 2564.28 CNY vs 4915.55 ± 2327.80 CNY; Z = 2.06, P = 0.04) compared to the control group. After IPTW adjustment, no statistically significant differences were found in intraoperative or postoperative bleeding rates between the groups. Quantile regression indicated no significant difference in operative time across quantiles (all P > 0.05). However, a significant association was observed at the 0.25 quantile with cost (coefficient = -382.282, P = 0.035), suggesting non-use of clips was associated with reduced cost in the lower-cost subgroup.
In this cohort, prophylactic hemoclip use does not significantly alter bleeding risk or procedural time for CSP of polyps ≥ 10 mm, but its utilization strategy may impact cost containment.
Core Tip: The routine use of prophylactic hemoclips following cold snare polypectomy for colorectal polyps ≥ 10 mm does not reduce the risk of bleeding but is associated with increased operative time and cost. This study, utilizing advanced statistical methods (inverse probability of treatment weighting, Firth regression, etc.), suggests that a non-clipping strategy yields comparable safety outcomes while potentially enhancing procedural efficiency and cost-effectiveness, challenging a common clinical practice.
- Citation: Li LD, Chen ZH, Zhong YS, He DL, Zhu DX, Xu JM, Jing JC. Safety of opening wounds after cold resection of colorectal cancer patients’ intestinal polyps ten mm or larger. World J Gastrointest Endosc 2026; 18(5): 120628
- URL: https://www.wjgnet.com/1948-5190/full/v18/i5/120628.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i5.120628
Colorectal cancer (CRC) is the third most commonly diagnosed cancer and the second leading cause of cancer-related deaths worldwide[1]. Colorectal polyps are significant precancerous lesions, and endoscopic screening with polypectomy can reduce the incidence and mortality of CRC[2].
Among the adverse events associated with polypectomy, post-polypectomy bleeding (PPB) is the most common, primarily including intraoperative immediate bleeding and postoperative delayed bleeding. The latter may necessitate additional interventions, including repeat colonoscopy, blood transfusion, or other hemostatic methods[3]. Current guidelines have not reached a consensus on hemostatic management for large polyps (10-20 mm) removed by cold snare polypectomy (CSP). The European Society of Gastrointestinal Endoscopy recommends piecemeal CSP or cold endoscopic mucosal resection for sessile serrated lesions of all sizes without suspected dysplasia, with low-quality evidence suggesting that routine hemoclip placement may not be necessary for lesions < 20 mm and for lesions ≥ 20 mm in the left colon[4]. The updated United States Multi-Society Task Force suggests cold or hot snare polypectomy (with or without submucosal injection) for removing 10-19 mm sessile lesions, but states there is insufficient evidence to support whether prophylactic clip placement or electrocoagulation is needed for CSP of 10-19 mm non-pedunculated colorectal polyps[5]. Considering the low complication rate of CSP for polyps ≥ 10 mm (0%-1.1%)[6-8], and the hypercoagulable state in cancer patients[9], we propose the following hypothesis: Prophylactic hemoclip use after CSP for colorectal polyps ≥ 10 mm may not provide additional benefit in reducing bleeding in patients with suspected CRC. Furthermore, omitting hemoclips has significant practical implications: It can save procedural time and surgical costs, and avoid interference with the operative field and tumor localization-especially in cases where lesion marking with clips is required for subsequent radical resection.
This clinical study employed a prospective cohort design with a historical control group to investigate whether prophylactic hemoclip use is necessary after CSP for colorectal polyps ≥ 10 mm in patients with CRC. The aim is to further define the boundaries for hemoclip application, reduce surgical costs, and improve surgical precision.
This was a single-center, nonrandomized controlled clinical study with a parallel-group design conducted at Zhongshan Hospital, comparing patients who received prophylactic clipping vs those who did not. This study specifically targeted patients with suspected CRC undergoing preoperative colonoscopy, in whom synchronous colorectal polyps amenable to CSP were identified and resected. A total of 110 patients were prospectively enrolled between November 2022 and October 2023, all of whom were subsequently confirmed to have CRC. During this period, prophylactic clip closure was not routinely performed after CSP. A historical control cohort of 94 patients, treated between November 2021 and November 2022, in whom prophylactic clip closure was routinely applied, was retrospectively included for comparison. The approximate allocation ratio between groups was determined by the number of eligible cases available during the study periods. The study followed an exploratory framework without predefined superiority, equivalence, or non-inferiority hypotheses. No formal sample size calculation was performed, as all consecutively eligible patients during the study period were included, given the partially retrospective design. No important changes to study design, outcomes, or analyses were made after study commencement. No interim analyses or stopping guidelines were applicable. No missing data were observed. No additional subgroup or sensitivity analyses were performed. As this was a non-randomized obse
Inclusion criteria: (1) Patients undergoing colonoscopy for suspected CRC and subsequently confirmed to have colorectal adenocarcinoma; (2) No prior antitumor treatment before colonoscopy, including surgery, radiotherapy, chemotherapy, targeted therapy, or immunotherapy; (3) Presence of at least one colorectal polyp ≥ 10 mm suitable for CSP, pathologically confirmed as low-grade adenoma or sessile serrated lesion without high-grade dysplasia or submucosal invasion; (4) Lesions assessed by image-enhanced endoscopy (e.g., Narrow Band Imaging, i-Scan) and classified as Kudo pit pattern types I–IV and Paris classification types 0-Is or 0-IIa; (5) Availability of complete preoperative clinical data, with coagulation parameters within normal or controllable ranges; (6) Postoperative pathological confirmation of stage I CRC; and (7) Provision of written informed consent.
Exclusion criteria: (1) Pregnancy or lactation; (2) Known coagulation disorders, defined as platelet count < 50 × 109/L or international normalized ratio > 1.5; (3) Use of antiplatelet or anticoagulant agents (e.g., aspirin, clopidogrel, rivaroxaban) within 7 days prior to the procedure; (4) Polyps suspected of high-grade dysplasia or deep submucosal invasion (e.g., Kudo pit pattern V, Paris 0-IIc); (5) Active gastrointestinal bleeding at the time of colonoscopy; (6) Severe dysfunction of major organs (e.g., hepatic, cardiac, renal, or pulmonary insufficiency); and (7) Incomplete clinical data, loss to follow-up, or transfer to another institution.
This study complied with the requirements of the World Medical Association Declaration of Helsinki. The study protocol was approved by the institutional ethics committee (No. B2022-421R) and registered in the Chinese Clinical Trial Registry (ChiCTR2200065019).
All subjects underwent colorectal CSP performed by the same endoscopists with over 3 years of experience in both periods. Colonoscopy was advanced to the cecum or the site of tumor stenosis to ensure complete examination. Lesions were carefully evaluated using image-enhanced endoscopy (e.g., chromoendoscopy, Narrow Band Imaging, or i-Scan), and eligibility for CSP was confirmed based on morphological criteria (Kudo pit pattern classification (types I-IV) and Paris classification types 0-Is or 0-IIa), indicating no features suggestive of deep submucosal invasion. The snare was positioned to encompass the entire polyp along with a 1-2 mm margin of surrounding normal mucosa, and resection was performed without electrocautery. En bloc resection was preferred whenever feasible; for larger lesions requiring piecemeal resection, the procedure was repeated until complete removal was achieved. Resected specimens were retrieved via the suction channel and immediately fixed in formalin for histopathological examination. After resection, the mucosal defect was irrigated with saline and carefully inspected for immediate PPB. Active bleeding requiring endoscopic hemostasis was defined as persistent oozing lasting ≥ 30 seconds despite irrigation, spurting or pulsatile bleeding, or bleeding that obscured the endoscopic field and required intervention. In the experimental group, hemoclips were applied only in cases of active bleeding, whereas in the control group, prophylactic hemoclips were routinely applied to all resection sites. A consistent and standardized perioperative protocol was adopted.
Baseline patient characteristics included age, sex, bowel preparation quality, as well as CSP polyp characteristics, including number, size, and location. The number of CSP polyps was defined as the total number of colorectal polyps resected by CSP during a single procedure per patient. Polyp size was defined as the maximum diameter of each lesion as estimated endoscopically. Bowel preparation quality was assessed using the Boston Bowel Preparation Scale (BBPS) according to current guidelines and was dichotomized as adequate (BBPS score ≥ 6) or inadequate (BBPS score < 6). Polyp location was categorized as right colon (cecum to transverse colon), left colon (splenic flexure to sigmoid colon), or rectum.
Procedural parameters included procedural time and procedural cost. Procedural time was defined as the duration from the initial placement of the snare at the target lesion to the completion of all CSP resections, including any additional time required for hemostatic interventions. Procedural cost was defined as the total direct medical cost incurred during the endoscopic procedure, including disposable devices (e.g., snares and hemoclips), medications, and endoscopy-related service fees. All costs were recorded in Chinese Yuan (CNY).
The primary complication of interest was PPB, which was categorized as immediate PPB (IPPB) and delayed PPB (DPPB). IPPB was defined as bleeding occurring during the procedure that persisted for more than 60 seconds or required endoscopic hemostatic intervention. DPPB was defined as bleeding occurring within 30 days after polypectomy, characterized by the presence of clinical bleeding symptoms (e.g., hematochezia or melena) together with endoscopic confirmation of active bleeding at the post-polypectomy site. IPPB was identified intraoperatively under direct endoscopic visualization. DPPB was identified through scheduled follow-up at 7 and 30 days after the procedure via telephone interviews and review of medical records, and was confirmed by repeat endoscopy when clinically indicated.
The statistical analysis for this study was reviewed by an expert in biomedical statistics before manuscript submission. All analyses were performed using SPSS software (version 22.0; IBM Corp., Armonk, NY, United States) and R software (version 4.4.2; R Foundation for Statistical Computing, Vienna, Austria). Continuous data were assessed for normality and presented as median and interquartile range due to a skewed distribution. Categorical data are presented as n (%). Between-group comparisons for continuous variables were conducted using the Mann-Whitney U test, with effect size reported as the Hodges-Lehmann median difference and its 95%CI. For categorical variables, the χ2 test was used. For outcomes with expected frequencies < 5 (postoperative immediate and delayed bleeding), Fisher’s exact test was employed, with effect size reported as the risk difference and its 95%CI. A two-sided P value < 0.05 was considered statistically significant.
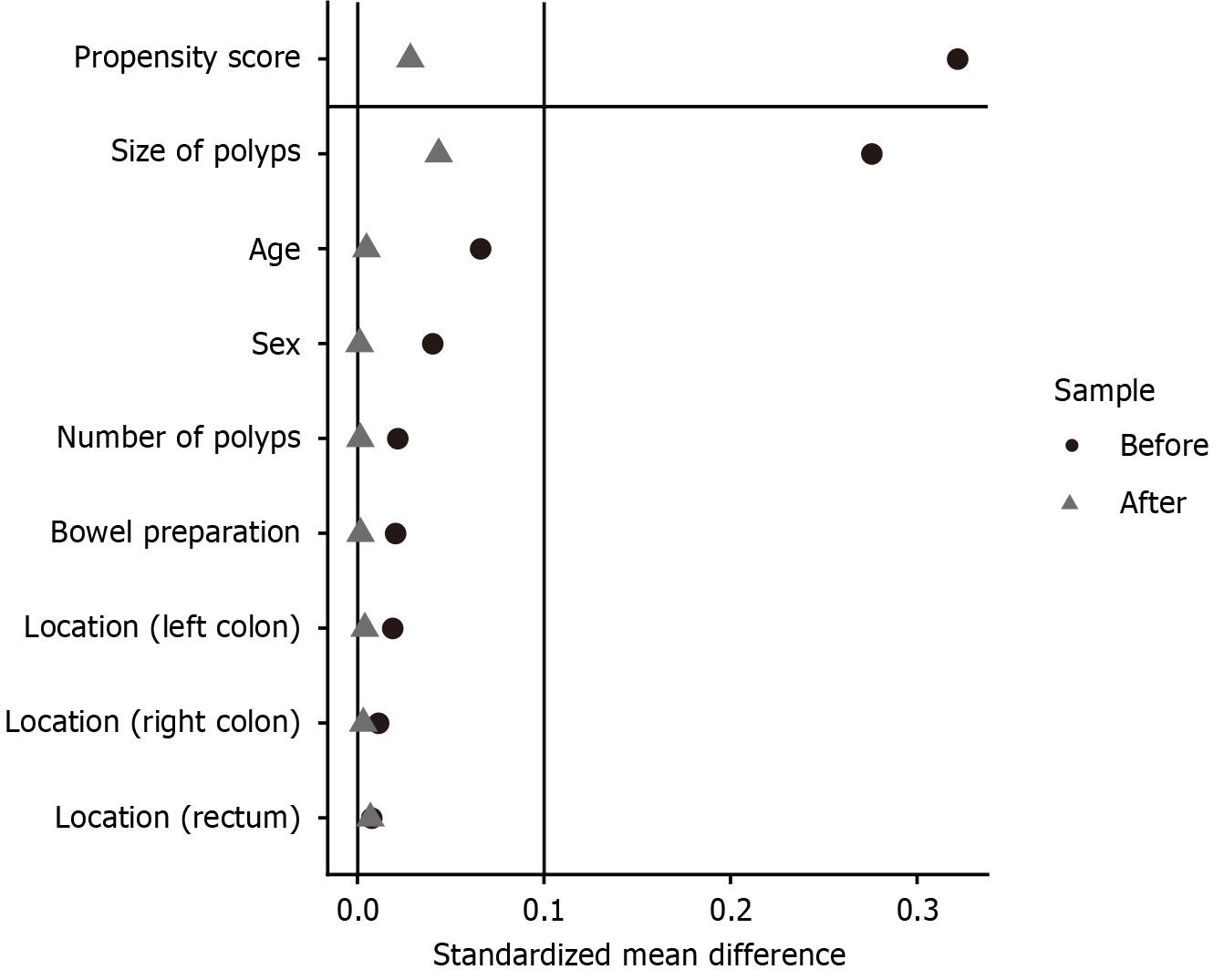
To control for potential confounding bias, inverse probability of treatment weighting (IPTW) was applied. A multivariable logistic regression model was fitted to estimate propensity scores, incorporating the following baseline covariates: Age, gender, polyp size, polyp location, polyp number, comorbidities, and other surgical characteristics. The propensity score represented the conditional probability of receiving the intervention. Stabilized weights were calculated as 1/propensity score for the intervention group and 1/(1-propensity score) for the control group. Covariate balance after IPTW was assessed using standardized mean differences (SMD), with an SMD < 0.10 indicating adequate balance.
Given the low incidence (< 10%) of bleeding events, Firth’s penalized likelihood logistic regression model was used to analyze intraoperative and postoperative bleeding outcomes within the IPTW-weighted cohort to mitigate small-sample bias. To assess the impact of the intervention on the entire distribution of operative time and cost, IPTW-weighted quantile regression analyses were performed at the 0.25, 0.50, and 0.75 quantiles. The standard errors for the quantile regression coefficients were estimated using bootstrap resampling with 1000 repetitions to ensure robustness.
After screening, 204 patients with 315 polyps removed by CSP were included. All participants had complete outcome data at the 30-day follow-up, and all interventions were delivered as intended. Before IPTW adjustment, there were no statistically significant differences between the two groups in terms of gender, age, bowel preparation adequacy, polyp number, or polyp size (P value > 0.05), as detailed in Table 1. After IPTW, imbalanced standardized mean differences were effectively corrected, as shown in Figure 1. All the pathological types of the polyps were confirmed as tubular adenoma with low-grade intraepithelial neoplasia. Standard post-polypectomy care, including observation for immediate bleeding and routine follow-up, was provided to all patients in both groups.
| Variable | Before IPTW | After IPTW | |||||||
| Experimental group | Control group | Z/χ² | P value | SMD | Experimental group | Control group | SMD | ||
| Age (year) | 66.218 ± 9.293 | 65.596 ± 9.591 | Z = | 0.359 | 0.066 | 65.948 ± 9.652 | 65.940 ± 9.551 | 0.001 | |
| Male (%) | 81 (73.636) | 73 (77.660) | χ² = 0.253 | 0.615 | 0.094 | 76.602 | 76.518 | -0.001 | |
| Bowel preparation (%) | 19 (17.273) | 21 (19.149) | χ² = 0.036 | 0.597 | 0.100 | 18.675 | 18.431 | 0.002 | |
| Number of CSP polyps | 1.555 ± 1.282 | 1.532 ± 0.758 | Z = 1.273 | 0.2032 | 0.021 | 1.514 ± 1.216 | 1.517 ± 0.732 | -0.003 | |
| Size of CSP polyps (cm) | 1.191 ± 0.381 | 1.102 ± 0.253 | Z = 1.825 | 0.068 | 0.276 | 1.162 ± 0.383 | 1.147 ± 0.312 | 0.045 | |
| Location of CSP polyps (%) | Left | 57 (51.818) | 48 (51.064) | χ² = 0.120 | 0.939 | 0.050 | 52.138 | 51.432 | -0.004 |
| Right | 34 (30.909) | 28 (29.787) | 30.251 | 30.590 | |||||
| Rectum | 19 (17.273) | 18 (19.149) | 17.611 | 17.978 | |||||
Compared to the control group, there were no statistically significant differences in intraoperative bleeding rate or postoperative bleeding rate (P > 0.05). However, the experimental group showed a significant reduction in both CSP procedural time (mean ± SD: Experimental group 25.31 ± 16.75 minutes vs control group 29.31 ± 14.97 minutes, Z = 2.49, P = 0.01) and procedural cost (mean ± SD: Experimental group 4604.75 ± 2564.28 CNY vs control group 4915.55 ± 2327.80 CNY, Z = 2.06, P = 0.04); the differences were statistically significant (P < 0.05). Details are shown in Tables 2 and 3. Notably, all bleeding events in this study were confined to the left colon subgroup, and no other serious complications, such as perforation occurred throughout the study.
| Variable | Experimental group | Control group | RD | 95%CI | P value |
| IPPB (%) | 0.909 | 1.064 | 0.15 | -4.03-4.97 | 1.000 |
| DPPB (%) | 0.909 | 0.000 | -0.91 | -4.99-3.05 | 0.461 |
| Variable | Experimental group | Control group | Z/χ² | Hodges-Lehmann median difference | 95%CI | P value |
| Procedural time | 25.309 ± 16.749 | 29.309 ± 14.973 | Z = 2.487 | 4.00 | 1.00-8.00 | 0.013 |
| Procedural cost | 4604.753 ± 2564.277 | 4915.547 ± 2327.799 | Z = 2.055 | 442.04 | 13.45-845.87 | 0.03992 |
Analysis of bleeding outcomes: Considering the low incidence of bleeding events, Firth’s penalized likelihood-corrected logistic regression analysis was used to compare intraoperative and postoperative bleeding between the two groups after IPTW. The Firth logistic regression showed no significant differences between groups in intraoperative bleeding [odds ratio (OR) = 0.761, 95%CI: 0.059-8.943, P = 0.814] or postoperative bleeding (OR = 2.454, 95%CI: 0.124-361.945, P = 0.562). Specific results are presented in Table 4.
| Variable | OR | 95%CI | P value |
| IPPB | 0.761 | 0.059-8.943 | 0.814 |
| DPPB | 2.454 | 0.124-361.945 | 0.562 |
Analysis of surgical outcomes: For the analysis of surgical outcomes, as the residuals of all continuous variables did not follow a normal distribution, quantile regression models were employed to estimate effects at the 0.25, 0.50, and 0.75 quantiles. The results showed no statistically significant differences in operative time at any of the three quantiles (all P > 0.05), suggesting that hemoclip use had a limited impact on operative time. Regarding procedural cost, at the 0.25 quantile, the regression coefficient was -382.282 (95%CI: -821.976 to -19.902, P = 0.035), indicating a statistically significant association between not using hemoclips and lower procedural costs in the patient subgroup with lower costs. No significant differences were found at the other quantiles. Detailed data are shown in Table 5.
| Variable | Quantile | Coefficient | Std. error | t value | 95%CI | P value |
| Procedural time | 0.25 | -3.662 | 2.146 | -1.706 | -8.000 to -0.000 | 0.088 |
| 0.5 | -5.000 | 3.257 | -1.535 | -10.000 to 2.520 | 0.125 | |
| 0.75 | -3.997 | 2.933 | -1.363 | -8.576 to 3.521 | 0.173 | |
| Procedural cost | 0.25 | -382.282 | 181.332 | -2.108 | -821.976 to -19.902 | 0.035 |
| 0.5 | -408.157 | 282.499 | -1.445 | -1164.250 to -19.521 | 0.149 | |
| 0.75 | -118.185 | 427.261 | -0.277 | -952.260 to 880.374 | 0.782 |
Colorectal polyps refer to protrusions from the mucosa into the intestinal lumen, primarily progressing to CRC via the adenoma-carcinoma sequence and the serrated neoplasia pathway[10]. These polyps can be detected and removed during endoscopy, thereby reducing the incidence of CRC[2]. For cancer patients, removing colorectal polyps can effectively prevent the vast majority of CRC deaths[11].
Bleeding is the most common adverse event following colorectal polypectomy. Current guidelines still lack sufficient evidence regarding whether hemoclips should be routinely used for CSP of 10-20 mm polyps. The incidence of bleeding after CSP is low. Existing evidence indicates that for 6-20 mm colorectal polyps, CSP has an equal or higher rate of intraoperative bleeding compared to hot snare polypectomy, but postoperative bleeding is relatively rare[7,12,13], suggesting a high safety profile. Some studies suggest that hemoclips offer limited benefit in large polypectomies. A study by Feagins et al[14] found that for polypectomy of polyps ≥ 10 mm, hemoclip use did not affect the proportion of significant DPPB events. A meta-analysis by Yu et al[15] investigating prophylactic clipping stratified by factors such as location found that clipping did not provide additional protective benefit against DPPB for colorectal polyps < 20 mm but was effective in reducing DPPB for polyps > 20 mm.
To further explore the boundaries of hemoclip use, this study focused specifically on patients with CRC, a population characterized by a distinct hypercoagulable state, fundamentally different from the general population[9]. Venous thromboembolism is common in cancer patients, with a risk approximately 15 times higher than in non-cancer patients[16]. It has been demonstrated that cancer co-opts wound healing mechanisms to promote its own growth, activating regulators of wound repair in the patient’s body[17], which may contribute to wound closure, potentially reducing PPB risk, and decreasing the necessity for hemoclips. This study found low rates of intraoperative and postoperative bleeding in CRC patients, consistent with the characteristics of this population.
Several studies have suggested that routine prophylactic clipping may increase procedural cost without clear clinical benefit. Decision-analytic models have demonstrated that routine prophylactic clipping is not cost-effective in average-risk patients and is highly sensitive to clip cost and bleeding risk[18,19]. A study by Shah and colleagues[20] suggested that prophylactic closure of right colon polypectomy sites would be cost-saving only if the clip cost was less than $10.
Hemoclips placed near tumors may also impact postoperative recovery after radical resection for CRC. The wound environment and high cytokine concentrations in cancer patients may theoretically facilitate cancer cell migration from other sites to the wound area, potentially leading to wound dehiscence[21]. Stanczyk et al[22] found that poor scar tissue formation, cancer cell migration to the wound, and inadequate inflammatory cell proliferation can impair the healing process. Single-cell wound healing relies on cell distortion and contraction, and a high-tension environment may increase the difficulty of this process. Hemoclips may also potentially affect local blood supply to the wound, slowing cellular proliferation and repair, and increasing the risk of anastomotic bleeding. In clinical practice, besides prophylaxis, hemoclips are also used for tumor localization post-resection. However, placing metal clips during colonic polypectomy may interfere with the intraoperative field of view, prolong procedure time, and even affect local healing processes.
This study has several limitations. First, given the sequential nature of the study design, a potential learning-curve effect cannot be excluded, which may have contributed to the shorter procedure time observed in the prospective group. Although efforts were made to maintain operator consistency, increasing familiarity with the technique over time might have influenced procedural efficiency. Second, this was a single-center study with a relatively limited sample size, which may affect the generalizability of the findings. Finally, the applicability of our findings should be interpreted with caution, and further large-scale, multicenter studies are needed to validate these results.
In summary, in CRC patients undergoing preoperative colonoscopy prior to radical resection, the removal of colon polyps ≥ 10 mm did not demonstrate a significant benefit in reducing bleeding incidence, while it was associated with increased procedural time and costs. As the first study to evaluate CRC patients as an independent cohort, this work provides preliminary evidence to refine the indications for hemoclip use in 10-19 mm polyps beyond current guideline frameworks, particularly considering the potential interference of clips with tumor localization. Further large-scale, multicenter studies are warranted to validate these findings and better define the optimal use of hemoclips in this setting.
The authors thank Dr. Zhi-Peng Qi for helpful discussions and guidance.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69712] [Article Influence: 13942.4] [Reference Citation Analysis (45)] |
| 2. | Brenner H, Heisser T, Cardoso R, Hoffmeister M. Reduction in colorectal cancer incidence by screening endoscopy. Nat Rev Gastroenterol Hepatol. 2024;21:125-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 54] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 3. | O’Mara MA, Emanuel PG, Tabibzadeh A, Duve RJ, Galati JS, Laynor G, Gross S, Gross SA. The Use of Clips to Prevent Post-Polypectomy Bleeding: A Clinical Review. J Clin Gastroenterol. 2024;58:739-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (2)] |
| 4. | Ferlitsch M, Hassan C, Bisschops R, Bhandari P, Dinis-Ribeiro M, Risio M, Paspatis GA, Moss A, Libânio D, Lorenzo-Zúñiga V, Voiosu AM, Rutter MD, Pellisé M, Moons LMG, Probst A, Awadie H, Amato A, Takeuchi Y, Repici A, Rahmi G, Koecklin HU, Albéniz E, Rockenbauer LM, Waldmann E, Messmann H, Triantafyllou K, Jover R, Gralnek IM, Dekker E, Bourke MJ. Colorectal polypectomy and endoscopic mucosal resection: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2024. Endoscopy. 2024;56:516-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 175] [Article Influence: 87.5] [Reference Citation Analysis (2)] |
| 5. | Kaltenbach T, Anderson JC, Burke CA, Dominitz JA, Gupta S, Lieberman D, Robertson DJ, Shaukat A, Syngal S, Rex DK. Endoscopic Removal of Colorectal Lesions-Recommendations by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2020;158:1095-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 234] [Article Influence: 39.0] [Reference Citation Analysis (0)] |
| 6. | Kimoto Y, Sakai E, Inamoto R, Kurebayashi M, Takayanagi S, Hirata T, Suzuki Y, Ishii R, Konishi T, Kanda K, Negishi R, Takita M, Ono K, Minato Y, Muramoto T, Ohata K. Safety and Efficacy of Cold Snare Polypectomy Without Submucosal Injection for Large Sessile Serrated Lesions: A Prospective Study. Clin Gastroenterol Hepatol. 2022;20:e132-e138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (2)] |
| 7. | Mangira D, Raftopoulos S, Vogrin S, Hartley I, Mack A, Gazelakis K, Nalankilli K, Trinh A, Metz AJ, Appleyard M, Grimpen F, Elliott T, Brown G, Moss A. Effectiveness and safety of cold snare polypectomy and cold endoscopic mucosal resection for nonpedunculated colorectal polyps of 10-19 mm: a multicenter observational cohort study. Endoscopy. 2023;55:627-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 8. | Thoguluva Chandrasekar V, Spadaccini M, Aziz M, Maselli R, Hassan S, Fuccio L, Duvvuri A, Frazzoni L, Desai M, Fugazza A, Jegadeesan R, Colombo M, Dasari CS, Hassan C, Sharma P, Repici A. Cold snare endoscopic resection of nonpedunculated colorectal polyps larger than 10 mm: a systematic review and pooled-analysis. Gastrointest Endosc. 2019;89:929-936.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 94] [Article Influence: 13.4] [Reference Citation Analysis (1)] |
| 9. | Nasser NJ, Fox J, Agbarya A. Potential Mechanisms of Cancer-Related Hypercoagulability. Cancers (Basel). 2020;12:566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 10. | Keum N, Giovannucci E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat Rev Gastroenterol Hepatol. 2019;16:713-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2156] [Cited by in RCA: 1842] [Article Influence: 263.1] [Reference Citation Analysis (4)] |
| 11. | Gupta S, Sidhu M, Shahidi N, Vosko S, McKay O, Bahin FF, Zahid S, Whitfield A, Byth K, Brown G, Lee EYT, Williams SJ, Burgess NG, Bourke MJ. Effect of prophylactic endoscopic clip placement on clinically significant post-endoscopic mucosal resection bleeding in the right colon: a single-centre, randomised controlled trial. Lancet Gastroenterol Hepatol. 2022;7:152-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 12. | Ito T, Takahashi K, Tanabe H, Sato K, Goto M, Sato T, Tanaka K, Utsumi T, Fujinaga A, Kawamoto T, Yanagawa N, Moriichi K, Fujiya M, Okumura T. Safety and efficacy of cold snare polypectomy for small colorectal polyps: A prospective randomized control trial and one-year follow-up study. Medicine (Baltimore). 2021;100:e26296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Winston K, Maulahela H, Raharjo DE, Tjoa K, Jonlean R. A Comparative Analysis of the Efficacy and Safety of Hot Snare Polypectomy and Cold Snare Polypectomy for Removing Small Colorectal Polyps: A Systematic Review and Meta-Analysis. Cureus. 2023;15:e38713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Feagins LA, Smith AD, Kim D, Halai A, Duttala S, Chebaa B, Lunsford T, Vizuete J, Mara M, Mascarenhas R, Meghani R, Kundrotas L, Dunbar KB, Cipher DJ, Harford WV, Spechler SJ. Efficacy of Prophylactic Hemoclips in Prevention of Delayed Post-Polypectomy Bleeding in Patients With Large Colonic Polyps. Gastroenterology. 2019;157:967-976.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 15. | Yu Z, Albéniz E, Hu J, Li P, Li Q, Hu Y, Chen J, Wang J. Prevention of delayed post-polypectomy bleeding by prophylactic clipping after endoscopic colorectal polypectomy: a meta-analysis. Int J Colorectal Dis. 2022;37:2229-2236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 16. | Khan F, Tritschler T, Marx CE, Lanting V, Rochwerg B, Tran A, Fernando SM, Lorenzetti DL, Wunsch H, Holodinsky J, Fiest K, Stelfox HT, Delluc A, Fergusson DA, Le Gal G, Wells PS, Wang TF, Sanfilippo K, van Es N, Schrag D, Connors JM, Carrier M. Predictors of recurrent venous thromboembolism and bleeding in patients with cancer: a meta-analysis. Eur Heart J. 2026;47:1917-1929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Cangkrama M, Wietecha M, Werner S. Wound Repair, Scar Formation, and Cancer: Converging on Activin. Trends Mol Med. 2020;26:1107-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 18. | Sonaiya S, Patel R, Parikh C, Chun M, Narwan A, Yagnik K, Patel P, Confer B, Khara H, Mohan BP. Prophylactic clipping versus no clipping after endoscopic mucosal resection of large nonpedunculated colon polyps: a cost-effectiveness analysis. Gastrointest Endosc. 2026;103:494-504.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 19. | Parikh ND, Zanocco K, Keswani RN, Gawron AJ. A cost-efficacy decision analysis of prophylactic clip placement after endoscopic removal of large polyps. Clin Gastroenterol Hepatol. 2013;11:1319-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 20. | Shah ED, Pohl H, Rex DK, Wallace MB, Crockett SD, Morales SJ, Feagins LA, Law R. Valuing innovative endoscopic techniques: prophylactic clip closure after endoscopic resection of large colon polyps. Gastrointest Endosc. 2020;91:1353-1360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 21. | Witte MB, Barbul A. General principles of wound healing. Surg Clin North Am. 1997;77:509-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 579] [Cited by in RCA: 454] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 22. | Stanczyk M, Olszewski W, Gewartowska M, Maruszynski M. Lack of healing reaction in the cancer wound. Pol J Pathol. 2014;65:267-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |