Published online Mar 16, 2026. doi: 10.4253/wjge.v18.i3.117820
Revised: January 5, 2026
Accepted: February 3, 2026
Published online: March 16, 2026
Processing time: 86 Days and 17 Hours
Traditional gastrointestinal endoscopy (GIE) apprenticeship training raises concerns regarding patient safety, prompting growing adoption of simulation-based training approaches. Virtual reality (VR) simulators provide a risk-free, learner-centered environment in which trainees can develop and refine endoscopic skills prior to clinical practice. Although VR-based training has been increasingly implemented worldwide, its efficacy relative to conventional training methods has yet to be conclusively established.
To evaluate the effectiveness and outcomes of VR-based training in GIE through a systematic review of interventional studies.
This systematic review followed PRISMA 2020 guidelines. PubMed, ScienceDirect, and Google Scholar were searched through June 2025 using Boolean combinations of terms related to VR, simulation, and GIE. Eligible studies were interventional trials evaluating VR-based training that reported changes in endoscopist competency. Procedures included esophagogastroduodenoscopy, colonoscopy, endoscopic retrograde cholangiopancreatography, and endoscopic ultrasound.
VR-based training consistently improved trainee competency, procedural independence, and patient safety compared with baseline performance, with outcomes comparable to conventional training. In esophagogastroduodenoscopy, VR simulation significantly increased independent procedure completion rates (odds ratio: 65.7; 95% confidence interval: 20.1-214.4) and achieved similar procedure times to traditional training (10.5 minutes vs 12.4 minutes). In endoscopic retrograde cholangiopancreatography, VR effectively differentiated skill levels, with experts outperforming novices in key metrics such as papilla visualization and cannulation time (P < 0.05). Endoscopic ultrasound competency improved when VR was combined with clinical training (64.53 ± 4.91 vs 60.09 ± 5.49; P = 0.028). Colonoscopy studies reported higher pass rates and improved objective skill scores in VR-trained groups (100% vs 88.9% pass rate in proficiency-based VR training).
These findings support integrating VR simulation as an adjunct to conventional endoscopy training and un
Core Tip: This article synthesizes evidence from 30 interventional studies involving 901 clinicians to evaluate the efficacy of virtual reality (VR) training in gastrointestinal endoscopy. VR simulation improves trainee competency, procedural independence, and patient safety across key procedures, including esophagogastroduodenoscopy, colonoscopy, endoscopic retrograde cholangiopancreatography, and endoscopic ultrasound. Performance gains with VR were comparable to those achieved with conventional training, supporting its role as an adjunct in endoscopic education. Heterogeneity in study design limited meta-analysis, underscoring the need for standardized VR training protocols and further research to assess long-term clinical outcomes.
- Citation: Gadour E, Miutescu B, Nica C, Taheri E, Al Saeed Z, Raheem B, Facciorusso A, AlQahtani MS. Virtual reality training for gastrointestinal endoscopy: A systematic review of efficacy and outcomes. World J Gastrointest Endosc 2026; 18(3): 117820
- URL: https://www.wjgnet.com/1948-5190/full/v18/i3/117820.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i3.117820
Gastrointestinal endoscopy (GIE) is widely used for the diagnosis and management of gastrointestinal (GI) disorders[1]. Given its central role in clinical practice, there is an increasing demand for highly skilled endoscopists. Traditionally, endoscopy training has relied on an apprenticeship model involving supervised procedures performed on patients. However, heightened awareness of patient safety has driven greater adoption of simulation-based training. Simulation allows novice endoscopists to practice and refine technical skills in a learner-centered, risk-free environment[1,2]. This approach reduces the risk of patient harm and enhances safety by ensuring that trainees achieve proficiency before clinical practice[2]. Accordingly, effective implementation of simulation training and successful transfer of acquired skills to real-world clinical settings are critical objectives of modern endoscopic education[2,3].
The first mechanical simulators for endoscopy training were introduced in the 20th century. Over time, these tools have evolved into sophisticated computer-based systems capable of providing real-time feedback to trainees[4]. These advanced platforms generate a virtual reality (VR) environment in which endoscopists can visualize procedural anatomy and receive immediate feedback on key technical parameters, such as applied force and loop formation during endo
VR has been used in endoscopy training for more than a decade, with its adoption expanding worldwide. However, early evidence regarding its effectiveness has been mixed[2]. While available data support VR as a supplement to conventional training, there is no consensus on its ability to replace traditional apprenticeship-based education, a conclusion echoed by prior systematic reviews and meta-analyses[5]. Accordingly, we conducted an updated systematic review to synthesize emerging evidence on the role of VR in training GI endoscopists.
This article was conducted in accordance with the PRISMA 2020 guidelines[6]. The review protocol was registered in the International Prospective Register of Systematic Reviews under registration number CRD420251071274.
A comprehensive literature search was conducted independently by two authors to identify relevant studies published up to June 2025. PubMed, Web of Science, and Google Scholar were searched using database-specific strategies. Boolean operators were used to combine keywords as follows: (“simulation” OR “simulator” OR “simulation based”) AND (“virtual reality” OR “augmented Reality” OR “VR”) AND (“gastrointestinal” OR “GI”) AND (“endoscopy” OR “upper endoscopy” OR “colonoscopy” OR “sigmoidoscopy” OR “endoscopic retrograde pancreatography” OR “endoscopic ultrasound”). Reference lists of included studies were manually screened to identify additional eligible articles.
Retrieved studies were assessed against predefined eligibility criteria prior to inclusion. Studies were included if they: (1) Evaluated VR-based training in GI endoscopic procedures; (2) Were interventional in design; (3) Reported changes in endoscopist competency following VR training; and (4) Were primary studies, including randomized clinical trials and nonrandomized interventional studies.
Studies were excluded if they: (1) Were published in languages other than English; (2) Did not incorporate VR as an endoscopy training intervention; (3) Did not involve GIE as the primary procedure; or (4) Failed to report relevant outcome measures.
Data extraction was independently performed by two reviewers. Any discrepancies were resolved through discussion until consensus was reached. In accordance with PRISMA guidelines, records were screened in multiple stages, beginning with title and abstract review to exclude irrelevant studies, followed by full-text assessment for eligibility. All studies meeting the inclusion criteria were retained for data extraction. Extracted data included author information, study setting, sample size, VR technology used, type of GIE, trainee experience level, intervention characteristics, and reported out
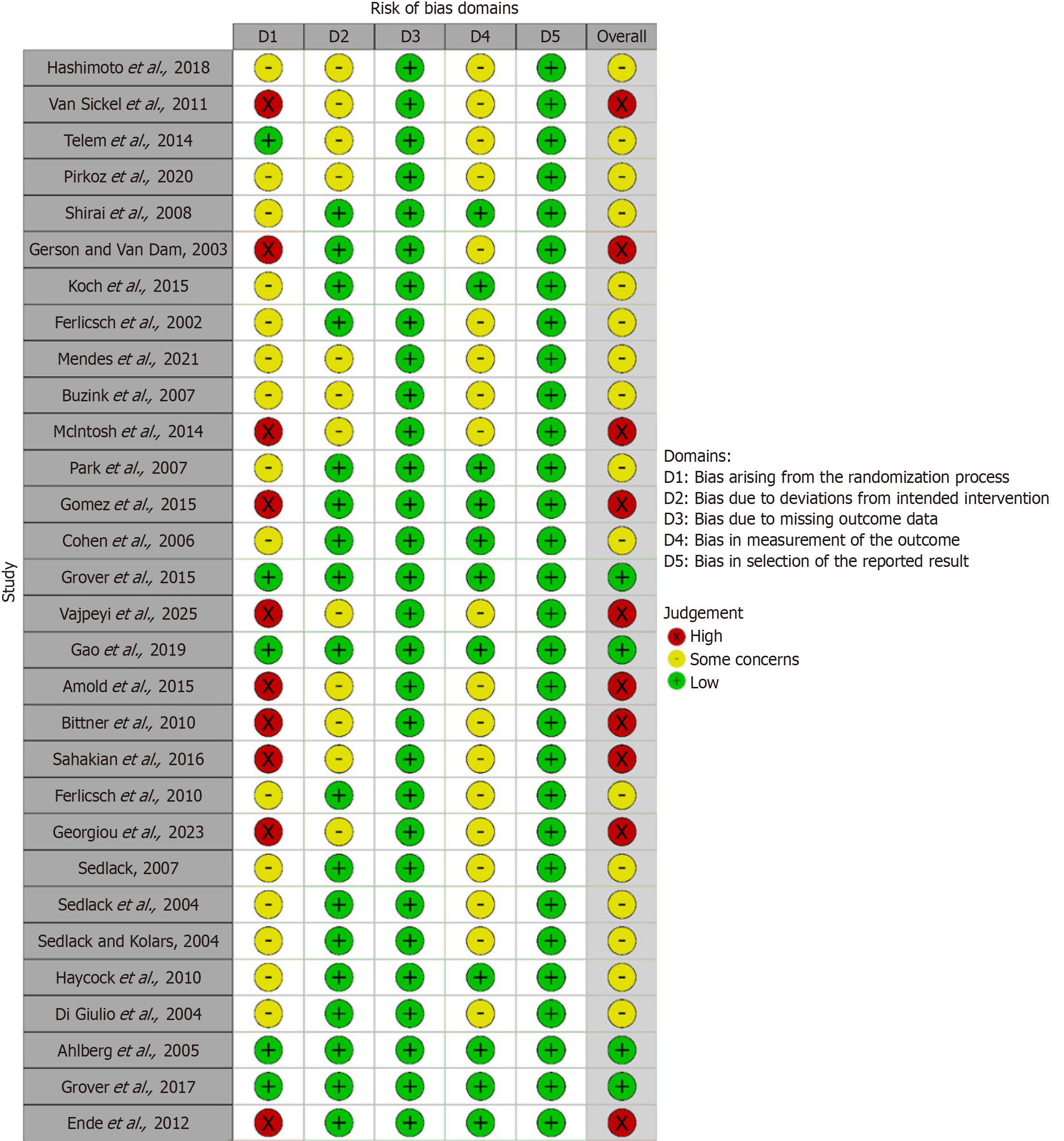
Risk of bias in the included trials was assessed using the Risk of Bias 2 tool. This tool evaluates bias across five domains: The randomization process, deviations from intended interventions, blinding, outcome measurement, and selective outcome reporting. An overall risk-of-bias judgment was derived from domain-level assessments. Studies were classified as high risk of bias if one or more domains were rated as high risk, as having some concerns if one or more domains raised concerns, and as low risk of bias only if all domains were rated as low risk.
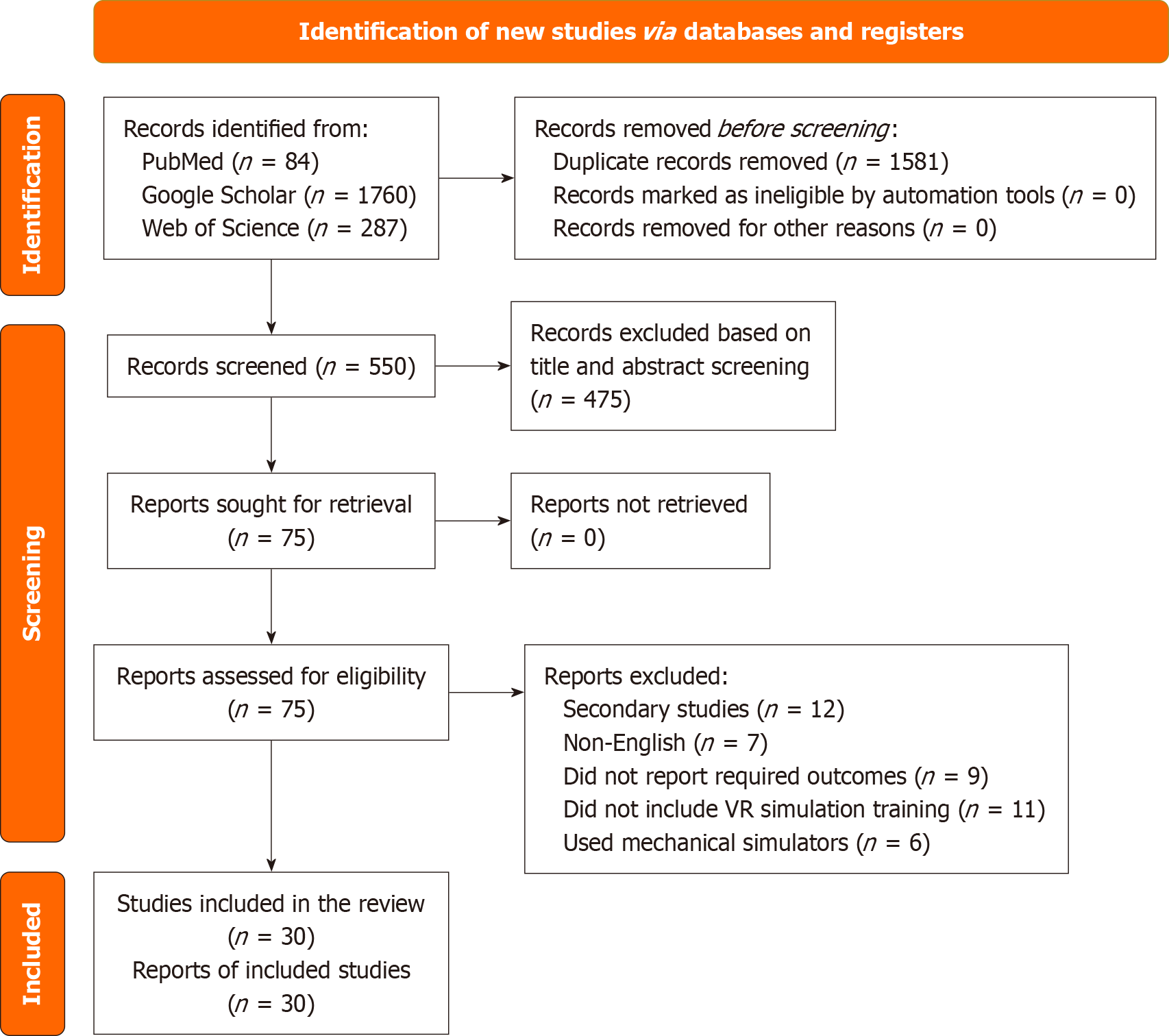
A total of 2131 articles were identified across the databases. After removal of 1581 duplicates, 550 articles were screened for relevance, of which 475 were excluded. Seventy-five full-text articles were assessed for eligibility, and 30 studies met the inclusion criteria and were included in the review. Excluded studies comprised 12 secondary studies, 7 non-English publications, 9 studies that did not report relevant outcomes, 11 that did not include simulation-based training, and 6 that used mechanical simulators. The study selection process is summarized in the PRISMA flow diagram (Figure 1).
This article included 30 trials conducted across diverse geographic regions, including North America, Europe, South America, and Asia. All studies utilized VR-based simulators, such as GI mentor I and II, ENDO VR, AccuTouch colo
| Ref. | Setting | Study design | Sample size | Level of training |
| Piskorz et al[4], 2020 | Argentina | Pre- and post-test trial | 126 | PGY1 gastroenterology residents |
| Van Sickle et al[5], 2011 | United States | Pre- and post-test trial | 41 | PGY1-4 general surgery residents |
| Telem et al[6], 2014 | United States | Pre- and post-test trial | 9 | Novice surgical interns |
| Shirai et al[7], 2008 | Japan | RCT | 20 | PGY1 and PGY2 residents in the gastroenterology and hepatology department |
| Gerson and Van Dam[8], 2003 | United States | RCT | 16 | Internal medicine residents |
| Koch et al[9], 2015 | Netherlands | RCT | 18 | Residents starting their gastroenterology training |
| Ferlitsch et al[10], 2002 | Austria | RCT | 24 | Medical residents and experienced endoscopists |
| Silva Mendes et al[11], 2022 | Portugal | Pre- and post-test trial | 23 | First-year gastroenterology residents |
| Buzink et al[12], 2007 | Netherlands | Pre- and post-test trial | 30 | Medical interns or residents in training |
| McIntosh et al[13], 2014 | United Kingdom | RCT | 18 | Residents PGY2 to PGY4 |
| Park et al[14], 2007 | Canada | RCT | 24 | Residents in internal medicine and gastroenterology in PGY1 to PGY3 |
| Gomez et al[15], 2015 | United States | RCT | 27 | Surgical PGY1 residents |
| Cohen et al[16], 2006 | United States | RCT | 45 | Gastroenterology fellows |
| Grover et al[17], 2015 | United States | RCT | 34 | Residents in internal medicine, gastroenterology, and general surgery |
| Vajpeyi et al[18], 2025 | Canada | Pre-test post-test trial | 33 | Eight expert endoscopists and 25 novice endoscopists |
| Gao et al[19], 2019 | China | RCT | 27 | Clinical doctors |
| Arnold et al[20], 2015 | Denmark | Pre-test post-test trial | 37 | Senior doctors, medical doctors, and medical students |
| Bittner et al[21], 2010 | United States | Pre-test post-test trial | 12 | Six were novice endoscopists, and 6 were expert endoscopists |
| Sahakian et al[22], 2016 | United States | NRCT | 10 | Four gastroenterology faculty and six fellows |
| Ferlitsch et al[23], 2010 | Austria | RCT | 28 | 28 residents |
| Georgiou et al[24], 2023 | Bulgaria | NRCT | 37 | 7 expert endoscopists and 31 novices |
| Sedlack[25], 2007 | United States | RCT | 29 | Gastroenterology fellows with different levels of experience |
| Sedlack et al[26], 2004 | United States | RCT | 38 | PGY2 internal medicine residents |
| Sedlack and Kolars[27], 2004 | United States | RCT | 8 | First-year GI fellows |
| Haycock et al[28], 2010 | Netherlands, Italy, and the United Kingdom | RCT | 36 | Novice colonoscopists |
| Di Giulio et al[29], 2004 | Italy | RCT | 22 | Gastroenterology fellows |
| Ahlberg et al[30], 2005 | Sweden | RCT | 22 | 12 residents and 10 surgeons |
| Grover et al[31], 2017 | Canada | RCT | 37 | Novice endoscopists |
| Ende et al[32], 2012 | Germany | RCT | 28 | Medical and surgical residents |
| Ref. | Type of virtual training | Endoscopic procedure | Description of procedure | Number of sessions | Outcome assessment |
| Van Sickle et al[5], 2011 | GI mentor II | Colonoscopy | Basic colonoscopy with no anatomical or pathological variation | The trainees needed to achieve proficiency in two tasks before proceeding to another level of training in a maximum of 10 trials | Performance data, training data, and subjective self-evaluation data |
| Telem et al[6], 2014 | GI mentor | Colonoscopy | 12-hour GI mentor independent training | 2-hour sessions each week for 6 weeks | Performance based on the GAGES-C criteria |
| Piskorz et al[4], 2020 | GI mentor II | Gastroscopy and colonoscopy | 8-hour work day | - | Performance data |
| Shirai et al[7], 2008 | GI mentor II | EGD | 1-hour training sessions | Five sessions over two weeks | The performance time taken by each trainee group to reach the various stages of EGD |
| Gerson and Van Dam[8], 2003 | - | Sigmoidoscopy | The virtual reality group had unlimited time for 2 weeks. The traditional bedside group had only ten sessions to practice | - | Performance of the residents, including the number of procedures completed independently, those requiring assistance, flexure recognition, and retroflexion |
| Koch et al[9], 2015 | GI mentor II | Colonoscopy | One session included at least five consecutive colonoscopies | They included 10-20 sessions distributed over 5 weeks to 10 weeks | The insertion depth and the rate of cecal intubation during patient-based assessments |
| Ferlitsch et al[10], 2002 | GI mentor | Gastroscopy and colonoscopy | 2-hour sessions | 5 sessions a week, for 3 weeks | Performance of the trainees in the endobubble, endobasket, and virtual endoscopy cases |
| Silva Mendes et al[11], 2022 | GI mentor II | EGD and colonoscopy | 5-hour sessions | One session each week for about 3-4 weeks | Cognitive evaluation of the resident’s performance, endoscopy skill assessment in both EGD and colonoscopy |
| Buzink et al[12], 2007 | GI mentor II | Colonoscopy | Not described | 1 session per day for five consecutive days | Performance time |
| McIntosh et al[13], 2014 | GI mentor II | Colonoscopy | The training was to be carried out for a total of between 10 hours and 20 hours | The sessions were distributed over four weeks | Number of proctor assists required per colonoscopy, the procedure time, and the rate of successful cecal intubation |
| Park et al[14], 2007 | AccuTouch colonoscopy simulator | Colonoscopy | The training period was for a period of 2 hours to 3 hours | Not specified | The performance data includes caecum intubation, lack of flaws, and global ratings as assessed by an independent evaluator |
| Gomez et al[15], 2015 | GI mentor II and Kyoto Kagaku colonoscopy | Colonoscopy | - | The total training was for 3 weeks | Performance time and the evaluator rating using the GAGES scores |
| Cohen et al[16], 2006 | GI mentor | Colonoscopy | 2-hour session on the simulators | Five sessions over 8 weeks | Objective and subjective competence of the fellows, and the discomfort scores |
| Grover et al[17], 2015 | EndoVR virtual reality endoscopy simulator | Colonoscopy | 8 hours of supervised endoscopy sessions | Not reported | JAG DOPS scale scores and performance data |
| Vajpeyi et al[18], 2025 | M40 colonoscope training simulator | Colonoscopy | - | - | Performance data, including performance time |
| Gao et al[19], 2019 | GI mentor II | EUS | - | All the training was carried out over 8 weeks | Test scores after completion of training |
| Arnold et al[20], 2015 | GI mentor II | ERCP | - | - | Motion movement as demonstrated by the different groups of endoscopists according to the level of expertise |
| Bittner et al[21], 2010 | GI mentor II | ERCP | They performed supervised 30-minute sessions | Each participant completed three cases | Validity of the VR tool as a simulator for ERCP |
| Sahakian et al[22], 2016 | Endo VR | ERCP | The participant had unlimited time to complete cases | Each participant completed 6 cases | Procedure time and patient discomfort |
| Ferlitsch et al[23], 2010 | GI mentor | Upper GI endoscopy | 2-hour sessions | The participants completed training sessions for as long as 20 hours | Patient discomfort and pain |
| Georgiou et al[24], 2023 | GI mentor | ERCP | 3 procedures | - | Performance time |
| Sedlack[25], 2007 | GI Mentor | EGD | 15-minute introduction session, then the participant completed the training for 6 hours | The total simulation training included up to 20 simulated cases | Performance data, including procedure time and endoscopy skills |
| Sedlack et al[26], 2004 | AccuTouch VR | Sigmoidoscopy | The participants had simulation training for a total of 3 hours | The total simulation training included 8-10 simulated cases | Subjective and objective assessment of the resident's sigmoidoscopy competency |
| Sedlack and Kolars[27], 2004 | AccuTouch VR | Colonoscopy | The participants received simulation training for about 6 hours | The simulation involved 20-25 cases spread over two days | Objective assessment of the fellows’ performance, including performance time |
| Haycock et al[28], 2010 | Endo TS-1 Olympus | Colonoscopy | The participants received four half-day sessions, leading to 16 hours of simulation training | The sessions were distributed over four days | Colonoscopy proficiency as assessed using the JAG DOPS and Global score |
| Di Giulio et al[29], 2004 | GI mentor | EGD | The participants completed 10 hours of simulation training | The simulation involved 10 cases to be covered in 3 sessions to 5 sessions | Procedure time, rate of successful intubation, and the reasons for failed or assisted intubations |
| Ahlberg et al[30], 2005 | AccuTouch VR | Colonoscopy | - | - | Procedure time and reason for procedure termination, if applicable |
| Grover et al[31], 2017 | Accutouch VR | Colonoscopy | The participants received 4 hours of interactive didactic sessions and 6 hours of supervised simulator training | - | The performance of endoscopists in the two different topics was assessed by the JAG DOPS assessment tool |
| Ende et al[32], 2012 | GI mentor, plastic phantom, and compactEASIE | EGD | The simulation training consisted of 2-hour sessions for a total of 10 hours | Five training sessions distributed over five weeks | Performance data including procedure time and the number of instances where residents required assistance |
| Hashimoto et al[33], 2018 | GI mentor | Colonoscopy | Each participant had to complete a repetitive task in colonoscopy for the FES program | Ten repetitions for each task in the repetitions group. The proficiency group repeated each task until they met the proficiency benchmark in two consecutive tasks | Pass rate and FES scores of each of the participants |
Figure 2 summarizes the risk of bias across the included studies. Only four studies were judged to have a low risk of bias. Among the remaining studies, concerns most commonly arose from limitations in the randomization process and lack of blinding. Notably, 10 studies were rated as having a high risk of bias due to absence of randomization.
Six included studies[8,12-16] evaluated the use of VR simulation in EGD training. Measures of post-training competency varied across studies, and only two reported quantitative trainee performance scores following VR training. Ende et al[32] compared VR simulation, clinical training alone, and clinical training supplemented with VR, finding that all approaches significantly improved manual skills from baseline (P < 0.05), with no significant differences between groups. In contrast, Shirai et al[7] compared VR-based and conventional training and reported that a significantly higher proportion of trainees in the traditional training group required assistance to complete the procedure, as indicated by a 1-point rating, compared with the VR group.
Two studies assessed rates of independent procedure completion. Di Giulio et al[29] reported that simulation-based training significantly increased the likelihood of independent completion (odds ratio = 65.7; 95% confidence interval: 20.1-214.4). Similar findings were reported by Shirai et al[7].
All included studies assessed differences in procedure time across intervention groups. Each study reported a significant reduction in procedure time from baseline following training. However, Di Giulio et al[29] found no significant difference between the VR and non-VR simulation groups (10.5 minutes vs 12.4 minutes, respectively). Similarly, Shirai et al[7] reported comparable procedure times between VR and conventional training groups (14.40 minutes vs 14.05 minutes, respectively). Ende et al[32] also observed no significant differences when comparing VR simulation alone, clinical training alone, or combined training approaches (P = 0.405).
Four included studies evaluated the use of VR simulation in ERCP training[20-24], primarily examining performance differences between expert and novice endoscopists. Across studies, procedure times and task-specific performance consistently varied by expertise level. Georgiou et al[24] compared experts and novices across three ERCP scenarios. During bile duct stone removal, total procedure time did not differ between groups (273 ± 83 seconds vs 273 ± 38 seconds; P = 0.99), although experts required significantly less time to visualize the papilla (89 ± 29 seconds vs 124 ± 45 seconds; P = 0.02) and achieve deep cannulation (127 ± 27 seconds vs 160 ± 54 seconds; P = 0.028). In cystic duct leak management, overall procedure time was similar (P = 0.415); however, experts were significantly faster in papilla visualization (P = 0.008), deep cannulation (P < 0.001), diagnosis (P = 0.001), sphincterotomy (P = 0.001), and procedure completion (P = 0.01). No significant differences were observed for hilar stenosis across measured parameters[24]. Bittner et al[21] similarly reported superior performance by experts compared with novices across most ERCP metrics, supporting the face validity of VR simulation. Consistent findings were reported by Arnold et al[20] and Sahakian et al[22], demon
Gao et al[19] uniquely evaluated the use of VR simulation in EUS training. The study compared combined VR simulation and clinical training with clinical training alone. EUS competency was assessed with a written examination scored out of 100. Trainees receiving combined training achieved significantly higher scores than those undergoing clinical training alone (64.53 ± 4.91 vs 60.09 ± 5.49; P = 0.028). Analysis of variance further demonstrated that VR simulation significantly improved EUS skills regardless of baseline endoscopy experience (P = 0.035)[19].
Most included studies evaluated the impact of VR simulation on colonoscopy training[6,9,13-18,27,28,30], whereas only two focused exclusively on sigmoidoscopy[12,29]. Outcomes and control conditions varied across studies, and en
Parallel-arm trials also evaluated the proportion of trainees who completed the colonoscopy independently. McIntosh et al[13] assessed the need for assistance during colonoscopy and found that trainees in the simulator group required significantly fewer assists than those in the control group (1.94 vs 3.43; P = 0.004).
McIntosh et al[13] compared colonoscope insertion times between VR simulator and control groups and found no statistically significant difference between trainees (14.4 vs 14.6; P = 0.37). No other included studies reported comparative procedure time outcomes between groups.
Gerson and Van Dam[8] compared VR simulator training with traditional instruction and found that the traditional teaching group achieved significantly higher competency scores (rated on of 5-point scale) than the simulator cohort (3.8 ± 0.2 vs 2.9 ± 0.2; P < 0.001). A significantly greater proportion of trainees in the traditional group also completed sigmoidoscopy independently (72% vs 29%; P = 0.001)[8]. In contrast, Sedlack[25] reported no significant difference between simulator and control groups in independent procedure completion (8 vs 8; P = 0.893) or procedure time per case (24.10 vs 24 ± 1.1; P > 0.05).
VR simulators have been increasingly integrated into GIE training over recent years. Rather than serving as a replacement for traditional apprenticeship-based methods, VR simulation functions as an adjunct that enhances conventional endoscopic training[34]. In this review, VR simulation is defined as computer-based simulation technology that enables interactive, immersive experiences designed to replicate procedural and clinical aspects of endoscopy.
Our review included studies evaluating the use of VR simulation across upper GI endoscopic procedures, including ERCP, EUS, and EGD. In EGD training, the evidence suggests that VR simulation improves trainee independence compared with conventional training and enhances novice competency relative to baseline performance. These findings are supported by a Cochrane review reporting that VR-trained trainees completed significantly more procedures in
In ERCP training, the studies included in this review primarily evaluated the validity of VR simulators in distinguishing expert from novice performance. Across studies, expert endoscopists consistently outperformed novices across multiple ERCP metrics, particularly in the management of pancreatic pathologies such as cystic duct leakage[24]. These findings support the construct validity of VR simulation as a training and assessment tool for ERCP. However, additional studies are needed to determine whether VR-based training leads to measurable improvements in procedural skills and competency over time. Such evidence will be critical to guide the optimal integration of VR simulation into ERCP training curricula.
In colonoscopy and sigmoidoscopy training, most studies in this review demonstrated significant improvements in endoscopist competency following VR simulation. Several studies also reported higher rates of independent colonoscopy completion among VR-trained trainees compared with controls. However, findings were not entirely consistent. Qiao et al[35] reported no significant difference in independent colonoscopy completion between simulation-based and traditional training groups (89.3% vs 88.9%; P = 0.41). Similarly, sigmoidoscopy outcomes were heterogeneous; one study found higher independent completion rates with traditional instruction[33], whereas another reported no significant difference between training modalities[25]. These inconsistencies highlight variability in study design, outcome measures, and trainee experience, underscoring the need for standardized assessment frameworks in endoscopic training research.
The included studies displayed substantial methodological heterogeneity in trainee experience and specialty, assessed outcomes, and VR intervention designs. This variability precluded quantitative meta-analysis and limited our ability to identify the most effective strategies for implementing VR simulation in endoscopy training.
This article demonstrates that VR simulation is an effective training modality for GIE, with performance outcomes generally comparable to those of traditional training methods. These findings support integrating VR simulation into endoscopy training curricula to prepare trainees before patient-based practice and enhance patient safety. However, substantial heterogeneity in VR training approaches across existing studies limits definitive conclusions regarding optimal implementation. Future research should focus on developing and evaluating standardized VR training fra
| 1. | Fan X, Luo B, Yang L. [Role of Virtual Reality in Gastrointestinal Endoscopy Training and Teaching]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2024;55:315-320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 2. | Khan R, Plahouras J, Johnston BC, Scaffidi MA, Grover SC, Walsh CM. Virtual reality simulation training for health professions trainees in gastrointestinal endoscopy. Cochrane Database Syst Rev. 2018;8:CD008237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 3. | Dunkin B, Adrales GL, Apelgren K, Mellinger JD. Surgical simulation: a current review. Surg Endosc. 2007;21:357-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 144] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 4. | Piskorz MM, Wonaga A, Bortot L, Linares ME, Araya V, Olmos JI, Gardey M, Perretta C, Olmos JA. Impact of a Virtual Endoscopy Training Curriculum in Novice Endoscopists: First Experience in Argentina. Dig Dis Sci. 2020;65:3072-3078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Van Sickle KR, Buck L, Willis R, Mangram A, Truitt MS, Shabahang M, Thomas S, Trombetta L, Dunkin B, Scott D. A multicenter, simulation-based skills training collaborative using shared GI Mentor II systems: results from the Texas Association of Surgical Skills Laboratories (TASSL) flexible endoscopy curriculum. Surg Endosc. 2011;25:2980-2986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 6. | Telem DA, Rattner DW, Gee DW. Endoscopic simulator curriculum improves colonoscopy performance in novice surgical interns as demonstrated in a swine model. Surg Endosc. 2014;28:1494-1499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Shirai Y, Yoshida T, Shiraishi R, Okamoto T, Nakamura H, Harada T, Nishikawa J, Sakaida I. Prospective randomized study on the use of a computer-based endoscopic simulator for training in esophagogastroduodenoscopy. J Gastroenterol Hepatol. 2008;23:1046-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 51] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 8. | Gerson LB, Van Dam J. A prospective randomized trial comparing a virtual reality simulator to bedside teaching for training in sigmoidoscopy. Endoscopy. 2003;35:569-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 76] [Article Influence: 3.3] [Reference Citation Analysis (2)] |
| 9. | Koch AD, Ekkelenkamp VE, Haringsma J, Schoon EJ, de Man RA, Kuipers EJ. Simulated colonoscopy training leads to improved performance during patient-based assessment. Gastrointest Endosc. 2015;81:630-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 10. | Ferlitsch A, Glauninger P, Gupper A, Schillinger M, Haefner M, Gangl A, Schoefl R. Evaluation of a virtual endoscopy simulator for training in gastrointestinal endoscopy. Endoscopy. 2002;34:698-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 107] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 11. | Silva Mendes S, Areia M, Dinis Ribeiro M, Rolanda C. The Impact of a Structured Virtual Reality Simulation Training Curriculum for Novice Endoscopists. GE Port J Gastroenterol. 2022;29:385-392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 12. | Buzink SN, Koch AD, Heemskerk J, Botden SM, Goossens RH, de Ridder H, Schoon EJ, Jakimowicz JJ. Acquiring basic endoscopy skills by training on the GI Mentor II. Surg Endosc. 2007;21:1996-2003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 13. | McIntosh KS, Gregor JC, Khanna NV. Computer-based virtual reality colonoscopy simulation improves patient-based colonoscopy performance. Can J Gastroenterol Hepatol. 2014;28:203-206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Park J, MacRae H, Musselman LJ, Rossos P, Hamstra SJ, Wolman S, Reznick RK. Randomized controlled trial of virtual reality simulator training: transfer to live patients. Am J Surg. 2007;194:205-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 137] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 15. | Gomez PP, Willis RE, Van Sickle K. Evaluation of two flexible colonoscopy simulators and transfer of skills into clinical practice. J Surg Educ. 2015;72:220-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 16. | Cohen J, Cohen SA, Vora KC, Xue X, Burdick JS, Bank S, Bini EJ, Bodenheimer H, Cerulli M, Gerdes H, Greenwald D, Gress F, Grosman I, Hawes R, Mullin G, Schnoll-Sussman F, Starpoli A, Stevens P, Tenner S, Villanueva G. Multicenter, randomized, controlled trial of virtual-reality simulator training in acquisition of competency in colonoscopy. Gastrointest Endosc. 2006;64:361-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 155] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 17. | Grover SC, Garg A, Scaffidi MA, Yu JJ, Plener IS, Yong E, Cino M, Grantcharov TP, Walsh CM. Impact of a simulation training curriculum on technical and nontechnical skills in colonoscopy: a randomized trial. Gastrointest Endosc. 2015;82:1072-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 84] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 18. | Vajpeyi A, Naidu AS, Hawel JD, Schlachta CM, Patel RV. A multi-modal training environment for colonoscopy with pressure feedback. Surg Endosc. 2025;39:960-969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Gao J, Fang J, Jin Z, Wang D, Li Z. Use of simulator for EUS training in the diagnosis of pancreatobiliary diseases. Endosc Ultrasound. 2019;8:25-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 20. | Arnold SH, Svendsen MB, Konge L, Svendsen LB, Preisler L. Three-dimensional motion tracking correlates with skill level in upper gastrointestinal endoscopy. Endoscopy. 2015;47:825-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Bittner JG 4th, Mellinger JD, Imam T, Schade RR, Macfadyen BV Jr. Face and construct validity of a computer-based virtual reality simulator for ERCP. Gastrointest Endosc. 2010;71:357-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 22. | Sahakian AB, Laine L, Jamidar PA, Siddiqui UD, Duffy A, Ciarleglio MM, Deng Y, Nagar A, Aslanian HR. Can a Computerized Simulator Assess Skill Level and Improvement in Performance of ERCP? Dig Dis Sci. 2016;61:722-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Ferlitsch A, Schoefl R, Puespoek A, Miehsler W, Schoeniger-Hekele M, Hofer H, Gangl A, Homoncik M. Effect of virtual endoscopy simulator training on performance of upper gastrointestinal endoscopy in patients: a randomized controlled trial. Endoscopy. 2010;42:1049-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | Georgiou K, Boyanov N, Antonakis P, Thanasas D, Sandblom G, Enochsson L. Validity of a virtual reality endoscopic retrograde cholangiopancreatography simulator: can it distinguish experts from novices? Front Surg. 2023;10:1289197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Sedlack RE. Validation of computer simulation training for esophagogastroduodenoscopy: Pilot study. J Gastroenterol Hepatol. 2007;22:1214-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 26. | Sedlack RE, Kolars JC, Alexander JA. Computer simulation training enhances patient comfort during endoscopy. Clin Gastroenterol Hepatol. 2004;2:348-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 113] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 27. | Sedlack RE, Kolars JC. Computer simulator training enhances the competency of gastroenterology fellows at colonoscopy: results of a pilot study. Am J Gastroenterol. 2004;99:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 201] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 28. | Haycock A, Koch AD, Familiari P, van Delft F, Dekker E, Petruzziello L, Haringsma J, Thomas-Gibson S. Training and transfer of colonoscopy skills: a multinational, randomized, blinded, controlled trial of simulator versus bedside training. Gastrointest Endosc. 2010;71:298-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 88] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 29. | Di Giulio E, Fregonese D, Casetti T, Cestari R, Chilovi F, D'Ambra G, Di Matteo G, Ficano L, Delle Fave G. Training with a computer-based simulator achieves basic manual skills required for upper endoscopy: a randomized controlled trial. Gastrointest Endosc. 2004;60:196-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 75] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Ahlberg G, Hultcrantz R, Jaramillo E, Lindblom A, Arvidsson D. Virtual reality colonoscopy simulation: a compulsory practice for the future colonoscopist? Endoscopy. 2005;37:1198-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 97] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 31. | Grover SC, Scaffidi MA, Khan R, Garg A, Al-Mazroui A, Alomani T, Yu JJ, Plener IS, Al-Awamy M, Yong EL, Cino M, Ravindran NC, Zasowski M, Grantcharov TP, Walsh CM. Progressive learning in endoscopy simulation training improves clinical performance: a blinded randomized trial. Gastrointest Endosc. 2017;86:881-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 32. | Ende A, Zopf Y, Konturek P, Naegel A, Hahn EG, Matthes K, Maiss J. Strategies for training in diagnostic upper endoscopy: a prospective, randomized trial. Gastrointest Endosc. 2012;75:254-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 33. | Hashimoto DA, Petrusa E, Phitayakorn R, Valle C, Casey B, Gee D. A proficiency-based virtual reality endoscopy curriculum improves performance on the fundamentals of endoscopic surgery examination. Surg Endosc. 2018;32:1397-1404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Gadour E, Miutescu B, Hassan Z, Aljahdli ES, Raees K. Advancements in the diagnosis of biliopancreatic diseases: A comparative review and study on future insights. World J Gastrointest Endosc. 2025;17:103391. [PubMed] [DOI] [Full Text] |
| 35. | Qiao W, Bai Y, Lv R, Zhang W, Chen Y, Lei S, Zhi F. The effect of virtual endoscopy simulator training on novices: a systematic review. PLoS One. 2014;9:e89224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |